The Dose of Somatostatin Analogues during Pre-Surgical Treatment Is a Key Factor to Achieve Surgical Remission in Acromegaly
Abstract
1. Introduction
2. Materials and Methods
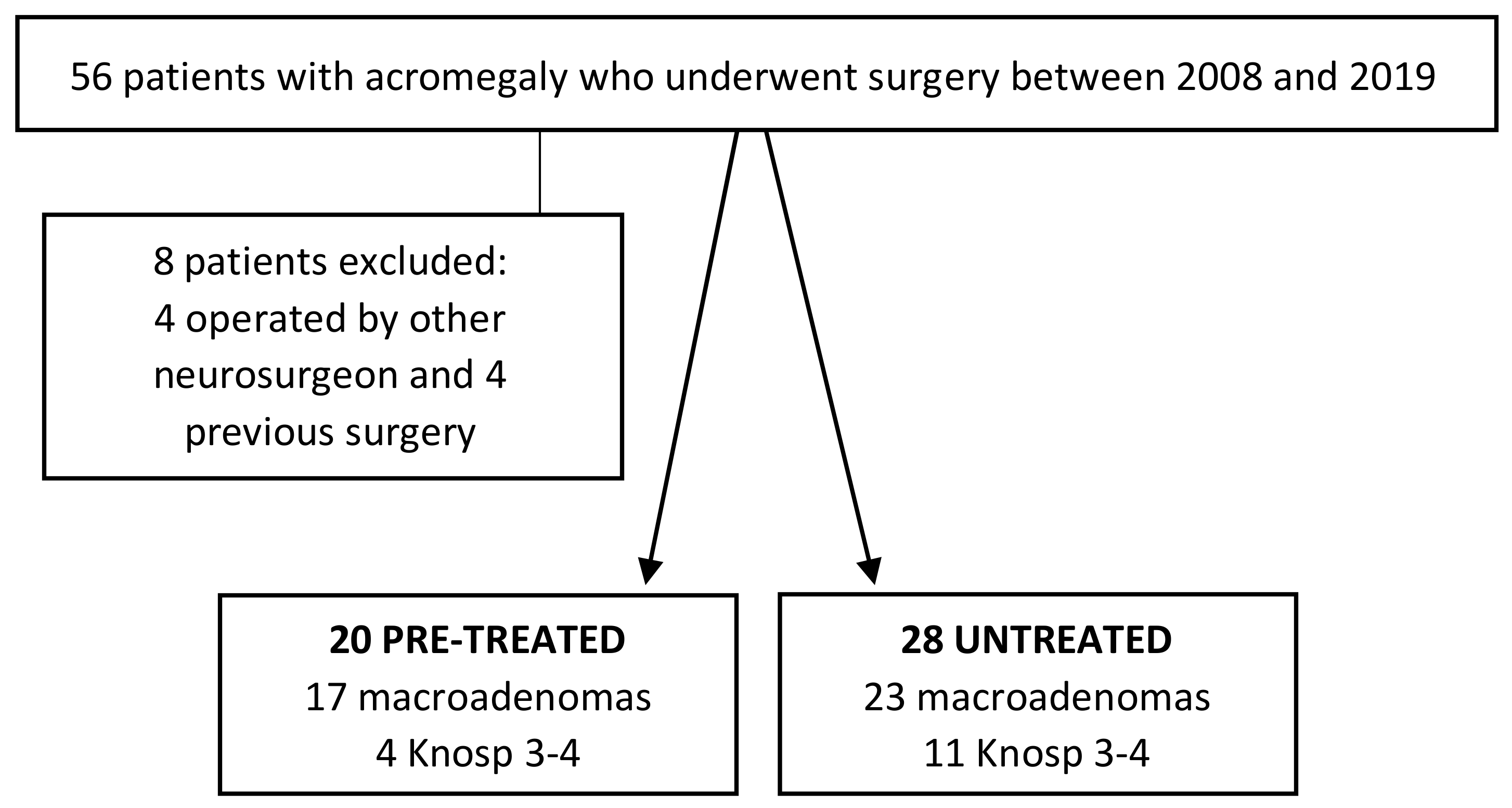
2.1. Patients
2.2. Assays and Remission Criteria
2.3. Clinical Definitions
2.4. Radiological Assessment
2.5. Surgical Procedure and Histological Analysis
2.6. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Surgical Remission (2010 Criteria)
3.3. Surgical and Endocrine Complications and Tumour Consistency
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bolfi, F.; Neves, A.F.; Boguszewski, C.; Nunes-Nogueira, V.S. Mortality in acromegaly decreased in the last decade: A systematic review and meta-analysis. Eur. J. Endocrinol. 2019, 181, L5–L6. [Google Scholar] [CrossRef] [PubMed]
- Katznelson, L.; Laws, E.R.; Melmed, S.; Molitch, M.E.; Murad, M.H.; Utz, A.; Wass, J.A.H. Acromegaly: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2014, 99, 3933–3951. [Google Scholar] [CrossRef] [PubMed]
- Araujo-Castro, M.; Pascual-Corrales, E.; Martínez-Vaello, V.; Saiz, G.B.; De Silva, J.Q.; Cancela, A.A.; Cano, A.M.G.; Berrocal, V.R. Predictive model of surgical remission in acromegaly: Age, presurgical GH levels and Knosp grade as the best predictors of surgical remission. J. Endocrinol. Investig. 2021, 44, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.H.; Oh, M.C.; Lee, E.J.; Kim, S.H. Predicting Long-term Remission by Measuring Immediate Postoperative Growth Hormone Levels and Oral Glucose Tolerance Test in Acromegaly. Neurosurgery 2012, 70, 1106–1113. [Google Scholar] [CrossRef] [PubMed]
- Araujo-Castro, M.; Pascual-Corrales, E.; Millan, J.S.M.S.; Rebolleda, G.; Pian, H.; Ruz-Caracuel, I.; Granados, G.D.L.S.; Urzaiz, L.L.; Escobar-Morreale, H.; Berrocal, V.R. Postoperative management of patients with pituitary tumors submitted to pituitary surgery. Experience of a Spanish Pituitary Tumor Center of Excellence. Endocrine 2020, 69, 5–17. [Google Scholar] [CrossRef]
- Freda, P.; Katznelson, L.; Van Der Lely, A.-J.; Reyes, C.M.; Zhao, S.; Rabinowitz, D. Long-Acting Somatostatin Analog Therapy of Acromegaly: A Meta-Analysis. J. Clin. Endocrinol. Metab. 2005, 90, 4465–4473. [Google Scholar] [CrossRef]
- Caron, P.J.; Bevan, J.S.; Petersenn, S.; Flanagan, D.; Tabarin, A.; Prévost, G.; Maisonobe, P.; Clermont, A.; on behalf of the PRIMARYS Investigators. Tumor Shrinkage with Lanreotide Autogel 120 mg as Primary Therapy in Acromegaly: Results of a Prospective Multicenter Clinical Trial. J. Clin. Endocrinol. Metab. 2014, 99, 1282–1290. [Google Scholar] [CrossRef] [PubMed]
