1. Introduction
The fundamental goal of radiotherapy is to deliver deadly doses to the tumor while avoiding the unnecessary dose toward the surrounding healthy tissues. This conundrum is particularly challenging, especially for percutaneous therapy [
1,
2]. There are also issues of metastatic cancer treatment difficulties, second primary cancers induced by radiotherapy, effects of cancer and microenvironment interactions, and organ motion during radiotherapy, which might cause treatment complications [
2,
3,
4].
Currently, there are many ideas on using a combination of radiotherapy with novel chemotherapy as the key strategy to curb the major radiotherapy dilemma [
5]. In chemoradiotherapy, researchers formulated ways to improve drug delivery and intensify radiosensitivity at cancer sites [
5]. In recent decades, ideas of nanoparticles (NPs) application in radiotherapy, as well as chemotherapy, have been expanding. One of the best received experiments is using metallic nanoparticles (NPs) as dose enhancers [
6,
7,
8]. Although there are NPs synthesized as natural polymer or lipid NPs, metallic nanoparticles are preferred, as they have high atomic number (Z), which could enhance the radiation-induced cell death [
9,
10].
Commonly referred to as nanoparticle-enhanced radiotherapy, the direct benefit of this treatment technique is the ability to increase the probability of physical-related radiation interactions. According to Guo, physical dose enhancement (DE) is the direct and linear response from increased radiation interaction with NPs [
11]. The photoelectric effect is the dominant interaction process during low-energy photon irradiations. Compton interaction dominates at the photon energy of >0.5 MeV, while pair-production starts at an energy of 1.022 MeV [
12]. As a result of photo absorption at the K- and L-shells, the high-Z metallic NPs release energy in the form of secondary electrons (photoelectrons, Auger electrons) and secondary photons (fluorescence) at a highly localized manner to their immediate surroundings, thus creating DE. In a biological scenario, the local DE by high-Z NPs within the tumor creates a contrast against the surrounding low-Z tissue, which would then reduce normal tissue complications while simultaneously increasing the tumor control probability [
13,
14].
Among the widely proposed metallic NPs in medical research are gold, silver, platinum, iron oxide and bismuth oxide NPs [
15,
16,
17,
18,
19,
20,
21]. To date, gold (Au; Z = 79) has remained the most studied element for DE because of its high Z and exceptional characteristic [
22,
23]. Bismuth, having a high-Z number (Bi; Z = 83), is said to be more admirable than Au due to its high X-ray absorption coefficient and affordability [
24,
25,
26].
Metallic NPs have been investigated for their radiosensitizing properties on cancer cells. Many research works on NPs in preclinical phases are up and coming. It was found that gold NPs (AuNPs) produced different radiosensitization effects in bovine aortic endothelial cells at different X-rays energies (30 to 100 keV) [
16]. The presence of AuNPs had also successfully amplified the radiation effects of brachytherapy with the iridium-192 (
192Ir) source and clinical megavoltage photon and electron in HeLa cervical cancer cells [
27]. In addition, different sizes of dendritic platinum NPs (PtNPs) have shown a sensitization factor of 1.77 to 2.31 in HeLa cells under 6 MV photon beam [
28]; meanwhile, a factor of 1.10 to 1.23 was indicated in HCT-116 colon carcinoma cells under 150 MeV proton beam [
18]. In the past few years, new metallic NPs of bismuth oxide NPs (BiONPs) were suggested as radiosensitizers [
29], and there have been several other follow-up research works in 9L gliosarcoma [
29] MCF-7 breast cancer [
20] and HeLa cells [
30] under clinical radiotherapy beams. More importantly, bismuth, as the core of BiONPs, has the advantage of being biodegradable due to its long-standing history in medicine. Its NP form could be readily oxidized and dissolved at physiological conditions and discharged from the human body as soluble bismuth ions [
31,
32]. Recently, BiONPs effects have been compared in MCF-7 and MDA-MB-231 breast cancer cells and NIH/3T3 normal cells [
21,
25,
33]. The influences of BiONPs when combined with conventional drugs, such as cisplatin, as well as a natural compound, such as baicalein-rich fraction, had revealed that the combinations could increase radiation doses of clinical radiotherapy beams in cancer cells compared to normal cells [
21,
25].
