Corrosion Barrier Coatings: Progress and Perspectives of the Chemical Route
Abstract
:1. Introduction
2. Sol–Gel Processing
3. Silanes
4. ORMOSIL
- a.
- Precursors and Reactions
- b.
- Organically Modified Silane
- c.
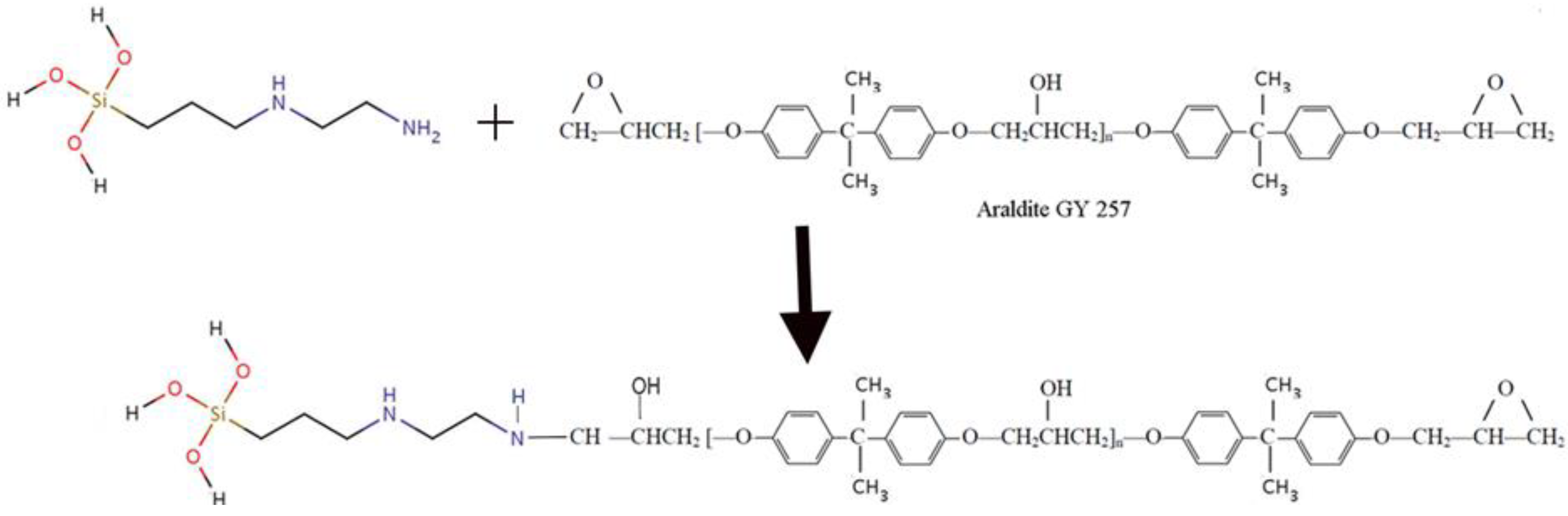
- Epoxy Resin
- d.
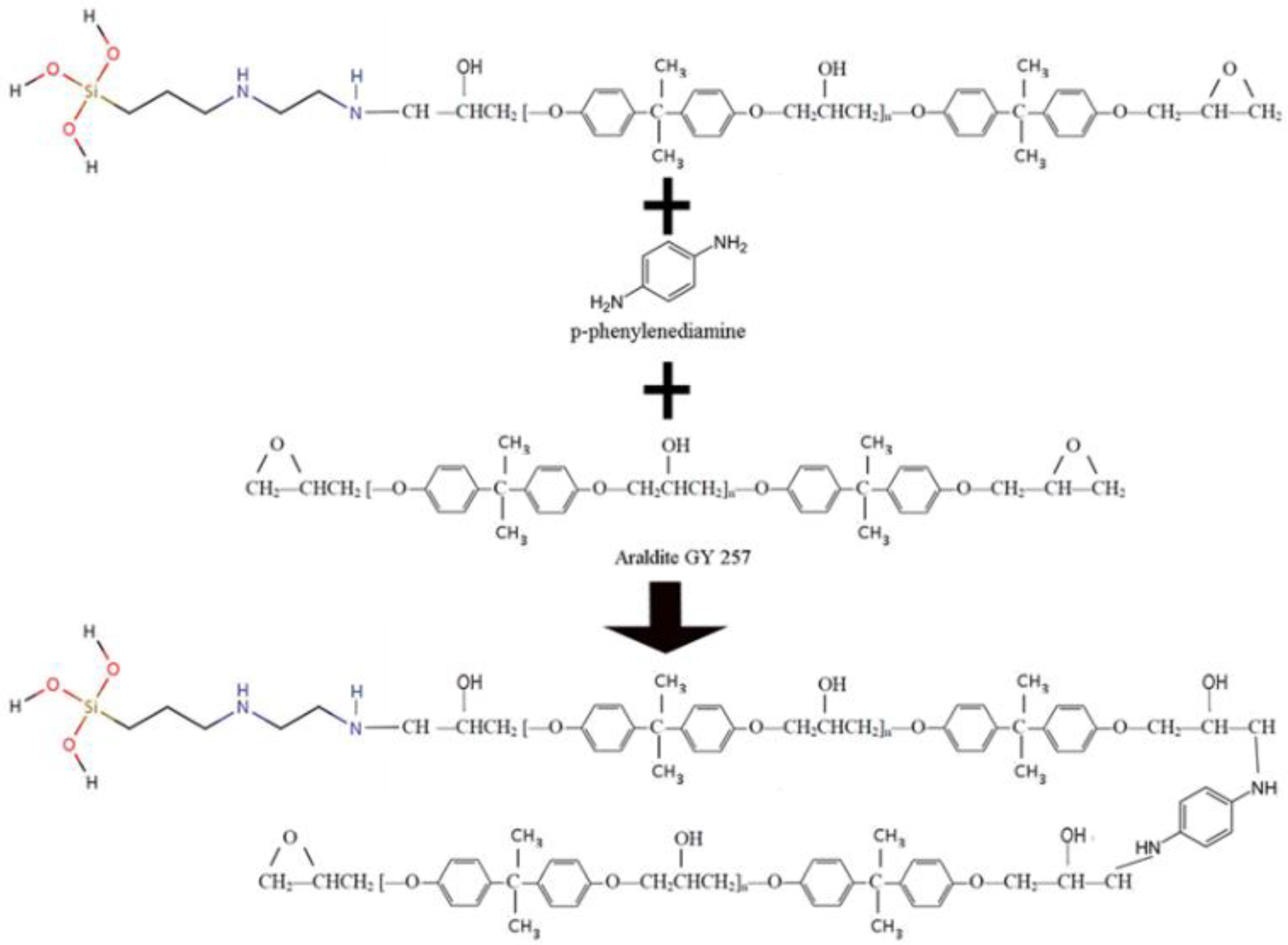
- Hardener
- e.
- Solvents
- f.
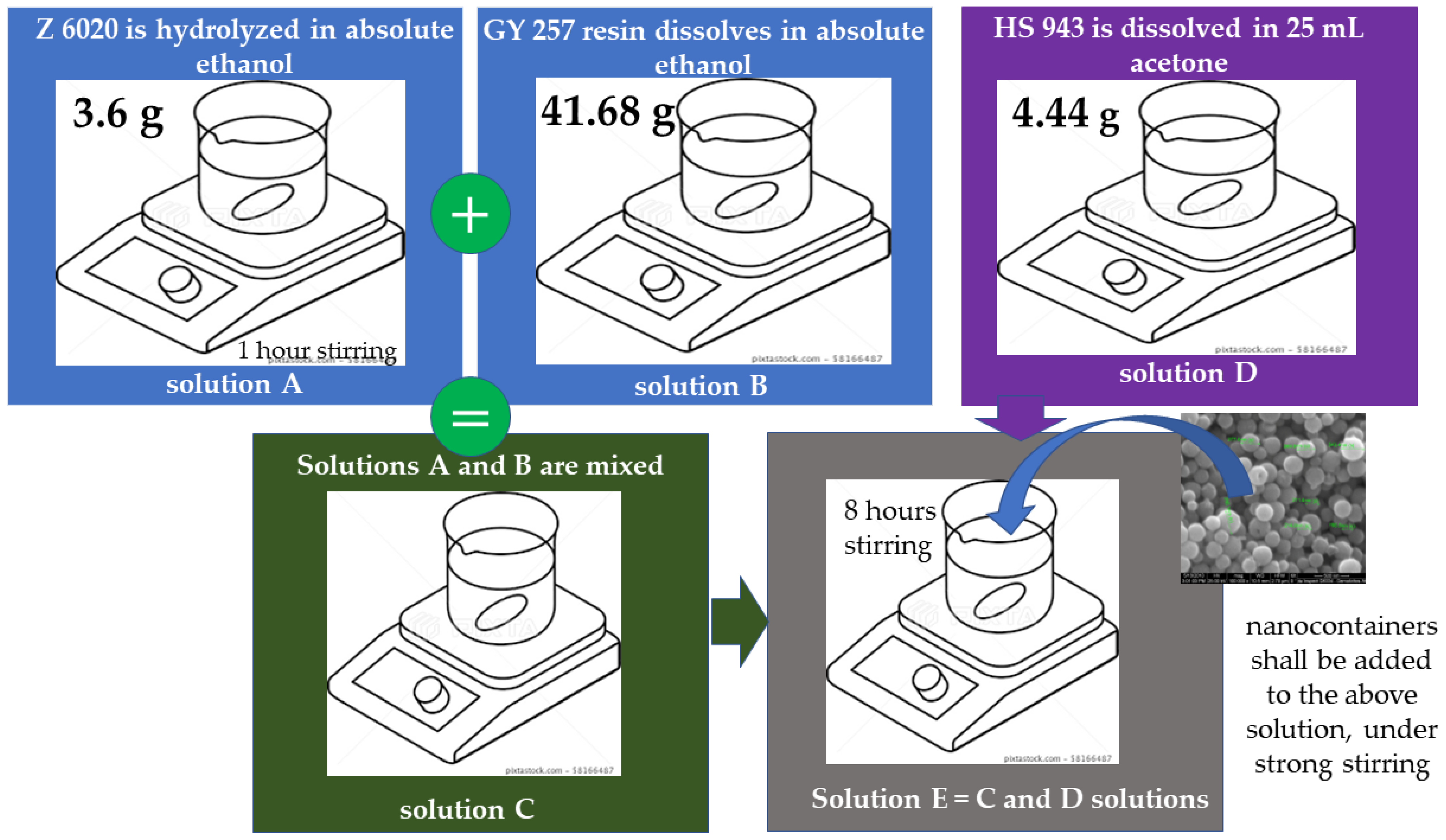
- Preparation of ORMOSIL
5. Water-Based Commercial Sol-Gel CBCs
6. Inhibitors
6.1. Inorganic
6.2. Organic
7. Split Cell Technique
8. Electrochemical Noise (EN) Method
9. Scanning Vibrating Electrode Technique (SVET)
10. Organic Inhibitors in Nanocontainers
11. Nanotechnology
11.1. Silanes and Nanoparticles
11.2. CBCs with Nanocontainers
12. Corrosion Sensors
13. Conclusions with Perspectives
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Park, R.M.; Bena, J.F.; Stayner, L.T.; Smith, R.J.; Gibb, H.J.; Lees, P.S.J. Hexavalent chromium and lung cancer in the chromate industry: A quantitative risk assessment. Risk Anal. 2004, 24, 1099–1108. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, D.; Hou, P.; Pan, J.; Zhao, X.; Hou, B. Preparation and Characterization of Polyelectrolyte-Modified Attapulgite as Nanocontainers for Protection of Carbon Steel. J. Electrochem. Soc. 2018, 165, C907–C915. [Google Scholar] [CrossRef]
- Chen, D.; Ray, A.K. Removal of toxic metal ions from wastewater by semiconductor photocatalysis. Chem. Eng. Sci. 2001, 56, 1561–1570. [Google Scholar] [CrossRef]
- Hoffmann, L.; Grinderslev, M. Mass Flow Analysis of Chromium and Chromium Compounds; Environmental Project, No. 793; The Danish Environmental Protection Agency: Copenhagen, Denmark, 2003. [Google Scholar]
- Authorisation and Restriction of Chemicals (REACH). 1907/2006 (Regula); Repealing Council Regulation (EEC) No 793/93 and Commission Regulation (EC) No 1488/94; EU-OSHA: Brussels, Belgium, 2006. [Google Scholar]
- European Commission. Amending 340/2008; L 139/1; EU-OSHA: Brussels, Belgium, 2018; Volume 2016. [Google Scholar]
- Qi, J. Trivalent Chromium Conversion Coatings on Al and Al-Cu Alloys. PhD Thesis, The University of Manchester, Manchester, UK, 2015. [Google Scholar]
- Qi, J.; Gao, L.; Li, Y.; Wang, Z.; Thompson, G.E.; Skeldon, P. An Optimized Trivalent Chromium Conversion Coating Process for AA2024-T351 Alloy. J. Electrochem. Soc. 2017, 164, C390–C395. [Google Scholar] [CrossRef]
- Guo, Y.; Frankel, G.S. Active corrosion inhibition of AA2024-T3 by trivalent chrome process treatment. Corrosion 2012, 68, 045002-1–045002-10. [Google Scholar] [CrossRef]
- Guo, Y.; Frankel, G.S. Characterization of trivalent chromium process coating on AA2024-T3. Surf. Coat. Technol. 2012, 206, 3895–3902. [Google Scholar] [CrossRef]
- Santa Coloma, P.; Izagirre, U.; Belaustegi, Y.; Jorcin, J.B.; Cano, F.J.; Lapeña, N. Chromium-free conversion coatings based on inorganic salts (Zr/Ti/Mn/Mo) for aluminum alloys used in aircraft applications. Appl. Surf. Sci. 2015, 345, 24–35. [Google Scholar] [CrossRef] [Green Version]
- Chidambaram, D.; Clayton, C.R.; Halada, G.P. The role of hexafluorozirconate in the formation of chromate conversion coatings on aluminum alloys. Electrochim. Acta 2006, 51, 2862–2871. [Google Scholar] [CrossRef]
- Qi, J.T.; Hashimoto, T.; Walton, J.R.; Zhou, X.; Skeldon, P.; Thompson, G.E. Trivalent chromium conversion coating formation on aluminium. Surf. Coat. Technol. 2015, 280, 317–329. [Google Scholar] [CrossRef]
- Corporation, H.; Heights, M.; States, U.; Corporation, H.C.; Capital, H.; Interlomas, C.U.; Huller, R.K.; District, N. Henkel Ready-to-Use Surface Treatments for the Aerospace Industry; Henkel: Dusseldorf, Germany, 2018; Volume 8340. [Google Scholar]
- Socomore. “Socosurf TCS.” Aluminum Surface Treatment. Available online: https://www.socomore.com/en/contact-us (accessed on 18 April 2022).
- Pearlstein, F.; Agarwala, V.S. Trivalent chromium solutions for applying chemical conversion coatings to aluminum alloys or for sealing anodized aluminum. Plat. Surf. Finish. 1994, 81, 50–55. [Google Scholar]
- Li, L.; Swain, G.M. Formation and structure of trivalent chromium process coatings on aluminum alloys 6061 and 7075. Corrosion 2013, 69, 1205–1216. [Google Scholar] [CrossRef]
- Qi, J.; Gao, L.; Liu, Y.; Liu, B.; Hashimoto, T.; Wang, Z.; Thompson, G.E. Chromate Formed in a Trivalent Chromium Conversion Coating on Aluminum. J. Electrochem. Soc. 2017, 164, C442–C449. [Google Scholar] [CrossRef]
- Qi, J.; Światowska, J.; Skeldon, P.; Marcus, P. Chromium valence change in trivalent chromium conversion coatings on aluminium deposited under applied potentials. Corros. Sci. 2020, 167, 108482. [Google Scholar] [CrossRef]
- Eastmond, D.A.; MacGregor, J.T.; Slesinski, R.S. Trivalent chromium: Assessing the genotoxic risk of an essential trace element and widely used human and animal nutritional supplement. Crit. Rev. Toxicol. 2008, 38, 173–190. [Google Scholar] [CrossRef] [PubMed]
- Ambreen, K.; Khan, F.H.; Bhadauria, S.; Kumar, S. Genotoxicity and oxidative stress in chromium-exposed tannery workers in North India. Toxicol. Ind. Health 2014, 30, 405–414. [Google Scholar] [CrossRef]
- Blasiak, J.; Kowalik, J. A comparison of the in vitro genotoxicity of tri- and hexavalent chromium. Mutat. Res.-Genet. Toxicol. Environ. Mutagen. 2000, 469, 135–145. [Google Scholar] [CrossRef]
- Speetjens, J.K.; Collins, R.A.; Vincent, J.B.; Woski, S.A. The nutritional supplement chromium(III) tris(picolinate) cleaves DNA. Chem. Res. Toxicol. 1999, 12, 483–487. [Google Scholar] [CrossRef]
- Stearns, D.M.; Wise, J.P.; Patierno, S.R.; Wetterhahn, K.E. Chromium(III) picolinate produces chromosome damage in Chinese hamster ovary cells. FASEB J. 1995, 9, 1643–1648. [Google Scholar] [CrossRef]
- Coryell, V.H.; Stearns, D.M. Molecular analysis of hprt mutations induced by chromium picolinate in CHO AA8 cells. Mutat. Res.-Genet. Toxicol. Environ. Mutagen. 2006, 610, 114–123. [Google Scholar] [CrossRef]
- Wang, C.; Jiang, F.; Wang, F. Cerium chemical conversion coating for aluminum alloy 2024-T3 and its corrosion resistance. Corrosion 2004, 60, 237–243. [Google Scholar] [CrossRef]
- Giles, T.R.; Vonk, D.; Favero, S.-L. The Next Generation of Conversion Coatings. In Proceedings of the Encontro e Exposição Brasileira de Tratamento de Superficie III INTERFINISH Latino Americano, São Paulo, Brazil, 11–14 April 2012; pp. 172–181. [Google Scholar]
