Microbiome of Sri Lankan Coral Reefs: An Indian Ocean Island Subjected to a Gradient of Natural and Anthropogenic Impacts
Abstract
:1. Introduction
2. Materials and Methods
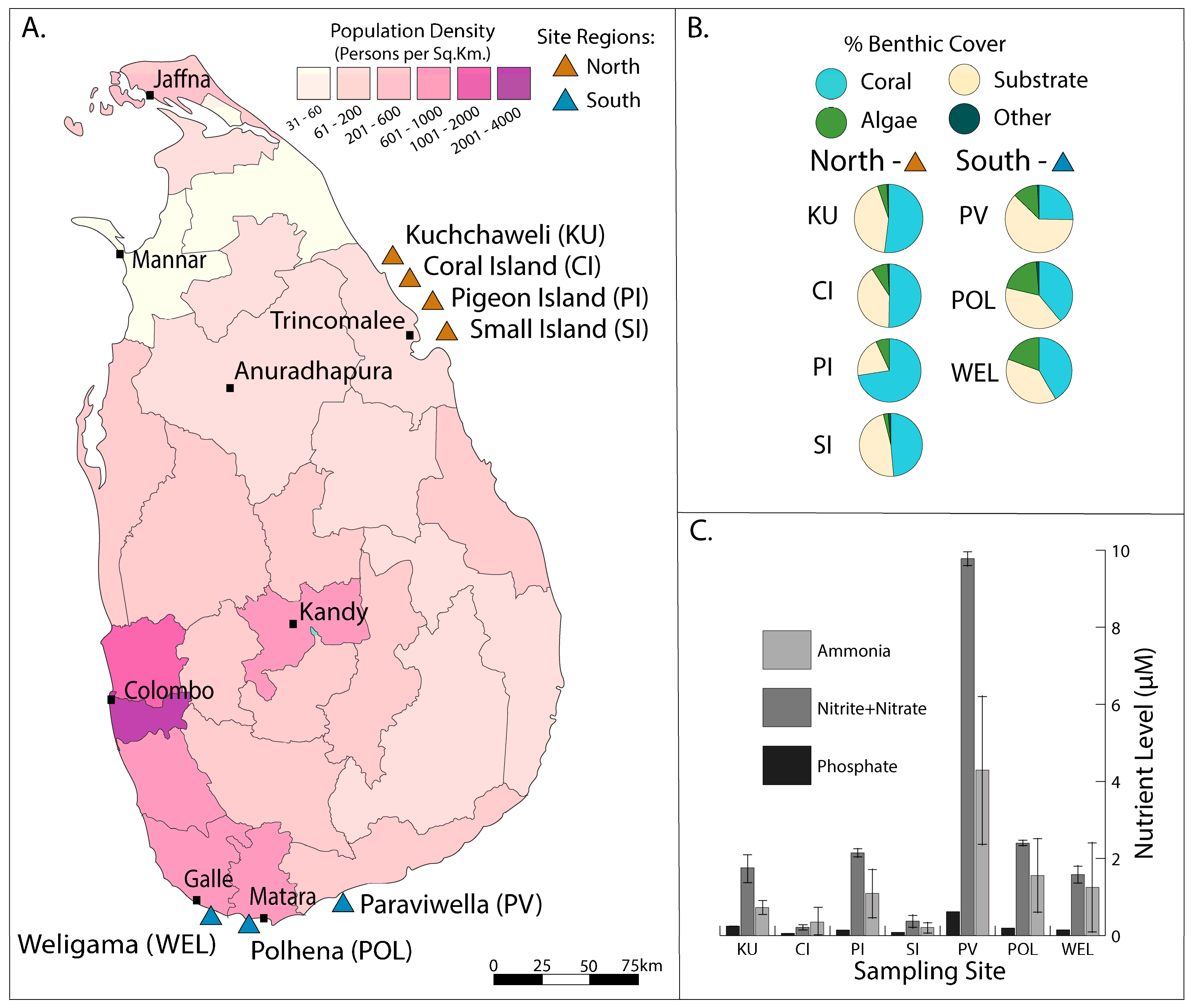
2.1. Reef Locations Selected for Sample Collection
2.2. Water Sample Collection and Processing
2.3. Population Density of Sri Lanka
2.4. Percent Benthic Cover Determination
2.5. Microbial and Archaea Abundances
2.6. Environmental Parameters and Nutrient Concentrations
2.7. DNA Extraction
2.8. DNA Sequencing
2.9. Functional Annotation of Metagenomes
2.10. Microbial Taxonomy Identification
2.11. Statistics
3. Results
3.1. Reef Site Characteristics
3.1.1. Reef Water Chemistry
3.1.2. Microbial Community Structure and Function
4. Discussion
4.1. Coral Reefs Status in South Asian Region
4.2. Population Densities of South Asian Region
4.3. Sri Lankan Reefs and Long Term Impacts
4.4. Microbial Taxonomic Community Composition Structure of Sri Lanka Northeast and South Regions of Sri Lanka
4.5. Need of a Comprehensive Study to Understand the Energy Utilization by Microbial System in Relation with Benthic Coral Cover to Eliminate Limitations of This First Baseline Data Set
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Site | SRA Number | BioSample Numbr |
|---|---|---|
| CI | SRS169408 | SAMN12385965 |
| KU | SRS169407 | SAMN12385964 |
| PI | SRS169405 | SAMN12385966 |
| POL | SRS169404 | SAMN12385969 |
| PV | SRS169403 | SAMN12385968 |
| SI | SRS169406 | SAMN12385967 |
| WEL | SRS169402 | SAMN12385970 |
References
- Sri Lanka. The World Fact Book; Central Intelligence Agency: Washington, DC, USA, 2021. Available online: https://www.cia.gov/the-world-factbook/countries/sri-lanka/ (accessed on 15 November 2022).
- Olsen, S.; Sadacharan, D.; Samarakoon, J.I.; White, A.T.; Wickremaratne, H.J.M.; Wijeratne, M.S. (Eds.) Coastal 2000: Recommendations for a Resources Management Strategy for Sri Lanka’s Coastal Regions, Volumes I and II; CRC Technical Report No. 2033; Coastal Resource Management Project, Coast Conservation Department, Coastal Resource Center, University of Rhode Island: Narragansett, RI, USA, 1992; pp. 21, 81. [Google Scholar]
- Obura, D.O.; Tamelander, J.; Linden, O. Coastal Oceans Research and Development in the Indian Ocean: Status Report 2008; CORDIO: Mombasa, Kenya, 2008. [Google Scholar]
- Ridely, S.O. The coral faunas of Ceylon with description of new species. Annu. Mag. Nat. Hist. 1883, 11, 250–262. [Google Scholar] [CrossRef]
- Ortmann, A. Beobacthungen an Steinkorallen von der Sudukuste Ceylons; Zoologische Jahrbücher: Jena, Germany, 1889; pp. 493–590. [Google Scholar]