Meanwhile, some of the NPs have been investigated in vivo for radiotherapy. Iron oxide NPs in mice exhibited biocompatibility and could be excreted through the liver [
34]. Compared with AuNPs and BiONPs, iron oxide NPs have a lower X-ray absorption coefficient due to their lower Z number (Fe; Z = 26), yet are still practical for DE applications [
35,
36]. When radiation was combined with superparamagnetic iron oxide NPs (SPIONs) in mice, radiosensitization was verified, as tumor growth rate was significantly decreased [
37]. The SPIONs could also double as dose enhancers and contrast agents for magnetic resonance imaging (MRI). In the advent of MRI-guided radiotherapy accelerators, the use of multifunctional iron NPs, such as SPIONs, would become very useful in managing cancer patients undergoing radiotherapy [
38].
Besides, AuNPs have been demonstrated to have antioxidant properties [
39]. A novel protein sulfonic acid reactive AuNPs was reported to improve CT imaging sensitivity and X-rays radiation effects due to the AuNPs uptake and prolonged retention in the living mice [
40]. Previous in silico studies of AuNPs in radiotherapy using Monte Carlo simulations had also confirmed the dose enhancement in photon and electron beams [
41] and brachytherapy with various sources [
42,
43]. Compared to other simulated NPs, such as platinum, silver, iodine and iron oxide, AuNPs showed the highest dose enhancement for skin therapy with radiation [
44]. Simulations using 3D radiochromic dosimeter and Monte Carlo also discovered the potential of BiONPs’ innovative radiosensitizer in clinical radiotherapy applications [
45,
46].
The radiosensitization process affects the tumor’s cellular function by damaging the deoxyribonucleic acid (DNA) directly through single- and double-strand breaks or indirectly via free radical production, such as reactive oxygen species (ROS) through radiolysis. The irradiated tumor cells would then succumb to these damages through many cell death pathways (mitotic catastrophe, apoptosis) after treatment [
47]. To further enhance these cell deaths and overcome the limitation of intrinsic tumor radio resistance, researchers have been trying to incorporate radiobiological interventions as a combination to radiotherapy [
48].
Although more easily compared to the chemical (i.e., ROS generation, polymerization) and biological (i.e., protein repair blocking) concepts of DE, it is often tricky to measure physical DE strictly. The reason is that most of the measurement techniques are indirect and require the assistance of chemical or biological processes. For instance, the gel dosimetry system responds toward OH
• radicals generated during radiolysis [
49]. Apart from being expensive and time consuming, using gels means that the measured dose enhancement factor (DEF) may be contaminated with chemical enhancement signals from polymerization reactions with catalytically active NPs [
11]. In this study, we proposed radiochromic films (RCFs) as an alternate option for detecting NP-originated physical DE (GAFCHROMIC™ EBT3; Ashland Specialty Ingredients, Bridgewater, NJ, USA). RCF belongs to a class of two-dimensional chemical dosimeters and is well known for its high spatial resolution and near water equivalence [
22,
23,
24]. Despite operating through the polymerisation of active layer (lithium salt of pentacosa-10, 12-diynoic acid; LiPCDA), the active layer is sandwiched and protected by two chemically inert surfaces of matte polyester substrates. This feature allows for the isolation of physical DE signals from chemical enhancement. Thus, the possibility of additional polymerization of RCFs due to secondary electrons or photons released from NPs can be speculated to be contributed solely by physical DE effects. This approach also has the clear advantage of rapid dose acquisition (minimum 24 h post-irradiation) and the capability of providing direct visualization of enhanced energy deposition [
11,
50].