- Kapic, E.; Heights, S.; Goodreau, B.H.; Romeo; Bobadilla, A.; Oak, R.; Febbraro, M.; Knoxville, T. Processes and Compositions for Improving Corrosion Performance of Zirconumoxde Pretreated Zinc Surfaces. US Patent 9,573,162, 21 February 2017. [Google Scholar]
- Vonk, D.R.; Kapic, E.; Sienkowski, M.L. Metal Pretreatment Composition Containing Zirconium, Copper, and Metal Chelatingagents and Related Coatings on Metal Substrates. US Patent 9,284,460, 15 March 2016. [Google Scholar]
- Matsukawa, M.; Shi, N.; Makino, K.; Shi, Y.; Toshiaki Shimakura, I. Chemical Conversion Coating Agent and Surface-Treated Metal. U.S. Patent US7510612B2, 31 March 2009. [Google Scholar]
- Šekularac, G.; Milošev, I. Electrochemical Behavior and Self-Sealing Ability of Zirconium Conversion Coating Applied on Aluminum Alloy 3005 in 0.5 M NaCl Solution. J. Electrochem. Soc. 2020, 167, 021509. [Google Scholar] [CrossRef]
- Doerre, M.; Hibbitts, L.; Patrick, G.; Akafuah, N.K. Advances in automotive conversion coatings during pretreatment of the body structure: A review. Coatings 2018, 8, 405. [Google Scholar] [CrossRef] [Green Version]
- Milošev, I.; Frankel, G.S. Review—Conversion Coatings Based on Zirconium and/or Titanium. J. Electrochem. Soc. 2018, 165, C127–C144. [Google Scholar] [CrossRef]
- Peng, D.; Wu, J.; Yan, X.; Du, X.; Yan, Y.; Li, X. The formation and corrosion behavior of a zirconium-based conversion coating on the aluminum alloy AA6061. J. Coat. Technol. Res. 2016, 13, 837–850. [Google Scholar] [CrossRef]
- Asemani, H.R.; Ahmadi, P.; Sarabi, A.A.; Eivaz Mohammadloo, H. Effect of zirconium conversion coating: Adhesion and anti-corrosion properties of epoxy organic coating containing zinc aluminum polyphosphate (ZAPP) pigment on carbon mild steel. Prog. Org. Coat. 2016, 94, 18–27. [Google Scholar] [CrossRef]
- Taheri, P.; Laha, P.; Terryn, H.; Mol, J.M.C. An in situ study of zirconium-based conversion treatment on zinc surfaces. Appl. Surf. Sci. 2015, 356, 837–843. [Google Scholar] [CrossRef]
- Gray, J.E.; Luan, B. Protective coatings on magnesium and its alloys—A critical review. J. Alloys Compd. 2002, 336, 88–113. [Google Scholar] [CrossRef]
- Šekularac, G.; Kovač, J.; Milošev, I. Prolonged protection, by zirconium conversion coatings, of AlSi7Mg0.3 aluminium alloy in chloride solution. Corros. Sci. 2020, 169, 108615. [Google Scholar] [CrossRef]
- Golru, S.S.; Attar, M.M.; Ramezanzadeh, B. Morphological analysis and corrosion performance of zirconium based conversion coating on the aluminum alloy 1050. J. Ind. Eng. Chem. 2015, 24, 233–244. [Google Scholar] [CrossRef]
- Kordas, G. Nanocontainers Against Biofouling and Corrosion Degradation of Materials: A Short Review With Prospects. Front. Nanotechnol. 2022, 4, 813908. [Google Scholar] [CrossRef]
- Díaz, B.; Härkönen, E.; Światowska, J.; Maurice, V.; Seyeux, A.; Marcus, P.; Ritala, M. Low-temperature atomic layer deposition of Al2O3 thin coatings for corrosion protection of steel: Surface and electrochemical analysis. Corros. Sci. 2011, 53, 2168–2175. [Google Scholar] [CrossRef]
- Garg, R.; Rajagopalan, N.; Pyeon, M.; Gönüllü, Y.; Fischer, T.; Khanna, A.S.; Mathur, S. Plasma CVD grown Al2O3 and MgAl2O4 coatings for corrosion protection applications. Surf. Coat. Technol. 2018, 356, 49–55. [Google Scholar] [CrossRef]
- Wang, D.; Bierwagen, G.P. Sol-gel coatings on metals for corrosion protection. Prog. Org. Coat. 2009, 64, 327–338. [Google Scholar] [CrossRef]
- Figueira, R.B.; Silva, C.J.R.; Pereira, E.V. Organic–inorganic hybrid sol–gel coatings for metal corrosion protection: A review of recent progress. J. Coat. Technol. Res. 2015, 12, 1–35. [Google Scholar] [CrossRef]
- Catubig, R.; Hughes, A.E.; Cole, I.S.; Hinton, B.R.W.; Forsyth, M. The use of cerium and praseodymium mercaptoacetate as thiol-containing inhibitors for AA2024-T3. Corros. Sci. 2014, 81, 45–53. [Google Scholar] [CrossRef]
- Ahamad, I.; Prasad, R.; Quraishi, M.A. Thermodynamic, electrochemical and quantum chemical investigation of some Schiff bases as corrosion inhibitors for mild steel in hydrochloric acid solutions. Corros. Sci. 2010, 52, 933–942. [Google Scholar] [CrossRef]
- Kartsonakis, I.A.; Balaskas, A.C.; Kordas, G.C. Influence of cerium molybdate containers on the corrosion performance of epoxy coated aluminium alloys 2024-T3. Corros. Sci. 2011, 53, 3771–3779. [Google Scholar] [CrossRef]
- Zheludkevich, M.; Tedim, J.; Ferreira, M. “Smart” coatings for active corrosion protection based on multi-functional micro and nanocontainers. Corros. Sci. 2015, 82, 314–323. [Google Scholar] [CrossRef]
- Wang, T.; Du, J.; Ye, S.; Tan, L.; Fu, J. Triple-Stimuli-Responsive Smart Nanocontainers Enhanced Self-Healing Anticorrosion Coatings for Protection of Aluminum Alloy. ACS Appl. Mater. Interfaces 2019, 11, 4425–4438. [Google Scholar] [CrossRef]
- Malinskii, Y.M.; Prokopenko, V.; Ivanova, N.; Kargin, V. A Investigation of Self-Healing of Cracks in Polymers. Mekhanika Polim. 1970, 2, 271–275. [Google Scholar]
- Wool, R.P. Material response and reversible cracks in viscoelastic polymers. Polym. Eng. Sci. 1978, 18, 1057–1061. [Google Scholar] [CrossRef]
- Wool, R.P.; OʹConnor, K.M. A theory crack healing in polymers. J. Appl. Phys. 1981, 52, 5953. [Google Scholar] [CrossRef]
- White, S.R.; Sottos, N.R.; Geubelle, P.H.; Moore, J.S.; Kessler, M.R.; Sriram, S.R.; Brown, E.N.; Viswanathan, S. Autonomic Healing of Polymer Composites. Nature 2001, 409, 794. [Google Scholar] [CrossRef] [PubMed]
- Brinker, C.J.; Scherer, G.W. Sol-Gel Science The Physics and Chemistry of Sol-Gel Processing; Academic Press, Inc.: Boston, UK, 1990; ISBN 978-0-12-134970-7. [Google Scholar]
- Brinker, C.J. Hydrolysis and condensation of silicates: Effect on structure. J. Non. Cryst. Solids 1998, 100, 31–50. [Google Scholar] [CrossRef] [Green Version]
- Innocenzi, P.C.; Guglielmi, M.; Gobbin, M.; Colombo, P. Coating of metals by the sol-gel dip-coating method. J. Eur. Ceram. Soc. 1992, 10, 431–436. [Google Scholar] [CrossRef]
- Deflorian, F.; Rossi, S.; Fedel, M.; Ecco, L.G.; Paganica, R.; Bastarolo, M.; Mathiazhagan, A.; Joseph, R.; Chen, X.; Hong, R.; et al. Corrosion protection properties of organofunctional silanes—An overview. Prog. Org. Coat. 2005, 51, 259–269. [Google Scholar]
- Palomino, L.M.; Suegama, P.H.; Aoki, I.V.; Fatima Montemor, M.; De Melo, H.G. Electrochemical study of modified non-functional bis-silane layers on Al alloy 2024-T3. Corros. Sci. 2008, 50, 1258–1266. [Google Scholar] [CrossRef]
