2.1. Scintillator Development
The formulation of the resin was based on five basic components; a monomer, a crosslinker, a photo-initiator to induce photopolymerization, and primary and secondary fluorescent dyes to produce light from radiation excitation. Several compounds were evaluated as part of the formulation development for each component.
In developing a 3D printable scintillator resin, the monomer forms the base of the resin and is the largest component by mass. The monomer needs to have good optical clarity at the scintillation wavelength, exhibit high solubility for fluorescent dyes, and not lead to the quenching of the scintillation light. Monomers, such as vinyltoluene or styrene, which are primarily used in plastic scintillators [
14,
15], were found to exhibit low reactivity during photopolymerization [
12,
16,
17]. Attempts to use these types of monomers resulted in cure times of >30 min to multiple hours, which is not acceptable for 3D-printing applications. Methacrylate and acrylate compounds exhibited much higher reactivity, on the order of 10s of seconds, which is suitable for 3D-printing applications. Attempts to find an off-the-shelf aromatic methacrylate or acrylate were unsuccessful. Compounds such as ethylene glycol phenyl ether acrylate, phenyl acrylate, or benzyl methacrylate resulted in either poor hardness, making mechanical polishing difficult, or poor light yield, as was the case for benzyl methacrylate which was <1000 photons per MeVee [
18]. In lieu of a suitable aromatic methacrylate, or acrylate monomer, the compound isobornyl acrylate (IBOA) was chosen due to its optical clarity, low cost, high boiling point, and high glass transition temperature. For this work, ≥85% technical-grade IBOA from Sigma Aldrich with 200 ppm monomethyl ether hydroquinone inhibitor was used.
Crosslinkers are needed to increase hardness and decrease the gel time of the photocured resins. Multiple crosslinkers were tested during this work, such as difunctionized 1,6-hexanediol dimethacrylate (HDDMA), bisphenol-A dimethacrylate (BPADMA), divinylbenzene (DVB), and tetrafunctionized crosslinkers, such as pentaerythritol tetraacrylate (PETA). The HDDMA was ≥90% pure and contained 100 ppm hydroquinone as an inhibitor, BPADMA was >98% pure with no inhibitor, PETA contained 350 ppm monomethyl ether hydroquinone as the inhibitor, and DVB was 80% pure. The photo-initiator used to start the polymerization from incident 405 nm light was TPO from Sigma Aldrich, which was 97% pure.All compounds were used as received.
For the production of light yield in the plastic scintillators, the solvents and fluors tested were EJ-309, diisopropyl naphthalene (DIN) from Eljen Technology (Sweetwater, TX, USA) (base EJ-309 solvent with no fluors), and PPO from Sigma Aldrich, which was of scintillation grade and 99% pure.1,4-Bis(2-methylstyryl)benzene (Bis-MSB) from Sigma Aldrich (St. Louis, MO, USA), diphenyl stilbene (DPS) from Luxottica Exciton (Lockbourne, OH, USA), and Exalite 416 from Luxottica Exciton were evaluated as wavelength shifters.
The compounds for the desired formulation to be tested were weighed and placed into a 28 × 57 mm (20 mL) reaction vial. The compounds were thoroughly mixed together using an alternating combination of an ultrasonic bath and hot plate to gently warm the mixture. To reduce oxygen-induced quenching, the reaction vial was transferred to a glove box pressurized with a nitrogen atmosphere (<0.1 ppm O and <1 ppm HO). Dissolved oxygen was removed by sparging with nitrogen for 10 min, and the reaction vial was sealed before removal from the glove box. During this process, care was taken to minimize exposure to ambient light, as the resin polymerizes with light. Next, the resin solution was cured using a commercially available 405 nm Formlabs (Somerville, MA, USA) Form Cure (estimated to be about 25 mW/cm) and Dymax (Torrington, CT, USA) BlueWave AX-550 VisiCure (800 mW/cm).Finally, all solid scintillators were sanded and polished using 180 grit, 400 grit, 1000 grit, and 2000 grit sandpaper and a 300 nm EXTEC alumina slurry. The dimensions of the solid scintillators were ~2.5 cm diameter and cm thick.
