Top and Side Leakage Effects on Thermoregulation and Moisture Retention with Facemask Wearing
Abstract
1. Introduction
- (1)
- Developing a unified ambiance–mask–face–airway model with varying gaps that account for cyclic breathing, heat transfer, vapor transport, and phase change.
- (2)
- Validating the computational model using experimental methods, including Schlieren imaging of exhaled flows, leakage velocities using an anemometer, infrared imaging of facial/mask temperatures, and real-time temperature using thermal probes.
- (3)
- Analyzing variations in temperature and vapor trapping beneath the mask under different leakage conditions (Top and Side) in comparison to scenarios with no mask and mask-wearing with a perfect fit.
- (4)
- Examining airflow dynamics responsible for the spatiotemporal variations in facial temperatures, particularly at key facial regions. such as the philtrum, chin, nose bridge, and cheeks, as well as in the proximity of the gaps.
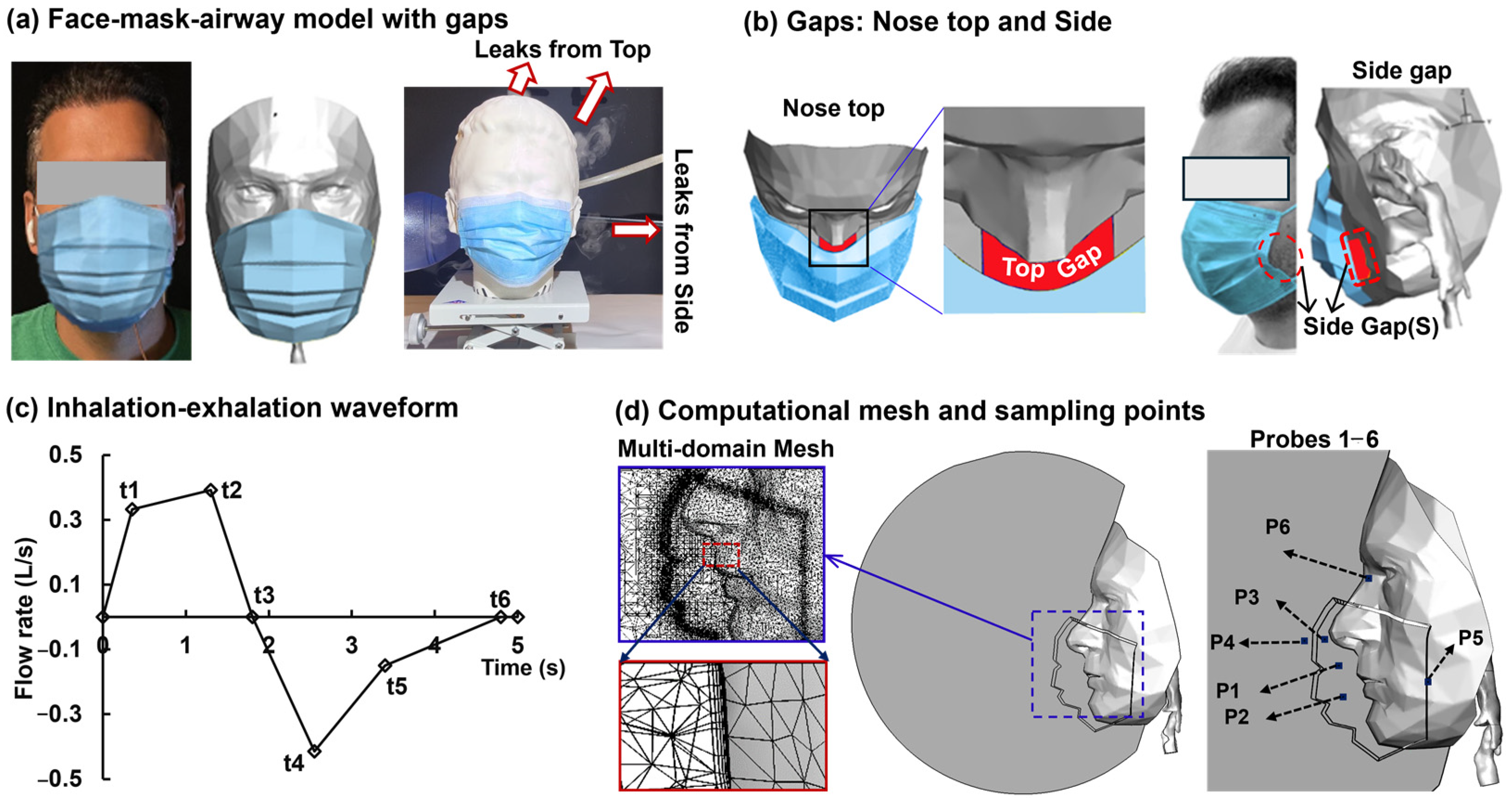
2. Materials and Methods
2.1. Integrated Respiratory–Facial Interface Simulation
2.2. Facemask Properties
2.3. Boundary Conditions
2.4. Numerical Methods
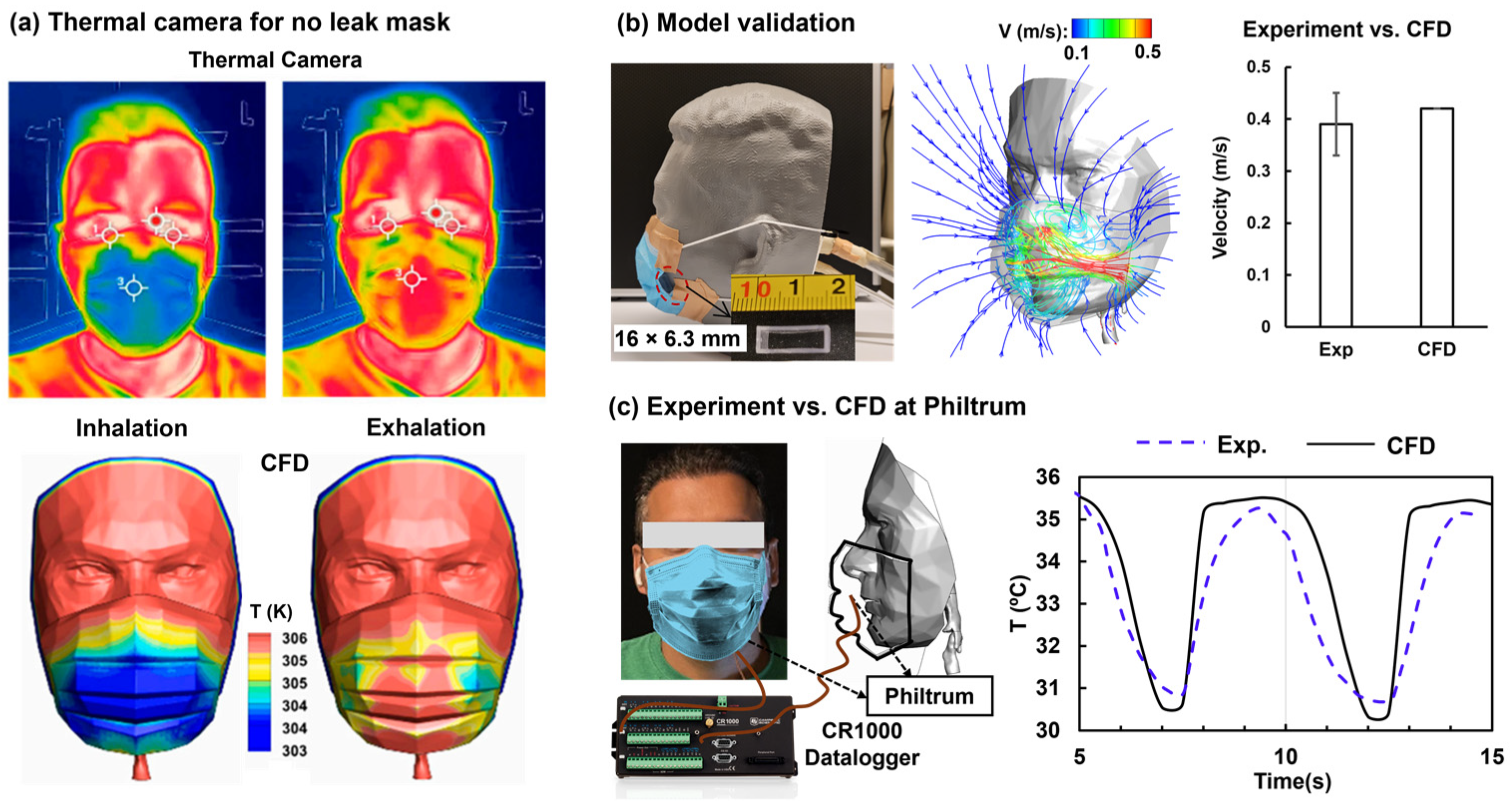
2.5. Experimental Methods
3. Results