- Colao, A.; Auriemma, R.S.; Pivonello, R. The effects of somatostatin analogue therapy on pituitary tumor volume in patients with acromegaly. Pituitary 2016, 19, 210–221. [Google Scholar] [CrossRef]
- Ben-Shlomo, A.; Melmed, S. The Role of Pharmacotherapy in Perioperative Management of Patients with Acromegaly. J. Clin. Endocrinol. Metab. 2003, 88, 963–968. [Google Scholar] [CrossRef]
- Losa, M.; Bollerslev, J. Pros and cons in endocrine practice: Pre-surgical treatment with somatostatin analogues in acromegaly. Endocrine 2016, 52, 451–457. [Google Scholar] [CrossRef]
- Fleseriu, M.; Hoffman, A.R.; Katznelson, L. American Association of Clinical Endocrinologists and American College of Endocrinology Disease State Clinical Review: Management of Acromegaly Patients: What is the Role of Pre-Operative Medical Therapy? Endocr. Pr. 2015, 21, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Stevenaert, A.; Beckers, A. Presurgical Octreotide: Treatment in acromegaly. Metabolism 1996, 45, 72–74. [Google Scholar] [CrossRef]
- Ferone, D.; Gatto, F.; Minuto, F. Pre-surgical treatment with somatostatin analogues in patients with acromegaly: The case for. J. Endocrinol. Investig. 2012, 35, 613–615. [Google Scholar] [CrossRef] [PubMed]
- Carlsen, S.M.; Lund-Johansen, M.; Schreiner, T.; Aanderud, S.; Johannesen, Ø.; Svartberg, J.; Cooper, J.G.; Hald, J.K.; Fougner, S.L.; Bollerslev, J. Preoperative Octreotide Treatment in Newly Diagnosed Acromegalic Patients with Macroadenomas Increases Cure Short-Term Postoperative Rates: A Prospective, Randomized Trial. J. Clin. Endocrinol. Metab. 2008, 93, 2984–2990. [Google Scholar] [CrossRef]
- Lv, L.; Hu, Y.; Zhou, P.; Zhang, S.; Yin, S.; Zhang, N.; Jiang, S. Presurgical treatment with somatostatin analogues in growth hormone-secreting pituitary adenomas: A long-term single-center experience. Clin. Neurol. Neurosurg. 2018, 167, 24–30. [Google Scholar] [CrossRef]
- Maiza, J.C.; Vezzosi, D.; Matta, M.; Donadille, F.; Loubes-Lacroix, F.; Cournot, M.; Bennet, A.; Caron, P. Long-term (up to 18 years) effects on GH/IGF-1 hypersecretion and tumour size of primary somatostatin analogue (SSTa) therapy in patients with GH-secreting pituitary adenoma responsive to SSTa. Clin. Endocrinol. 2007, 67, 282–289. [Google Scholar] [CrossRef]
- Zhang, L.; Wu, X.; Yan, Y.; Qian, J.; Lu, Y.; Luo, C. Preoperative somatostatin analogs treatment in acromegalic patients with macroadenomas. A meta-analysis. Brain Dev. 2015, 37, 181–190. [Google Scholar] [CrossRef]
