Administration of Polyphenol-Rich Sugarcane Extract Alleviates Deficits Induced by Amyloid-Beta1–42 (Aβ1–42) in Transgenic C. elegans
Abstract
1. Introduction
2. Materials and Methods
2.1. Polyphenol-Rich Sugarcane Extract and Treatment
2.2. Culture Conditions for Escherichia coli OP50 and Maintenance of C. elegans
2.3. Lifespan Assay
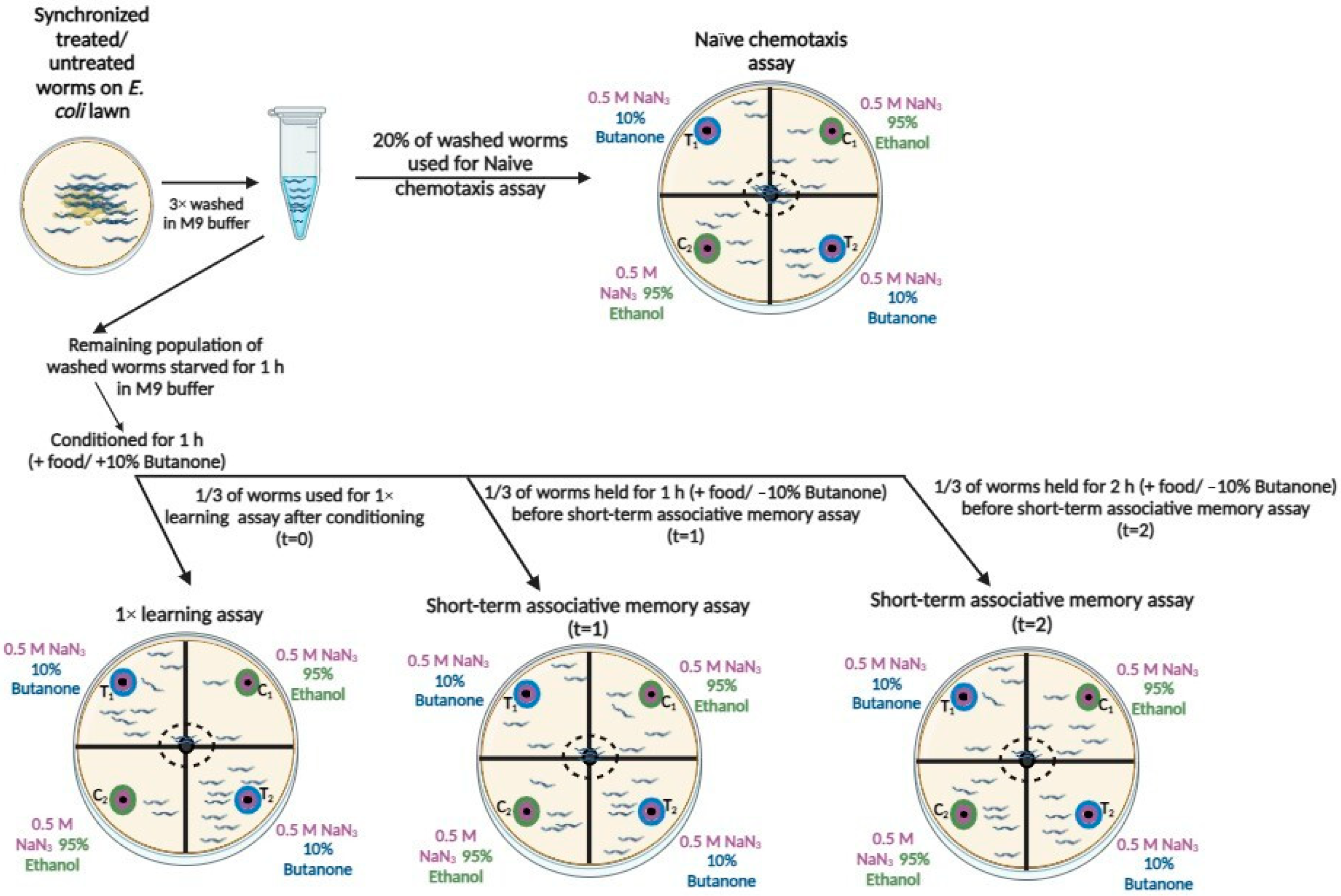
2.4. Chemotaxis Behavior, Learning, and Short-Term Associative Memory
2.5. Quantitative Detection of Aβ Fibrils Using Thioflavin-T Staining
2.6. Statistical Analysis
3. Results
3.1. PRSE Supplementation Restores Lifespan in Pan-Neuronal Aβ1–42 Expressing C. elegans
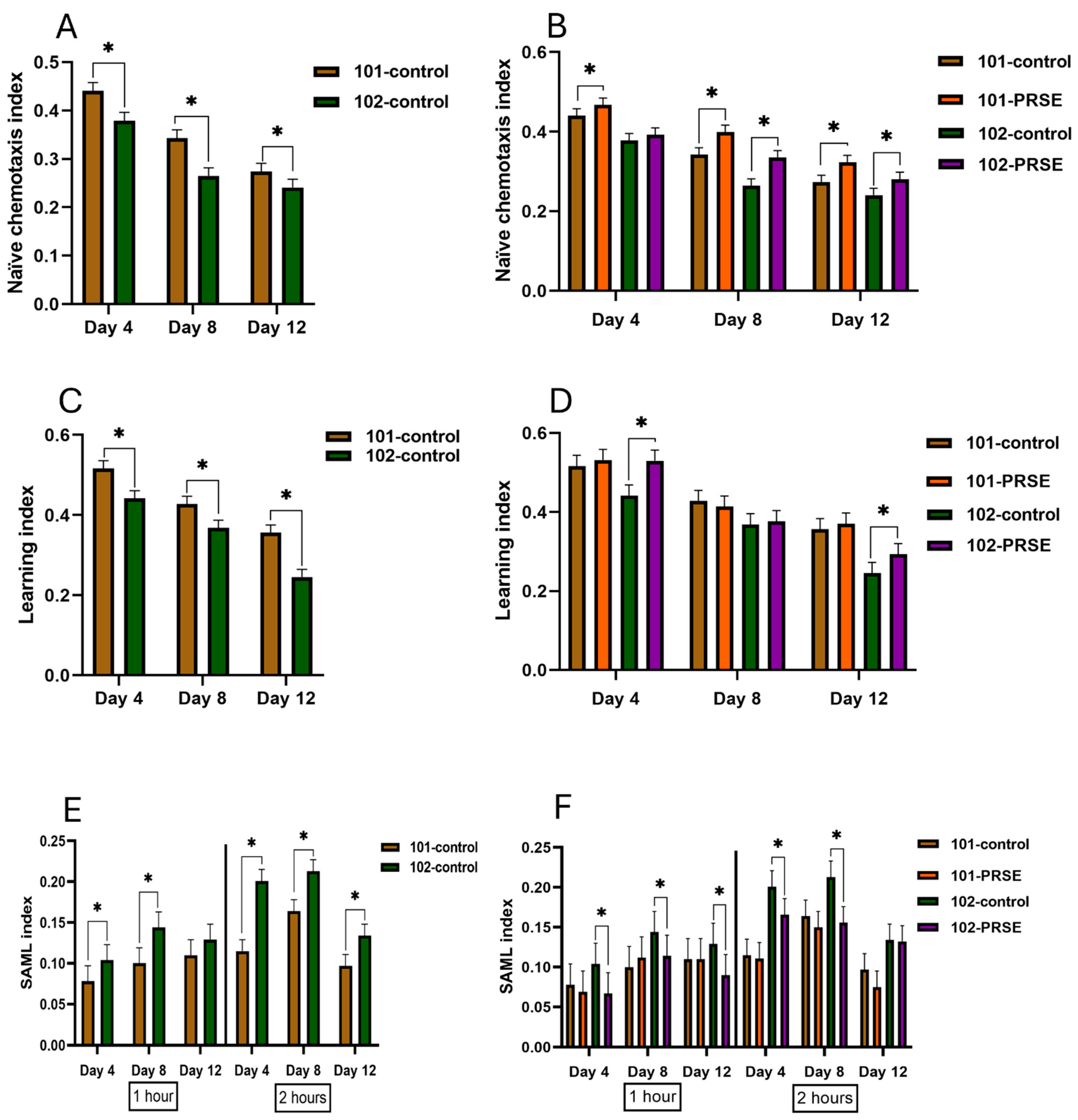
3.2. PRSE Supplement Intervention Improved Chemosensation, Learning, and Memory Loss of C. elegans
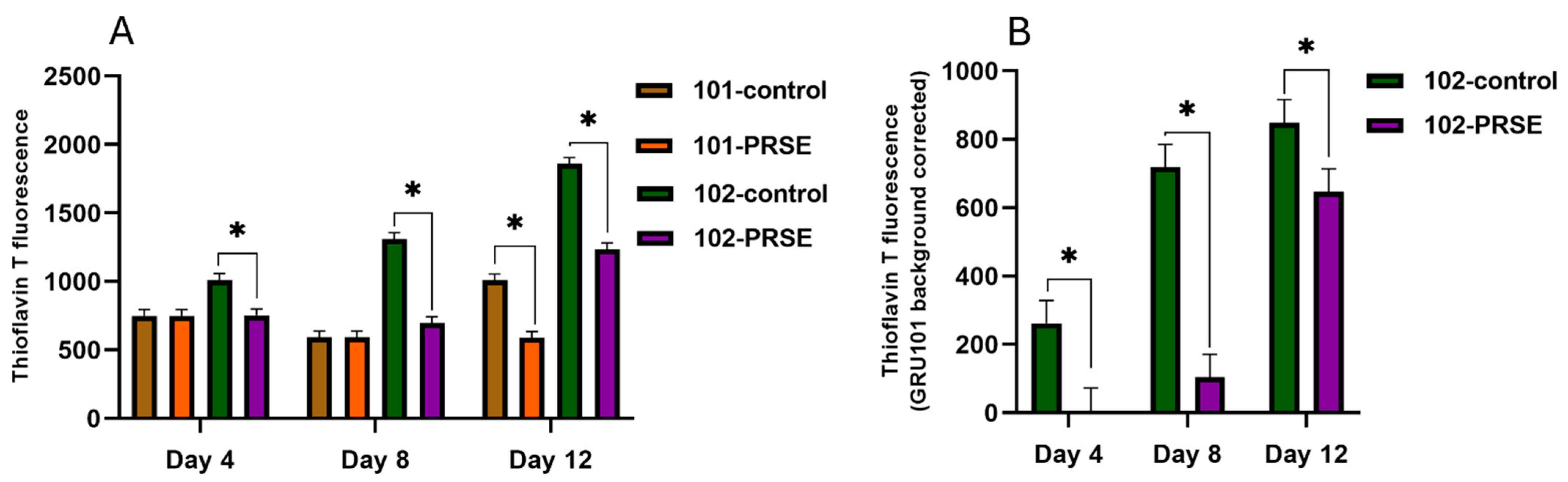
3.3. PRSE Supplementation Decreased Aβ Fibril Accumulation in Pan-Neuronal Aβ1–42-Expressing Worms During the Young, Middle, and Old-Aged Stages
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Butterfield, D.A.; Drake, J.; Pocernich, C.; Castegna, A. Evidence of Oxidative Damage in Alzheimer’s Disease Brain: Central Role for Amyloid β-Peptide. Trends Mol. Med. 2001, 7, 548–554. [Google Scholar] [CrossRef]
- Reddy, P.H.; Beal, M.F. Amyloid Beta, Mitochondrial Dysfunction and Synaptic Damage: Implications for Cognitive Decline in Aging and Alzheimer’s Disease. Trends Mol. Med. 2008, 14, 45–53. [Google Scholar] [CrossRef]
- Perez Ortiz, J.M.; Swerdlow, R.H. Mitochondrial Dysfunction in Alzheimer’s Disease: Role in Pathogenesis and Novel Therapeutic Opportunities. Br. J. Pharmacol. 2019, 176, 3489–3507. [Google Scholar] [CrossRef]
- Monsalvo-Maraver, L.Á.; Maya-López, M.; Rangel-López, E.; Túnez, I.; Tinkov, A.A.; Skalny, A.; Ferrer, B.; Aschner, M.; Santamaría, A. Amyloid Beta Peptide-Mediated Alterations in Mitochondrial Dynamicsand Its Implications for Alzheimer’s Disease. CNS Neurol. Disord. Drug Targets 2023, 22, 1039–1056. [Google Scholar] [CrossRef]
