Initial Inoculum and the Severity of COVID-19: A Mathematical Modeling Study of the Dose-Response of SARS-CoV-2 Infections
Abstract
:1. Introduction
2. Materials and Methods
2.1. Mathematical Model
2.2. Measurements
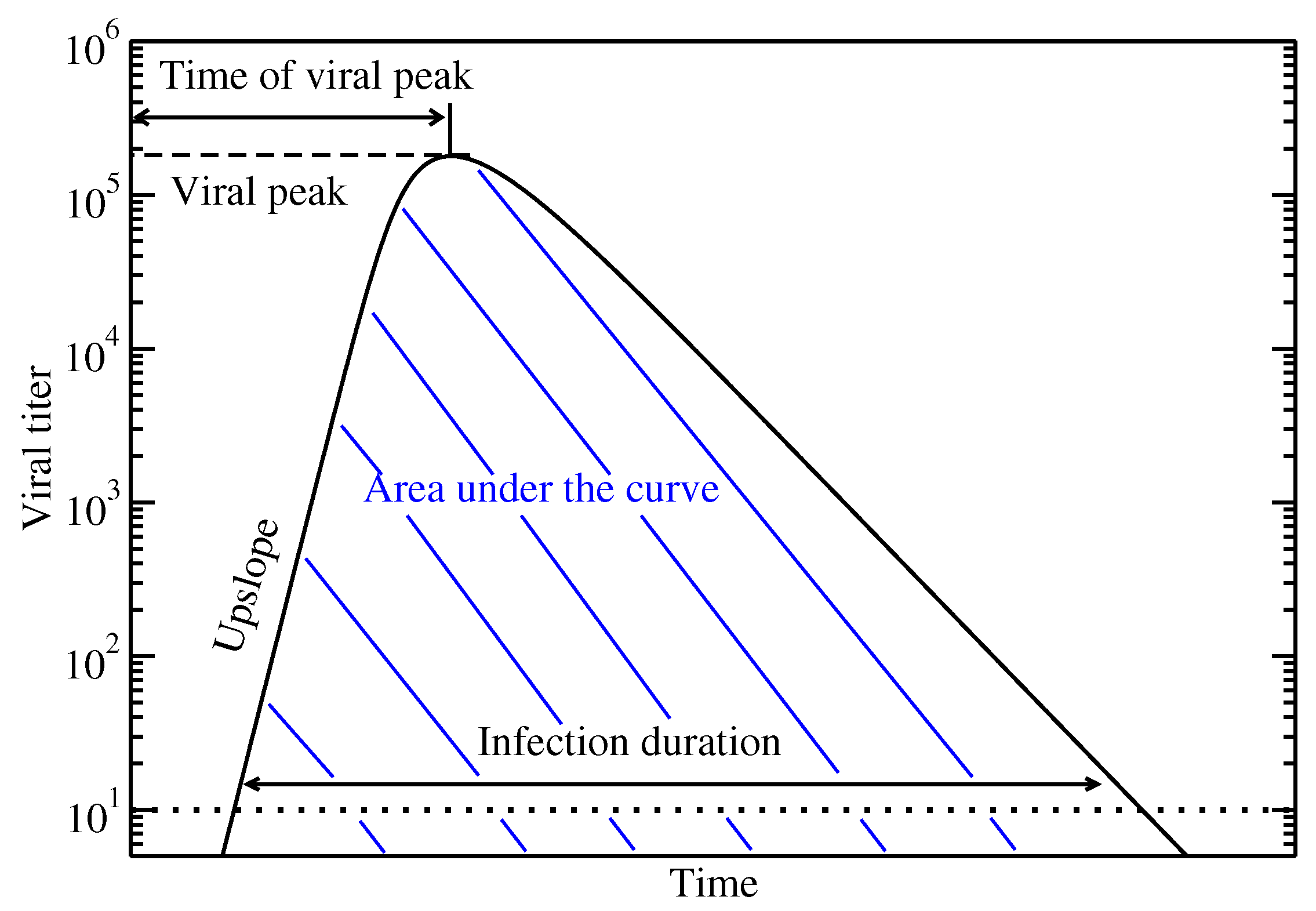
- Peak viral load: The maximum amount of virus is commonly used as an indicator of the transmissibility of an infection [42].