- Quabbe, H.-J. Presurgical octreotide treatment in acromegaly: No improvement of final growth hormone (GH) concentration and pituitary function. A long-term case-control study. Acta Neurochir. 2005, 147, 485–493. [Google Scholar] [CrossRef]
- Losa, M.; Mortini, P.; Urbaz, L.; Ribotto, P.; Castrignanò, T.; Giovanelli, M. Presurgical treatment with somatostatin analogs in patients with acromegaly: Effects on the remission and complication rates. J. Neurosurg. 2006, 104, 899–906. [Google Scholar] [CrossRef]
- Melmed, S.; Sternberg, R.; Cook, D.; Klibanski, A.; Chanson, P.; Bonert, V.; Vance, M.L.; Rhew, D.; Kleinberg, D.; Barkan, A. A Critical Analysis of Pituitary Tumor Shrinkage during Primary Medical Therapy in Acromegaly. J. Clin. Endocrinol. Metab. 2005, 90, 4405–4410. [Google Scholar] [CrossRef]
- Giustina, A.; Chanson, P.; Bronstein, M.D.; Klibanski, A.; Lamberts, S.; Casanueva, F.F.; Trainer, P.; Ghigo, E.; Ho, K.; Melmed, S. A Consensus on Criteria for Cure of Acromegaly. J. Clin. Endocrinol. Metab. 2010, 95, 3141–3148. [Google Scholar] [CrossRef] [PubMed]
- Araujo-Castro, M.; Pascual-Corrales, E.; Cancela, A.A.; Duque, S.G.; Urzaiz, L.L.; Berrocal, V.R. Status and clinical and radiological predictive factors of presurgical anterior pituitary function in pituitary adenomas. Study of 232 patients. Endocrine 2020, 70, 584–592. [Google Scholar] [CrossRef] [PubMed]
- Araujo-Castro, M.; Pian, H.; Ruz-Caracuel, I.; Cancela, A.A.; Pascual-Corrales, E.; Berrocal, V.R. Presurgical somatostatin receptor ligand treatment does not affect tumor consistency in GH-secreting pituitary macroadenomas. Endocr. Connect. 2021, 10, 102–109. [Google Scholar] [CrossRef]
- Biermasz, N.R.; Van Dulken, H.; Roelfsema, F. Direct Postoperative and Follow-Up Results of Transsphenoidal Surgery in 19 Acromegalic Patients Pretreated with Octreotide Compared to Those in Untreated Matched Controls. J. Clin. Endocrinol. Metab. 1999, 84, 3551–3555. [Google Scholar] [CrossRef][Green Version]
- Albarel, F.; Castinetti, F.; Morange, I.; Guibert, N.; Graillon, T.; Dufour, H.; Brue, T. Pre-surgical medical treatment, a major prognostic factor for long-term remission in acromegaly. Pituitary 2018, 21, 615–623. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Li, G.; Jiang, S.; Bao, X.; Wang, R. Preoperative Somatostatin Analogues in Patients with Newly-diagnosed Acromegaly: A Systematic Review and Meta-analysis of Comparative Studies. Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef]