- Pillai, C.S.G. Stony corals of the seas around India. In Proceedings of the Symposium on Corals and Coral Reefs, Marine Biological Association of India, Mandapam Camp, India, 12–16 January 1969; pp. 191–216. [Google Scholar]
- Arulananthan, A.; Herath, V.; Kuganathan, S.; Upasanta, A.; Harishchandra, A. The Status of the Coral Reefs of the Jaffna Peninsula (Northern Sri Lanka), with 36 Coral Species New to Sri Lanka Confirmed by DNA Bar-Coding. Oceans 2021, 2, 509–529. [Google Scholar] [CrossRef]
- CRC Sheppard. Coral reefs of the western Indian Ocean: On overview. In Coral Reefs of the Indian Ocean: Their Ecology and Conservation; Oxford University Press: Oxford, UK, 2000; pp. 3–38. [Google Scholar]
- Ohman, M.C.; Linden, O.; Rajasuriya, A. Human disturbances on coral reefs in Sri Lanka: A case study. Ambio (J. Hum. Environ. Res. Manag.) 1993, 22, 474–480. [Google Scholar]
- Rajasuriya, A. Status of coral reefs in Sri Lanka in the aftermath of the 1998 coral bleaching event and 2004 tsunami. In Coral Reef Degradation in the Indian Ocean: Status Report 2005; Souter, D., Linden, O., Eds.; CORDIO, Department of Biology and Environmental Science, University of Kalmar: Småland, Sweden, 2005; pp. 83–96. [Google Scholar]
- Rajasuriya, A.; Karunaratne, C. Post-bleaching status of the coral reefs of Sri Lanka. Coral reef degradation in the Indian Ocean. In Coral Reef Degradation in the Indian Ocean: Status Report 2000; Souter, D., Obura, D., Linden, O., Eds.; CORDIO, SAREC Marine Science Program, Stockholm University: Stockholm, Sweden, 2000; pp. 54–63. [Google Scholar]
- Helen, P. Future Effects of Climate Change on Coral Reefs and Mangroves in South Asia. In Coral Reef Degradation in the Indian Ocean Status Report Linden; Souter, O.D., Wlihelmsson, D., Obura, D., Eds.; Kalmar University: Växjö, Sweden, 2002; pp. 167–175. [Google Scholar]
- Fairoz, M.F.M. Impacts of Natural Processes and Anthropogenic Activities of Two Fringing Coral Reefs of Southern Sri Lanka; Faculty of Fisheries and Marine Science and Technology, University of Ruhuna: Matara, Sri Lanka, 2006. [Google Scholar]
- Rajasuriya, A.; De Silva, M.W.R.N.; Ohman, M.C. Coral reefs of Sri Lanka, human disturbance and management issues. Ambio 1995, 24, 428–437. [Google Scholar]
- Tamelander, J.; Rajasuriya, A. Status of Coral Reefs in South Asia: Bangladesh, Chagos, India, Maldives and Sri Lanka. In Status of Coral Reefs of the World: 2008; Wilkinson, C.R., Ed.; Global Coral Reef Monitoring Network and Reef and Rainforest Research Centre: Townsville, Australia, 2008; p. 296. [Google Scholar]
