An Experimental Method to Determine the Interstitial Splitting Forces and Thermal Load Input Induced by Self-Tapping and Self-Drilling Bone Screws: A Pilot Study
Abstract
:1. Introduction
2. Materials and Methods
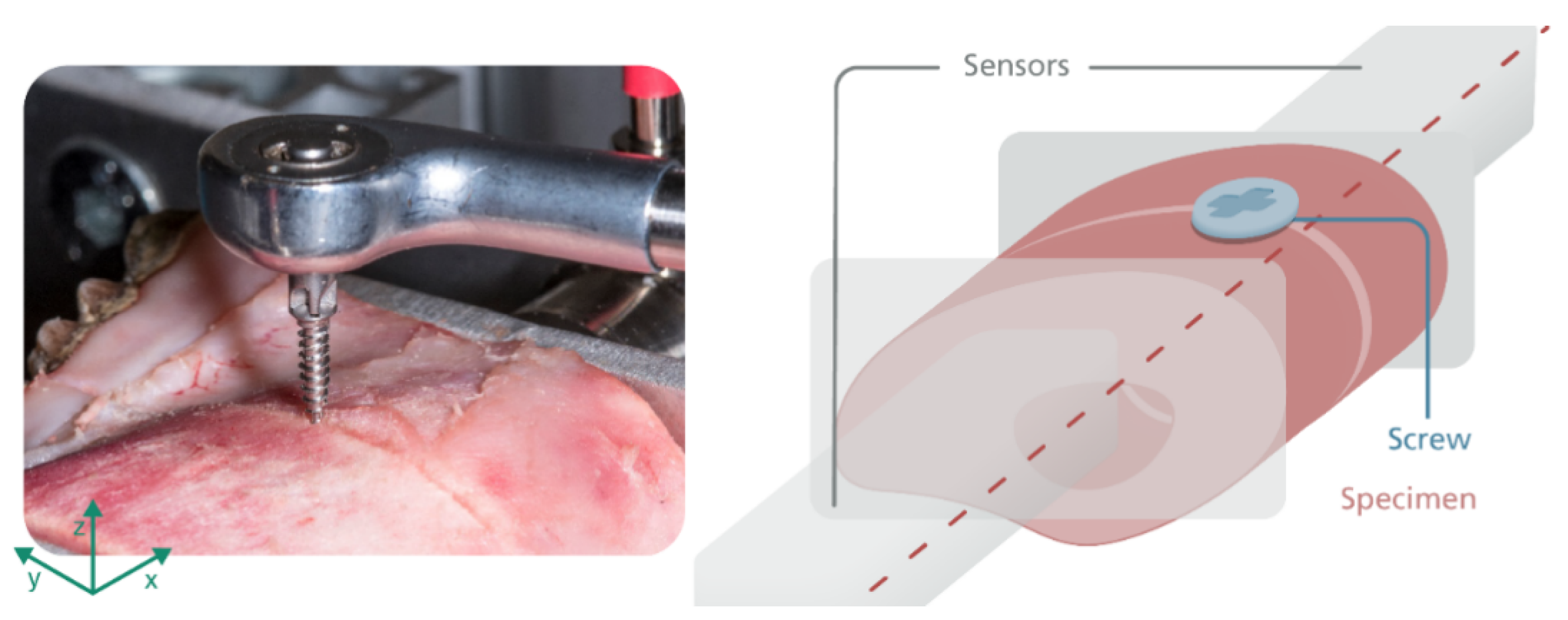
2.1. Investigation of the Splitting Force Induced by Osteosynthesis Screws
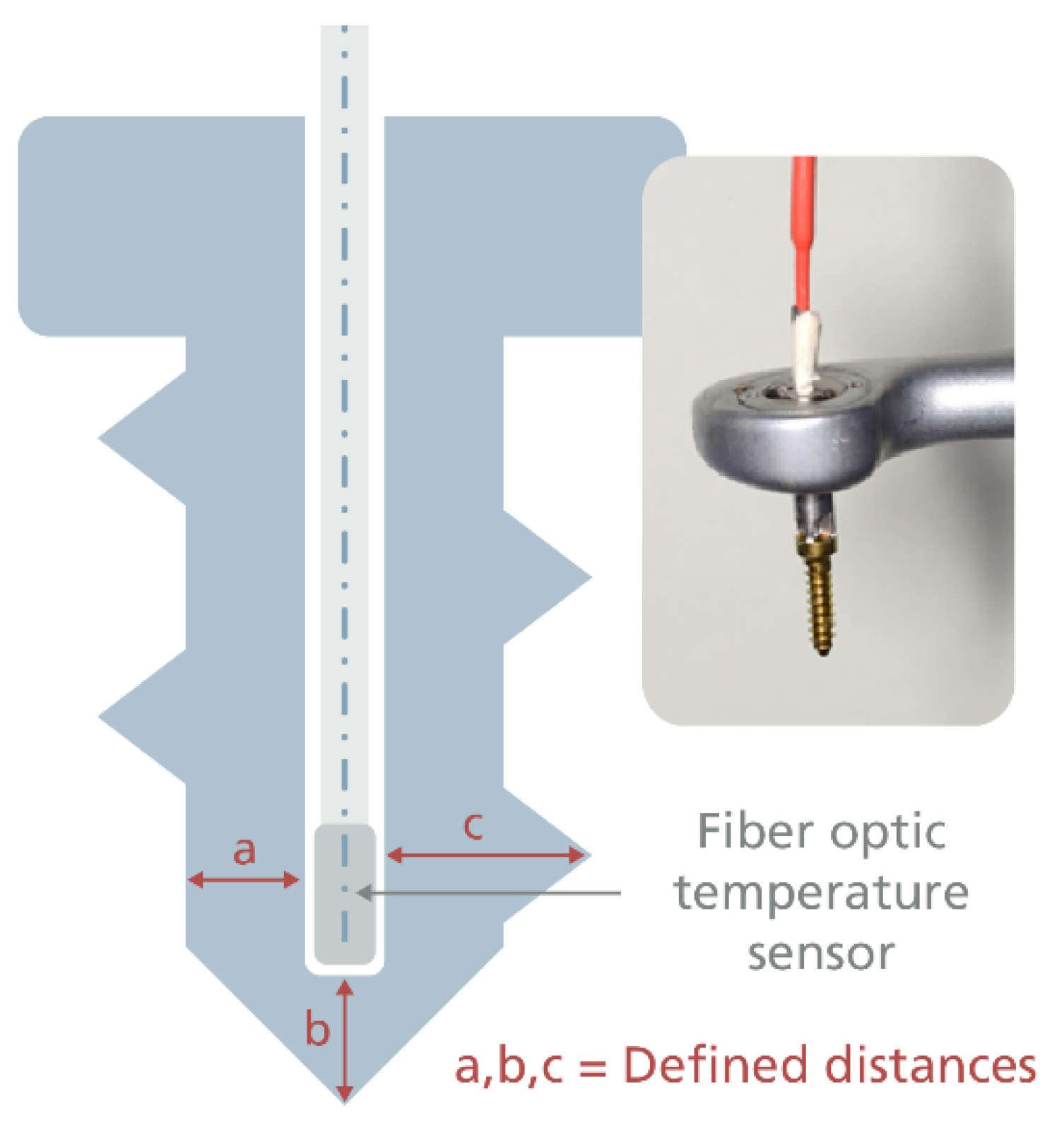
2.2. Temperature Measurement
3. Results
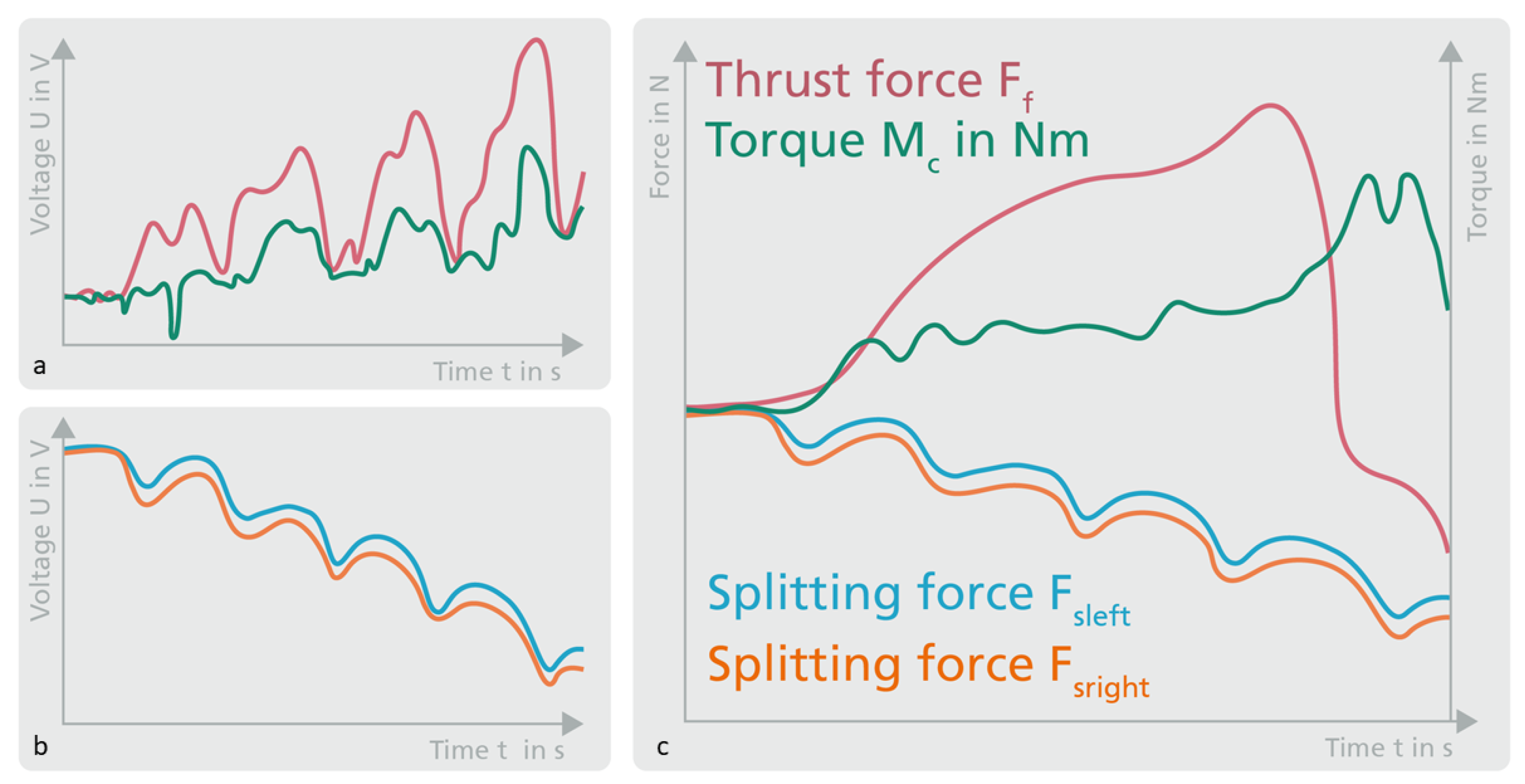
3.1. Splitting Force Measurement
3.2. Temperature Measurement
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Neumayer, B. Zeit-Drehmoment-Diagramme selbstschneidender Osteosyntheseschrauben unterschiedlicher Länge bei Abrißversuchen in Aluminium. Laryngo-Rhino-Otologie 1994, 73, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Baumgaertel, S.; Razavi, M.R.; Hans, M.G. Mini-implant anchorage for the orthodontic practitioner. Am. J. Orthod. Dentofac. Orthop. 2008, 133, 621–627. [Google Scholar] [CrossRef]
- Ellis, E.; McFadden, D.; Simon, P.; Throckmorton, G. Surgical complications with open treatment of mandibular condylar process fractures. J. Oral Maxillofac. Surg. 2000, 58, 950–958. [Google Scholar] [CrossRef] [PubMed]
- Schneider, M.; Lauer, G.; Eckelt, U. Surgical treatment of fractures of the mandibular condyle: A comparison of long-term results following different approaches—Functional, axiographical, and radiological findings. J. Cranio-Maxillofac. Surg. 2007, 35, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Möhlhenrich, S.C.; Modabber, A.; Steiner, T.; Mitchell, D.A.; Hölzle, F. Heat generation and drill wear during dental implant site preparation: Systematic review. Br. J. Oral Maxillofac. Surg. 2015, 53, 679–689. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Ruiz, R.A.; Calvo-Guirado, J.L.; Romanos, G.E. Effects of occlusal forces on the peri-implant-bone interface stability. Periodontology 2000 2019, 81, 179–193. [Google Scholar] [CrossRef] [PubMed]