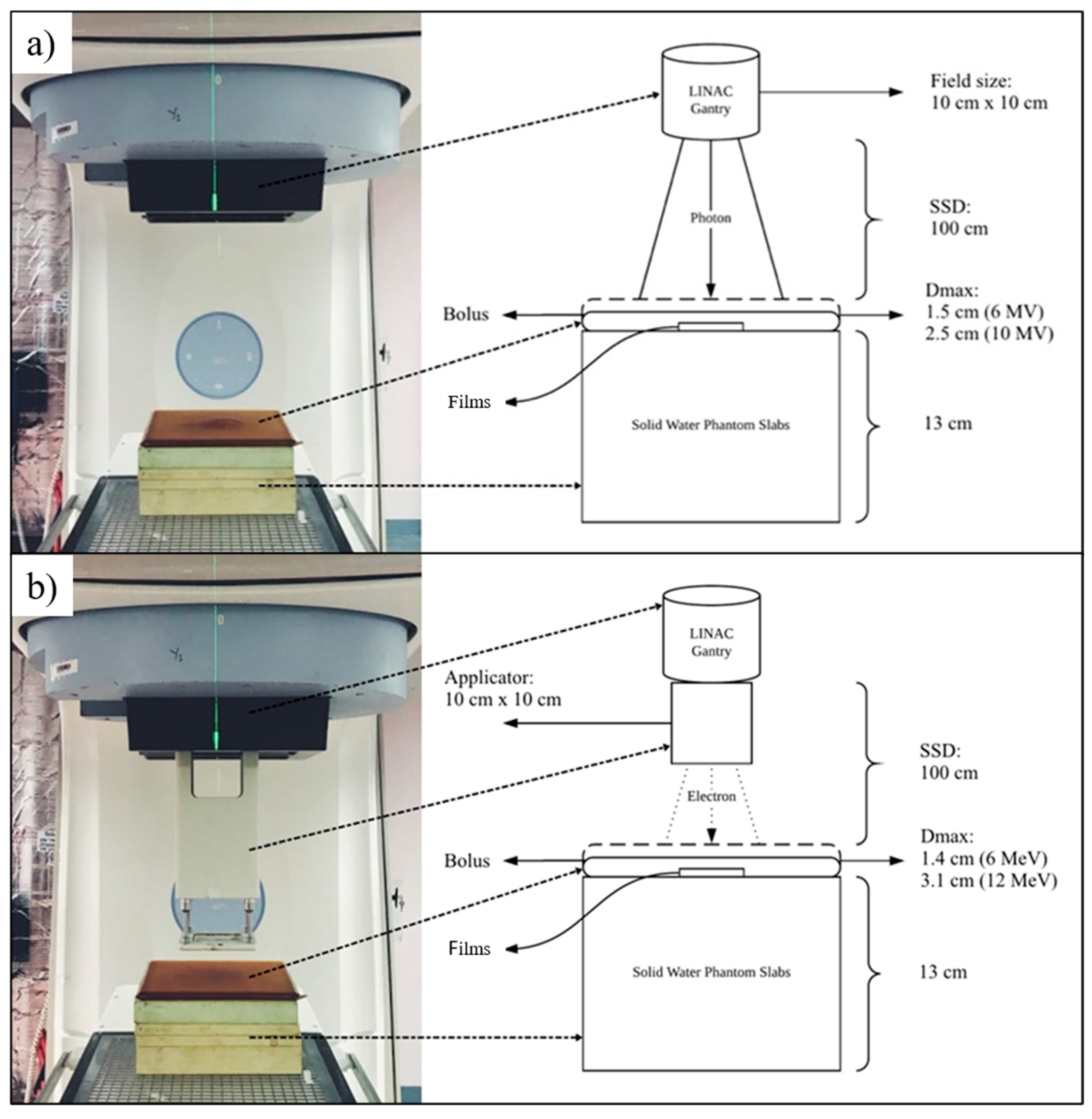
The purpose of this study is to investigate the quantification of physical DE using Gafchromic™ EBT3 RCFs with three different types of NPs: AuNPs, SPIONs and BiONPs, as to compare the effects of NPs with different atomic numbers. The present study is the first experimental study that uses different clinical radiotherapy beams to compare different types of NPs. The dosimetry characterization was carried out using standard film calibration protocols for radiotherapy beams of clinically relevant energies. Precise calibration function and accurate dose measurements are set for the RCF dosimetry system using comprehensive uncertainty analysis. DEF values collected from our experimental setup are then compared against analytical-theoretical DEF calculations from the method outlined by Corde et al. [
51] and Roeske et al. [
35] for a similar NP concentration and beam spectrum. This study also aims to provide, for the first time, a head-to-head comparison of physical DE quantification between different NPs in a film dosimetry system.