- Wilhelmsson, D.; Haputhantri, S.S.K.; Rajasuriya, A.; Vidanage, S.P. Monitoring the Trends of Marine Ornamental Fish Collection in Sri Lanka. In Coral Reef Degradation in the Indian Ocean: Status Report 2002; CORDIO, Department of Biology and Environmental Science, University of Kalmar: Kalmar, Sweden, 2002; pp. 158–166. ISBN 91-973959-2-7. [Google Scholar]
- Rajasuriya, A.; White, A.T. Coral Reefs of Sri Lanka: Review of Their Extent, Condition, and Management Status. Coast. Manag. 1995, 23, 77–90. [Google Scholar] [CrossRef]
- Anne-Kathrin, G. Time for a New Approach: Ending Protracted Displacement of Sri Lanka; Norwegian Refugee Council/Internal Displacement Monitoring Centre, Colombo (NRC/IDMC): Oslo, Norway, 2015. [Google Scholar]
- Haas, A.F.; Fairoz, M.F.M.; Kelly, L.W.; Nelson, C.E.; Dinsdale, E.A.; Edwards, R.A.; Giles, S.; Hatay, M.; Hisakawa, N.; Knowles, B.; et al. Global microbialization of coral reefs. Nat. Microbiol. 2016, 1, 16042. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.E.; Shaw, M.; Edwards, R.A.; Obura, D.; Pantos, O.; Sala, E.; Sandin, S.A.; Smriga, S.; Hatay, M.; Rohwer, F.L. Indirect effects of algae on coral: Algae-mediated, microbe-induced coral mortality. Ecol. Lett. 2006, 9, 835–845. [Google Scholar] [CrossRef] [PubMed]
- Dinsdale, E.A.; Pantos, O.; Smriga, S.; Edwards, R.; Angly, F.; Wegley, L.; Hatay, M.; Hall, D.; Brown, E.; Haynes, M.; et al. Microbial Ecology of Four Coral Atolls in the Northern Line Islands. PLoS ONE 2008, 3, e1584. [Google Scholar] [CrossRef]
- Haas, A.F.; Knowles, B.; Lim, Y.W.; Somera, T.M.; Kelly, L.W.; Hatay, M.; Rohwer, F. Unraveling the Unseen Players in the Ocean—A Field Guide to Water Chemistry and Marine Microbiology. J. Vis. Exp. 2014, 93, e52131. [Google Scholar] [CrossRef]
- Kelly, L.W.; Williams, G.J.; Barott, K.L.; Carlson, C.A.; Dinsdale, E.A.; Edwards, R.A.; Haas, A.F.; Haynes, M.; Lim, Y.W.; McDole, T.; et al. Local genomic adaptation of coral reef-associated microbiomes to gradients of natural variability and anthropogenic stressors. Proc. Natl. Acad. Sci. USA 2014, 111, 10227–10232. [Google Scholar] [CrossRef]
- McDole, T.; Nulton, J.; Barott, K.L.; Felts, B.; Hand, C.; Hatay, M.; Lee, H.; Nadon, M.O.; Nosrat, B.; Salamon, P.; et al. Assessing Coral Reefs on a Pacific-Wide Scale Using the Microbialization Score. PLoS ONE 2012, 7, e43233. [Google Scholar] [CrossRef] [PubMed]
- Department of Census and Statistics. Population Atlas of Sri Lanka 2012; Department of Census and Statistics (DCS) and Ministry of Finance and Planning: Colombo, Sri Lanka, 2012.