- Noble, B.S.; Peet, N.; Stevens, H.Y.; Brabbs, A.; Mosley, J.R.; Reilly, G.C.; Reeve, J.; Skerry, T.M.; Lanyon, L.E. Mechanical loading: Biphasic osteocyte survival and targeting of osteoclasts for bone destruction in rat cortical bone. Am. J. Physiol. Physiol. 2003, 284, C934–C943. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verborgt, O.; Gibson, G.J.; Schaffler, M.B. Loss of Osteocyte Integrity in Association with Microdamage and Bone Remodeling After Fatigue In Vivo. J. Bone Miner. Res. 2000, 15, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Takano-Yamamoto, T. Osteocyte function under compressive mechanical force. Jpn. Dent. Sci. Rev. 2014, 50, 29–39. [Google Scholar] [CrossRef] [Green Version]
- Kobayashi, Y.; Hashimoto, F.; Miyamoto, H.; Kanaoka, K.; Miyazaki-Kawashita, Y.; Nakashima, T.; Shibata, M.; Kobayashi, K.; Kato, Y.; Sakai, H. Force-Induced Osteoclast Apoptosis In Vivo Is Accompanied by Elevation in Transforming Growth Factor β and Osteoprotegerin Expression. J. Bone Miner. Res. 2000, 15, 1924–1934. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Halldin, A.; Jimbo, R.; Johansson, C.B.; Wennerberg, A.; Jacobsson, M.; Albrektsson, T.; Hansson, S. Implant Stability and Bone Remodeling after 3 and 13 Days of Implantation with an Initial Static Strain. Clin. Implant Dent. Relat. Res. 2014, 16, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Ikar, M.; Grobecker-Karl, T.; Karl, M.; Steiner, C. Mechanical stress during implant surgery and its effects on marginal bone: A literature review. Quintessence Int. 2019, 51, 2–10. [Google Scholar] [CrossRef]
- Bashutski, J.D.; D’Silva, N.J.; Wang, H.-L. Implant Compression Necrosis: Current Understanding and Case Report. J. Periodontol. 2009, 80, 700–704. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ikumi, N.; Suzawa, T.; Yoshimura, K.; Kamijo, R. Bone Response to Static Compressive Stress at Bone-Implant Interface: A Pilot Study of Critical Static Compressive Stress. Int. J. Oral Maxillofac. Implant. 