- Mao, Z.-G.; Zhu, Y.-H.; Tang, H.-L.; Wang, D.-Y.; Zhou, J.; He, D.-S.; Lan, H.; Luo, B.-N.; Wang, H.-J. Preoperative lanreotide treatment in acromegalic patients with macroadenomas increases short-term postoperative cure rates: A prospective, randomised trial. Eur. J. Endocrinol. 2010, 162, 661–666. [Google Scholar] [CrossRef]
- Pita-Gutiérrez, F.; Pértega-Díaz, S.; Pita-Fernandez, S.; Pena, L.; Lugo, G.; Sangiao-Alvarellos, S.; Cordido, F. Place of Preoperative Treatment of Acromegaly with Somatostatin Analog on Surgical Outcome: A Systematic Review and Meta-Analysis. PLoS ONE 2013, 8, e61523. [Google Scholar] [CrossRef]
- Nunes, V.S.; Correa, J.M.S.; Puga, M.E.S.; Silva, E.M.K.; Boguszewski, C. Preoperative somatostatin analogues versus direct transsphenoidal surgery for newly-diagnosed acromegaly patients: A systematic review and meta-analysis using the GRADE system. Pituitary 2014, 18, 500–508. [Google Scholar] [CrossRef]
- Alexopoulou, O.; Abrams, P.; Verhelst, J.; Poppe, K.; Velkeniers, B.; Abs, R.; Maiter, D. Efficacy and tolerability of lanreotide Autogel therapy in acromegalic patients previously treated with octreotide LAR. Eur. J. Endocrinol. 2004, 151, 317–324. [Google Scholar] [CrossRef]
- Andries, M.; Glintborg, D.; Kvistborg, A.; Hagen, C.; Andersen, M. A 12-month randomized crossover study on the effects of Lanreotide Autogel and Octreotide long-acting repeatable on GH and IGF-l in patients with acromegaly. Clin. Endocrinol. 2007, 68, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Caron, P.; Bex, M.; Cullen, D.R.; Feldt-Rasmussen, U.; Alfonso, A.M.P.; Pynka, S.; Rácz, K.; Schopohl, J.; Tabarin, A.; Välimäki, M.J.; et al. One-year follow-up of patients with acromegaly treated with fixed or titrated doses of lanreotide AutogelR. Clin. Endocrinol. 2004, 60, 734–740. [Google Scholar] [CrossRef] [PubMed]
- Sassolas, G.; Harris, A.G.; James-Deidier, A. Long Term Effect of Incremental Doses of the Somatostatin Analog SMS 201–995 in 58 Acromegalic Patients*. J. Clin. Endocrinol. Metab. 1990, 71, 391–397. [Google Scholar] [CrossRef] [PubMed]
- Colao, A.; Ferone, D.; Cappabianca, P.; Caro, M.L.D.B.D.; Marzullo, P.; Monticelli, A.; Alfieri, A.; Merola, B.; Cali, A.; De Divitiis, E.; et al. Effect of Octreotide Pretreatment on Surgical Outcome in Acromegaly. J. Clin. Endocrinol. Metab. 1997, 82, 3308–3314. [Google Scholar] [CrossRef] [PubMed]
- Gadelha, M.R.; Bronstein, M.D.; Brue, T.; Coculescu, M.; Fleseriu, M.; Guitelman, M.; Pronin, V.; Raverot, G.; Shimon, I.; Lievre, K.K.; et al. Pasireotide versus continued treatment with octreotide or lanreotide in patients with inadequately controlled acromegaly (PAOLA): A randomised, phase 3 trial. Lancet Diabetes Endocrinol. 2014, 2, 875–884. [Google Scholar] [CrossRef]