- Montemor, M.F.; Cabral, A.M.; Zheludkevich, M.L.; Ferreira, M.G.S. The corrosion resistance of hot dip galvanized steel pretreated with Bis-functional silanes modified with microsilica. Surf. Coat. Technol. 2006, 200, 2875–2885. [Google Scholar] [CrossRef]
- Franquet, A.; Biesemans, M.; Willem, R.; Terryn, H.; Vereecken, J. Multinuclear 1D- and 2D-NMR study of the hydrolysis and condensation of bis-1,2-(triethoxysilyl)ethane. J. Adhes. Sci. Technol. 2004, 18, 765–778. [Google Scholar] [CrossRef]
- Palanivel, V.M. Modified Silane Thin Films as an Aternative to Chromates for Corrosion Protection of AA2024-T3 Alloy. Ph.D. Thesis, University of Cincinnati, Cincinnatim, OH, USA, 1957. Volume 9. [Google Scholar]
- Figueira, R.B.; Fontinha, I.R.; Silva, C.J.R.; Pereira, E.V.; Yang, K.; van Ooij, W.J.; Seth, A.; Mugada, T.; Pan, G.; Schaefer, D.W.; et al. Hybrid sol-gel coatings: Smart and green materials for corrosion mitigation. Prog. Org. Coat. 2016, 63, 283–292. [Google Scholar] [CrossRef] [Green Version]
- Trabelsi, W.; Dhouibi, L.; Triki, E.; Ferreira, M.G.S.; Montemor, M.F.; Cabral, A.M.; Duarte, R.G.; Montemor, M.F.; Zheludkevich, M.L.; Ferreira, M.G.S.; et al. Corrosion protection properties of organofunctional silanes—An overview. Prog. Org. Coat. 2005, 48, 639–664. [Google Scholar]
- Mirabedini, S.M.; Thompson, G.E.; Moradian, S.; Scantlebury, J.D. Corrosion performance of powder coated aluminium using EIS. Prog. Org. Coat. 2003, 46, 112–120. [Google Scholar] [CrossRef]
- Franquet, A.; Terryn, H.; Vereecken, J. IRSE study on effect of thermal curing on the chemistry and thickness of organosilane films coated on aluminium. Appl. Surf. Sci. 2003, 211, 259–269. [Google Scholar] [CrossRef]
- Aïssa, B.; Therriault, D.; Haddad, E.; Jamroz, W. Self-healing materials systems: Overview of major approaches and recent developed technologies. Adv. Mater. Sci. Eng. 2012, 2012, 854203. [Google Scholar] [CrossRef] [Green Version]
- Ji, W.G.; Hu, J.M.; Liu, L.; Zhang, J.Q.; Cao, C.N. Improving the corrosion performance of epoxy coatings by chemical modification with silane monomers. Surf. Coat. Technol. 2007, 201, 4789–4795. [Google Scholar] [CrossRef]
- Kim, H.; Jang, J. Corrosion protection and adhesion promotion for polyimide/copper system using silane-modified polymeric materials. Polymer 2000, 41, 6553–6561. [Google Scholar] [CrossRef]
- Chen, M.A.; Xie, X.; Zhang, X.M. Interactions of BTESPT silane and maleic anhydride grafted polypropylene with epoxy and application to improve adhesive durability between epoxy and aluminium sheet. Prog. Org. Coat. 2009, 66, 40–51. [Google Scholar] [CrossRef]
- Cabral, A.; Duarte, R.G.; Montemor, M.F.; Zheludkevich, M.L.; Ferreira, M.G.S. Analytical characterisation and corrosion behaviour of bis-[triethoxysilylpropyl]tetrasulphide pre-treated AA2024-T3. Corros. Sci. 2005, 47, 869–881. [Google Scholar] [CrossRef]
- Montoya, P.; Martins, C.R.; De Melo, H.G.; Aoki, I.V.; Jaramillo, F.; Calderón, J.A. Synthesis of polypyrrole-magnetite/silane coatings on steel and assessment of anticorrosive properties. Electrochim. Acta 2014, 124, 100–108. [Google Scholar] [CrossRef]
- Nguyen Thi Le, H.; Bernard, M.C.; Garcia-Renaud, B.; Deslouis, C. Raman spectroscopy analysis of polypyrrole films as protective coatings on iron. Synth. Met. 2004, 140, 287–293. [Google Scholar] [CrossRef]
- Herrasti, P.; Recio, F.J.; Ocón, P.; Fatás, E. Effect of the polymer layers and bilayers on the corrosion behaviour of mild steel: Comparison with polymers containing Zn microparticles. Prog. Org. Coat. 2005, 54, 285–291. [Google Scholar] [CrossRef]
- Van Schaftinghen, T.; Deslouis, C.; Hubin, A.; Terryn, H. Influence of the surface pre-treatment prior to the film synthesis, on the corrosion protection of iron with polypyrrole films. Electrochim. Acta 2006, 51, 1695–1703. [Google Scholar] [CrossRef]
- Giacomelli, C.; Giacomelli, F.C.; Baptista, J.A.A.; Spinelli, A. The effect of oxalic acid on the corrosion of carbon steel. Anti-Corros. Methods Mater. 2004, 51, 105–111. [Google Scholar] [CrossRef]
- Asan, A.; Kabasakaloglu, M. Electrochemical and corrosion behaviors of mild steel coated with polypyrrole. Mater. Sci. 2003, 39, 643–651. [Google Scholar] [CrossRef]
- Rahman, S.U. Corrosion protection of steel by catalyzed polypyrrole films. Surf. Coat. Technol. 2011, 205, 3035–3042. [Google Scholar] [CrossRef]
- Koene, L.; Hamer, W.J.; De Wit, J.H.W. Electrochemical behaviour of poly(pyrrole) coatings on steel. J. Appl. Electrochem. 2006, 36, 545–556. [Google Scholar] [CrossRef]
- Grgur, B.N.; Krstajić, N.V.; Vojnović, M.V.; Lačnjevac, Č.; Gajić-Krstajić, L. The influence of polypyrrole films on the corrosion behavior of iron in acid sulfate solutions. Prog. Org. Coat. 1998, 33, 1–6. [Google Scholar] [CrossRef]
- Grgur, B.N.; Živković, P.; Gvozdenović, M.M. Kinetics of the mild steel corrosion protection by polypyrrole-oxalate coating in sulfuric acid solution. Prog. Org. Coat. 2006, 56, 240–247. [Google Scholar] [CrossRef]
- Ocón, P.; Cristobal, A.B.; Herrasti, P.; Fatas, E. Corrosion performance of conducting polymer coatings applied on mild steel. Corros. Sci. 2005, 47, 649–662. [Google Scholar] [CrossRef]
- Roy, I. Synthesis, Surface Modification, Characterization, and Biomedical In Vitro Applications of Organically Modified Silica (ORMOSIL) Nanoparticles. In Nanoparticles in Biology and Medicine: Methods and Protocols; Soloviev, M., Ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2012; Volume 906, pp. 365–379. ISBN 9781617799532. [Google Scholar]
- Kordas, G. Nanocontainers (CeO2): Synthesis, Characterization, Properties, and Anti-corrosive Application. ACS Symp. Ser. 2021, 1404, 177–185. [Google Scholar]
- Kordas, G. Nanocontainers for Enhanced Protection of HDG Steel Used in Concrete. Materials 2022, 2, 1–12. [Google Scholar]
- Metroke, T.L.; Apblett, A. Effect of solvent dilution on corrosion protective properties of Ormosil coatings on 2024-T3 aluminum alloy. Prog. Org. Coat. 2004, 51, 36–46. [Google Scholar] [CrossRef]
- Metroke, T.L.; Kachurina, O.; Knobbe, E.T. Spectroscopic and corrosion resistance characterization of GLYMO-TEOS Ormosil coatings for aluminum alloy corrosion inhibition. Prog. Org. Coat. 2002, 44, 295–305. [Google Scholar] [CrossRef]
- Program, E.E.S.T.C. Non-Chromate Aluminum Pretreatments Phase II Interim Report. Available online: http://www.iaindy.com/Documents/ncapii.pdf (accessed on 18 April 2022).