2015, 30, 827–833. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blaß, H.J.; Bejtka, I.; Uibel, T. Tragfähigkeit Von Verbindungen Mit Selbstbohrenden Holzschrauben Mit Vollgewinde. In Karlsruher Berichte zum Ingenieurholzbau; Band 4; Universitätsverlag Karlsruhe: Karlsruhe, Germany, 2006. [Google Scholar]
- Eriksson, A.; Albrektsson, T.; Grane, B.; McQueen, D. Thermal injury to bone. Int. J. Oral Surg. 1982, 11, 115–121. [Google Scholar] [CrossRef]
- Li, S.; Chien, S.; Brånemark, P.I. Heat shock-induced necrosis and apoptosis in osteoblasts. J. Orthop. Res. 1999, 17, 891–899. [Google Scholar] [CrossRef] [PubMed]
- Pellicer-Chover, H.; Peñarrocha-Oltra, D.; Aloy-Prosper, A.; Sanchis-Gonzalez, J.; Peñarrocha-Diago, M. Comparison of peri-implant bone loss between conventional drilling with irrigation versus low-speed drilling without irrigation. Med. Oral Patol. Oral Cir. Bucal 2017, 22, e730. [Google Scholar] [CrossRef]
- Ben Achour, A.; Petto, C.; Meißner, H.; Hipp, D.; Nestler, A.; Lauer, G.; Teicher, U. The Influence of Thrust Force on the Vitality of Bone Chips Harvested for Autologous Augmentation during Dental Implantation. Materials 2019, 12, 3695. [Google Scholar] [CrossRef] [Green Version]
- Oh, J.-H.; Fang, Y.; Jeong, S.-M.; Choi, B.-H. The effect of low-speed drilling without irrigation on heat generation: An experimental study. J. Korean Assoc. Oral Maxillofac. Surg. 2016, 42, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Löhr, J.; Gellrich, N.; Büscher, P.; Wahl, D.; Rahn, B.A. Vergleichende In-vitro-Untersuchungen von selbstbohrenden und selbstschneidenden Schrauben. Mund-Kiefer-und Gesichtschirurgie 2000, 4, 159–163. [Google Scholar] [CrossRef]
- Baumgart, F.W.; Cordey, J.; Morikawa, K.; Perren, S.; Rahn, B.; Schavan, R.; Snyder, S. AO/ASIF self-tapping screws (STS). Injury 1993, 24, 1–17. [Google Scholar] [CrossRef]