- Bhatia, V.; Sharma, S. Role of Mitochondrial Dysfunction, Oxidative Stress and Autophagy in Progression of Alzheimer’s Disease. J. Neurol. Sci. 2021, 421, 117253. [Google Scholar] [CrossRef]
- Varadarajan, S.; Yatin, S.; Aksenova, M.; Butterfield, D.A. Review: Alzheimer’s Amyloid β-Peptide-Associated Free Radical Oxidative Stress and Neurotoxicity. J. Struct. Biol. 2000, 130, 184–208. [Google Scholar] [CrossRef]
- Babaei Abraki, S.; Chavoshi-Nezhad, S. Mitochondrial Defects and Oxidative Stress in Alzheimer Disease. Neurosci. J. Shefaye Khatam 2014, 2, 85–94. [Google Scholar] [CrossRef]
- Scarmeas, N.; Stern, Y.; Tang, M.; Mayeux, R.; Luchsinger, J.A. Mediterranean Diet and Risk for Alzheimer’s Disease. Ann. Neurol. 2006, 59, 912–921. [Google Scholar] [CrossRef]
- Singh, B.; Parsaik, A.K.; Mielke, M.M.; Erwin, P.J.; Knopman, D.S.; Petersen, R.C.; Roberts, R.O. Association of Mediterranean Diet with Mild Cognitive Impairment and Alzheimer’s Disease: A Systematic Review and Meta-Analysis. J. Alzheimer’s Dis. 2014, 39, 271–282. [Google Scholar] [CrossRef]
- Munasinghe, M.; Almotayri, A.; Kolivas, D.; Thomas, J.; Heydarian, D.; Jois, M. Cocoa Supplementation Reduces Amyloid-Beta1–42 (Aβ1–42) Induced Deficits in a Transgenic C. elegans. Nutr. Healthy Aging 2021, 6, 117–130. [Google Scholar] [CrossRef]
- Heydarian, D.; Flavel, M.; Munasinghe, M.; Almotayri, A.; Jois, M.; Thomas, J. Early Exposure to Polyphenol-Rich Sugarcane Extract (PRSE) Mitigates Aging While Enhancing Thermotolerance in C. elegans. J. Ageing Longev. 2024, 4, 15–27. [Google Scholar] [CrossRef]
- Heydarian, D.; Flavel, M.; Munasinghe, M.; Jois, M.; Thomas, J. Improving Cognitive and Chemosensory Function in Caenorhabditis Elegans Through Polyphenol-Rich Sugarcane Extract. Stresses 2024, 4, 816–826. [Google Scholar] [CrossRef]
- Malito, E.; Hulse, R.E.; Tang, W.-J. Amyloid β-Degrading Cryptidases: Insulin Degrading Enzyme, Presequence Peptidase, and Neprilysin. Cell. Mol. Life Sci. 2008, 65, 2574–2585. [Google Scholar] [CrossRef]
- Deseo, M.A.; Elkins, A.; Rochfort, S.; Kitchen, B. Antioxidant Activity and Polyphenol Composition of Sugarcane Molasses Extract. Food Chem. 2020, 314, 126180. [Google Scholar] [CrossRef]