- Chung, Y.J.; Jeanjaquet, S.L.; Kendig, M.W. Normal Probability Function. US Patent 6,579,472 B2, 17 June 2003. [Google Scholar]
- Fedrizzi, L.; Fürbeth, W.; Montemor, F. (Eds.) Self-Healing Properties of New Surface Treatments; NUMBER 58; European Federation of Corrosion, Institute of Materials, Minerals, and Mining: Wakefield, UK, 2011; Volume 47, ISBN 9781906540364. [Google Scholar]
- Davenport, A.J.; Isaacs, H.S.; Kendig, M.W. XANES investigation of the role of cerium compounds as corrosion inhibitors for aluminum. Corros. Sci. 1991, 32, 653–663. [Google Scholar] [CrossRef]
- Pourbaix, M. Atlas of Electrochemical Equilibria in Aqueous Solutions. NACE Int. 1974, 307, 194. [Google Scholar]
- Hayes, S.A.; Yu, P.; O’Keefe, T.J.; O’Keefe, M.J.; Stoffer, J.O. The Phase Stability of Cerium Species in Aqueous Systems. J. Electrochem. Soc. 2002, 149, C623. [Google Scholar] [CrossRef]
- Aldykiewicz, A.J.; Davenport, A.J.; Isaacs, H.S. Studies of the Formation of Cerium-Rich Protective Films Using X-Ray Absorption Near-Edge Spectroscopy and Rotating Disk Electrode Methods. J. Electrochem. Soc. 1996, 143, 147–154. [Google Scholar] [CrossRef]
- Böhm, S.; Greef, R.; McMurray, H.N.; Powell, S.M.; Worsley, D.A. Kinetic and Mechanistic Studies of Rare Earth-Rich Protective Film Formation Using In Situ Ellipsometry. J. Electrochem. Soc. 2000, 147, 3286. [Google Scholar] [CrossRef]
- Brusciotti, F.; Batan, A.; De Graeve, I.; Wenkin, M.; Biessemans, M.; Willem, R.; Reniers, F.; Pireaux, J.J.; Piens, M.; Vereecken, J.; et al. Characterization of thin water-based silane pre-treatments on aluminium with the incorporation of nano-dispersed CeO2 particles. Surf. Coat. Technol. 2010, 205, 603–613. [Google Scholar] [CrossRef]
- Cambon, J.B.; Esteban, J.; Ansart, F.; Bonino, J.P.; Turq, V.; Santagneli, S.H.; Santilli, C.V.; Pulcinelli, S.H. Effect of cerium on structure modifications of a hybrid sol-gel coating, its mechanical properties and anti-corrosion behavior. Mater. Res. Bull. 2012, 47, 3170–3176. [Google Scholar] [CrossRef] [Green Version]
- Cabral, A.M.; Trabelsi, W.; Serra, R.; Montemor, M.F.; Zheludkevich, M.L.; Ferreira, M.G.S. The corrosion resistance of hot dip galvanised steel and AA2024-T3 pre-treated with bis-[triethoxysilylpropyl] tetrasulfide solutions doped with Ce(NO3)3. Corros. Sci. 2006, 48, 3740–3758. [Google Scholar] [CrossRef]
- Montemor, M.F.; Ferreira, M.G.S. Cerium salt activated nanoparticles as fillers for silane films: Evaluation of the corrosion inhibition performance on galvanised steel substrates. Electrochim. Acta 2007, 52, 6976–6987. [Google Scholar] [CrossRef]
- Farzi, G.; Davoodi, A.; Ahmadi, A.; Neisiany, R.E.; Anwer, M.K.; Aboudzadeh, M.A. Encapsulation of Cerium Nitrate within Poly(urea-formaldehyde) Microcapsules for the Development of Self-Healing Epoxy-Based Coating. ACS Omega 2021, 6, 31147–31153. [Google Scholar] [CrossRef]
- Wang, H.; Akid, R. Encapsulated cerium nitrate inhibitors to provide high-performance anti-corrosion sol-gel coatings on mild steel. Corros. Sci. 2008, 50, 1142–1148. [Google Scholar] [CrossRef]
- Trabelsi, W.; Cecilio, P.; Ferreira, M.G.S.; Montemor, M.F. Electrochemical assessment of the self-healing properties of Ce-doped silane solutions for the pre-treatment of galvanised steel substrates. Prog. Org. Coat. 2005, 54, 276–284. [Google Scholar] [CrossRef]
- Pepe, A.; Aparicio, M.; Durán, A.; Ceré, S. Cerium hybrid silica coatings on stainless steel AISI 304 substrate. J. Sol-Gel Sci. Technol. 2006, 39, 131–138. [Google Scholar] [CrossRef]
- Pepe, A.; Aparicio, M.; Ceré, S.; Durán, A. Preparation and characterization of cerium doped silica sol-gel coatings on glass and aluminum substrates. J. Non. Cryst. Solids 2004, 348, 162–171. [Google Scholar] [CrossRef]
- Trabelsi, W.; Triki, E.; Dhouibi, L.; Ferreira, M.G.S.; Zheludkevich, M.L.; Montemor, M.F. The use of pre-treatments based on doped silane solutions for improved corrosion resistance of galvanised steel substrates. Surf. Coat. Technol. 2006, 200, 4240–4250. [Google Scholar] [CrossRef]
- Voevodin, N.N.; Grebasch, N.T.; Soto, W.S.; Arnold, F.E.; Donley, M.S. Potentiodynamic evaluation of sol-gel coatings with inorganic inhibitors. Surf. Coat. Technol. 2001, 140, 24–28. [Google Scholar] [CrossRef]
- Morozova, M.; Kluson, P.; Krysa, J.; Zlamal, M.; Solcova, O.; Kment, S.; Steck, T. Role of the template molecular structure on the photo-electrochemical functionality of the sol-gel titania thin films. J. Sol-Gel Sci. Technol. 2009, 52, 398–407. [Google Scholar] [CrossRef]
- Schem, M.; Schmidt, T.; Gerwann, J.; Wittmar, M.; Veith, M.; Thompson, G.E.; Molchan, I.S.; Hashimoto, T.; Skeldon, P.; Phani, A.R.; et al. CeO2-filled sol-gel coatings for corrosion protection of AA2024-T3 aluminium alloy. Corros. Sci. 2009, 51, 2304–2315. [Google Scholar] [CrossRef]
- Zhu, D.; Van Ooij, W.J. Corrosion protection of metals by water-based silane mixtures of bis-[trimethoxysilylpropyl]amine and vinyltriacetoxysilane. Prog. Org. Coat. 2004, 49, 42–53. [Google Scholar] [CrossRef]
- Owczarek, E. Methods of modifying anticorrosive protective properties of silane films. Acta Phys. Pol. A 2019, 135, 147–152. [Google Scholar] [CrossRef]
- Yu, Z.; Hu, J.; Meng, H. A Review of Recent Developments in Coating Systems for Hot-Dip Galvanized Steel. Front. Mater. 2020, 7, 74. [Google Scholar] [CrossRef] [Green Version]
- Kozaderov, O.; Shikhaliev, K.; Prabhakar, C.; Tripathi, A.; Shevtsov, D.; Kruzhilin, A.; Komarova, E.; Potapov, A.; Zartsyn, I.; Kuznetsov, Y. Corrosion of α-Brass in Solutions Containing Chloride Ions and 3-Mercaptoalkyl-5-amino-lfi-l,2,4-triazoles. Appl. Sci. 2019, 9, 2821. [Google Scholar] [CrossRef] [Green Version]
- Özcan, M.; Dehri, I.; Erbil, M. Organic sulphur-containing compounds as corrosion inhibitors for mild steel in acidic media: Correlation between inhibition efficiency and chemical structure. Appl. Surf. Sci. 2004, 236, 155–164. [Google Scholar] [CrossRef]
- Marconato, J.C.; Bulhões, L.O.; Temperin, M.L. A spectroelectrochemical study of the inhibition of the electrode process on copper by 2-mercaptobenzothiazole in ethanolic solutions. Electrochim. Acta 1997, 43, 771–780. [Google Scholar] [CrossRef]
- Ebenso, E.E.; Isabirye, D.A.; Eddy, N.O. Adsorption and quantum chemical studies on the inhibition potentials of some thiosemicarbazides for the corrosion of mild steel in acidic medium. Int. J. Mol. Sci. 2010, 11, 2473–2498. [Google Scholar] [CrossRef]
- Balaskas, A.C.; Curioni, M.; Thompson, G.E. Corrosion protection mechanism of 2-mercaptobenzothiazole and its potential synergistic effect with cerium ions for treatment of AA 2024-T3. J. Electroanal. Chem. 2020, 863, 114081. [Google Scholar] [CrossRef]
- Lamaka, S.V.; Zheludkevich, M.L.; Yasakau, K.A.; Montemor, M.F.; Ferreira, M.G.S.S. High effective organic corrosion inhibitors for 2024 aluminium alloy. Electrochim. Acta 2007, 52, 7231–7247. [Google Scholar] [CrossRef]
- Tan, B.; Zhang, S.; Cao, X.; Fu, A.; Guo, L.; Marzouki, R.; Li, W. Insight into the anti-corrosion performance of two food flavors as eco-friendly and ultra-high performance inhibitors for copper in sulfuric acid medium. J. Colloid Interface Sci. 2022, 609, 838–851. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.; Lan, W.; Zhang, S.; Deng, H.; Qiang, Y.; Fu, A.; Ran, Y.; Xiong, J.; Marzouki, R.; Li, W. Passiflora edulia Sims leaves Extract as renewable and degradable inhibitor for copper in sulfuric acid solution. Colloids Surf. A Physicochem. Eng. Asp. 2022, 645, 128892. [Google Scholar] [CrossRef]
- Balaskas, A.; Curioni, M.; Thompson, G. Effectiveness of 2-mercaptobenzothiazole, 8-hydroxyquinoline and benzotriazol as corrosion inhibitors on AA 2024-T3 assessed by electrochemical methos. Surf. Interface Anal. 2015, 47, 1029–1039. [Google Scholar] [CrossRef]
- Curioni, M.; Balaskas, A.C.; Thompson, G.E. An alternative to the use of a zero resistance ammeter for electrochemical noise measurement: Theoretical analysis, experimental validation and evaluation of electrode asymmetry. Corros. Sci. 2013, 77, 281–291. [Google Scholar] [CrossRef] [Green Version]