- Time of viral peak: This is the time between the start of the infection and the peak of the virus and can give an indication of how quickly the virus is replicating.
- Viral upslope: Viral upslope is the exponential growth rate of the viral titer before the peak is reached and is another indication of how quickly the virus is spreading from cell to cell.
- Infection duration: The infection duration is indicative of how long an infected patient might test positive for presence of the virus. Note that the threshold used here is 10 virions based on a 10 RNA copies/ml detection threshold for the experimental data [45] that is converted to individual virions.
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ABM | Agent based model |
| AUC | Area under the curve |
| CUDA | Compute unified device architecture |
| MOI | Multiplicity of infection |
| ODE | Ordinary differential equation |
| PDM | Partial differential equation model |
| SARS-CoV-2 | Severe acute respiratory syndrome coronavirus 2 |
| SHIV | Simian-human immunodeficiency virus |
References
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 2020, 395, 507–513. [Google Scholar] [CrossRef] [Green Version]
- Wu, F.; Zhao, S.; Yu, B.; Chen, Y.M.; Wang, W.; Song, Z.G.; Hu, Y.; Tao, Z.W.; Tian, J.H.; Pei, Y.Y.; et al. A new coronavirus associated with human respiratory disease in China. Nature 2020, 579, 265–271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, L.; Shen, L.; Fan, J.; Gu, F.; Hu, M.; An, Y.; Zhou, Q.; Fan, H.; Bi, J. Clinical features of patients with coronavirus disease 2019 from a designated hospital in Beijing, China. J. Med. Virol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Goyal, P.; Choi, J.J.; Pinheiro, L.C.; Schenck, E.J.; Chen, R.; Jabri, A.; Satlin, M.J.; Campion, T.R.; Nahid, M.; Ringel, J.B.; et al. Clinical Characteristics of Covid-19 in New York City. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Deng, L.; Zhang, L.; Cai, Y.; Cheung, C.W.; Xia, Z. Review of the Clinical Characteristics of Coronavirus Disease 2019 (COVID-19). J. Gen. Intern. Med. 2020. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, W.; Yi, G.Y.; Zhu, Y. Estimation of the basic reproduction number, average incubation time, asymptomatic infection rate, and case fatality rate for COVID-19: Meta-analysis and sensitivity analysis. J. Med. Virol. 2020. [Google Scholar] [CrossRef]
- Ilie, P.C.; Stefanescu, S.; Smith, L. The role of vitamin D in the prevention of coronavirus disease 2019 infection and mortality. Aging Clin. Exp. Res. 2020. [Google Scholar] [CrossRef]
- Liu, Y.; Liao, W.; Wan, L.; Xiang, T.; Zhang, W. Correlation Between Relative Nasopharyngeal Virus RNA Load and Lymphocyte Count Disease Severityin Patients with COVID-19. Viral Immunol. 2020. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.; Li, S.; Liu, J.; Liang, B.; Wang, X.; Wang, H.; Li, W.; Tong, Q.; Yi, J.; Zhao, L.; et al. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBiomed. 2020, 55, 102763. [Google Scholar] [CrossRef]
- Zhang, J.; Dong, X.; Cao, Y.; Yuan, Y.; Yang, Y.; Yan, Y.; Akdis, C.A.; Gao, Y. Clinical characteristics of 140 patients infected with SARS-CoV-2 in Wuhan, China. Allergy 2020. [Google Scholar] [CrossRef]