- Preskitt, L.B.; Vroom, P.S.; Smith, C.M. A Rapid Ecological Assessment (REA) Quantitative Survey Method for Benthic Algae Using Photoquadrats with Scuba. Pac. Sci. 2004, 58, 201–209. [Google Scholar] [CrossRef]
- Mathew, D.R. A Field Guide to the Seashores of Eastern Africa and the Western Indian Ocean Islands, 3rd ed.; Swedeish International Development Agency: Stockholm, Sweden, 2011; 464p, ISBN 9987897797. [Google Scholar]
- Veron, J.E.N.; Stafford-Smith, M. Corals of the World; Australian Institute of Marine Science: Townsville, Australia, 2000.
- Schmieder, R.; Edwards, R. Quality control and preprocessing of metagenomic datasets. Bioinformatics 2011, 27, 863–864. [Google Scholar] [CrossRef]
- Silva, G.G.Z.; Green, K.T.; Dutilh, B.E.; Edwards, R.A. SUPER-FOCUS: A tool for agile functional analysis of shotgun metagenomic data. Bioinformatics 2016, 32, 354–361. [Google Scholar] [CrossRef]
- Zhao, Y.; Tang, H.; Ye, Y. RAPSearch2: A fast and memory-efficient protein similarity search tool for next-generation sequencing data. Bioinformatics 2012, 28, 125–126. [Google Scholar] [CrossRef]
- Underwood, A.J. Experiments in Ecology: Their Logical Design and Interpretation Using Analysis of Variance; Cambridge University Press: Cambridge, UK, 1996. [Google Scholar]
- Menzel, P.; Ng, K.L.; Krogh, A. Fast and sensitive taxonomic classification for metagenomics with Kaiju. Nat. Commun. 2016, 7, 11257. [Google Scholar] [CrossRef]
- Ihaka, R.; Gentleman, R. R: A Language for Data Analysis and Graphics. J. Comput. Graph. Stat. 1996, 5, 299–314. [Google Scholar] [CrossRef]
- José Cláudio, F.; Demétrio, C.G.B.; Allaman, I.B. Biplot of Multivariate Data Based on Principal Components Analysis. 2013. Available online: https://search.r-project.org/CRAN/refmans/bpca/html/bpca.html (accessed on 6 June 2019).
- Lê, S.; Josse, J.; Husson, F. FactoMineR: An R Package for Multivariate Analysis. J. Stat. Softw. 2008, 25, 1–18. [Google Scholar] [CrossRef]
- The R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Souter, D.; Planes, S.; Wicquart, J.; Logan, M.; Obura, D.; Staub, F. Status of Coral Reefs of the World: 2020 Report. Global Coral Reef Monitoring Network (GCRMN)/International Coral Reef Initiative (ICRI). 2021. Available online: https://gcrmn.net/2020-report/ (accessed on 10 May 2021).