3.1. Computional Model Validation
3.2. Airflows
3.2.1. No Mask (NM)
3.2.2. Airflow and Pressure Visualization in Mask-Wearing Cases
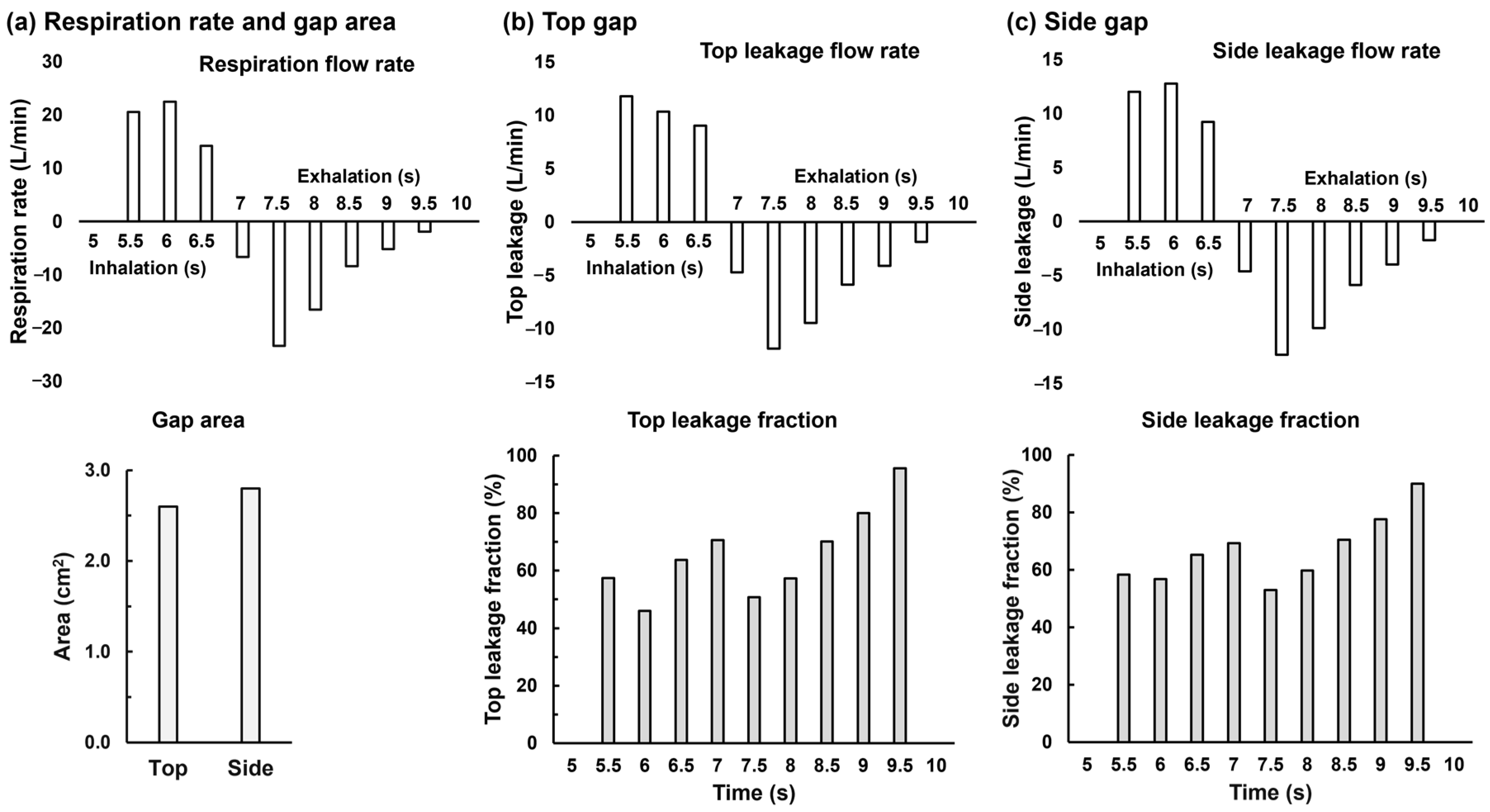
3.2.3. Leakage Flow Quantification
3.3. Leakage-Elicited Thermo-Humidity Regulations
3.3.1. No Leak
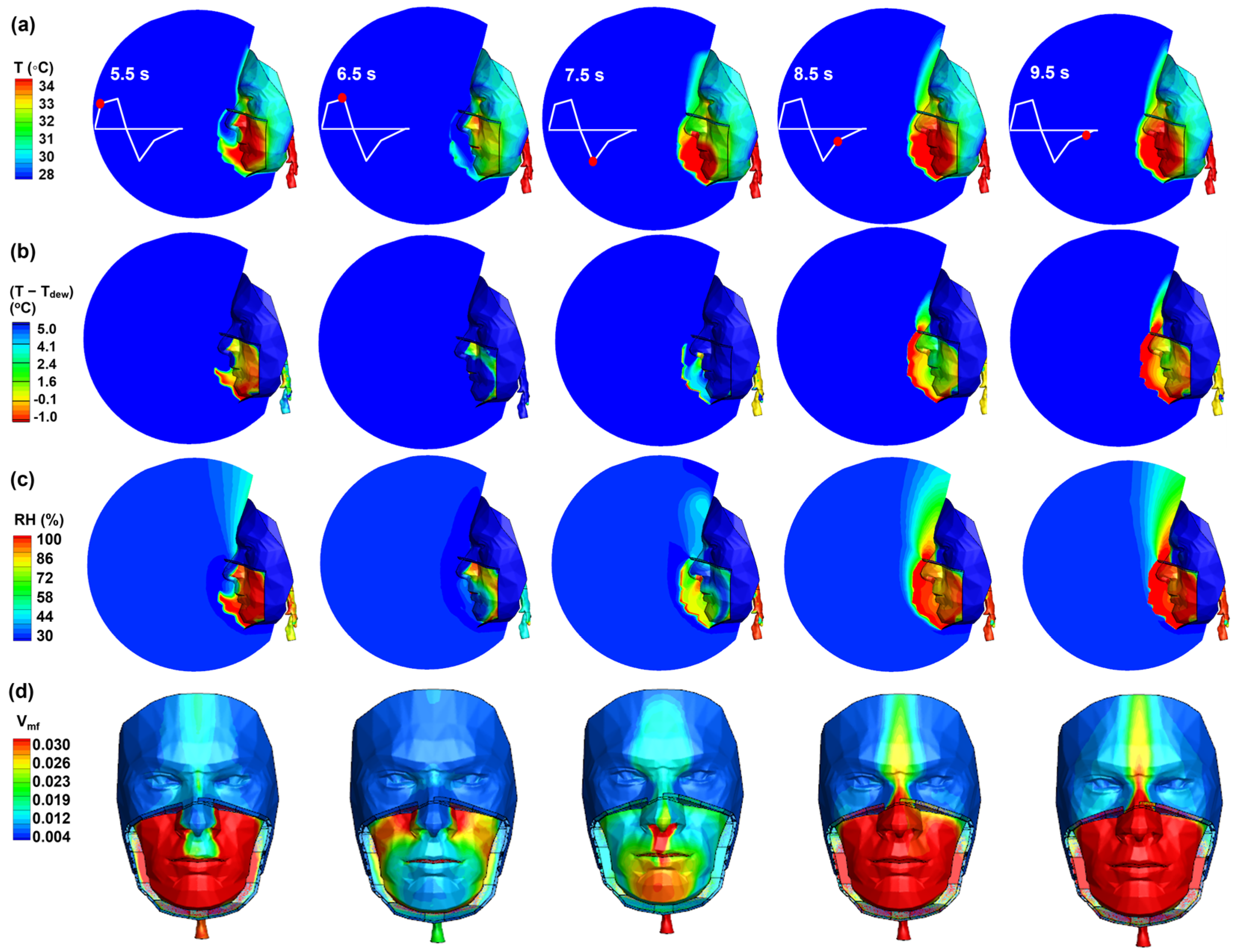
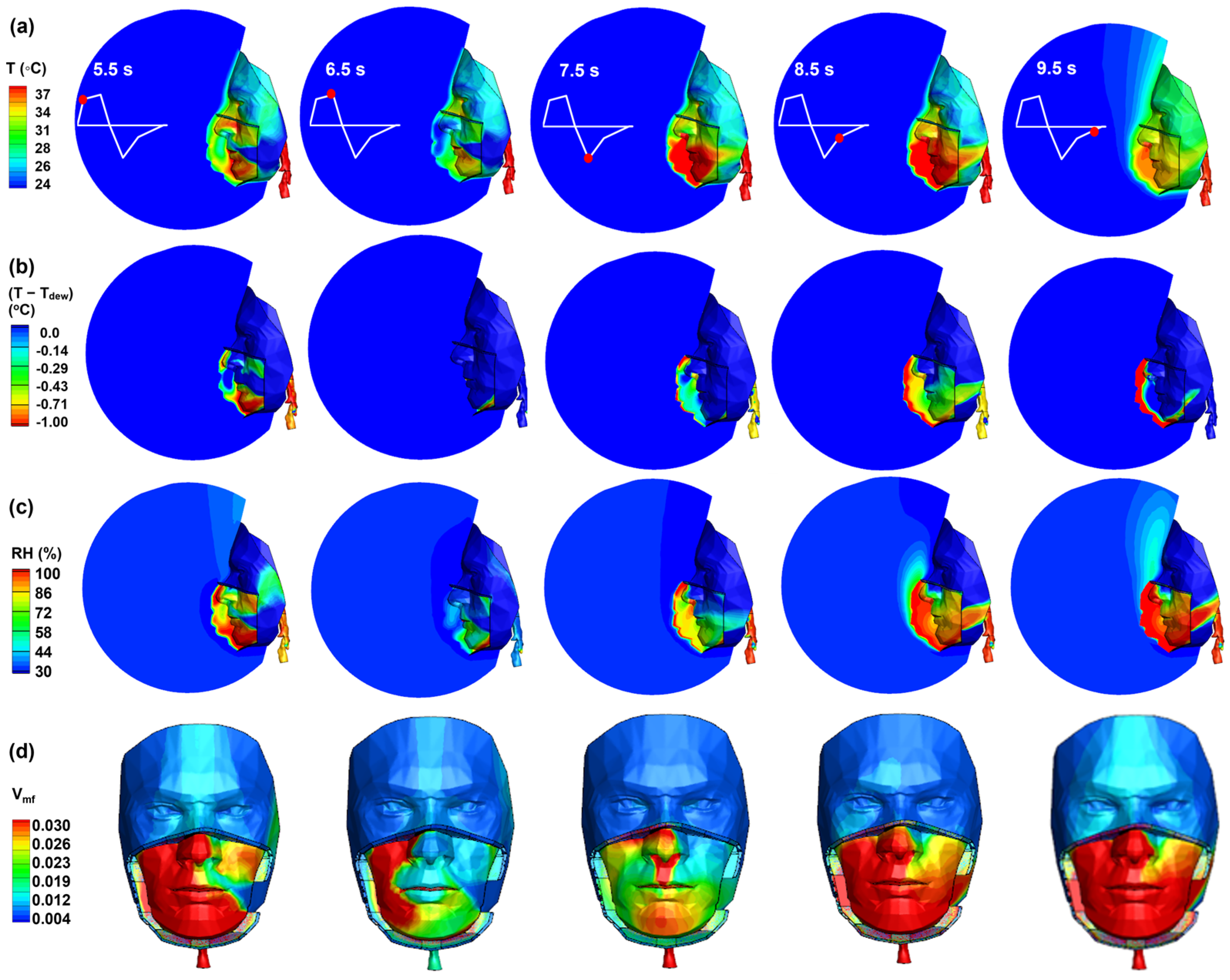
3.3.2. Top Gap
3.3.3. Side Gap
3.4. Quantitative Analysis of Thermo-Humidity Variables
4. Discussion
4.1. Leakage Flows and Thermoregulation
4.2. Moisture Accumulation and Implications
4.3. Limitations
5. Conclusions
- (1)
- The balance between natural and forced convection plays an important role in regulating thermo-humidity underneath the mask and is sensitive to leakage flows.
- (2)
- For a given gap, the leakage fraction varies significantly within one breathing cycle.
- (3)
- In the no-leak scenario, the nose is the major region that benefits least from inhalation cooling and thus experiences excess thermal stress throughout the breathing cycle. Leakage flows mitigate this stress during inhalation in both top and side gap cases.
- (4)
- Vapor condensation occurs in the mask filter medium during exhalation for all mask-wearing cases, causing moisture retention in the filter.