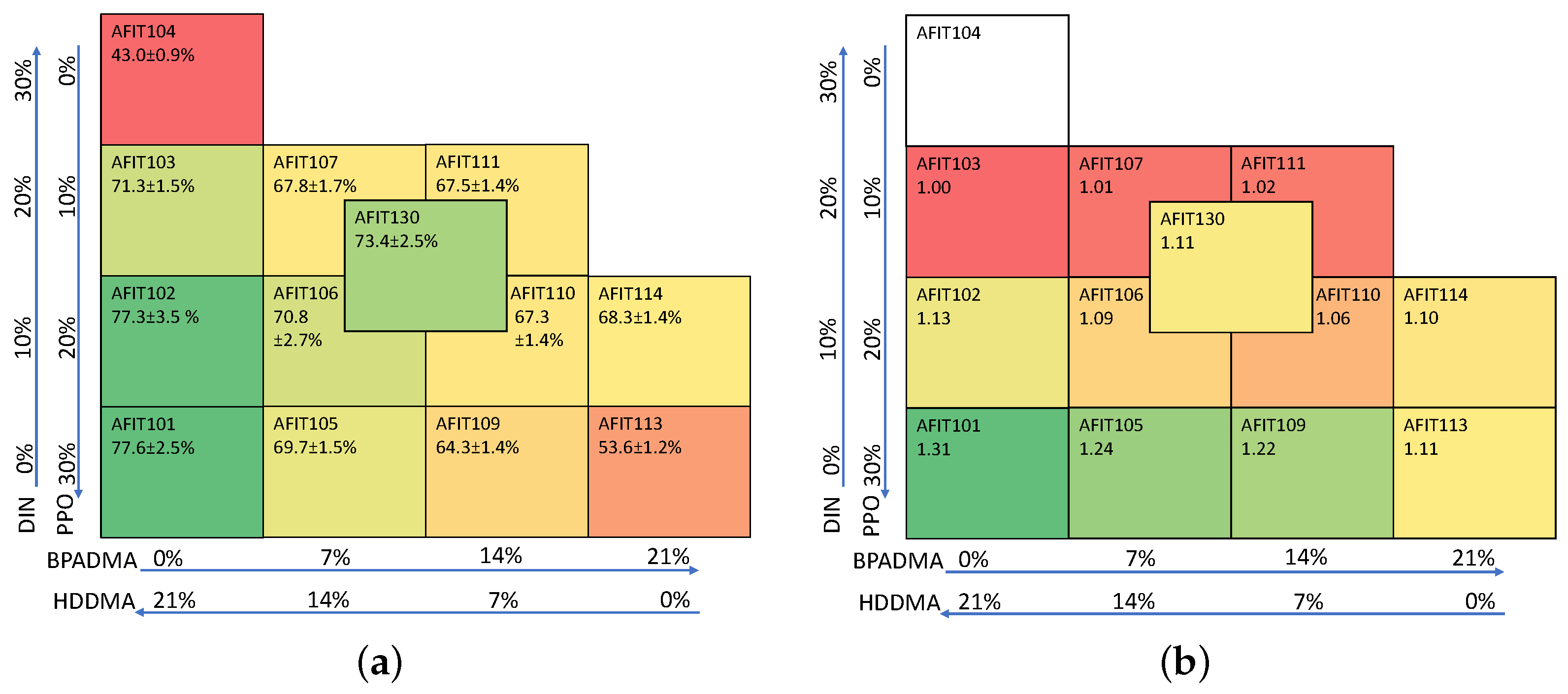
Each formulation was given a name in the format “AFITXXX” in order to track the various formulations, and all are listed in
Appendix A.
2.2. Scintillator Characterization
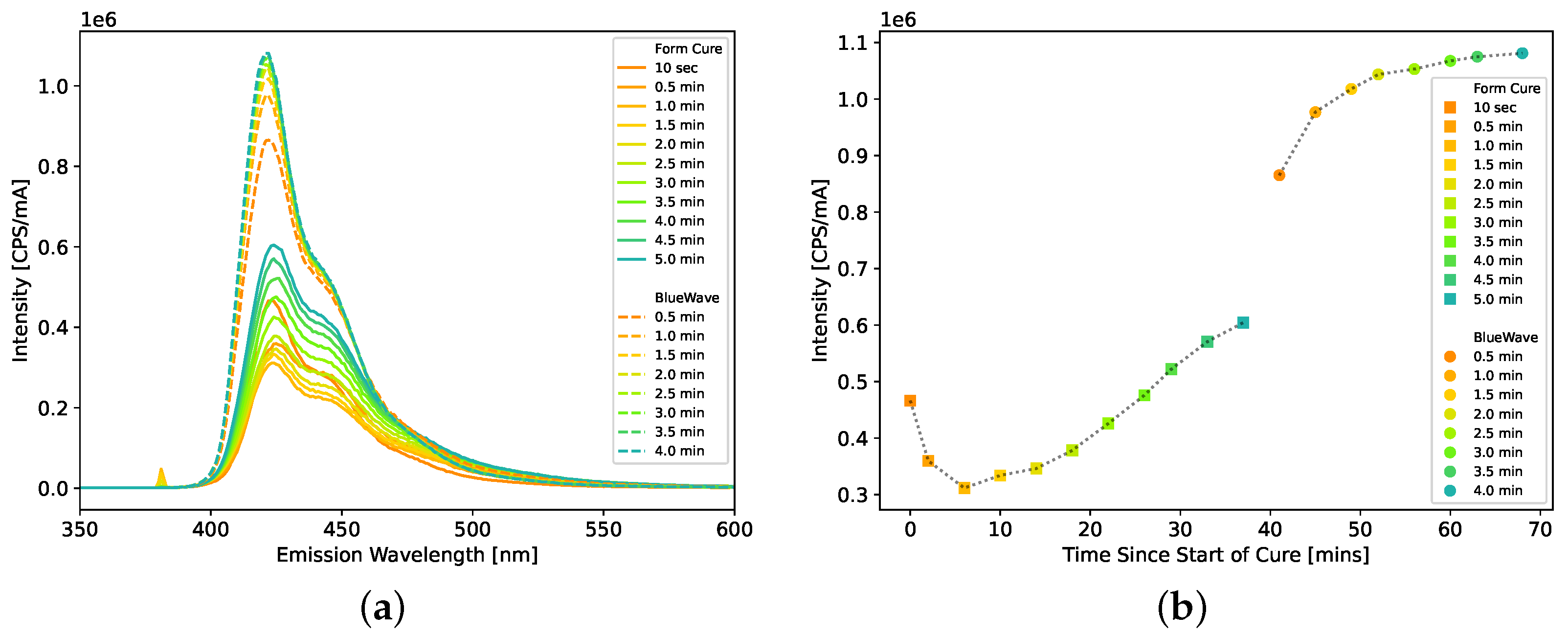
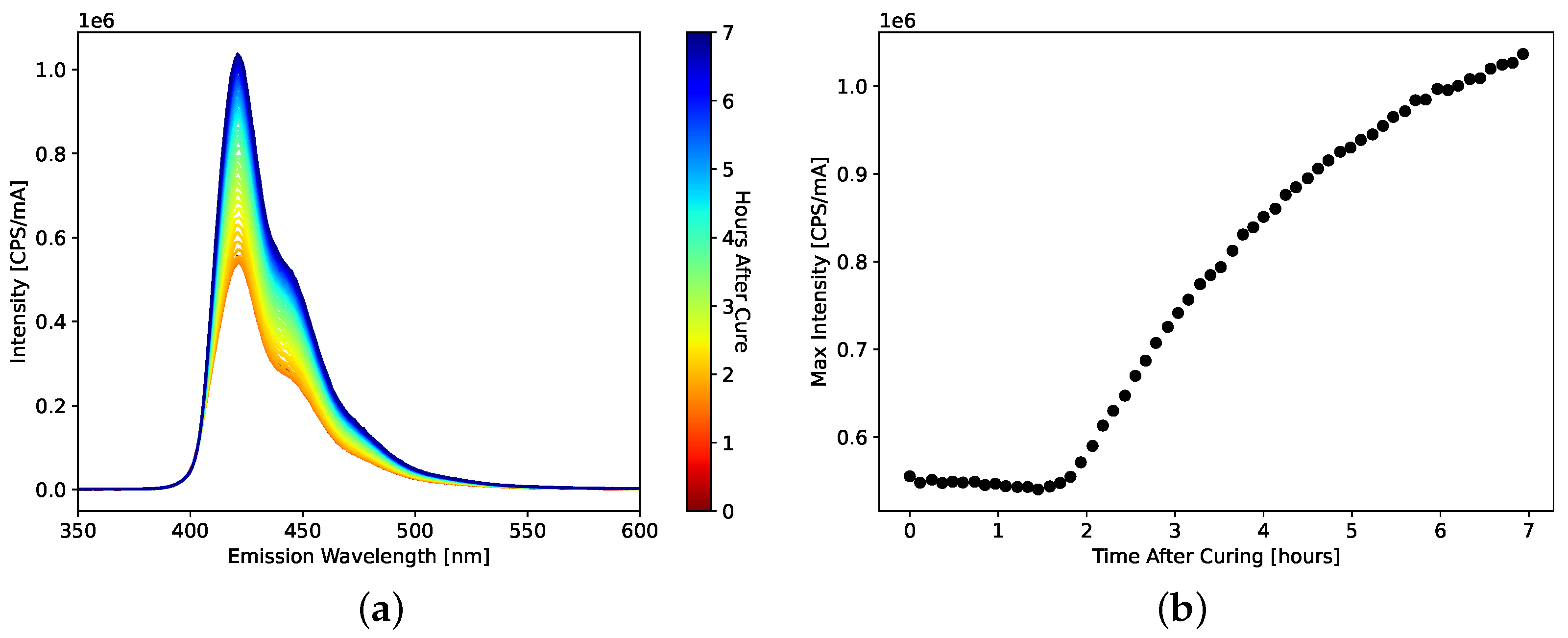
For each scintillator, the hardness of the plastic scintillators was measured using a Shore A durometer. For the curing studies, the light emission spectrum of the scintillators was measured by a Horiba (Kyoto, Japan) FluoroMax Plus with an excitation wavelength of 380 nm.