- Haas, A.F.; Nelson, C.; Kelly, L.W.; Carlson, C.A.; Rohwer, F.; Leichter, J.J.; Wyatt, A.; Smith, J.E. Effects of Coral Reef Benthic Primary Producers on Dissolved Organic Carbon and Microbial Activity. PLoS ONE 2011, 6, e27973. [Google Scholar] [CrossRef] [PubMed]
- López-Pérez, M.; Gonzaga, A.; Martin-Cuadrado, A.-B.; Onyshchenko, O.; Ghavidel, A.; Ghai, R.; Rodriguez-Valera, F. Genomes of surface isolates of Alteromonas macleodii: The life of a widespread marine opportunistic copiotroph. Sci. Rep. 2012, 2, 696. [Google Scholar] [CrossRef] [PubMed]
- Nelson, C.; Goldberg, S.J.; Kelly, L.W.; Haas, A.F.; E Smith, J.; Rohwer, F.; A Carlson, C. Coral and macroalgal exudates vary in neutral sugar composition and differentially enrich reef bacterioplankton lineages. ISME J. 2013, 7, 962–979. [Google Scholar] [CrossRef] [PubMed]
- Roach, T.N.; Abieri, M.L.; George, E.E.; Knowles, B.; Naliboff, D.S.; Smurthwaite, C.A.; Kelly, L.W.; Haas, A.F.; Rohwer, F.L. Microbial bioenergetics of coral-algal interactions. PeerJ 2017, 5, e3423. [Google Scholar] [CrossRef] [PubMed]
- Fairoz, M.F.M. Coral Reefs and Blue Economy. In Blue Economy; Urban, E.R., Jr., Ittekkot, V., Eds.; Springer: Singapore, 2022. [Google Scholar]


| Site | Coordinate North | Coordinate East |
|---|---|---|
| Kutchchaweli (KU) Trincomalee District | 08°49′45.48″ N | 81°6′15.958″ E |
| Coral Island (CI) Trincomalee District | 08°44′31.931″ N | 81°10′45.073″ E |
| Pigeon Island (PI) Trincomalee, District | 08°43′18.764″ N | 81°12′16.509″ E |
| Small Island (SI) Trincomalee, District | 08°42′48.232″ N | 81°12′3.06″ E |
| Paraviwella (PV) Hambantota District | 06° 1′ 17.503″ N | 80°48′8.191″ E |
| Polhena (POL) Matara District | 05°56′5.65″ N | 80°31′32.305″ E |
| Weligama (WEL) Matara District | 05°34′24.996″ N | 80°15′10.548″ E |
| Region | Site | Live Coral | Macroalgae | Substrate ** | Other * |
|---|---|---|---|---|---|
| Northeast | KU | 52.6 ± 7.4 | 10.4 ± 1.9 | 31.5 ± 2.2 | 5.4 ± 2.15 |
| CI | 50.3 ± 4.1 | 8.4 ± 4.7 | 36.0 ± 1.5 | 5.2 ± 1.65 | |
| PI | 69.7 ± 6.9 | 8.9 ± 3.0 | 17.9 ± 3.9 | 3.3 ± 4.0 | |
| SI | 49.7 ± 8.4 | 6.4 ± 0.4 | 38.5 ± 0.8 | 5.3 ± 2.6 | |
| South | PV | 16.4 ± 10.1 | 10.1 ± 3.3 | 68.3 ± 5.4 | 5.1 ± 2.2 |
| POL | 31.4 ± 12.8 | 12.8 ± 2.9 | 49.4 ± 1.0 | 6.2 ± 2.5 | |
| WEL | 36.2 ± 14.9 | 14.9 ± 3.4 | 46.5 ± 3.5 | 2.2 ± 4.7 |
| Coral Reef Site. | Sample Month | Temp/°C Range | Salinity (ppt) | Phosphate µM | Nitrite + Nitrate µM | Ammonia µM |
|---|---|---|---|---|---|---|
| Kutchchaweli (KU) | June 2015 | 30–31.5 | 28 | 0.24 ± 0.02 | 1.76 ± 0.37 | 0.72 ± 0.19 |
| Coral Island (CI) | 28 | 0.06 ± 0.006 | 0.21 ± 0.06 | 0.34 ± 0.44 | ||
| Pigeon Island (PI) Trincomalee | 28 | 0.14 ± 0.02 | 2.14 ± 0.09 | 1.08 ± 0.66 | ||