- (5)
- When considering thermo-humidity variations at a matrix of sampling points, unique patterns are observed that differentiate the no-leak, top-gap, and side-gap cases, suggesting the feasibility of leveraging multi-point thermo-humidity measurements to quantify mask fit and/or leakage fraction.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Scheid, J.L.; Lupien, S.P.; Ford, G.S.; West, S.L. Commentary: Physiological and psychological impact of face mask usage during the COVID-19 pandemic. Int. J. Environ. Res. Public. Health 2020, 17, 6655. [Google Scholar] [PubMed]
- Cucinotta, D.; Vanelli, M. WHO declares COVID-19 a pandemic. Acta Biomed. 2020, 91, 157–160. [Google Scholar] [PubMed]
- Buzzin, A.; Domènech-Gil, G.; Fraschetti, E.; Giovine, E.; Puglisi, D.; Caputo, D. Assessing the consequences of prolonged usage of disposable face masks. Sci. Rep. 2022, 12, 16796. [Google Scholar]
- Stadnytskyi, V.; Bax, C.E.; Bax, A.; Anfinrud, P. The airborne lifetime of small speech droplets and their potential importance in SARS-CoV-2 transmission. PNAS 2020, 117, 11875–11877. [Google Scholar] [CrossRef]
- Alsved, M.; Matamis, A.; Bohlin, R.; Richter, M.; Bengtsson, P.-E.; Fraenkel, C.-J.; Medstrand, P.; Löndahl, J. Exhaled respiratory particles during singing and talking. Aerosol Sci. Technol. 2020, 54, 1245–1248. [Google Scholar] [CrossRef]
- Cates, V.C.; Marullo, A.L.; Isakovich, R.; Bird, J.D.; Keess, J.L.; Ricord, K.C.; Leslie, K.M.; Janssens, A.A.; Bhardwaj, R.; Day, T.A. Comparison of mild physiological effects of surgical masks and N95 respirators over 60 min at rest. J. Appl. Physiol. 2023, 135, 227–237. [Google Scholar] [CrossRef]
- Gyapong, F.; Debrah, E.; Oforiwaa, M.; Isawumi, A.; Mosi, L. Challenges and adverse effects of wearing face masks in the COVID-19 era. Challenges 2022, 13, 67. [Google Scholar] [CrossRef]
- Pan, X.; Li, X.; Kong, P.; Wang, L.; Deng, R.; Wen, B.; Xiao, L.; Song, H.; Sun, Y.; Zhou, H. Assessment of use and fit of face masks among individuals in public during the COVID-19 pandemic in China. JAMA Netw. Open 2021, 4, e212574. [Google Scholar] [CrossRef]
- Su, W.-C.; Lee, J.; Xi, J.; Zhang, K. Investigation of mask efficiency for loose-fitting masks against ultrafine particles and effect on airway deposition efficiency. Aerosol Air Qual. Res. 2022, 22, 210228. [Google Scholar] [CrossRef]
- Schmitt, J.; Wang, J. A critical review on the role of leakages in the facemask protection against SARS-CoV-2 infection with consideration of vaccination and virus variants. Indoor Air 2022, 32, e13127. [Google Scholar] [CrossRef]
- Barari, K.; Si, X.; Xi, J. Impacts of mask wearing and leakages on cyclic respiratory flows and facial thermoregulation. Fluids 2023, 9, 9. [Google Scholar] [CrossRef]
- Xi, J.; Barari, K.; Si, X.A.; Abdollahzadeh Jamalabadi, M.Y.; Park, J.H.; Rein, M. Inspiratory leakage flow fraction for surgical masks with varying gaps and filter materials. Phys. Fluids 2022, 34, 041908. [Google Scholar]
- Dai, X.; Wang, Z.; Zheng, W.; He, Z.; Wang, Y. Numerical simulation of the thermo-mechanical coupling behavior of skin tissue exposed to repetitive pulse laser irradiation. Int. J. Heat. Mass. Transf. 2024, 234, 126063. [Google Scholar]
- Paiva-Santos, A.C.; Gonçalves, T.; Peixoto, D.; Pires, P.C.; Velsankar, K.; Jha, N.K.; Chavda, V.P.; Mohammad, I.S.; Cefali, L.C.; Mazzola, P.G. Rosacea topical treatment and care: From traditional to new drug delivery systems. Mol. Pharm. 2023, 20, 3804–3828. [Google Scholar] [PubMed]
- Rathi, S.K.; Dsouza, J.M. Maskne: A new acne variant in COVID-19 era. Indian. J. Dermatol. 2022, 67, 552–555. [Google Scholar] [CrossRef]
- Ipaki, B.; Merrikhpour, Z.; Taheri Rizi, M.S.; Torkashvand, S. A study on usability and design parameters in face mask: Concept design of UVW face mask for COVID-19 protection. Hum. Factors Ergon. Manuf. 2021, 31, 664–678. [Google Scholar]
- Li, Y.; Tokura, H.; Guo, Y.; Wong, A.; Wong, T.; Chung, J.; Newton, E. Effects of wearing N95 and surgical facemasks on heart rate, thermal stress and subjective sensations. Int. Arch. Occup. Environ. Health 2005, 78, 501–509. [Google Scholar]
- Scarano, A.; Inchingolo, F.; Lorusso, F. Facial skin temperature and discomfort when wearing protective face masks: Thermal infrared imaging evaluation and hands moving the mask. Int. J. Environ. Res. Public. Health 2020, 17, 4624. [Google Scholar]