- Balaskas, A.C.; Curioni, M.; Thompson, G.E. Evaluation of Inhibitor Performance by Electrochemical Methods: Comparative Study of Nitrate Salts on AA 2024-T3. J. Electrochem. Soc. 2014, 161, C389–C394. [Google Scholar] [CrossRef]
- Li, S.M.; Zhang, H.R.; Liu, J.H. Corrosion behavior of aluminum alloy 2024-T3 by 8-hydroxy-quinoline and its derivative in 3.5% chloride solution. Trans. Nonferrous Met. Soc. China 2007, 17, 318–325. [Google Scholar] [CrossRef]
- Wang, A.Q.; Golden, T.D. Anodic Electrodeposition of Cerium Oxide Thin Films. J. Electrochem. Soc. 2003, 150, C616. [Google Scholar] [CrossRef]
- Kartsonakis, I.; Daniilidis, I.; Kordas, G. Encapsulation of the corrosion inhibitor 8-hydroxyquinoline into ceria nanocontainers. J. Sol-Gel Sci. Technol. 2008, 48, 24–31. [Google Scholar] [CrossRef]
- Liu, L.; Hu, J.M.; Zhang, J.Q.; Cao, C.N. Improving the formation and protective properties of silane films by the combined use of electrodeposition and nanoparticles incorporation. Electrochim. Acta 2006, 52, 538–545. [Google Scholar] [CrossRef]
- Phanasgaonkar, A.; Raja, V.S. Influence of curing temperature, silica nanoparticles- and cerium on surface morphology and corrosion behaviour of hybrid silane coatings on mild steel. Surf. Coat. Technol. 2009, 203, 2260–2271. [Google Scholar] [CrossRef]
- Chen, Y.; Jin, L.; Xie, Y. Sol-Gel Processing of Organic-Inorganic Nanocomposite Protective Coatings. J. Sol-Gel Sci. Technol. 1998, 13, 735–738. [Google Scholar] [CrossRef]
- Wu, L.; Yang, H.; Wang, H.; Lu, J. Electrosynthesis of cyclic carbonates from CO2 and epoxides on a reusable copper nanoparticle cathode. RSC Adv. 2015, 5, 23189–23192. [Google Scholar] [CrossRef]
- Asadi, N.; Naderi, R.; Saremi, M.; Arman, S.Y.; Fedel, M.; Deflorian, F. Study of corrosion protection of mild steel by eco-friendly silane sol-gel coating. J. Sol-Gel Sci. Technol. 2014, 70, 329–338. [Google Scholar] [CrossRef]
- Ramezanzadeh, B.; Ahmadi, A.; Mahdavian, M. Enhancement of the corrosion protection performance and cathodic delamination resistance of epoxy coating through treatment of steel substrate by a novel nanometric sol-gel based silane composite film filled with functionalized graphene oxide nanosheets. Corros. Sci. 2016, 109, 182–205. [Google Scholar] [CrossRef]
- Zheludkevich, M.L.; Serra, R.; Montemor, M.F.; Yasakau, K.A.; Salvado, I.M.M.; Ferreira, M.G.S. Nanostructured sol-gel coatings doped with cerium nitrate as pre-treatments for AA2024-T3 Corrosion protection performance. Electrochim. Acta 2005, 51, 208–217. [Google Scholar] [CrossRef]
- Cant, N.E.; Critchley, K.; Zhang, H.L.; Evans, S.D. Surface functionalisation for the self-assembly of nanoparticle/polymer multilayer films. Thin Solid Films 2003, 426, 31–39. [Google Scholar] [CrossRef]
- Palanivel, V.; Zhu, D.; Van Ooij, W.J. Nanoparticle-filled silane films as chromate replacements for aluminum alloys. Prog. Org. Coat. 2003, 47, 384–392. [Google Scholar] [CrossRef]
- Carraro, M.; Gross, S. Hybrid materials based on the embedding of organically modified transition metal oxoclusters or polyoxometalates into polymers for functional applications: A review. Materials 2014, 7, 3956–3989. [Google Scholar] [CrossRef] [Green Version]
- Zheludkevich, M.L.; Serra, R.; Montemor, M.F.; Ferreira, M.G.S. Oxide nanoparticle reservoirs for storage and prolonged release of the corrosion inhibitors. Electrochem. Commun. 2005, 7, 836–840. [Google Scholar] [CrossRef]
- Susac, D.; Leung, C.W.; Sun, X.; Wong, K.C.; Mitchell, K.A.R. Comparison of a chromic acid and a BTSE final rinse applied to phosphated 2024-T3 aluminum alloy. Surf. Coat. Technol. 2004, 187, 216–224. [Google Scholar] [CrossRef]
- Biedunkiewicz, A.; Gordon, N.; Straszko, J.; Tamir, S. Kinetics of thermal oxidation of titanium carbide and its carbon nano-composites in dry air atmosphere. J. Therm. Anal. Calorim. 2007, 88, 717–722. [Google Scholar] [CrossRef]
- Wittmar, A.; Wittmar, M.; Caparrotti, H.; Veith, M. The influence of the inhibitor particle sizes to the corrosion properties of hybrid sol-gel coatings. J. Sol-Gel Sci. Technol. 2011, 59, 621–628. [Google Scholar] [CrossRef]
- Trenado, C.; Wittmar, M.; Veith, M.; Rosero-Navarro, N.C.; Aparicio, M.; Durn, A.; Castro, Y.; Strauss, D.J. Multiscale numerical modeling of Ce3+-inhibitor release from novel corrosion protection coatings. Model. Simul. Mater. Sci. Eng. 2011, 19, 025009. [Google Scholar] [CrossRef] [Green Version]
- Rosero-Navarro, N.C.; Paussa, L.; Andreatta, F.; Castro, Y.; Durán, A.; Aparicio, M.; Fedrizzi, L. Optimization of hybrid sol-gel coatings by combination of layers with complementary properties for corrosion protection of AA2024. Prog. Org. Coat. 2010, 69, 167–174. [Google Scholar] [CrossRef]
- Palermo, E.F.; Lee, D.K.; Ramamoorthy, A.; Kuroda, K. Role of cationic group structure in membrane binding and disruption by amphiphilic copolymers. J. Phys. Chem. B 2011, 115, 366–375. [Google Scholar] [CrossRef] [Green Version]
- Gite, V.V.; Tatiya, P.D.; Marathe, R.J.; Mahulikar, P.P.; Hundiwale, D.G. Microencapsulation of quinoline as a corrosion inhibitor in polyurea microcapsules for application in anticorrosive PU coatings. Prog. Org. Coat. 2015, 83, 11–18. [Google Scholar] [CrossRef]
- Kartsonakis, I.A.; Balaskas, A.C.; Kordas, G.C. Influence of TiO2 nanocontainers on hybrid organic-inorganic coatings for corrosion protection of magnesium alloy. Int. J. Struct. Integr. 2013, 4, 127–142. [Google Scholar] [CrossRef]
- Szabó, T.; Molnár-Nagy, L.; Bognár, J.; Nyikos, L.; Telegdi, J. Self-healing microcapsules and slow release microspheres in paints. Prog. Org. Coat. 2011, 72, 52–57. [Google Scholar] [CrossRef]
- Samadzadeh, M.; Boura, S.H.; Peikari, M.; Kasiriha, S.M.; Ashrafi, A. A review on self-healing coatings based on micro/nanocapsules. Prog. Org. Coat. 2010, 68, 159–164. [Google Scholar] [CrossRef]
- Thanawala, K.; Mutneja, N.; Khanna, A.S.; Singh Raman, R.K. Development of self-healing coatings based on linseed oil as autonomous repairing agent for corrosion resistance. Materials 2014, 7, 7324–7338. [Google Scholar] [CrossRef] [Green Version]
- Kartsonakis, I.A.; Kontogiani, P.; Pappas, G.S.; Kordas, G. Photocatalytic action of cerium molybdate and iron-titanium oxide hollow nanospheres on Escherichia coli. J. Nanoparticle Res. 2013, 15, 1759. [Google Scholar] [CrossRef]
- Kartsonakis, I.A.; Koumoulos, E.P.; Charitidis, C.A.; Kordas, G. Hybrid organic-inorganic coatings including nanocontainers for corrosion protection of magnesium alloy ZK30 Nanostructured Materials 2012. Special Issue Editors: Juan Manuel Rojo, Vasileios Koutsos. J. Nanoparticle Res. 2013, 15, 1871. [Google Scholar] [CrossRef]
- Kordas, G.C.; Balaskas, A.C.; Kartsonakis, I.A.; Efthimiadou, E.K. A Raman study of 8-Hydroxyquinoline release from loaded TiO2 nanocontainer. Int. J. Struct. Integr. 2013, 4, 121–126. [Google Scholar] [CrossRef]
- Parameswaranpillai, J.; Salim, N.V.; Pulikkalparambil, H.; Rangappa, S.M.; Siengchin, I.; Habil, S. Micro- and Nano-containers for Smart Applications; Springer Nature Singapore Pte Ltd.: Singapore, 2022; ISBN 9789811681455. [Google Scholar]
- Rule, J.D.; Sottos, N.R.; White, S.R. Effect of microcapsule size on the performance of self-healing polymers. Polymer 2007, 48, 3520–3529. [Google Scholar] [CrossRef]
- Borisova, D.; Möhwald, H.; Shchukin, D.G. Influence of embedded nanocontainers on the efficiency of active anticorrosive coatings for aluminum alloys part II: Influence of nanocontainer position. ACS Appl. Mater. Interfaces 2013, 5, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Kartsonakis, I.A.; Athanasopoulou, E.; Snihirova, D.; Martins, B.; Koklioti, M.A.; Montemor, M.F.; Kordas, G.; Charitidis, C.A. Multifunctional epoxy coatings combining a mixture of traps and inhibitor loaded nanocontainers for corrosion protection of AA2024-T3. Corros. Sci. 2014, 85, 147–159. [Google Scholar] [CrossRef]