- Yang, Y.; Shen, C.; Li, J.; Yuan, J.; Yang, M.; Wang, F.; Li, G.; Li, Y.; Xing, L.; Peng, L.; et al. Exuberant elevation of IP-10, MCP-3 and IL-1ra during SARS-CoV-2 infection is associated with disease severity and fatal outcome. medRxiv 2020. [Google Scholar]
- Borghesi, A.; Zigliani, A.; Masciullo, R.; Golemi, S.; Maculotti, P.; Farina, D.; Maroldi, R. Radiographic severity index in COVID-19 pneumonia: Relationship to age and sex in 783 Italian patients. Radiol. Medica 2020, 125, 461–464. [Google Scholar] [CrossRef] [PubMed]
- Little, P.; Read, R.C.; Amlôt, R.; Chadborn, T.; Rice, C.; Bostock, J.; Yardley, L. Reducing risks from coronavirus transmission in the home—the role of viral load. Brit. Med. J. 2020, 369, m1728. [Google Scholar] [CrossRef] [PubMed]
- Guallar, M.P.; Meirino, R.; Donat-Vargas, C.; Corral, O.; Jouve, N.; Soriano, V. Inoculum at the time of SARS-CoV-2 exposure and risk of disease severity. Intl. J. Infect. Dis. 2020, 97, 290–292. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, M.; Beyrer, C.; Goosby, E. Masks Do More Than Protect Others During COVID-19: Reducing the Inoculum of SARS-CoV-2 to Protect the Wearer. J. Gen. Intern. Med. 2020. [Google Scholar] [CrossRef]
- Paulo, A.C.; Correia-Neves, M.; Domingos, T.; Murta, A.G.; Pedrosa, J. Influenza Infectious Dose May Explain the High Mortality of the Second and Third Wave of 1918–1919 Influenza Pandemic. PLoS ONE 2010, 5, e11655. [Google Scholar] [CrossRef] [Green Version]
- Price, I.; Mochan-Keef, E.D.; Swigon, D.; Ermentrout, B.G.; Lukens, S.; Toapanta, F.R.; Ross, T.M.; Clermont, G. The inflammatory response to influenza A virus (H1N1): An experimental and mathematical study. Phys. Rev. Lett. 2015, 374, 83–93. [Google Scholar] [CrossRef] [Green Version]
- Moore, J.R.; Ahmed, H.; Manicassamy, B.; Garcia-Sastre, A.; Handel, A.; Antia, R. Varying Inoculum Dose to Assess the Roles of the Immune Response and Target Cell Depletion by the Pathogen in Control of Acute Viral Infections. Bull. Math. Biol. 2020, 82, 35. [Google Scholar] [CrossRef]
- Wethington, D.; Harder, O.; Uppulury, K.; Stewart, W.C.; Chen, P.; King, T.; Reynolds, S.D.; Perelson, A.S.; Peeples, M.E.; Niewiesk, S.; et al. Mathematical modelling identifies the role of adaptive immunity as a key controller of respiratory syncytial virus in cotton rats. J. Roy. Soc. Interface 2019, 16, 20190389. [Google Scholar] [CrossRef]
- Li, Y.; Handel, A. Modeling inoculum dose dependent patterns of acute virus infections. J. Theor. Biol. 2014, 347, 63–73. [Google Scholar] [CrossRef]
- Go, N.; Belloc, C.; Bidot, C.; Touzeau, S. Why, when and how should exposure be considered at the within-host scale? A modelling contribution to PRRSv infection. Math. Med. Biol. 2019, 36, 179–206. [Google Scholar] [CrossRef] [PubMed]