- Kauffman, A.; Parsons, L.; Stein, G.; Wills, A.; Kaletsky, R.; Murphy, C. C. elegans Positive Butanone Learning, Short-Term, and Long-Term Associative Memory Assays. J. Vis. Exp. 2011, 49, 2490. [Google Scholar] [CrossRef]
- Margie, O.; Palmer, C.; Chin-Sang, I. C. elegans Chemotaxis Assay. J. Vis. Exp. 2013, 74, 50069. [Google Scholar] [CrossRef]
- Xin, L.; Yamujala, R.; Wang, Y.; Wang, H.; Wu, W.-H.; Lawton, M.A.; Long, C.; Di, R. Acetylcholineestarase-Inhibiting Alkaloids from Lycoris Radiata Delay Paralysis of Amyloid Beta-Expressing Transgenic C. elegans CL4176. PLoS ONE 2013, 8, e63874. [Google Scholar] [CrossRef]
- Ji, J.; Yang, X.; Flavel, M.; Shields, Z.P.-I.; Kitchen, B. Antioxidant and Anti-Diabetic Functions of a Polyphenol-Rich Sugarcane Extract. J. Am. Coll. Nutr. 2019, 38, 670–680. [Google Scholar] [CrossRef]
- Hewawansa, U.H.A.J.; Houghton, M.J.; Barber, E.; Costa, R.J.S.; Kitchen, B.; Williamson, G. Flavonoids and Phenolic Acids from Sugarcane: Distribution in the Plant, Changes during Processing, and Potential Benefits to Industry and Health. Compr. Rev. Food Sci. Food Saf. 2024, 23, e13307. [Google Scholar] [CrossRef]
- Singla, R.K.; Dubey, A.K.; Garg, A.; Sharma, R.K.; Fiorino, M.; Ameen, S.M.; Haddad, M.A.; Al-Hiary, M. Natural Polyphenols: Chemical Classification, Definition of Classes, Subcategories, and Structures. J. AOAC Int. 2019, 102, 1397–1400. [Google Scholar] [CrossRef]
- Liu, X.; Le Bourvellec, C.; Guyot, S.; Renard, C.M.G.C. Reactivity of Flavanols: Their Fate in Physical Food Processing and Recent Advances in Their Analysis by Depolymerization. Compr. Rev. Food Sci. Food Saf. 2021, 20, 4841–4880. [Google Scholar] [CrossRef]
- Di Lorenzo, C.; Colombo, F.; Biella, S.; Stockley, C.; Restani, P. Polyphenols and Human Health: The Role of Bioavailability. Nutrients 2021, 13, 273. [Google Scholar] [CrossRef]
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Rémésy, C. Bioavailability and Bioefficacy of Polyphenols in Humans. I. Review of 97 Bioavailability Studies. Am. J. Clin. Nutr. 2005, 81, 230S–242S. [Google Scholar] [CrossRef]