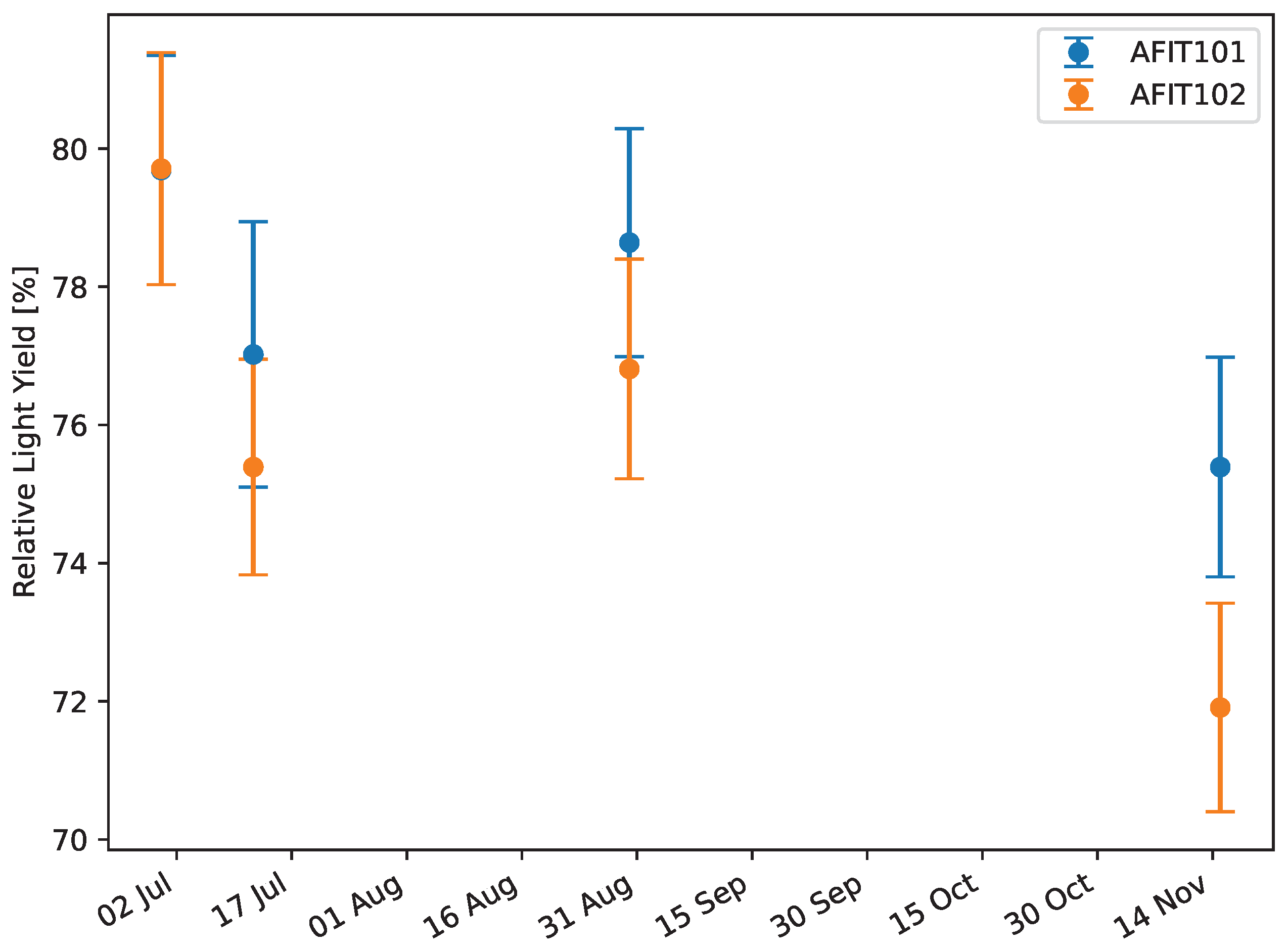
The relative light yield and PSD figure of merit (FoM) were measured for each scintillator. For the scintillators that leached (see
Section 3.2.2 for details), the hazy film was cleaned off using mild soap and water before the measurements. Each scintillator was wrapped with two layers of MIL-T-27730A PTFE tape and coupled to a 2” Hamamatsu (Shizuoka, Japan) R7724 PMT biased at −1050 V.The signals were recorded using CAEN (Viareggio, Italy) CoMPASS (version 1.5.3) software on a CAEN DT5730, a 500 MS/s, 14-bit waveform digitizer.