- Manicassamy, B.; Manicassamy, S.; Belicha-Villanueva, A.; Pisanelli, G.; Pulendran, B.; García-Sastre, A. Analysis of in vivo dynamics of influenza virus infection in mice using a GFP reporter virus. Proc. Natl. Acad. Sci. USA 2010, 107, 11531–11536. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ginsberg, H.; Horsfall, F. Quantitative aspects of the multiplication of influenza A virus in the mouse lung—Relation between the degree of viral multiplication and the extent of pneumonia. J. Exp. Med. 1952, 95, 135–145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iida, T.; Bang, F. Infection of the upper respiratory tract of mice with influenza A virus. Amer. J. Hyg. 1963, 77, 169–176. [Google Scholar]
- Ottolini, M.; Blanco, J.; Eichelberger, M.; Porter, D.; Pletneva, L.; Richardson, J.; Prince, G. The cotton rat provides a useful small-animal model for the study of influenza virus pathogenesis. J. Gen. Virol. 2005, 86, 2823–2830. [Google Scholar] [CrossRef]
- Prince, G.; Porter, D.; Jenson, A.; Horswood, R.; Chanock, R.; Ginsberg, H. Pathogenesis of adenovirus type-5 pneumonia in cotton rats (sigmodon-hispidus). J. Virol. 1993, 67, 101–111. [Google Scholar] [CrossRef] [Green Version]
- Ottolini, M.; Porter, D.; Hemming, V.; Hensen, S.; Sami, I.; Prince, G. Semi-permissive replication and functional aspects of the immune response in a cotton rat model of human parainfluenza virus type 3 infection. J. Gen. Virol. 1996, 77, 1739–1743. [Google Scholar] [CrossRef]
- Morawska, L.; Cao, J. Airborne transmission of SARS-CoV-2: The world should face the reality. Env. Intl. 2020, 139, 105730. [Google Scholar] [CrossRef]
- Bourouiba, L. Turbulent Gas Clouds and Respiratory Pathogen Emissions: Potential Implications for Reducing Transmission of COVID-19. JAMA 2020, 323, 1837–1838. [Google Scholar] [CrossRef]
- Bahl, P.; Doolan, C.; de Silva, C.; Chughtai, A.A.; Bourouiba, L.; MacIntyre, C.R. Airborne or Droplet Precautions for Health Workers Treating Coronavirus Disease 2019? J. Infect. Dis. 2020, 189. [Google Scholar]
- Mittal, R.; Ni, R.; Seo, J.H. The flow physics of COVID-19. J. Fluid Phys. 2020, 894, F2. [Google Scholar] [CrossRef]
- Kakizoe, Y.; Nakaoka, S.; Beauchemin, C.A.; Morita, S.; Mori, H.; Igarashi, T.; Aihara, K.; Miura, T.; Iwami, S. A method to determine the duration of the eclipse phase for in vitro infection with a highly pathogenic SHIV strain. Sci. Rep. 2015, 5, 10371. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beauchemin, C.A.; Miura, T.; Iwami, S. Duration of SHIV production by infected cells is not exponentially distributed: Implications for estimates of infection parameters and antiviral efficacy. Sci. Rep. 2017, 7, 42765. [Google Scholar] [CrossRef] [PubMed]
- Holder, B.P.; Liao, L.E.; Simon, P.; Boivin, G.; Beauchemin, C.A.A. Design considerations in building in silico equivalents of common experimental influenza virus assays and the benefits of such an approach. Autoimmunity 2011, 44. [Google Scholar] [CrossRef] [PubMed]
- Holder, B.P.; Beauchemin, C.A. Exploring the effect of biological delays in kinetic models of influenza within a host or cell culture. BMC Public Health 2011, 11, S10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pinky, L.; Dobrovolny, H.M. SARS-CoV-2 coinfections: Could influenza and the common cold be beneficial? J. Med. Virol. 2020, 1–8. [Google Scholar]