- Olasehinde, T.A.; Olaokun, O.O. The Beneficial Role of Apigenin against Cognitive and Neurobehavioural Dysfunction: A Systematic Review of Preclinical Investigations. Biomedicines 2024, 12, 178. [Google Scholar] [CrossRef]
- Balez, R.; Steiner, N.; Engel, M.; Muñoz, S.S.; Lum, J.S.; Wu, Y.; Wang, D.; Vallotton, P.; Sachdev, P.; O’Connor, M.; et al. Neuroprotective Effects of Apigenin against Inflammation, Neuronal Excitability and Apoptosis in an Induced Pluripotent Stem Cell Model of Alzheimer’s Disease. Sci. Rep. 2016, 6, 31450. [Google Scholar] [CrossRef]
- Nabavi, S.F.; Braidy, N.; Gortzi, O.; Sobarzo-Sanchez, E.; Daglia, M.; Skalicka-Woźniak, K.; Nabavi, S.M. Luteolin as an Anti-Inflammatory and Neuroprotective Agent: A Brief Review. Brain Res. Bull. 2015, 119, 1–11. [Google Scholar] [CrossRef]
- Kempuraj, D.; Thangavel, R.; Kempuraj, D.D.; Ahmed, M.E.; Selvakumar, G.P.; Raikwar, S.P.; Zaheer, S.A.; Iyer, S.S.; Govindarajan, R.; Chandrasekaran, P.N.; et al. Neuroprotective Effects of Flavone Luteolin in Neuroinflammation and Neurotrauma. BioFactors 2021, 47, 190–197. [Google Scholar] [CrossRef]
- Singh, N.K.; Bhushan, B.; Singh, P.; Sahu, K.K. Therapeutic Expedition of Luteolin against Brain-Related Disorders:An Updated Review. Comb. Chem. High Throughput Screen. 2024, 28, 371–391. [Google Scholar] [CrossRef]
- Goyal, A.; Solanki, K.; Verma, A. Luteolin: Nature’s Promising Warrior against Alzheimer’s and Parkinson’s Disease. J. Biochem. Mol. Toxicol. 2024, 38, e23619. [Google Scholar] [CrossRef]
- Dilberger, B.; Passon, M.; Asseburg, H.; Silaidos, C.V.; Schmitt, F.; Schmiedl, T.; Schieber, A.; Eckert, G.P. Polyphenols and Metabolites Enhance Survival in Rodents and Nematodes—Impact of Mitochondria. Nutrients 2019, 11, 1886. [Google Scholar] [CrossRef]
- Das, S.; Stark, L.; Musgrave, I.F.; Pukala, T.; Smid, S.D. Bioactive Polyphenol Interactions with β Amyloid: A Comparison of Binding Modelling, Effects on Fibril and Aggregate Formation and Neuroprotective Capacity. Food Funct. 2016, 7, 1138–1146. [Google Scholar] [CrossRef]