- Park, S.r.; Han, J.; Yeon, Y.M.; Kang, N.Y.; Kim, E. Effect of face mask on skin characteristics changes during the COVID-19 pandemic. Skin. Res. Technol. 2021, 27, 554–559. [Google Scholar]
- Mohamadi, F.; Fazeli, A. A review on applications of CFD modeling in COVID-19 pandemic. Arch. Comput. Methods Eng. 2022, 29, 3567–3586. [Google Scholar]
- Xi, J.; Si, X.A.; Nagarajan, R. Effects of mask-wearing on the inhalability and deposition of airborne SARS-CoV-2 aerosols in human upper airway. Phys. Fluids 2020, 32, 123312. [Google Scholar]
- Liao, M.; Liu, H.; Wang, X.; Hu, X.; Huang, Y.; Liu, X.; Brenan, K.; Mecha, J.; Nirmalan, M.; Lu, J.R. A technical review of face mask wearing in preventing respiratory COVID-19 transmission. Curr. Opin. Colloid. Interface Sci. 2021, 52, 101417. [Google Scholar] [CrossRef] [PubMed]
- O’Dowd, K.; Nair, K.M.; Forouzandeh, P.; Mathew, S.; Grant, J.; Moran, R.; Bartlett, J.; Bird, J.; Pillai, S.C. Face masks and respirators in the fight against the COVID-19 pandemic: A review of current materials, advances and future perspectives. Materials 2020, 13, 3363. [Google Scholar] [CrossRef] [PubMed]
- Goyal, A.; Reeves, D.B.; Thakkar, N.; Famulare, M.; Cardozo-Ojeda, E.F.; Mayer, B.T.; Schiffer, J.T. Slight reduction in SARS-CoV-2 exposure viral load due to masking results in a significant reduction in transmission with widespread implementation. Sci. Rep. 2021, 11, 11838. [Google Scholar] [CrossRef]
- Rasekh, M.; Pisapia, F.; Hafizi, S.; Rees, D. Filtering efficiency and design properties of medical- and non-medical-grade face masks: A multiscale modeling approach. Appl. Sci. 2024, 14, 4796. [Google Scholar] [CrossRef]
- Schmitt, J.; Wang, J. Quantitative modeling of the impact of facemasks and associated leakage on the airborne transmission of SARS-CoV-2. Sci. Rep. 2021, 11, 19403. [Google Scholar]
- Bukhari, A.E.; Dakhil, A.A.B.; Albrkheel, A.A.; Almutlq, M.M.; Alolayan, O.K.; Alqahtani, M.A.; Alsubaie, F.S.; Alessa, D.S.; Alzamil, F.M. Face mask-induced skin changes: A new common phenomenon during the coronavirus disease 2019 pandemic. Dermatol. Rep 2023, 15, 9526. [Google Scholar]
- Gericke, A.; Militký, J.; Venkataraman, M.; Steyn, H.; Vermaas, J. The effect of mask style and fabric selection on the comfort properties of fabric masks. Materials 2022, 15, 2559. [Google Scholar] [CrossRef]
- Lee, K.-P.; Yip, J.; Kan, C.-W.; Chiou, J.-C.; Yung, K.-F. Reusable face masks as alternative for disposable medical masks: Factors that affect their wear-comfort. Int. J. Environ. Res. Public. Health 2020, 17, 6623. [Google Scholar]
- Kwong, L.H.; Wilson, R.; Kumar, S.; Crider, Y.S.; Reyes Sanchez, Y.; Rempel, D.; Pillarisetti, A. Review of the breathability and filtration efficiency of common household materials for face masks. ACS Nano 2021, 15, 5904–5924. [Google Scholar]
- Cherrie, J.W.; Wang, S.; Mueller, W.; Wendelboe-Nelson, C.; Loh, M. In-mask temperature and humidity can validate respirator wear-time and indicate lung health status. J. Expo. Sci. Environ. Epidemiol. 2019, 29, 578–583. [Google Scholar] [CrossRef] [PubMed]
- Xi, J.; Kim, J.; Si, X.A.; Mckee, E.; Corley, R.A.; Kabilan, S.; Wang, S. CFD modeling and image analysis of exhaled aerosols due to a growing bronchial tumor: Towards non-invasive diagnosis and treatment of respiratory obstructive diseases. Theranostics 2015, 5, 443–455. [Google Scholar] [CrossRef] [PubMed]
- Talaat, K.; Xi, J.; Baldez, P.; Hecht, A. Radiation dosimetry of inhaled radioactive aerosols: CFPD and MCNP transport simulations of radionuclides in the lung. Sci. Rep. 2019, 9, 17450. [Google Scholar] [CrossRef] [PubMed]
- Cai, L.; Song, A.Y.; Wu, P.; Hsu, P.C.; Peng, Y.; Chen, J.; Liu, C.; Catrysse, P.B.; Liu, Y.; Yang, A.; et al. Warming up human body by nanoporous metallized polyethylene textile. Nat. Commun. 2017, 8, 496. [Google Scholar] [CrossRef]
- Noto, T.; Zhou, G.; Schuele, S.; Templer, J.; Zelano, C. Automated analysis of breathing waveforms using BreathMetrics: A respiratory signal processing toolbox. Chem. Senses 2018, 43, 583–597. [Google Scholar] [CrossRef]
- Romanovsky, A.A. Skin temperature: Its role in thermoregulation. Acta Physiol. 2014, 210, 498–507. [Google Scholar] [CrossRef]
- Li, C.; Guan, G.; Reif, R.; Huang, Z.; Wang, R.K. Determining elastic properties of skin by measuring surface waves from an impulse mechanical stimulus using phase-sensitive optical coherence tomography. J. R. Soc. Interface 2012, 9, 831–841. [Google Scholar] [CrossRef]