| Variable | Untreated (n = 28) | Pre-Treated (n = 20) | p Value * |
|---|---|---|---|
| Age (years) | 49.6 ± 2.3 | 52.2 ± 3.2 | 0.511 |
| Female sex | 64.3% (n = 18) | 70 % (n = 14) | 0.679 |
| Diabetes | 10.7% (n = 3) | 30.0% (n = 6) | 0.091 |
| Hypertension | 35.7% (n = 10) | 45.0% (n = 9) | 0.517 |
| Heart disease | 10.7% (n = 3) | 5.0% (n = 1) | 0.480 |
| Obesity | 14.3% (n = 4) | 10.0% (n = 2) | 0.658 |
| Sleep apnoea syndrome | 21.4% (n = 6) | 25.0% (n = 5) | 0.772 |
| Pituitary apoplexy | 3.6% (n = 1) | 0.0% (n = 0) | 0.333 |
| Visual involvement | 17.9% (n = 5) | 0.0% (n = 0) | 0.046 |
| Headache | 32.1% (n = 9) | 5.0% (n = 1) | 0.022 |
| Hypopituitarism | 21.4% (n = 6) | 15.0% (n = 3) | 0.574 |
| Presurgical GH (ng/mL) | 12.3 ± 3.1 | 10.5 ± 1.9 | 0.664 |
| Presurgical IGF1 (ng/mL) | 653.2 ± 58.0 | 686.5 ± 67.7 | 0.711 |
| Macroadenoma | 82.1% (n = 23) | 85.0% (n = 17) | 0.793 |
| Tumour size (mm) | 16.1 ± 1.7 | 14.3 ± 1.7 | 0.451 |
| Knosp grade 3–4 | 39.3% (n = 11) | 20.0% (n = 4) | 0.098 |
| Patients | 2010 Criteria | |||
|---|---|---|---|---|
| Pretreated | Untreated | P | Odds Ratio, 95% CI | |
| All patients (n = 48) | 60.0% (n = 12) | 50.0% (n = 14) | 0.493 | OR = 1.50 [0.47–4.79] |
| Microadenomas (n = 8) | 100% (n = 3) | 80% (n = 4) | 0.408 | NC |
| Macroadenomas (n = 40) | 52.9% (n = 9) | 43.5% (n = 10) | 0.554 | OR = 1.46 [0.41–5.15] |
| Knosp 2–3 pituitary adenomas (n = 16) | 0.0% (n = 0) | 18.2% (n = 2) | 0.360 | NC |
| All patients considering high SSAs dose (n = 48) | 78.6% (n = 11/14) | 44.1% (n = 15/34) | 0.029 | OR = 4.64 [1.09–19.7] |
| Macroadenomas considering high SSAs dose (n = 40) | 72.7% (n = 8/11) | 37.9% (n = 11/29) | 0.049 | OR = 4.39 [0.95–20.03] |
| All patients considering treatment ≥ 6 months with SSA (n = 48) | 63.6% (n = 7/11) | 51.4% (n = 19/37) | 0.473 | OR = 1.66 [0.41–6.64] |
| Macroadenomas considering treatment ≥ 6 months with SSA (n = 40) | 55.6% (n = 5/9) | 45.2% (n = 14/31) | 0.583 | OR = 1.52 [0.34–6.76] |
| All patients considering treatment with lanreotide (n = 48) | 71.4% (n = 10/14) | 47.1% (n = 16/34) | 0.124 | OR = 0.38 [0.06–2.28] |
| Macroadenomas considering treatment with lanreotide (n = 40) | 63.6% (n = 7/11) | 41.4% (n = 12/29) | 0.208 | OR = 0.5 [0.08–3.10] |
| All patients considering treatment with octreotide (n = 48) | 33.3% (n = 2/6) | 57.1% (n = 24/42) | 0.274 | OR = 2.81 [0.74–10.75] |
| Macroadenomas considering treatment with octreotide (n = 40) | 33.3% (n = 2/6) | 50.0% (n = 17/34) | 0.451 | OR = 2.48 [0.59–10.40] |