- Baruah, P.; Moorthy, H.; Ramesh, M.; Padhi, D.; Govindaraju, T. A Natural Polyphenol Activates and Enhances GPX4 to Mitigate Amyloid-β Induced Ferroptosis in Alzheimer’s Disease. Chem. Sci. 2023, 14, 9427–9438. [Google Scholar] [CrossRef]
- Vauzour, D.; Vafeiadou, K.; Rodriguez-Mateos, A.; Rendeiro, C.; Spencer, J.P.E. The Neuroprotective Potential of Flavonoids: A Multiplicity of Effects. Genes Nutr. 2008, 3, 115–126. [Google Scholar] [CrossRef]
- Fong, S.; Teo, E.; Ng, L.F.; Chen, C.-B.; Lakshmanan, L.N.; Tsoi, S.Y.; Moore, P.K.; Inoue, T.; Halliwell, B.; Gruber, J. Energy Crisis Precedes Global Metabolic Failure in a Novel Caenorhabditis Elegans Alzheimer Disease Model. Sci. Rep. 2016, 6, 33781. [Google Scholar] [CrossRef]
- Kang, N.; Luan, Y.; Jiang, Y.; Cheng, W.; Liu, Y.; Su, Z.; Liu, Y.; Tan, P. Neuroprotective Effects of Oligosaccharides in Rehmanniae Radix on Transgenic Caenorhabditis Elegans Models for Alzheimer’s Disease. Front. Pharmacol. 2022, 13, 878631. [Google Scholar] [CrossRef]
- Weng, Y.; Zhou, S.; Morillo, K.; Kaletsky, R.; Lin, S.; Murphy, C.T. The Neuron-Specific IIS/FOXO Transcriptome in Aged Animals Reveals Regulatory Mechanisms of Cognitive Aging. eLife 2024, 13, RP95621. [Google Scholar] [CrossRef]
- Mastrandreas, P.; Arnold, A.; Boglari, C.; De Quervain, D.J.-F.; Stetak, A.; Papassotiropoulos, A. (−)-Gossypol Inhibition of Musashi-Mediated Forgetting Improves Memory and Age-Dependent Memory Decline in Caenorhabditis Elegans. Mol. Neurobiol. 2023, 60, 820–835. [Google Scholar] [CrossRef]
- Kauffman, A.L.; Ashraf, J.M.; Corces-Zimmerman, M.R.; Landis, J.N.; Murphy, C.T. Insulin Signaling and Dietary Restriction Differentially Influence the Decline of Learning and Memory with Age. PLoS Biol. 2010, 8, e1000372. [Google Scholar] [CrossRef]
- Farr, S.A.; Price, T.O.; Dominguez, L.J.; Motisi, A.; Saiano, F.; Niehoff, M.L.; Morley, J.E.; Banks, W.A.; Ercal, N.; Barbagallo, M. Extra Virgin Olive Oil Improves Learning and Memory in SAMP8 Mice. J. Alzheimer’s Dis. 2012, 28, 81–92. [Google Scholar] [CrossRef]