- Krzak, M.; Tabor, Z.; Nowak, P.; Warszyński, P.; Karatzas, A.; Kartsonakis, I.A.; Kordas, G.C.; Warszy, P.; Karatzas, A.; Kartsonakis, I.A.; et al. Water diffusion in polymer coatings containing water-trapping particles. Part 2. Experimental verification of the mathematical model. Prog. Org. Coat. 2012, 75, 207–214. [Google Scholar] [CrossRef]
- Buchheit, R.G.; Guan, H.; Mahajanam, S.; Wong, F. Active corrosion protection and corrosion sensing in chromate-free organic coatings. Prog. Org. Coat. 2003, 47, 174–182. [Google Scholar] [CrossRef]
- Zheludkevich, M.L.; Poznyak, S.K.; Rodrigues, L.M.; Raps, D.; Hack, T.; Dick, L.F.; Nunes, T.; Ferreira, M.G.S.S. Active protection coatings with layered double hydroxide nanocontainers of corrosion inhibitor. Corros. Sci. 2010, 52, 602–611. [Google Scholar] [CrossRef]
- Tedim, J.; Kuznetsova, A.; Salak, A.N.; Montemor, F.; Snihirova, D.; Pilz, M.; Zheludkevich, M.L.; Ferreira, M.G.S. Zn-Al layered double hydroxides as chloride nanotraps in active protective coatings. Corros. Sci. 2012, 55, 1–4. [Google Scholar] [CrossRef]
- Montemor, M.F.; Snihirova, D.V.; Taryba, M.G.; Lamaka, S.V.; Kartsonakis, I.A.; Balaskas, A.C.; Kordas, G.C.; Tedim, J.; Kuznetsova, A.; Zheludkevich, M.L.; et al. Evaluation of self-healing ability in protective coatings modified with combinations of layered double hydroxides and cerium molibdate nanocontainers filled with corrosion inhibitors. Electrochim. Acta 2012, 60, 31–40. [Google Scholar] [CrossRef]
- Shchukin, B.D.G.; Zheludkevich, M.; Yasakau, K.; Lamaka, S.; Ferreira, M.G.S.; Möhwald, H. Layer-by-Layer Assembled Nanocontainers for Self-Healing Corrosion Protection. Adv. Mater. 2006, 18, 1672–1678. [Google Scholar] [CrossRef]
- Sonawane, S.H.; Bhanvase, B.A.; Jamali, A.A.; Dubey, S.K.; Kale, S.S.; Pinjari, D.V.; Kulkarni, R.D.; Gogate, P.R.; Pandit, A.B. Improved active anticorrosion coatings using layer-by-layer assembled ZnO nanocontainers with benzotriazole. Chem. Eng. J. 2012, 189, 464–472. [Google Scholar] [CrossRef]
- Bhanvase, B.A.; Patel, M.A.; Sonawane, S.H. Kinetic properties of layer-by-layer assembled cerium zinc molybdate nanocontainers during corrosion inhibition. Corros. Sci. 2014, 88, 170–177. [Google Scholar] [CrossRef]
- Balaskas, A.C.; Kartsonakis, I.; Bilalis, P.; Karatzas, A.; Kordas, G. Epoxy Coatings Containing Nanocontainers Loaded with Corrosion Inhibitors for Corrosion Protection of AA 2024-T3. ECS Meet. Abstr. 2011, MA2011-02, 1656. [Google Scholar] [CrossRef]
- Balaskas, A.C.; Hashimoto, T.; Curioni, M.; Thompson, G.E. Two-shell structured PMAA@CeO2 nanocontainers loaded with 2-mercaptobenzothiazole for corrosion protection of damaged epoxy coated AA 2024-T3. Nanoscale 2017, 9, 5499–5508. [Google Scholar] [CrossRef]
- Chenan, A.; Ramya, S.; George, R.P.; Kamachi Mudali, U. Hollow mesoporous zirconia nanocontainers for storing and controlled releasing of corrosion inhibitors. Ceram. Int. 2014, 40, 10457–10463. [Google Scholar] [CrossRef]
- Chenan, A.; Ramya, S.; George, R.P.; Mudali, U.K. 2-mercaptobenzothiazole-loaded hollow mesoporous silica-based hybrid coatings for corrosion protection of modified 9Cr-1Mo ferritic steel. Corrosion 2014, 70, 496–511. [Google Scholar] [CrossRef]
- Chen, T.; Chen, R.; Jin, Z.; Liu, J. Engineering hollow mesoporous silica nanocontainers with molecular switches for continuous self-healing anticorrosion coating. J. Mater. Chem. A 2015, 3, 9510–9516. [Google Scholar] [CrossRef]
- Qian, B.; Michailidis, M.; Bilton, M.; Hobson, T.; Zheng, Z.; Shchukin, D. Tannic complexes coated nanocontainers for controlled release of corrosion inhibitors in self-healing coatings. Electrochim. Acta 2019, 297, 1035–1041. [Google Scholar] [CrossRef]
- Manasa, S.; Jyothirmayi, A.; Siva, T.; Sathiyanarayanan, S.; Gobi, K.V.; Subasri, R. Effect of inhibitor loading into nanocontainer additives of self-healing corrosion protection coatings on aluminum alloy A356.0. J. Alloys Compd. 2017, 726, 969–977. [Google Scholar] [CrossRef]
- Manasa, S.; Siva, T.; Sathiyanarayanan, S.; Gobi, K.V.; Subasri, R. Montmorillonite nanoclay-based self-healing coatings on AA 2024-T4. J. Coat. Technol. Res. 2018, 15, 721–735. [Google Scholar] [CrossRef]
- He, Y.; Zhang, C.; Wu, F.; Xu, Z. Fabrication study of a new anticorrosion coating based on supramolecular nanocontainer. Synth. Met. 2016, 212, 186–194. [Google Scholar] [CrossRef]
- Shchukin, D.G.; Lamaka, S.V.; Yasakau, K.A.; Zheludkevich, M.L.; Ferreira, M.G.S.; Möhwald, H. Active anticorrosion coatings with halloysite nanocontainers. J. Phys. Chem. C 2008, 112, 958–964. [Google Scholar] [CrossRef]
- Yabuki, A.; Shiraiwa, T.; Fathona, I.W. pH-controlled self-healing polymer coatings with cellulose nanofibers providing an effective release of corrosion inhibitor. Corros. Sci. 2016, 103, 117–123. [Google Scholar] [CrossRef]
- Sauvant-Moynot, V.; Gonzalez, S.; Kittel, J.; Leal, D.A.; Riegel-Vidotti, I.C.; Ferreira, M.G.S.; Marino, C.E.B.; Lv, L.P.; Zhao, Y.; Vilbrandt, N.; et al. Engineering hollow mesoporous silica nanocontainers with molecular switches for continuous self-healing anticorrosion coating. Corros. Sci. 2018, 3, 5499–5508. [Google Scholar]
- Song, Y.; Jo, Y.; Lim, Y.; Cho, S.; Yu, H.; Ryu, B.; Lee, S.; Chung, C. Sunlight-Induced Self-Healing of a Microcapsule-Type Protective Coating. Appl. Mater. Interfaces 2013, 5, 378–384. [Google Scholar] [CrossRef]
- Zhao, Y.; Berger, R.; Landfester, K.; Crespy, D. Double Redox-Responsive Release of Encoded and Encapsulated Molecules from Patchy Nanocapsules. Small 2015, 11, 2995–2999. [Google Scholar] [CrossRef]
- Saji, V.S. Supramolecular concepts and approaches in corrosion and biofouling prevention. Corros. Rev. 2019, 37, 187–230. [Google Scholar] [CrossRef]
- Ding, C.; Xu, J.; Tong, L.; Gong, G.; Jiang, W.; Fu, J. Design and Fabrication of a Novel Stimulus-Feedback Anticorrosion Coating Featured by Rapid Self-healing Functionality for Protection of Magnesium Alloy. ACS Appl. Mater. Interfaces ’HVLJQ 2017, 9, 21034–21047. [Google Scholar] [CrossRef]
- Wheat, H.G.; Liu, G. Coatings for Early Corrosion Detection. ECS Meet. Abstr. 2010, MA2010-01, 903. [Google Scholar] [CrossRef]
- Frankel, G.S. Corrosion-sensing behavior of an acrylic-based coating system. Corrosion 1999, 55, 957–967. [Google Scholar]
- Sibi, M.P.; Zong, Z. Determination of corrosion on aluminum alloy under protective coatings using fluorescent probes. Prog. Org. Coat. 2003, 47, 8–15. [Google Scholar] [CrossRef]
- Augustyniak, A.; Tsavalas, J.; Ming, W. Early detection of steel corrosion via “turn-On” fluorescence in smart epoxy coatings. ACS Appl. Mater. Interfaces 2009, 1, 2618–2623. [Google Scholar] [CrossRef] [PubMed]
- Calle, M.; Li, W.; Buhrow, J.W.; Johnsey, M.N.; Jolley, S.T.; Benjamin, P.; Zhang, X.; Fitzpatrick, L.; Gillis, M.; Blanton, M.; et al. Environmentally Friendly Coating Technology for Autonomous Corrosion Control. In Proceedings of the 43rd Annual Waterborne Symposium, New Orleans, LA, USA, 31 January–5 February 2016; Volume 32899. [Google Scholar]
- Liu, G.; Wheat, H.G. Use of a Fluorescent Indicator in Monitoring Underlying Corrosion on Coated Aluminum 2024-T4. J. Electrochem. Soc. 2009, 156, C160. [Google Scholar] [CrossRef]
- Mata, D.; Scharnagl, N.; Lamaka, S.V.; Malheiro, E.; Maia, F.; Zheludkevich, M.L. Validating the early corrosion sensing functionality in poly (ether imide) coatings for enhanced protection of magnesium alloy AZ31. Corros. Sci. 2018, 140, 307–320. [Google Scholar] [CrossRef]
- Maia, F.; Tedim, J.; Bastos, A.C.; Ferreira, M.G.S.; Zheludkevich, M.L. Nanocontainer-based corrosion sensing coating. Nanotechnology 2013, 24, 415502. [Google Scholar] [CrossRef]
- Wankhede, R.G.; Morey, S.; Khanna, A.S.; Birbilis, N. Development of water-repellent organic-inorganic hybrid sol-gel coatings on aluminum using short chain perfluoro polymer emulsion. Appl. Surf. Sci. 2013, 283, 1051–1059. [Google Scholar] [CrossRef]
- Thermo Fisher Scientific ASTM B117. Mater. Saf. Data Sheet 2012, 4, 8–10.