- Guevara Morel, C.R.; van Reeuwijk, M.; Graf, T. Systematic investigation of non-Boussinesq effects in variable-density groundwater flow simulations. J. Contam. Hydrol. 2015, 183, 82–98. [Google Scholar] [CrossRef]
- Barari, K.; Thakkar, J.; Si, X.A.; Hajian, R.; Xi, J. Facemask vapor trapping, condensation, and thermoregulation. Int. J. Heat. Mass. Transf. 2024, 234, 126080. [Google Scholar] [CrossRef]
- Guan, X.; Li, X.; Yang, N.; Liu, M. CFD simulation of gas-liquid flow in stirred tanks: Effect of drag models. Chem. Eng. J. 2020, 386, 121554. [Google Scholar] [CrossRef]
- Wang, C.Y.; Cheng, P. A multiphase mixture model for multiphase, multicomponent transport in capillary porous media—I. Model development. Int. J. Heat. Mass. Transf. 1996, 39, 3607–3618. [Google Scholar]
- Wilcox, D.C. Formulation of the k-w Turbulence model revisited. AIAA J. 2008, 46, 2823–2838. [Google Scholar] [CrossRef]
- Si, X.A.; Talaat, M.; Xi, J. SARS COV-2 virus-laden droplets coughed from deep lungs: Numerical quantification in a single-path whole respiratory tract geometry. Phys. Fluids 2021, 33, 023306. [Google Scholar]
- Xi, J.; Zhao, W. Correlating exhaled aerosol images to small airway obstructive diseases: A study with dynamic mode decomposition and machine learning. PLoS ONE 2019, 14, e0211413. [Google Scholar] [CrossRef]
- Liu, J.; Hao, M.; Chen, S.; Yang, Y.; Li, J.; Mei, Q.; Bian, X.; Liu, K. Numerical evaluation of face masks for prevention of COVID-19 airborne transmission. Environ. Sci. Pollut. Res. Int. 2022, 29, 44939–44953. [Google Scholar]
- Sun, D.; Xu, J.; Chen, Q. Modeling of the evaporation and condensation phase-change problems with FLUENT. Numer. Heat. Transf. B Fundam. 2014, 66, 326–342. [Google Scholar] [CrossRef]
- Alduchov, O.A.; Eskridge, R.E. Improved magnus form approximation of saturation vapor pressure. J. Appl. Meteorol. 1996, 35, 601–609. [Google Scholar] [CrossRef]
- Lawrence, M.G. The relationship between relative humidity and the dewpoint temperature in moist air: A simple conversion and applications. Bull. Amer Meteor. Soc. 2005, 86, 225–234. [Google Scholar] [CrossRef]
- Verheyen, C.A.; Bourouiba, L. Associations between indoor relative humidity and global COVID-19 outcomes. J. R. Soc. Interface 2022, 19, 20210865. [Google Scholar]
- Pires, C. A pre-systematic review on the use of masks as a protection material for SARS-COV-2 during the COVID-19 pandemic. Int. J. Clin. Pract. 2021, 75, e14215. [Google Scholar]
- Yi, L.; Fengzhi, L.; Qingyong, Z. Numerical simulation of virus diffusion in facemask during breathing cycles. Int. J. Heat. Mass. Transf. 2005, 48, 4229–4242. [Google Scholar] [CrossRef] [PubMed]
- Siu, J.Y. Qualitative study on the shifting sociocultural meanings of the facemask in Hong Kong since the severe acute respiratory syndrome (SARS) outbreak: Implications for infection control in the post-SARS era. Int. J. Equity Health 2016, 15, 73. [Google Scholar] [CrossRef] [PubMed]
- Chaabna, K.; Doraiswamy, S.; Mamtani, R.; Cheema, S. Facemask use in community settings to prevent respiratory infection transmission: A rapid review and meta-analysis. Int. J. Infect. Dis. 2021, 104, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Xi, J.; Lei, L.R.; Zouzas, W.; April Si, X. Nasally inhaled therapeutics and vaccination for COVID-19: Developments and challenges. MedComm 2021, 2, 569–586. [Google Scholar] [CrossRef]
- Ni, R.; Michalski, M.H.; Brown, E.; Doan, N.; Zinter, J.; Ouellette, N.T.; Shepherd, G.M. Optimal directional volatile transport in retronasal olfaction. PNAS 2015, 112, 14700–14704. [Google Scholar] [CrossRef]
- Nuckols, M.L.; Zumrick, J.L.; Johnson, C.E. Heat and water vapor transport in the human upper airways at hyperbaric conditions. J. Biomech. Eng. 1983, 105, 24–30. [Google Scholar] [CrossRef]
- Guan, X.; Lin, J.; Han, J.; Gao, X.; Zhang, Y.; Hu, B.; Guidoin, R.; Wang, L. Prolonged use of surgical masks and respirators affects the protection and comfort for healthcare workers. Materials 2022, 15, 7918. [Google Scholar] [CrossRef]
- Tcharkhtchi, A.; Abbasnezhad, N.; Zarbini Seydani, M.; Zirak, N.; Farzaneh, S.; Shirinbayan, M. An overview of filtration efficiency through the masks: Mechanisms of the aerosols penetration. Bioact. Mater. 2021, 6, 106–122. [Google Scholar] [CrossRef]