- Liu, P.; Kemper, L.J.; Wang, J.; Zahs, K.R.; Ashe, K.H.; Pasinetti, G.M. Grape Seed Polyphenolic Extract Specifically Decreases Aβ*56 in the Brains of Tg2576 Mice. J. Alzheimer’s Dis. 2011, 26, 657–666. [Google Scholar] [CrossRef]
- Wang, G.; Chen, L.; Pan, X.; Chen, J.; Wang, L.; Wang, W.; Cheng, R.; Wu, F.; Feng, X.; Yu, Y.; et al. The Effect of Resveratrol on Beta Amyloid-Induced Memory Impairment Involves Inhibition of Phosphodiesterase-4 Related Signaling. Oncotarget 2016, 7, 17380–17392. [Google Scholar] [CrossRef] [PubMed]
- Rege, S.; Geetha, T.; Broderick, T.; Babu, J. Resveratrol Protects β Amyloid-Induced Oxidative Damage and Memory Associated Proteins in H19-7 Hippocampal Neuronal Cells. Curr. Alzheimer Res. 2015, 12, 147–156. [Google Scholar] [CrossRef]
- Choi, Y.-T.; Jung, C.-H.; Lee, S.-R.; Bae, J.-H.; Baek, W.-K.; Suh, M.-H.; Park, J.; Park, C.-W.; Suh, S.-I. The Green Tea Polyphenol (−)-Epigallocatechin Gallate Attenuates β-Amyloid-Induced Neurotoxicity in Cultured Hippocampal Neurons. Life Sci. 2001, 70, 603–614. [Google Scholar] [CrossRef]
- Xue, W.; Lei, Z.; Liu, B.; Guo, H.; Yan, W.; Jin, Y.N.; Yu, Y.V. Olfactory Dysfunction as an Early Pathogenic Indicator in C. elegans Models of Alzheimer’s and Polyglutamine Diseases. Front. Aging Neurosci. 2024, 16, 1462238. [Google Scholar] [CrossRef]
- Machino, K.; Link, C.D.; Wang, S.; Murakami, H.; Murakami, S. A Semi-Automated Motion-Tracking Analysis of Locomotion Speed in the C. elegans Transgenics Overexpressing Beta-Amyloid in Neurons. Front. Genet. 2014, 5, 202. [Google Scholar] [CrossRef]
- Coburn, C.; Gems, D. The Mysterious Case of the C. elegans Gut Granule: Death Fluorescence, Anthranilic Acid and the Kynurenine Pathway. Front. Genet. 2013, 4, 151. [Google Scholar] [CrossRef]
- Arad, E.; Green, H.; Jelinek, R.; Rapaport, H. Revisiting Thioflavin T (ThT) Fluorescence as a Marker of Protein Fibrillation—The Prominent Role of Electrostatic Interactions. J. Colloid Interface Sci. 2020, 573, 87–95. [Google Scholar] [CrossRef]