- Ghazali, M.; Roseiro, L.; Garruço, A.; Margalho, L.; Expedito, F. Pre-drilling vs. Self-drilling of Pin Bone Insertion—A Thermography Experimental Evaluation. In VipIMAGE 2017; Tavares, J., Natal Jorge, R., Eds.; Springer International Publishing: Cham, Switzerland, 2018; Volume 27, pp. 1063–1068. [Google Scholar] [CrossRef]
- Möhlhenrich, S.C.; Heussen, N.; Modabber, A.; Kniha, K.; Hölzle, F.; Wilmes, B.; Danesh, G.; Szalma, J. Influence of bone density, screw size and surgical procedure on orthodontic mini-implant placement—Part A: Temperature development. Int. J. Oral Maxillofac. Surg. 2021, 50, 555–564. [Google Scholar] [CrossRef] [PubMed]
- Matsuoka, M.; Motoyoshi, M.; Sakaguchi, M.; Shinohara, A.; Shigeede, T.; Saito, Y.; Matsuda, M.; Shimizu, N. Friction heat during self-drilling of an orthodontic miniscrew. Int. J. Oral Maxillofac. Surg. 2011, 40, 191–194. [Google Scholar] [CrossRef] [PubMed]
- Xu, W. Instrumentation and experiment design for in-vitro interface temperature measurement during the insertion of an orthopaedic implant. In Proceedings of the 10th International Conference on Control, Automation, Robotics and Vision, Hanoi, Vietnam, 1 April 2008; pp. 1773–1778. [Google Scholar] [CrossRef]
- Trullenque-Eriksson, A.; Guisado-Moya, B. Retrospective long-term evaluation of dental implants in totally and partially edentulous patients. Part I: Survival and marginal bone loss. Implant Dent. 2014, 23, 732–737. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.C.; Small, P.N.; Elian, N.; Tarnow, D. Screw loosening for standard and wide diameter implants in partially edentulous cases: 3- to 7-year longitudinal data. Implant Dent. 2004, 13, 245–250. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teicher, U.; Ben Achour, A.; Nestler, A.; Brosius, A.; Lauer, G. Process based analysis of manually controlled drilling processes for bone. AIP Conf. Proc. 2018, 1960, 070025. [Google Scholar] [CrossRef]