4. Discussion
Analytical calculations are one of the many methods used by researchers to predict physical DE by photons. Corde et al., for example, uses Equation (7) to estimate the DEF generated by 10 mg/mL iodine at monoenergetic photon energies of 0.001 MeV to 0.5 MeV [
51]. Roeske et al. studied the theoretical DE on multiple NP elements (Z = 25–90; 5 mg/mL) using clinically relevant photons of external beams and radionuclide sources [
35]. In a more recent publication, Srinivasan et al. used the mass energy absorption coefficient for soft tissue (ICRU-44) as the low Z interface instead of water to simulate the DEF generated by 7 mg/mL AuNPs [
71]. It is evident from the literature that the analytical calculations were conducted under varied conditions, i.e., element type, photon energy and concentration. Although it served as a challenge for us to conduct side-to-side comparisons, the results obtained in our calculations were in line with the trend of DEF observed in the studies mentioned above. In this paper, we first demonstrated that the incorporation of high Z component into the analytical calculation of absorbed dose generates theoretical DE. Bismuth oxide, characterized with the highest Z number among the three elements, offered the greatest
, followed by gold and then iron NPs. This is especially true for HDR
192Ir because of the larger amount of low-energy photons in its spectrum (0.296–0.608 MeV), where Z plays a major role in dose enhancement. In contrast, the
for 6 MV and 10 MV photons were far lower due to the dominance of interaction that is independent or weakly dependent on Z, i.e., Compton interaction.
To the best of our knowledge, this is the first study that calculates analytical
for different types of elements using Boone and Chavez’s total mass energy absorption data source via the XMuDat program. The referred data were chosen for this research because they cover all of the subsequent re-emission and reabsorption possibilities generated when an incidental photon travels through an absorber medium [
72]. In opposition to the more commonly used data source by Hubbell and Seltzer in the XCOM program/NIST website, the generated
may be underestimated because it has been reported to exclude the contribution of secondary photons, i.e., fluorescence [
35]. This could explain our findings, whereby the maximum
for AuNPs and BiONPs occur at a slightly lower energy (0.038 MeV) compared to those reported by Hubble and Seltzer’s (0.040 MeV) [
11,
12,
36]. After recalculation, the
for Au and Bi from our study (Au
= 9.364; Bi
= 26.251) were also found to be slightly higher than Hubble and Seltzer’s (Au
= 9.244; Bi
= 24.571), possibly indicating the added contribution of fluorescence. Nonetheless, the analytical approach is not ideal and is still lagging behind more sophisticated methods, such as the computerized Monte Carlo (MC) system. This data-intensive method is capable of simulating many more geometrical aspects of the DE. For example, MC such as GEANT4, MCNP5 and EGSnrc, can be programed to randomly sample the path of radiation-induced products, generate intricate geometries and model more complex interactions (backscattering of secondary electrons, Auger avalanches, succeeding Compton electrons/scattered photons interaction, plasmon excitations) [
73]. The analytical calculation here only considers a linear trajectory framework and assumes that all of the energy generated would deposit on the intended target. Such assumptions and simplifications were made to achieve the simplest method of predicting physical DE but may require further investigations with MC systems to cross-validate the findings.
Meanwhile, initial tests were conducted on the film dosimetry system to establish an optimized protocol for accurate calibration and DE measurements. The selected
parameter satisfied all of the fit tests and was found to produce an acceptable combined standard uncertainty (
= 1.0; <5.00% of radiotherapy accuracy) [
74]. Most film dosimetry studies have investigated the ability of high-Z contrast materials or metallic NPs to induce DE in both monochromatic and polychromatic low-energy photon beams (the majority of which use a single type of NPs) [
75,
76,
77,
78]. The presence of NP-loaded solution in the dosimeter’s environment resulted in an increase in absorbed dose. However, the under-coverage for deep-seated tumors by photons of this spectra limits their potential DE application. Any radiation dose delivered from this poorly penetrating beam would also have detrimental effects to the skin and skull if not properly filtered and tuned for intensity modality radiotherapy [
79]. Conversely, the photon modalities used in this study are supported by currently available clinical practice, but they seemed to be less in harmony with the sought-after photoelectric effect.