- Peng, Z.; Shi, J.; Xiao, X.; Hong, Y.; Li, X.; Zhang, W.; Cheng, Y.; Wang, Z.; Li, W.J.; Chen, J.; et al. Self-charging electrostatic face masks leveraging triboelectrification for prolonged air filtration. Nat. Commun. 2022, 13, 7835. [Google Scholar] [CrossRef]
- Morris, N.B.; Piil, J.F.; Christiansen, L.; Flouris, A.D.; Nybo, L. Prolonged facemask use in the heat worsens dyspnea without compromising motor-cognitive performance. Temperature 2021, 8, 160–165. [Google Scholar] [CrossRef]
- Yip, W.L.; Leung, L.P.; Lau, P.F.; Tong, H.K. The effect of wearing a face mask on body temperature. Hong Kong J. Emerg. Med. 2005, 12, 23–27. [Google Scholar]
- Delanghe, L.; Cauwenberghs, E.; Spacova, I.; De Boeck, I.; Van Beeck, W.; Pepermans, K.; Claes, I.; Vandenheuvel, D.; Verhoeven, V.; Lebeer, S. Cotton and surgical face masks in community settings: Bacterial contamination and face mask hygiene. Front. Med. 2021, 8, 732047. [Google Scholar]
- Howard, J.; Huang, A.; Li, Z.; Tufekci, Z.; Zdimal, V.; van der Westhuizen, H.M.; von Delft, A.; Price, A.; Fridman, L.; Tang, L.H.; et al. An evidence review of face masks against COVID-19. PNAS 2021, 118, e2014564118. [Google Scholar]
- Gupta, D. Living with in-mask micro-climate. Med. Hypotheses 2020, 144, 110010. [Google Scholar]
- Mehta, P.; Picken, H.; White, C.; Howarth, K.; Langridge, K.; Nazari, K.; Taylor, P.; Qutachi, O.; Chang, M.W.; Ahmad, Z. Engineering optimisation of commercial facemask formulations capable of improving skin moisturisation. Int. J. Cosmet. Sci. 2019, 41, 462–471. [Google Scholar] [CrossRef]
- Singh, S.; Shauloff, N.; Sharma, C.P.; Shimoni, R.; Arnusch, C.J.; Jelinek, R. Carbon dot-polymer nanoporous membrane for recyclable sunlight-sterilized facemasks. J. Colloid. Interface Sci. 2021, 592, 342–348. [Google Scholar]
- Sharifi, A.R.; Ardalan, S.; Tabatabaee, R.S.; Soleimani Gorgani, S.; Yousefi, H.; Omidfar, K.; Kiani, M.A.; Dincer, C.; Naghdi, T.; Golmohammadi, H. Smart wearable nanopaper patch for continuous multiplexed optical monitoring of sweat parameters. Anal. Chem. 2023, 95, 16098–16106. [Google Scholar]
- Bianco, G.M.; Marrocco, G. Sensorized facemask with moisture-sensitive RFID antenna. IEEE Sens. Lett. 2021, 5, 6000604. [Google Scholar]
- Si, X.; Xi, J.S.; Talaat, M.; Park, J.H.; Nagarajan, R.; Rein, M.; Xi, J. Visualization and quantification of facemask leakage flows and interpersonal transmission with varying face coverings. Fluids 2024, 9, 166. [Google Scholar] [CrossRef]
- Peko, L.; Ovadia-Blechman, Z.; Hoffer, O.; Gefen, A. Physiological measurements of facial skin response under personal protective equipment. J. Mech. Behav. Biomed. Mater. 2021, 120, 104566. [Google Scholar]
- Morabito, M.; Messeri, A.; Crisci, A.; Pratali, L.; Bonafede, M.; Marinaccio, A.; Group, W.C. Heat warning and public and workers’ health at the time of COVID-19 pandemic. Sci. Total Environ. 2020, 738, 140347. [Google Scholar] [PubMed]
- Shi, D.; Song, J.; Du, R.; Chan, P.W. Dual challenges of heat wave and protective facemask-induced thermal stress in Hong Kong. Build. Environ. 2021, 206, 108317. [Google Scholar] [CrossRef] [PubMed]
- Gholamreza, F.; Nadaraja, A.V.; Milani, A.S.; Golovin, K. Enhanced protection face masks do not adversely impact thermophysiological comfort. PLoS ONE 2022, 17, e0265126. [Google Scholar]




| Momentum | Multiphase | Thermal | |
|---|---|---|---|
| Inlet | NM1: 1.703, NM2: 0.56 1 m/s Waveform | Air: N/A, Vapor: 0.039 2 Water: 0.0 | 310.15 K |
| Face | No Slip | N/A 3 | Mixed 4 |
| Airway | No Slip | N/A | 310.15 K |
| Outlet (Ambient) | 0 Pa | Air: N/A, Vapor: 0.055, Water: 0.0 | 297.15 K |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barari, K.; Si, X.; Hajian, R.; Xi, J. Top and Side Leakage Effects on Thermoregulation and Moisture Retention with Facemask Wearing. J. Respir. 2025, 5, 5. https://doi.org/10.3390/jor5020005
Barari K, Si X, Hajian R, Xi J. Top and Side Leakage Effects on Thermoregulation and Moisture Retention with Facemask Wearing. Journal of Respiration. 2025; 5(2):5. https://doi.org/10.3390/jor5020005
Chicago/Turabian StyleBarari, Kian, Xiuhua Si, Rozhin Hajian, and Jinxiang Xi. 2025. "Top and Side Leakage Effects on Thermoregulation and Moisture Retention with Facemask Wearing" Journal of Respiration 5, no. 2: 5. https://doi.org/10.3390/jor5020005
APA StyleBarari, K., Si, X., Hajian, R., & Xi, J. (2025). Top and Side Leakage Effects on Thermoregulation and Moisture Retention with Facemask Wearing. Journal of Respiration, 5(2), 5. https://doi.org/10.3390/jor5020005