- Kordas, G. CuO (Bromosphaerol) and CeMo (8 Hydroxyquinoline) microcontainers incorporated into commercial marine paints. J. Am. Ceram. Soc. 2020, 103, 2340–2350. [Google Scholar] [CrossRef]
- Kordas, G. Novel Antifouling and Self-Healing Eco-Friendly Coatings for Marine Applications Enhancing the Performance of Commercial Marine Paints. In Engineering Failure Analysis; IntechOpen: London, UK, 2020; pp. 1–9. [Google Scholar]
- Kordas, G. Nanotechnology to improve the biofouling and corrosion performance of marine paints: From lab experiments to real tests in sea. Int. J. Phys. Res. Appl. 2019, 2, 33–37. [Google Scholar] [CrossRef] [Green Version]
- Kordas, G. Nanocontainers-Based Anti-Biofouling Coatings—A Pilot Study. In Supramolecular Chemistry in Corrosion and Biofouling Protection; Saji, V.S., Ed.; CRC Press: London, UK, 2021; pp. 383–392. ISBN 9781003169130. [Google Scholar]
- Thomas, S.; Surendran, A. (Eds.) Self-Healing Polymer-Based Systems; Elsevier: Amsterdam, The Netherlands, 2020; ISBN 9780128184509. [Google Scholar]

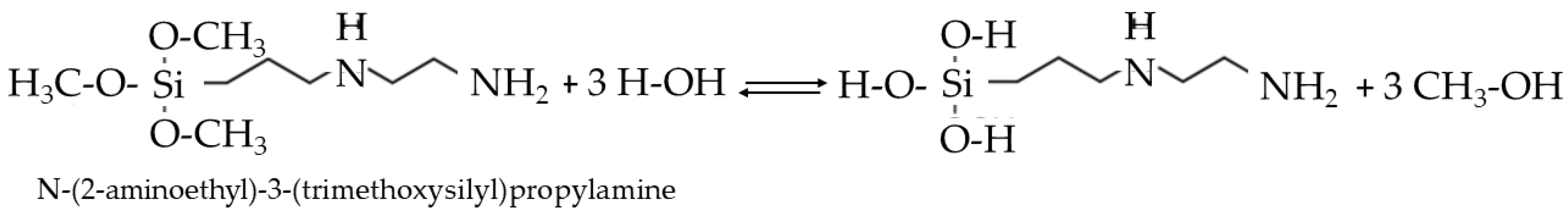
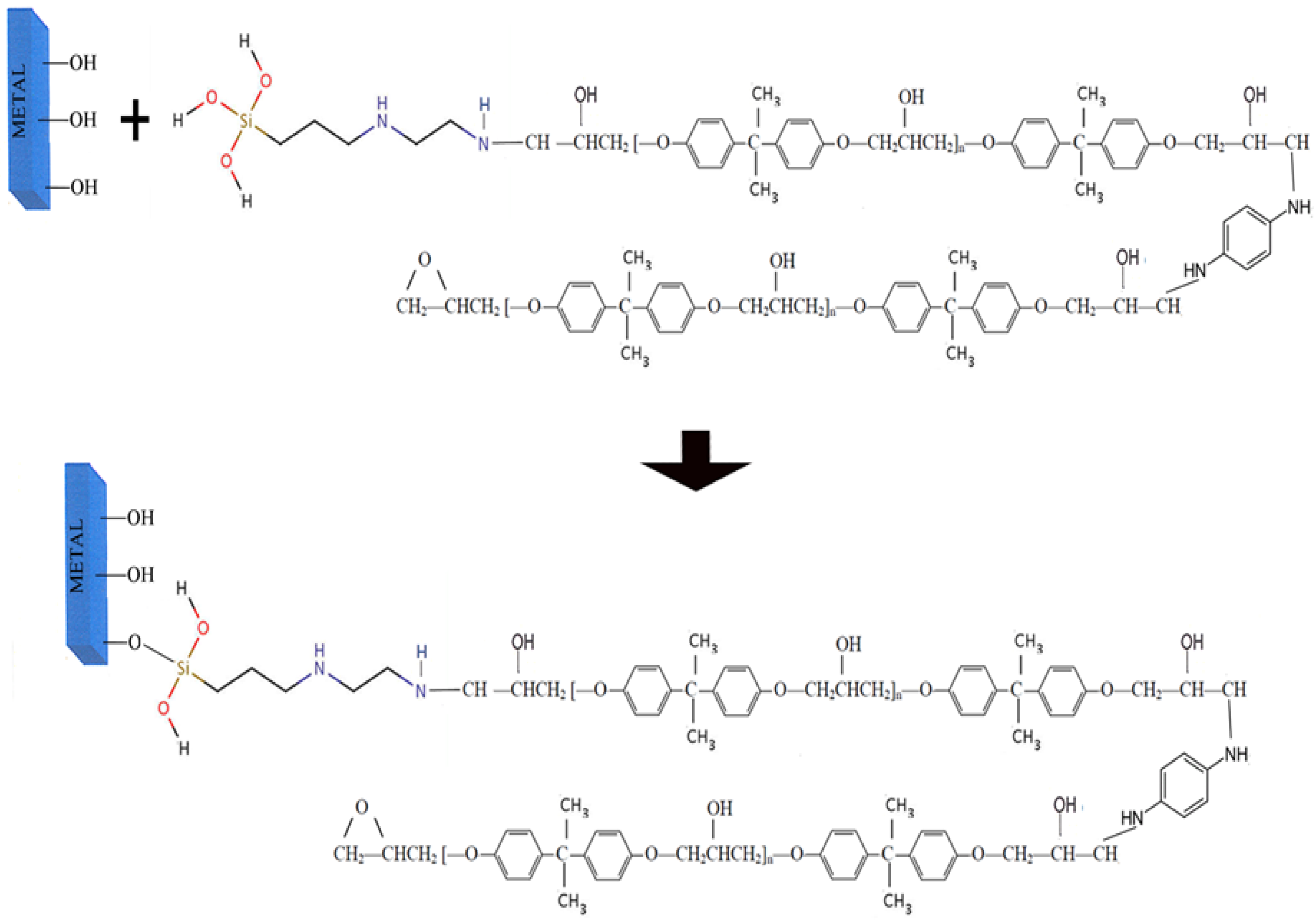
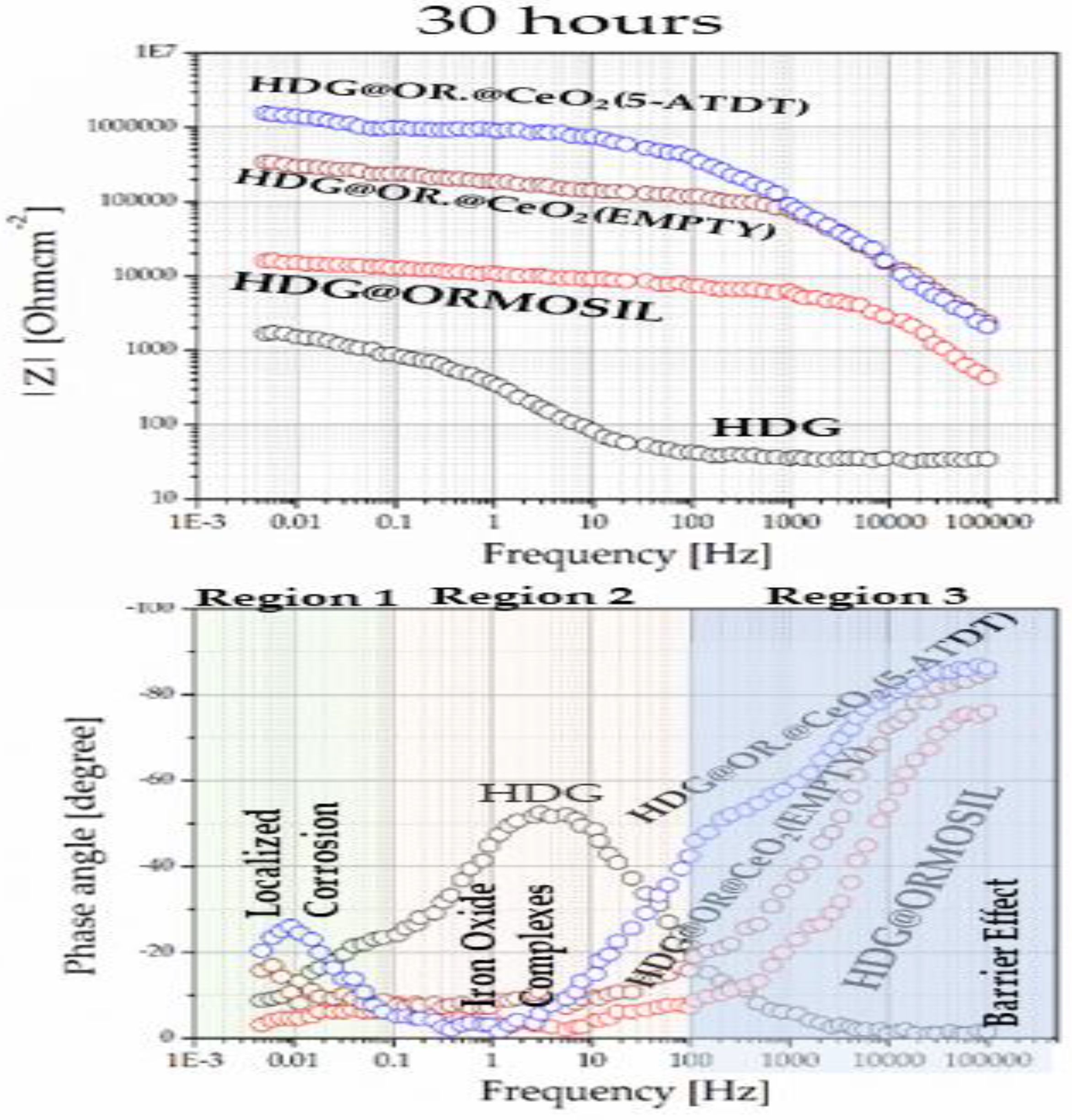



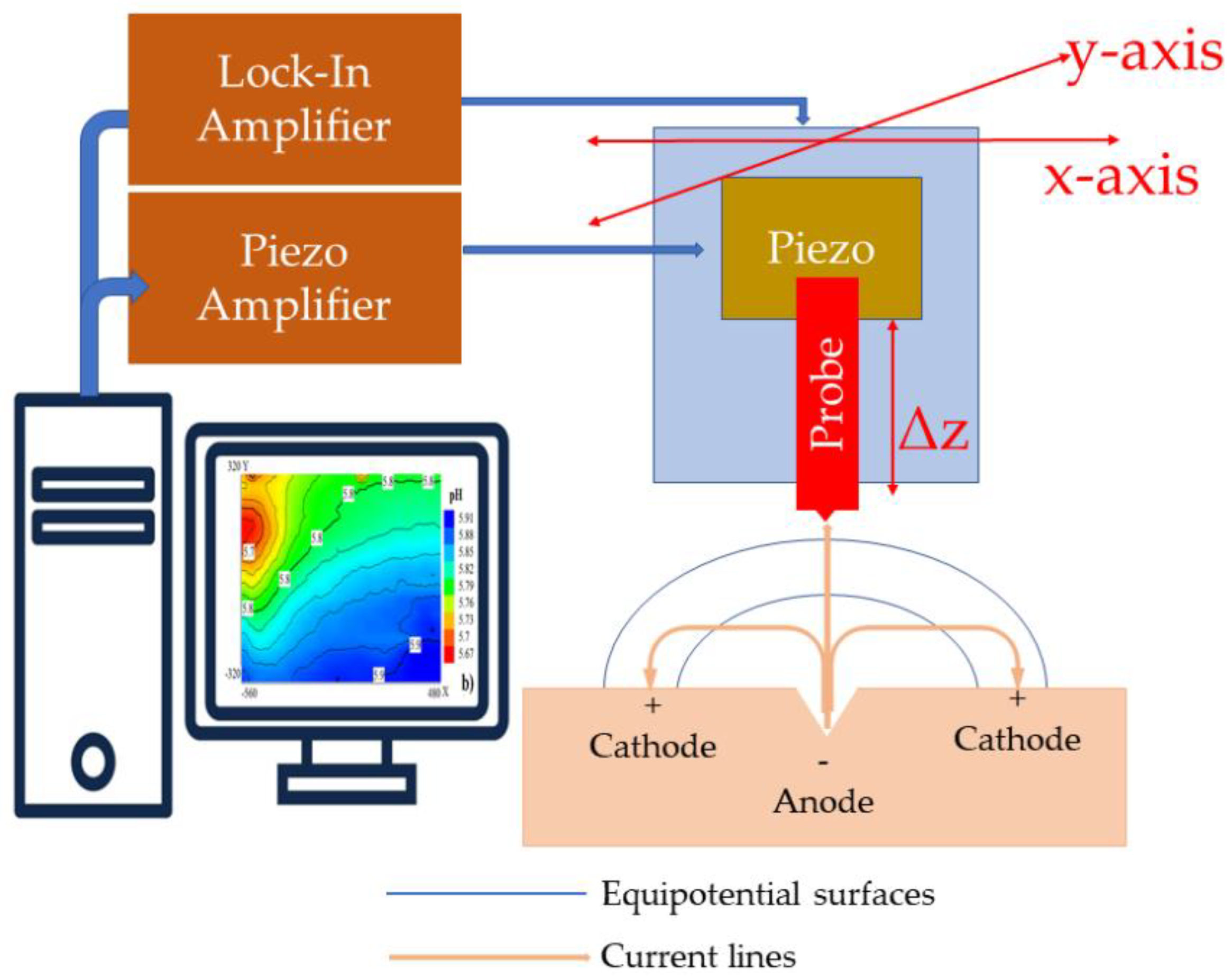





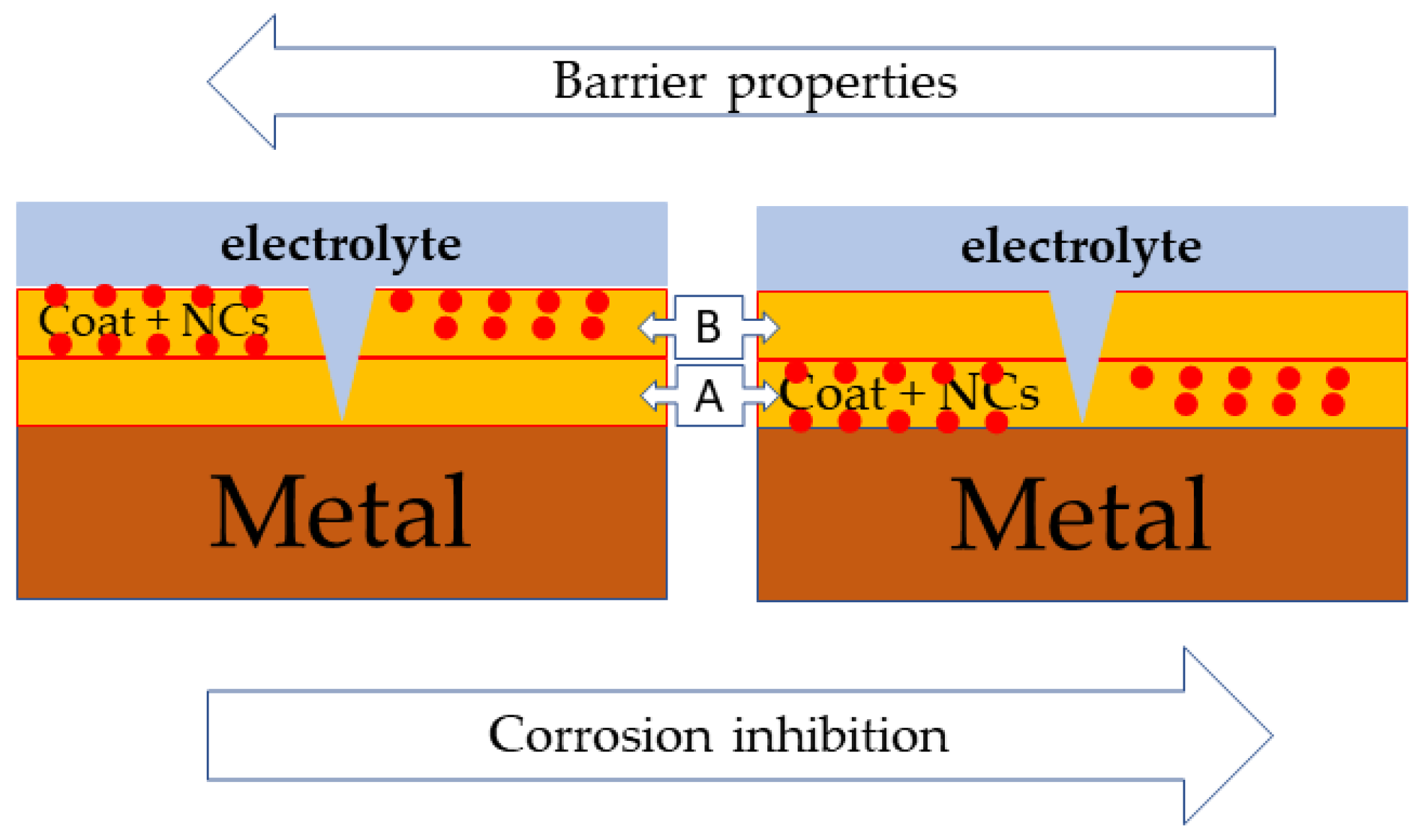








| Material | Structure |
|---|---|
| Vinyl trimethoxy silane |  |
| Bis-l,2-(trimethoxysilyl) ethane |  |