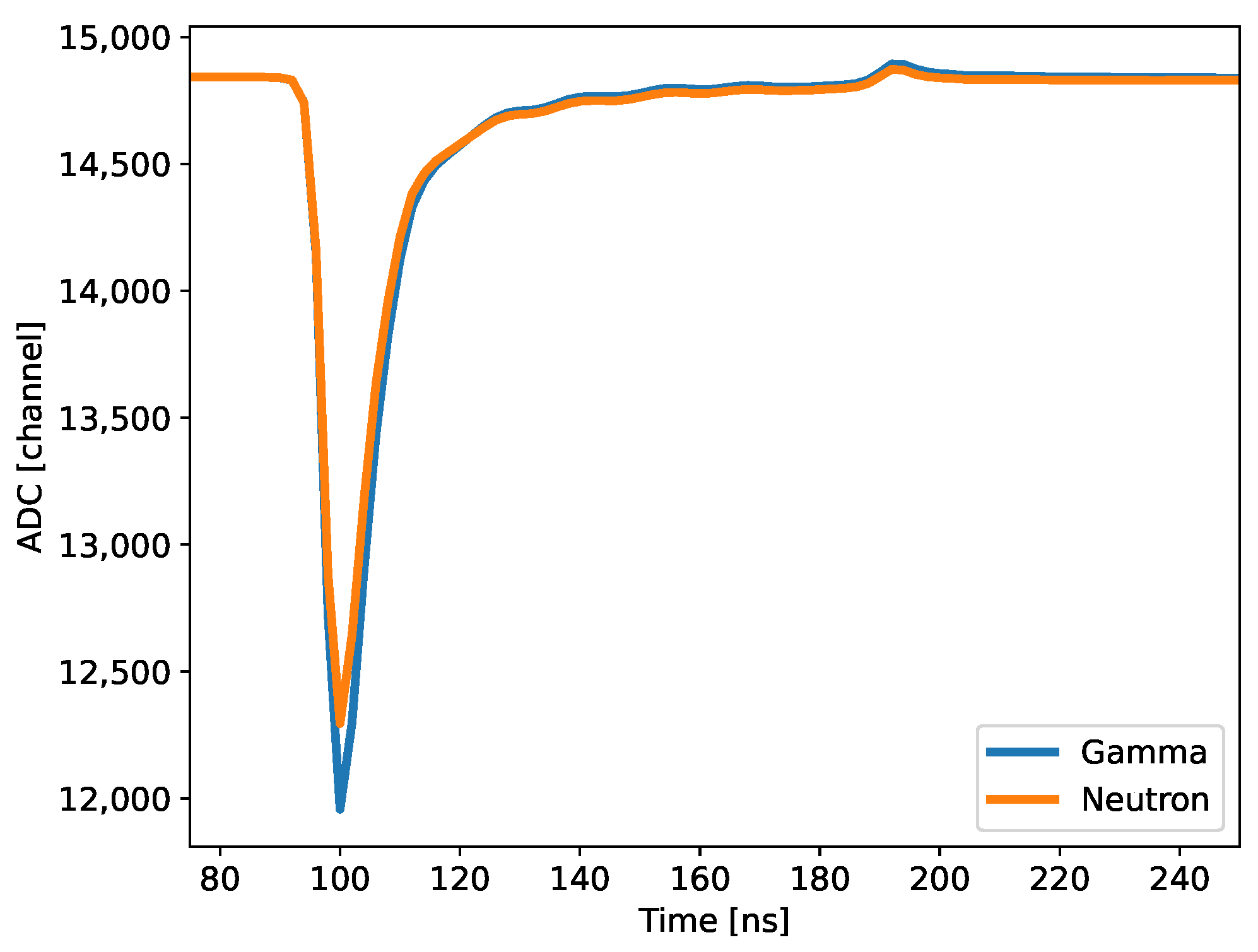
Figure 1 plots examples of neutron and gamma waveforms from the AFIT101 plastic scintillator formulation as recorded by the digitizer. The pulse shape is similar to that of other organic scintillators. For each waveform, the pretrigger was set to 48 samples (96 ns), and the baseline was calculated using the first 40 samples (80 ns). The long gate was set to 400 ns, and the short gate was varied to optimize the PSD FoM for each scintillator. The short gate averaged approximately 23 samples (46 ns). Both the long and short gates began immediately after the 40-sample baseline window.