It was demonstrated in
Figure 6 that the
for water only and SPIONs were in fair agreement with
(from
Figure 5) across all photon beams. SPIONs, having a relatively low Z number, were expected not to produce substantial physical DE [
80], and most of the radiosensitization effects observed in biological samples during SPIONs-photon irradiation were dominated by the effects of ROS [
36,
81]. The
for AuNPs and BiONPs, on the other hand, were found not to be in agreement with the
(except for 6 MV photon). The
also showed no significant difference among the different radiotherapeutic techniques within the same NPs. These discrepancies can be attributed to the fact that the
considers all the energy generated from the NP–photon interaction would be deposited at the intended target, whereas the detection geometry limits the
. In our experimental setting, the target site is the active layer of EBT3 films. Based on the
and the statistical test, we concluded that no considerable
were able to be recorded by the active layer, and they are said to occur mainly in three parts:
The abundant, low-energy electrons, i.e., Auger electrons produced by NPs had nanometric trajectory range and were hypothesized to be physically absorbed or attenuated by the outer layer of matte polyester substrate;
Highly energetic electrons, i.e., photoelectrons generated by the NPs at the specific concentration, with trajectory range of >125 μm, were sparse;
The measured due to photoelectrons are lower than their associated uncertainty and statistically indistinguishable from the control experiment.
Regardless, the high amounts of ROS during AuNPs and BiONPs irradiation with photons were the primary cause of DE (low cell survival fractions), rather than the physical contributors [
1,
16,
25].
The focus on NP dose enhancers has been attributed mainly to their interaction with photon radiation. As the number of heavy-particle facilities [
82] is growing, so are the studies investigating DE by NPs during heavy-particle irradiations. Proton radiotherapy has a unique advantage over conventional photon radiotherapy because of the Bragg peak, which allows for excellent dose conformity to the tumor. In a pioneering study by Kim et al., the group demonstrated that AuNPs and SPIONs were capable of enhancing proton irradiation in vivo, as they observed a 75% to 90% mouse tumor volume reduction, following an intravenous NP injection with 41.7 MeV proton irradiation [
83]. Among the contributors suggested for prompting DE during proton irradiations are particle-induced X-ray emission (PIXE), particle-induced gamma-ray emission (PIGE), secondary electrons, nuclear activation products, i.e., ions (neutrons, electrons, protons, deuterons or tritons) and recoil nuclei—on top of chemical–biological responses, such as ROS [
83,
84].
Until now, there has only been a handful of research attempts to assess the physical DE by NP–proton interaction using film dosimetry [
84,
85]. The film measurements conducted in this study showed no measurable DE across all types of NPs under 150 MeV proton irradiation—which shares a similar conclusion with Cho et al. [
84]. In the same way as photons, protons are also capable of generating an avalanche of secondary electron emission from high-Z NPs through impact ionisation and ensuing Auger cascades [
6]. Nevertheless, MC simulations have shown that the secondary electrons produced from proton-irradiated NPs travel in a much shorter range than those generated from photon-irradiated NPs. The use of EBT3 RCFs may provide an adequate detection geometry for highly energetic PIXE/PIGE but most definitely not suitable for the weaker and shorter-ranged Auger electrons due to the thick polyester substrate. Surprisingly, Ahmad et al. managed to observe a substantial
(1.210 or 21.0% DE for 5.5 mg Au/mL; 1.260 or 26.0% DE for 1.1 mg Au/mL) using EBT3 films under proton irradiation [
85]. A plausible explanation for the high
might be due to the different proton beam energy (226 MeV), concentration and NP size (50.7 nm). Further validation studies with the same experimental setup are required to confirm the findings.
The increased cell deaths or tumor volume reduction shown in biological studies during proton irradiation were also mainly due to the enhanced ROS generation [
8], with the latest simulations suggesting the most considerable proton-induced ROS enhancement occurring over 50 nm from the NPs’ surface [
86]. Our in vitro model (with HCT116 human colon cancer cell) confirmed the idea, as the addition of iron-, gold-, bismuth oxide- and platinum-based NPs during proton irradiation was found to spike the amount of ROS [
17]. The proposed mechanisms of enhanced ROS generation in cells were attributed primarily to the emission of secondary electrons from NPs. Therefore, to comprehensively and accurately quantify DE effects exclusively from the physical standpoint, a combination of chemistry and physics modeling via MC with biological work is much needed.