- Aerssens, J.; Boonen, S.; Lowet, G.; Dequeker, J. Interspecies differences in bone composition, density, and quality: Potential implications for in vivo bone research. Endocrinology 1998, 139, 663–670. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.; Upadhyay, M.; Liu, S.; Roberts, E.; Neace, W.P.; Nanda, R. Microdamage of the cortical bone during mini-implant insertion with self-drilling and self-tapping techniques: A randomized controlled trial. Am. J. Orthod. Dentofac. Orthop. 2012, 141, 538–546. [Google Scholar] [CrossRef]
- Wright, T.M.; Hayes, W.C. Fracture mechanics parameters for compact bone-Effects of density and specimen thickness. J. Biomech. 1977, 10, 419–430. [Google Scholar] [CrossRef]
- Zipprich, H.; Weigl, P.; König, E.; Toderas, A.; Balaban, Ü.; Ratka, C. Heat Generation at the Implant–Bone Interface by Insertion of Ceramic and Titanium Implants. J. Clin. Med. 2019, 8, 1541. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kronenberg, M. Machining Science and Application—Theory and Practice for Operation and Development of Machining Processes; Pergamon Press: Oxford, NY, USA, 1966; p. 53. [Google Scholar]
- Kauschinger, B.; Schroeder, S. Uncertain Parameters in Thermal Machine-Tool Models and Methods to Design their Metrological Adjustment Process. Appl. Mech. Mater. 2015, 794, 379–386. [Google Scholar] [CrossRef]
- Robles-Linares, J.A.; Axinte, D.; Liao, Z.; Gameros, A. Machining-induced thermal damage in cortical bone: Necrosis and micro-mechanical integrity. Mater. Des. 2021, 197, 109215. [Google Scholar] [CrossRef]






| Saw Blade | ||
|---|---|---|
| Specification | Value | Units |
| Tool material | 12C27® (equivalent to 1.4034/1.4037) | |
| Tool length | 1065 | mm |
| Number of teeth zsaw | 24 | |
| Tooth width b | 0.4 | mm |
| Cutting speed vc_saw | 44 | m/min |
| Feed rate vf_saw | Manual | |
| Angled screw instrument | ||
| Gear ratio | 13:1 | |