| Bis-[trimethoxysilylpropyl]amine |  |
| (3-Aminopropyl)trimethoxysilane |  |
| 3-Glycidyloxypropyl- trimethoxysilane |  |
| 3-(Trimethoxysilyl)propyl methacrylate |  |
| 3-(2-Aminoethylamino)-propyldimethoxymethylsilane |  |
| 1,2-Bis(trimethoxysilyl)ethane |  |
| Inhibitors | TiO2 | CeMo | CeO2 |
|---|---|---|---|
| 8-hydroxyquinoline | 3.56 | 5.22 | 5.22 |
| 5-amino-1,3,4-thiadiazole-2-thiol | 18.90 | 46.45 | 21.91 |
| 2-mercaptobenzothiazole | 6.14 | 59.44 | 63.42 |
| 1-H-benzotriazole-4-sulfonic acid | - | 12.03 | - |
| 1-H-benzotriazole | - | - | 54.17 |
| p-toluenesulfonic acid | 41.27 | - | - |
| FAS-3 (3,3,3-trifluoropropyltrimethoxysilane) |  |
| FAS-17 (1H,1H,2H,2H-perfluorodecyltriethoxysilane) |  |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kordas, G. Corrosion Barrier Coatings: Progress and Perspectives of the Chemical Route. Corros. Mater. Degrad. 2022, 3, 376-413. https://doi.org/10.3390/cmd3030023
Kordas G. Corrosion Barrier Coatings: Progress and Perspectives of the Chemical Route. Corrosion and Materials Degradation. 2022; 3(3):376-413. https://doi.org/10.3390/cmd3030023
Chicago/Turabian StyleKordas, George. 2022. "Corrosion Barrier Coatings: Progress and Perspectives of the Chemical Route" Corrosion and Materials Degradation 3, no. 3: 376-413. https://doi.org/10.3390/cmd3030023