The relative light yield of the scintillators was compared to EJ-276 [
13] and measured using the Compton edges from the 0.511 MeV (0.341 MeV) and 1.275 MeV (1.062 MeV)
Na gamma rays and the 0.662 MeV (0.478 MeV)
Cs gamma ray. The EJ-276 plastic scintillator was a bare 1” diameter × 1” long right circular cylinder, dated 30 October 2020 from the manufacturer, which has known aging degradation [
15,
19,
20]. It was removed from its nitrogen-environment shipping container on 26 April 2021 for its first use in measurements.
The Compton edge location in the recorded spectrum was determined by fitting a half-Gaussian distribution to the edge as shown in
Figure 2 for the
Na 0.511 MeV (0.341 MeV) gamma ray in EJ-276. The Compton edge, which was used for comparing the relative light yield between scintillators, was calculated as
where
and
are the mean and standard deviation of the Gaussian distribution [
21]. For each scintillator, including EJ-276, the average and standard deviation of each of the three Compton edges were calculated from multiple measurements of each scintillator. The relative light yield was then calculated as the ratio of the average Compton edge for each scintillator to EJ-276 for each of the three Compton edges. The reported relative light yield is an average of the three relative light yields obtained for each scintillator for the three Compton edges measured.
This approach in locating the Compton edge introduces potential bias [
22] and does not take into account effects such as multiple scattering [
23]. Since this potential bias was comparable in the scintillators and EJ-276 measurements (<0.5% difference), calculating relative light yield minimizes the potential impact of this bias [
22].
Second, neutron and gamma-ray induced events were discriminated using the waveform charge integration PSD as shown in
Figure 3a [
24]. After calibrating the energy of the spectrum using the Compton edges from the 0.511 MeV, 0.662 MeV, and 1.275 MeV gamma rays, a PSD histogram was generated from the events between 450 and 550 keVee. The histogram was then fit with two Gaussians, shown in
Figure 3b, and the quality of the PSD was quantified using a FoM given as [
24,
25]
where
and FWHM are the mean and full-width at half-maximum for each of the Gaussian distributions, respectively.