| Intra coupling | DIN 1390/ISO-DIS 3964 | |
| Length Ltool | 19 | mm |
| Angle of attack | 90 | ° |
| Screw blade | Cross shape/cruciform | Anton Hipp GmbH |
| Diameter pilot drill ddrill | 1.5 | mm |
| Rotational speed n | 100 | rpm |
| Screws | ||
| Core diameter (max.) dcore | 1.35 | mm |
| Outer diameter douter | 2 | mm |
| Total Screw length Lscrew | 9 | mm |
| Pitch P | 0.75 | mm per round |
| Screw tip shape | Chamfer | Self-tapping (STS) |
| Sharp | Self-drilling (SDS) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ben Achour, A.; Petto, C.; Meißner, H.; Mostofa, A.; Teicher, U.; Haim, D.; Ihlenfeldt, S.; Lauer, G. An Experimental Method to Determine the Interstitial Splitting Forces and Thermal Load Input Induced by Self-Tapping and Self-Drilling Bone Screws: A Pilot Study. Biomechanics 2021, 1, 239-252. https://doi.org/10.3390/biomechanics1020020
Ben Achour A, Petto C, Meißner H, Mostofa A, Teicher U, Haim D, Ihlenfeldt S, Lauer G. An Experimental Method to Determine the Interstitial Splitting Forces and Thermal Load Input Induced by Self-Tapping and Self-Drilling Bone Screws: A Pilot Study. Biomechanics. 2021; 1(2):239-252. https://doi.org/10.3390/biomechanics1020020
Chicago/Turabian StyleBen Achour, Anas, Carola Petto, Heike Meißner, Anita Mostofa, Uwe Teicher, Dominik Haim, Steffen Ihlenfeldt, and Günter Lauer. 2021. "An Experimental Method to Determine the Interstitial Splitting Forces and Thermal Load Input Induced by Self-Tapping and Self-Drilling Bone Screws: A Pilot Study" Biomechanics 1, no. 2: 239-252. https://doi.org/10.3390/biomechanics1020020
APA StyleBen Achour, A., Petto, C., Meißner, H., Mostofa, A., Teicher, U., Haim, D., Ihlenfeldt, S., & Lauer, G. (2021). An Experimental Method to Determine the Interstitial Splitting Forces and Thermal Load Input Induced by Self-Tapping and Self-Drilling Bone Screws: A Pilot Study. Biomechanics, 1(2), 239-252. https://doi.org/10.3390/biomechanics1020020