- Komatsu, H.; Meurice, C.; Grasso, G.; Lippert, L.G.; Goldman, Y.E.; Axelsen, P.H. Variable Binding of Thioflavin T by Amyloid Fibrils. Biophys. J. 2017, 112, 198a. [Google Scholar] [CrossRef]
- Oba, R.; Ujike, N.; Ono, Y.; Okano, T.; Murakami, T. Label-Free Autofluorescence and Hyperspectral Imaging of Cerebral Amyloid-β Lesions in Aged Squirrel Monkeys. J. Vet. Diagn. Investig. 2024, 36, 41–45. [Google Scholar] [CrossRef]

| Treatment | Mean Lifespan (Days) | % Extension Compared to Control | Median Lifespan (Days) | % Extension Compared to Control | Maximum Lifespan (Days) |
|---|---|---|---|---|---|
| 101-control | 17.60 ± 0.33 | 18.00 ± 0.41 | 20.97 ± 0.20 | ||
| 102-control | 15.95 ± 0.43 a | 17.00 ± 0.60 | 19.90 ± 0.23 a | ||
| 101-PRSE | 18.18 ± 0.35 | 18.00 ± 0.50 | 21.37 ± 0.18 | ||
| 102-PRSE | 17.78 ± 0.36 b | 11 | 19.00 ± 0.52 ab | 6 a, 12 b | 21.60 ± 0.93 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heydarian, D.; Flavel, M.; Munasinghe, M.; Jois, M.; Thomas, J. Administration of Polyphenol-Rich Sugarcane Extract Alleviates Deficits Induced by Amyloid-Beta1–42 (Aβ1–42) in Transgenic C. elegans. J. Ageing Longev. 2025, 5, 12. https://doi.org/10.3390/jal5020012
Heydarian D, Flavel M, Munasinghe M, Jois M, Thomas J. Administration of Polyphenol-Rich Sugarcane Extract Alleviates Deficits Induced by Amyloid-Beta1–42 (Aβ1–42) in Transgenic C. elegans. Journal of Ageing and Longevity. 2025; 5(2):12. https://doi.org/10.3390/jal5020012
Chicago/Turabian StyleHeydarian, Deniz, Matthew Flavel, Mihiri Munasinghe, Markandeya Jois, and Jency Thomas. 2025. "Administration of Polyphenol-Rich Sugarcane Extract Alleviates Deficits Induced by Amyloid-Beta1–42 (Aβ1–42) in Transgenic C. elegans" Journal of Ageing and Longevity 5, no. 2: 12. https://doi.org/10.3390/jal5020012
APA StyleHeydarian, D., Flavel, M., Munasinghe, M., Jois, M., & Thomas, J. (2025). Administration of Polyphenol-Rich Sugarcane Extract Alleviates Deficits Induced by Amyloid-Beta1–42 (Aβ1–42) in Transgenic C. elegans. Journal of Ageing and Longevity, 5(2), 12. https://doi.org/10.3390/jal5020012