| Variable | Pre-Treated (n = 20) | Untreated (n = 28) | p Value |
|---|---|---|---|
| Surgical complications | 15.0% (n = 3) | 21.4% (n = 6) | 0.574 |
| New APD | 38.9% (n = 7) | 42.9% (n = 9) | 0.802 |
| Diabetes insipidus * | 15.0% (n = 3) | 21.4% (n = 6) | 0.574 |
| Hospitalization length stay | 6.7 ± 2.0 | 6.9 ± 1.4 | 0.937 |
| Hard tumours | 15.0% (n=3) | 28.6% (n = 8) | 0.270 |
| Variable | Pre-treated with High Doses (n = 14) | Untreated or low doses (n = 34) | p Value |
| Surgical complications | 14.3% (n = 2) | 20.6% (n = 7) | 0.611 |
| New APD | 25.0% (n = 3) | 48.2% (n = 13) | 0.175 |
| Diabetes insipidus | 21.4% (n = 3) | 17.7% (n = 6) | 0.760 |
| Hospitalization length stay | 7.46 ± 10.6 | 6.5 ± 6.20 | 0.711 |
| Hard tumours | 7.1% (n = 1) | 29.4% (n = 10) | 0.095 |
| Variable | Pre-treated >6 Months | Untreated or Pre-treated <6 Months | p Value |
| Surgical complications | 18.2% (n = 2) | 18.9% (n = 7) | 0.956 |
| New APD | 33.3% (n = 3) | 43.3% (n = 13) | 0.593 |
| Diabetes insipidus | 18.2% (n = 2) | 18.9% (n = 7) | 0.956 |
| Hospitalization length stay | 7.0 ± 10.99 | 6.72 ± 6.38 | 0.918 |
| Hard tumours | 18.2% (n = 2) | 24.3% (n = 9) | 0.670 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Araujo-Castro, M.; Pascual-Corrales, E.; Pian, H.; Ruz-Caracuel, I.; Acitores Cancela, A.; Duque, S.G.; Berrocal, V.R. The Dose of Somatostatin Analogues during Pre-Surgical Treatment Is a Key Factor to Achieve Surgical Remission in Acromegaly. Endocrines 2021, 2, 241-250. https://doi.org/10.3390/endocrines2030023
Araujo-Castro M, Pascual-Corrales E, Pian H, Ruz-Caracuel I, Acitores Cancela A, Duque SG, Berrocal VR. The Dose of Somatostatin Analogues during Pre-Surgical Treatment Is a Key Factor to Achieve Surgical Remission in Acromegaly. Endocrines. 2021; 2(3):241-250. https://doi.org/10.3390/endocrines2030023
Chicago/Turabian StyleAraujo-Castro, Marta, Eider Pascual-Corrales, Héctor Pian, Ignacio Ruz-Caracuel, Alberto Acitores Cancela, Sara García Duque, and Víctor Rodríguez Berrocal. 2021. "The Dose of Somatostatin Analogues during Pre-Surgical Treatment Is a Key Factor to Achieve Surgical Remission in Acromegaly" Endocrines 2, no. 3: 241-250. https://doi.org/10.3390/endocrines2030023
APA StyleAraujo-Castro, M., Pascual-Corrales, E., Pian, H., Ruz-Caracuel, I., Acitores Cancela, A., Duque, S. G., & Berrocal, V. R. (2021). The Dose of Somatostatin Analogues during Pre-Surgical Treatment Is a Key Factor to Achieve Surgical Remission in Acromegaly. Endocrines, 2(3), 241-250. https://doi.org/10.3390/endocrines2030023