- Handel, A.; Longini, I.M., Jr.; Antia, R. Neuraminidase Inhibitor Resistance in Influenza: Assessing the Danger of Its Generation and Spread. PLoS Comput. Biol. 2007, 3, 2456–2464. [Google Scholar] [CrossRef]
- Perelson, A.S.; Rong, L.; Hayden, F.G. Combination antiviral therapy for influenza: Predictions from modeling of human infections. J. Infect. Dis. 2012, 205, 1642–1645. [Google Scholar] [CrossRef] [Green Version]
- Dobrovolny, H.M.; Beauchemin, C.A. Modelling the Emergence of Influenza Drug Resistance: The Roles of Surface Proteins, the Immune Response and Antiviral Mechanisms. PLoS ONE 2017, 12, e0180582. [Google Scholar] [CrossRef] [Green Version]
- Pinilla, L.T.; Holder, B.P.; Abed, Y.; Boivin, G.; Beauchemin, C.A.A. The H275Y Neuraminidase Mutation of the Pandemic A/H1N1 Influenza Virus Lengthens the Eclipse Phase and Reduces Viral Output of Infected Cells, Potentially Compromising Fitness in Ferrets. J. Virol. 2012, 86, 10651–10660. [Google Scholar] [CrossRef] [Green Version]
- Cush, R.; Russo, P.; Kucukyavuz, Z.; Bu, Z.; Neau, D.; Shih, D.; Kucukyavuz, S.; Ricks, H. Rotational and translational diffusion of a rodlike virus in random coil polymer solutions. Macromolecules 1997, 30, 4920–4926. [Google Scholar] [CrossRef]
- Handel, A.; Longini, I.M.; Antia, R. Antiviral resistance and the control of pandemic influenza: The roles of stochasticity, evolution and model details. J. Theor. Biol. 2009, 256, 117–125. [Google Scholar] [CrossRef] [Green Version]
- Hayden, F.; Jennings, L.; Robson, R.; Schiff, G.; Jackson, H.; Rana, B.; McClelland, G.; Ipe, D.; Roberts, N.; Ward, P. Oral oseltamivir in human experimental influenza B infection. Antivir. Ther. 2000, 5, 205–213. [Google Scholar]
- Barroso, L.; Treanor, J.; Gubareva, L.; Hayden, F.G. Efficacy and tolerability of the oral neuraminidase inhibitor peramivir in experimental human influenza: Randomized, controlled trials for prophylaxis and treatment. Antivir. Ther. 2005, 10, 901–910. [Google Scholar]
- Gonçalves, A.; Bertrand, J.; Ke, R.; Comets, E.; de Lamballerie, X.; Malvy, D.; Pizzorno, A.; Terrier, O.; Calatrava, M.R.; Mentré, F.; et al. Timing of antiviral treatment initiation is critical to reduce SARS-CoV-2 viral load. CPT Pharmacomet. Syst. Pharmacol. 2020, 9, 509–514. [Google Scholar] [CrossRef]
- Zheng, S.; Fan, J.; Yu, F.; Feng, B.; Lou, B.; Zou, Q.; Xie, G.; Lin, S.; Wang, R.; Yang, X.; et al. Viral load dynamics and disease severity in patients infected with SARS-CoV-2 in Zhejiang province, China, January–March 2020: Retrospective cohort study. Brit. Med. J. 2020, 369, m1443. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Yan, L.M.; Wan, L.; Xiang, T.X.; Le, A.; Liu, J.M.; Peiris, M.; Poon, L.L.; Zhang, W. Viral dynamics in mild and severe cases of COVID-19. Lancet Infect. Dis. 2020, 20, 656–657. [Google Scholar] [CrossRef] [Green Version]
- To, K.K.W.; Tsang, O.T.; Leung, W.S.; Tam, A.R.; Wu, T.C.; Lung, D.C.; Yip, C.C.Y.; Cai, J.P.; Chan, J.M.C.; Chik, T.S.H.; et al. Temporal profiles of viral load in posterior oropharyngeal saliva samples and serum antibody responses during infection by SARS-CoV-2: An observational cohort study. Lancet 2020, 20, 565–574. [Google Scholar] [CrossRef] [Green Version]