2.3. Exploratory Formulation Studies
A step-by-step, trial-and-error approach was taken to identify promising combinations to achieve a formulation that would create a mechanically hard, optically clear plastic with high light yield and neutron/gamma-ray discrimination. While not an exhaustive search of the parameter space, the series of small studies led to a promising general formulation that was then parametrically optimized as described in
Section 2.4. All samples were approximately 10 g (~2.5 cm diameter and ~1.6 cm thick), and the formulations,
Table A1 and
Table A2, are shown in
Appendix A for the key steps and formulations described below.
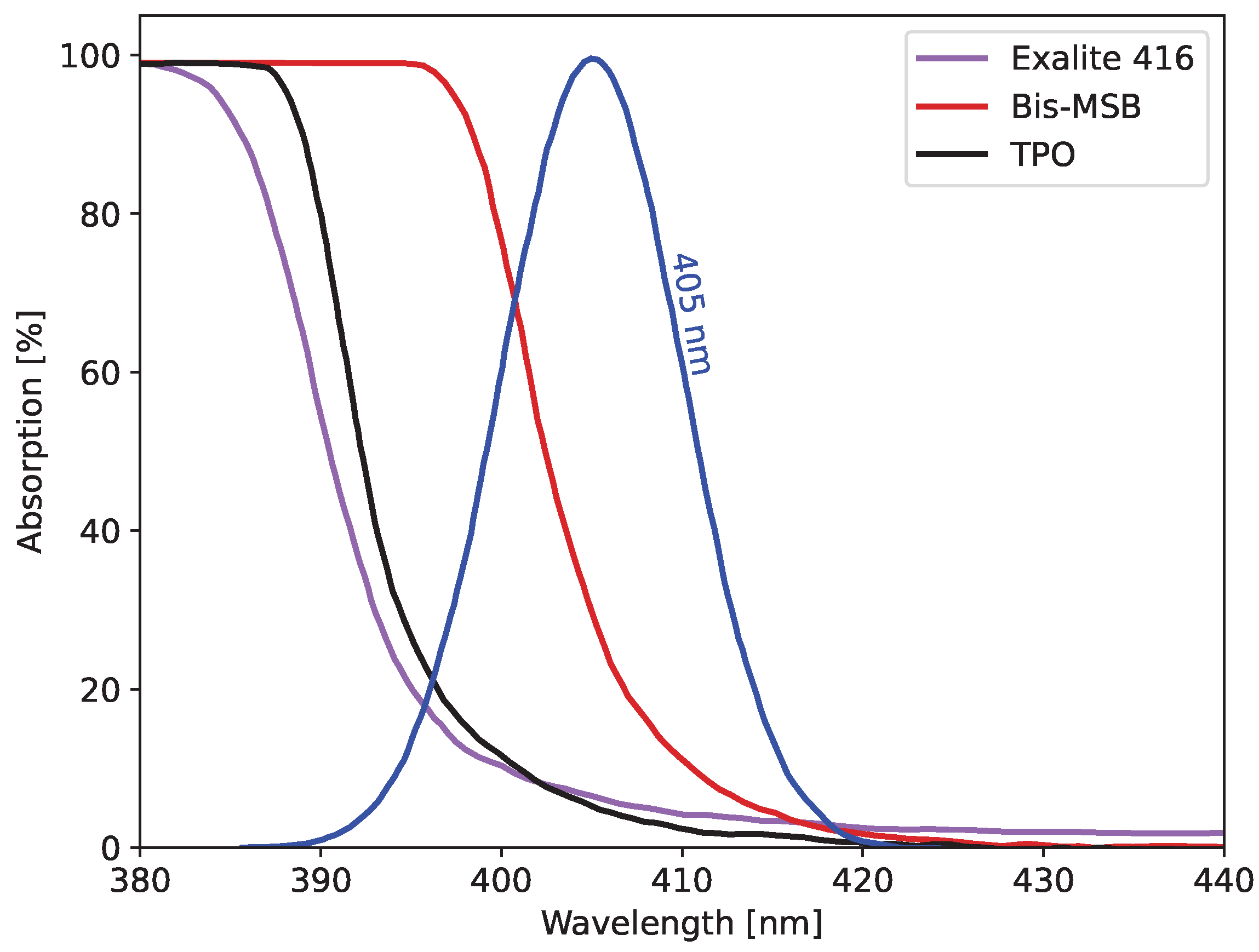
The first study explored the compatibility of different wave shifters with the 405 nm cure light and primary solvents. Three samples (AFIT002-004) were created with 0.2 wt% of Bis-MSB, Exalite 416, and DPS, respectively. The other chemicals in the scintillators were kept constant for all three. The DPS did not dissolve into the solution. The scintillator with Bis-MSB was solid throughout but softer in the center, while the Exalite 416 formulation was consistently hard throughout. It was determined that the Bis-MSB had a higher absorption at 405 nm, shown in
Figure 4, preventing the light from penetrating the entire depth of the resin and obtaining a complete cure. Therefore, Exalite 416 was selected as the wave shifter for all future formulations.
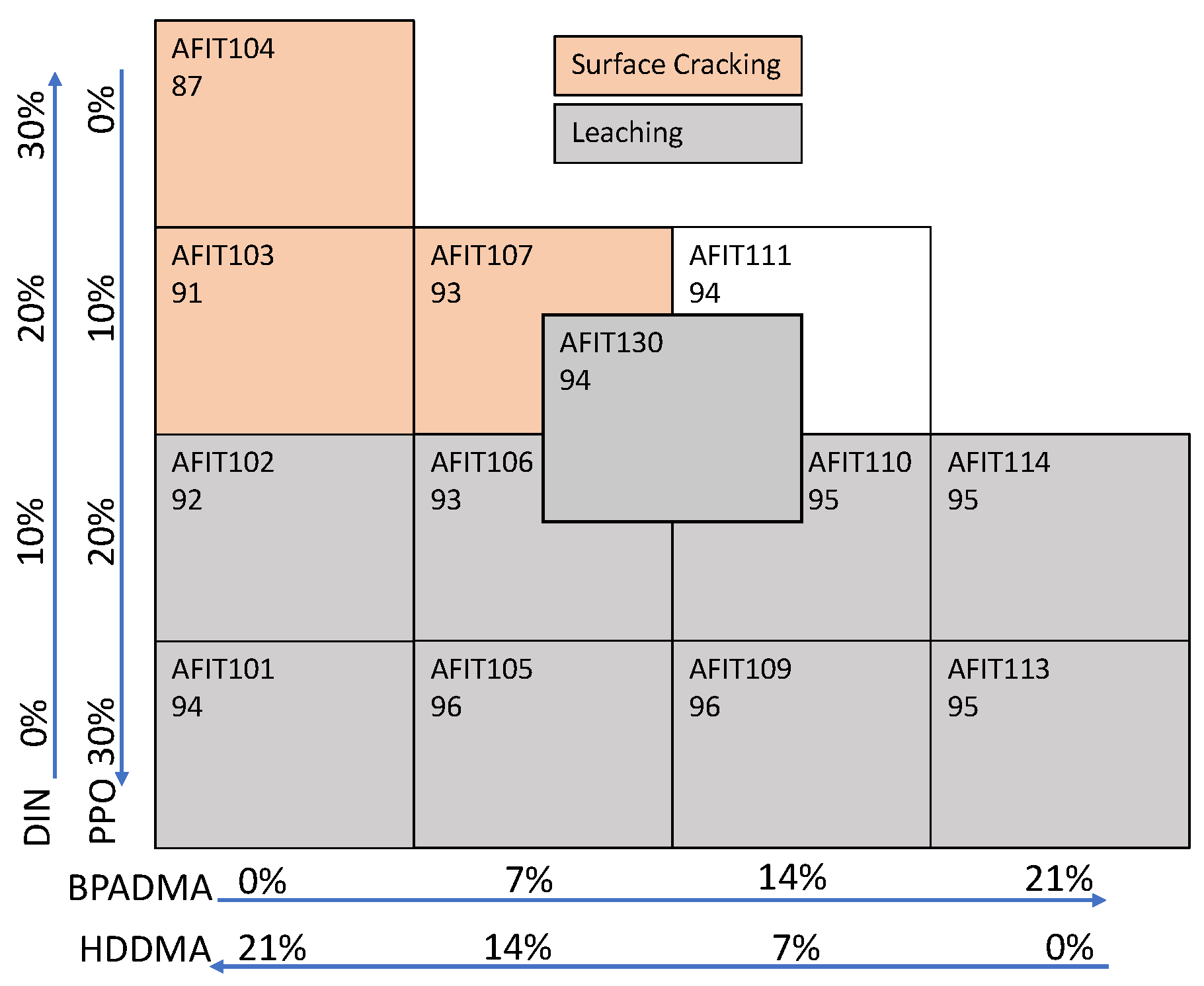
In the next step, the ratio of plastic-forming to light-yielding compounds was switched to 60/40 to increase the light yield of the scintillator. Reduced crosslinker concentration had previously been observed to result in soft, gummy-like cures, while high crosslinker concentrations led to cloudy scintillators. Therefore, the solid BPADMA was replaced with liquid HDDMA to explore formulations that lead to a harder, clearer plastic since the liquid HDDMA better dissolved and did not precipitate out of the solution. Three formulations with different IBOA/HDDMA ratios (70/30, 60/40, and 50/50) were explored. The formulations with the 60/40 and 50/50 IBOA/HDDMA ratios (AFIT013 and 014) yielded cloudy scintillators. The formulation with the 70/30 IBOA/HDDMA ratio (AFIT009) was clear but too soft, similar to hard rubber. PETA and DVB crosslinkers were also tried but did not provide better results than HDDMA or BPADMA. Additionally, all attempts to use DVB or any vinyl monomer significantly increased the required cure time.