| Small Island (SI) | 28 | 0.08 ± 0.008 | 0.37 ± 0.16 | 0.20 ± 0.17 | ||
| Paraviwella (PV) | February 2016 | 29.5–31 | 32 | 0.61 ± 0.01 | 9.78 ± 0.18 | 4.29 ± 2.00 |
| Polhena (POL) | February 2016 | 31 | 0.19 ± 0.005 | 2.39 ± 0.08 | 1.56 ± 0.99 | |
| Weligama (WEL) | March 2016 | 28 | 0.15 ± 0.006 | 1.58 ± 0.23 | 1.24 ± 1.20 |
| Site | Microbe (×105) per mL | % GC Content |
|---|---|---|
| KU | 8.3 | 47.0 |
| CI | 4.5 | 44.0 |
| PI | 2.9 | 46.6 |
| SI | 5.9 | 44.0 |
| PV | 1.4 | 47.9 |
| POL | 2.2 | 44.3 |
| WEL | 7.1 | 46.5 |
| Site | Post-QC Reads | Assigned | Unassigned | Bacteria | Eukaryota | Archaea | Viruses |
|---|---|---|---|---|---|---|---|
| KU | 806,743 | 497,202 | 309,541 | 487,156 | 3692 | 4491 | 1234 |
| CI | 373,567 | 212,173 | 161,394 | 199,761 | 5967 | 2377 | 3670 |
| PI | 361,566 | 193,905 | 167,661 | 188,378 | 2387 | 1016 | 1756 |
| SI | 623,758 | 294,601 | 329,157 | 275,545 | 8739 | 7727 | 1968 |
| PV | 326,126 | 234,300 | 91,826 | 230,327 | 1167 | 1441 | 1212 |
| POL | 1,058,343 | 487,603 | 570,740 | 443,761 | 19,877 | 15,254 | 7198 |
| WEL | 693,647 | 423,146 | 270,501 | 406,118 | 5261 | 10,069 | 869 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fairoz, M.F.M.; Green, K.T.; Sajith, K.N.M.; Chamika, W.A.S.; Kularathna, A.M.K.N.; Macherla, S.; Naliboff, D.S.; Cobián-Güemes, A.; Wegley-Kelly, L.; Rohwer, F. Microbiome of Sri Lankan Coral Reefs: An Indian Ocean Island Subjected to a Gradient of Natural and Anthropogenic Impacts. Oceans 2023, 4, 185-199. https://doi.org/10.3390/oceans4020013
Fairoz MFM, Green KT, Sajith KNM, Chamika WAS, Kularathna AMKN, Macherla S, Naliboff DS, Cobián-Güemes A, Wegley-Kelly L, Rohwer F. Microbiome of Sri Lankan Coral Reefs: An Indian Ocean Island Subjected to a Gradient of Natural and Anthropogenic Impacts. Oceans. 2023; 4(2):185-199. https://doi.org/10.3390/oceans4020013
Chicago/Turabian StyleFairoz, Mohamed F. M., Kevin T. Green, Kuwaja N. M. Sajith, Weerathunga A. S. Chamika, Amarasingha M. K. N. Kularathna, Saichetana Macherla, Douglas S. Naliboff, Ana Cobián-Güemes, Linda Wegley-Kelly, and Forest Rohwer. 2023. "Microbiome of Sri Lankan Coral Reefs: An Indian Ocean Island Subjected to a Gradient of Natural and Anthropogenic Impacts" Oceans 4, no. 2: 185-199. https://doi.org/10.3390/oceans4020013
APA StyleFairoz, M. F. M., Green, K. T., Sajith, K. N. M., Chamika, W. A. S., Kularathna, A. M. K. N., Macherla, S., Naliboff, D. S., Cobián-Güemes, A., Wegley-Kelly, L., & Rohwer, F. (2023). Microbiome of Sri Lankan Coral Reefs: An Indian Ocean Island Subjected to a Gradient of Natural and Anthropogenic Impacts. Oceans, 4(2), 185-199. https://doi.org/10.3390/oceans4020013