- Qi, L.; Yang, Y.; Jiang, D.; Tu, C.; Wan, L.; Chen, X.; Li, Z. Factors associated with the duration of viral shedding in adults with COVID-19 outside of Wuhan, China: A retrospective cohort study. Intl. J. Infect. Dis. 2020, 96, 531–537. [Google Scholar] [CrossRef]
- He, X.; Lau, E.H.Y.; Wu, P.; Deng, X.; Wang, J.; Hao, X.; Lau, Y.C.; Wong, J.Y.; Guan, Y.; Tan, X.; et al. Temporal dynamics in viral shedding and transmissibility of COVID-19. Nat. Med. 2020, 26, 672–675. [Google Scholar] [CrossRef] [Green Version]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Lee, P.H.; Tay, W.C.; Sutjipto, S.; Fong, S.W.; Ong, S.W.X.; Wei, W.E.; Chan, Y.; Ling, L.M.; Young, B.E.; Toh, M.P.H.; et al. Associations of viral ribonucleic acid (RNA) shedding patterns with clinical illness and immune responses in Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) infection. Clin. Trans. Immunol. 2020, 9, e1160. [Google Scholar] [CrossRef]
- Mancuso, P.; Venturelli, F.; Vicentini, M.; Perilli, C.; Larosa, E.; Bisaccia, E.; Bedeschi, E.; Zerbini, A.; Rossi, P.G. Temporal profile and determinants of viral shedding and of viral clearance confirmation on nasopharyngeal swabs from SARS-CoV-2-positive subjects: A population-based prospective cohort study in Reggio Emilia, Italy. BMJ Open 2020, 10, e040380. [Google Scholar] [CrossRef]
- Park, S.Y.; Yun, S.G.; Shin, J.W.; Lee, B.Y.; Son, H.J.; Lee, S.; Lee, E.; Kim, T.H. Persistent severe acute respiratory syndrome coronavirus 2 detection after resolution of coronavirus disease 2019-associated symptoms/signs. Korean J. Intern. Med. 2020, 35, 793–796. [Google Scholar] [CrossRef]
- Li, J.; Zhang, L.; Liu, B.; Song, D. Case Report: Viral Shedding for 60 Days in a Woman with COVID-19. Amer. J. Trop. Med. Hyg. 2020, 102, 1210–1213. [Google Scholar] [CrossRef]
- Zeng, Q.; Li, G.; Ji, F.; Ma, S.; Zhang, G.; Xu, J.; Lin, W.; Xu, G.; Zhang, G.; Li, G.; et al. Clinical course and treatment efficacy of COVID-19 near Hubei Province, China: A multicentre, retrospective study. Transbound. Emerg. Dis. 2020. [Google Scholar] [CrossRef]
- Long, Q.; Tang, X.; Shi, Q.; Li, Q.; Deng, H.; Yuan, J.; Hu, J.; Xu, W.; Zhang, Y.; Lv, F.; et al. Clinical and immunological assessment of asymptomatic SARS-CoV-2 infections. Nat. Med. 2020, 26, 1200–1204. [Google Scholar] [CrossRef]
- Chen, S.C.; You, S.H.; Ling, M.P.; Chio, C.P.; Liao, C.M. Use of Seasonal Influenza Virus Titer and Respiratory Symptom Score to Estimate Effective Human Contact Rates. J. Epidemiol. 2012, 22, 353–363. [Google Scholar] [CrossRef] [Green Version]
- Handel, A.; Rohani, P. Crossing the scale from within-host infection dynamics to between-host transmission fitness: A discussion of current assumptions and knowledge. Phil. Trans. R. Soc. B 2015, 370, 20140302. [Google Scholar] [CrossRef] [Green Version]
- DeVincenzo, J.; Tait, D.; Efthimiou, J.; Mori, J.; Kim, Y.; Thomas, E.; Wilson, L.; Harland, R.; Mathews, N.; Cockerill, S.; et al. A Randomized, Placebo-Controlled, Respiratory Syncytial Virus Human Challenge Study of the Antiviral Efficacy, Safety, and Pharmacokinetics of RV521, an Inhibitor of the RSV-F Protein. Antimicrob. Agents Chemother. 2020, 64, e01884-19. [Google Scholar] [CrossRef] [Green Version]