Therefore, an IBOA/HDDMA ratio of 70/30 was used for future formulations, and variations were made to improve the hardness and radiation detection performance. First, the amount of light-yielding compounds was reduced to improve the hardness of the plastic. Two formulations (AFIT016 and 017) were developed with 30 wt% (70 wt% IBOA and HDDMA) and 20 wt% (80 wt% IBOA and HDDMA) light-yielding compounds. The Exalite 416 and TPO were held the same at 0.2 wt% and 0.1 wt%, respectively. Second, the amount of Exalite 416 was doubled from 0.2 wt% (AFIT016) to 0.4 wt% (AFIT022). Third, an approximately 50/50 ratio of HDDMA and BPADMA (AFIT023) was tested as the crosslinker. Each of these was sequentially cured for 4–5 min in a Formlabs Form Cure, followed by 1 min in the Dymax BlueWave AX-550 VisiCure, and then sanded and polished.
An increase in the plastic-forming compounds to 70–80 wt% and the addition of the BPADMA crosslinker (AFIT023) resulted in scintillators that were harder than the baseline 60/40 (plastic-forming/light-yielding compounds) ratio (e.g., AFIT013). The radiation detection performance of these scintillators demonstrated reasonable light yield (58–65% of EJ-276) and PSD (FoM between 0.74 and 1.07). It was observed that increasing the light-yielding compounds above a combined total of 30 wt% did not provide more light yield, attributable to the scintillator’s reduced clarity, but it did improve the PSD performance. However, a reduction to 20 wt% of light-yielding compounds did result in a ~5% reduction in light yield and a ~16% reduction in PSD performance. Doubling the amount of Exalite 416 only slightly increased the light yield. Finally, the addition of BPADMA with HDDMA resulted in similar light yield and PSD performance. While it was observed that the scintillator with an 80/20 ratio of plastic-forming to light-yielding compounds was the clearest and hardest, it had a relatively low light yield and PSD FoM. Thus, a 70/30 ratio of plastic-forming to light-yielding compounds was carried forward.