- Hershberger, E.; Sloan, S.; Narayan, K.; Hay, C.A.; Smith, P.; Engler, R.; Jeeninga, R.; Smits, S.; Trevejo, J.; Shriver, Z.; et al. Safety and efficacy of monoclonal antibody VIS410 in adults with uncomplicated influenza A infection: Results from a randomized, double-blind, phase-2, placebo-controlled study. EBioMedicine 2019, 40, 574–582. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stevens, M.; Rusch, S.; DeVincenzo, J.; Kim, Y.; Harrison, L.; Meals, E.A.; Boyers, A.; Fok-Seang, J.; Huntjens, D.; Lounis, N.; et al. Antiviral Activity of Oral JNJ-53718678 in Healthy Adult Volunteers Challenged With Respiratory Syncytial Virus: A Placebo-Controlled Study. J. Infect. Dis. 2018, 218, 748–756. [Google Scholar] [CrossRef] [Green Version]
- DeVincenzo, J.P.; McClure, M.W.; Symons, J.A.; Fathi, H.; Westland, C.; Chanda, S.; Lambkin-Williams, R.; Smith, P.; Zhang, Q.; Beigelman, L.; et al. Activity of Oral ALS-008176 in a Respiratory Syncytial Virus Challenge Study. N. Engl. J. Med. 2015, 373, 2048–2058. [Google Scholar] [CrossRef] [Green Version]
- Hijano, D.R.; Vu, L.D.; Kauvar, L.M.; Tripp, R.A.; Polack, F.P.; Cormier, S.A. Role of Type I Interferon (IFN) in the Respiratory Syncytial Virus (RSV) Immune Response and Disease Severity. Front. Immunol. 2019, 10, 566. [Google Scholar] [CrossRef] [Green Version]
- Xu, W.; Xu, Z.; Huang, L.; Qin, E.Q.; Zhao, P.; Tu, B.; Shi, L.; Li, W.G.; Chen, W.W. Transcriptome Sequencing Identifies Novel Immune Response Genes Highly Related to the Severity of Human Adenovirus Type 55 Infection. Front. Microbiol. 2019, 10, 130. [Google Scholar] [CrossRef] [Green Version]
- Alosaimi, B.; Hamed, M.E.; Naeem, A.; Alsharef, A.A.; Al-Qahtani, S.Y.; Al-Dosari, K.M.; Alamri, A.A.; Al-Eisa, K.; Khojah, T.; Assiri, A.M.; et al. MERS-CoV infection is associated with downregulation of genes encoding Th1 and Th2 cytokines/chemokines and elevated inflammatory innate immune response in the lower respiratory tract. Cytokine 2020, 126, 154895. [Google Scholar] [CrossRef]
- Lin, L.; Lu, L.; Cao, W.; Li, T. Hypothesis for potential pathogenesis of SARS-CoV-2 infection-a review of immune changes in patients with viral pneumonia. Emerg. Microb. Infect. 2020, 9, 727–732. [Google Scholar] [CrossRef] [Green Version]
- Cao, W.; Li, T. COVID-19: Towards understanding of pathogenesis. Cell Res. 2020, 30, 367–369. [Google Scholar] [CrossRef]
- Zhu, H.; Rhee, J.W.; Cheng, P.; Waliany, S.; Chang, A.; Witteles, R.; Maecker, H.; Davis, M.M.; Nguyen, P.K.; Wu, S.M. Cardiovascular Complications in Patients with COVID-19: Consequences of Viral Toxicities and Host Immune Response. Curr. Cardiol. Rep. 2020, 22, 32. [Google Scholar] [CrossRef] [Green Version]
- Littwitz-Salomon, E.; Schimmer, S.; Dittmer, U. Dose of Retroviral Infection Determines Induction of Antiviral NK Cell Responses. J. Virol. 2017, 91, e01122-17. [Google Scholar] [CrossRef] [Green Version]
- Handel, A.; Li, Y.; McKay, B.; Pawelek, K.A.; Zarnitsyna, V.; Antia, R. Exploring the impact of inoculum dose on host immunity and morbidity to inform model-based vaccine design. PLoS Comput. Biol. 2018, 14, e1006505. [Google Scholar] [CrossRef] [Green Version]
- Redeker, A.; Welten, S.P.; Arens, R. Viral inoculum dose impacts memory T-cell inflation. Eur. J. Immunol. 2014, 44, 1046–1057. [Google Scholar] [CrossRef]
- Anderson, R.V.; McGill, J.; Legge, K.L. Quantification of the Frequency and Multiplicity of Infection of Respiratory- and Lymph Node-Resident Dendritic Cells During Influenza Virus Infection. PLoS ONE 2010, 5, e12902. [Google Scholar]
- Henriquez, K.M.; Hayney, M.S.; Xie, Y.; Zhang, Z.; Barrett, B. Association of Interleukin-8 and Neutrophils with Nasal Symptom Severity During Acute Respiratory Infection. J. Med. Virol. 2015, 87, 330–337. [Google Scholar] [CrossRef] [Green Version]
- Dobrovolny, H.M.; Reddy, M.B.; Kamal, M.A.; Rayner, C.R.; Beauchemin, C.A. Assessing mathematical models of influenza infections using features of the immune response. PLoS ONE 2013, 8, e57088. [Google Scholar] [CrossRef]
- Canini, L.; Carrat, F. Population Modeling of Influenza A/H1N1 Virus Kinetics and Symptom Dynamics. J. Virol. 2011, 85, 2764–2770. [Google Scholar] [CrossRef] [Green Version]
- Haghnegahdar, A.; Zhao, J.; Feng, Y. Lung aerosol dynamics of airborne influenza A virus-laden droplets and the resultant immune system responses: An in silico study. J. Aero. Sci. 2019, 134, 34–55. [Google Scholar] [CrossRef]
- COVID 19 Investigation. Clinical and virologic characteristics of the first 12 patients with coronavirus disease 2019 (COVID-19) in the United States. Nature Sci. 2020. [Google Scholar]
- González-Parra, G.; Dobrovolny, H.M. The rate of viral transfer between upper and lower respiratory tracts determines RSV illness duration. J. Math. Biol. 2019, 79, 467–483. [Google Scholar] [CrossRef]
- Quirouette, C.; Younis, N.P.; Reddy, M.B.; Beauchemin, C.A.A. A mathematical model describing the localization and spread of influenza A virus infection within the human respiratory tract. PLoS Comput. Biol. 2020, 16, e1007705. [Google Scholar]


| Parameter | Meaning | Value |
|---|---|---|
| Infection rate | 84.0 | |
| Mean eclipse duration | ||
| Eclipse shape parameter | 30 | |
| Mean infectious lifespan | ||
| Infectious shape parameter | 100 | |
| Viral production rate | 19,900 | |
| Viral clearance rate | 0.00490 | |
| Diffusion coefficient |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fain, B.; Dobrovolny, H.M. Initial Inoculum and the Severity of COVID-19: A Mathematical Modeling Study of the Dose-Response of SARS-CoV-2 Infections. Epidemiologia 2020, 1, 5-15. https://doi.org/10.3390/epidemiologia1010003
Fain B, Dobrovolny HM. Initial Inoculum and the Severity of COVID-19: A Mathematical Modeling Study of the Dose-Response of SARS-CoV-2 Infections. Epidemiologia. 2020; 1(1):5-15. https://doi.org/10.3390/epidemiologia1010003
Chicago/Turabian StyleFain, Baylor, and Hana M. Dobrovolny. 2020. "Initial Inoculum and the Severity of COVID-19: A Mathematical Modeling Study of the Dose-Response of SARS-CoV-2 Infections" Epidemiologia 1, no. 1: 5-15. https://doi.org/10.3390/epidemiologia1010003






