Strain Engineering of ZrO2@TiO2 Core@shell Nanoparticle Photocatalysts
Abstract
1. Introduction
2. Materials and Methods
3. Results
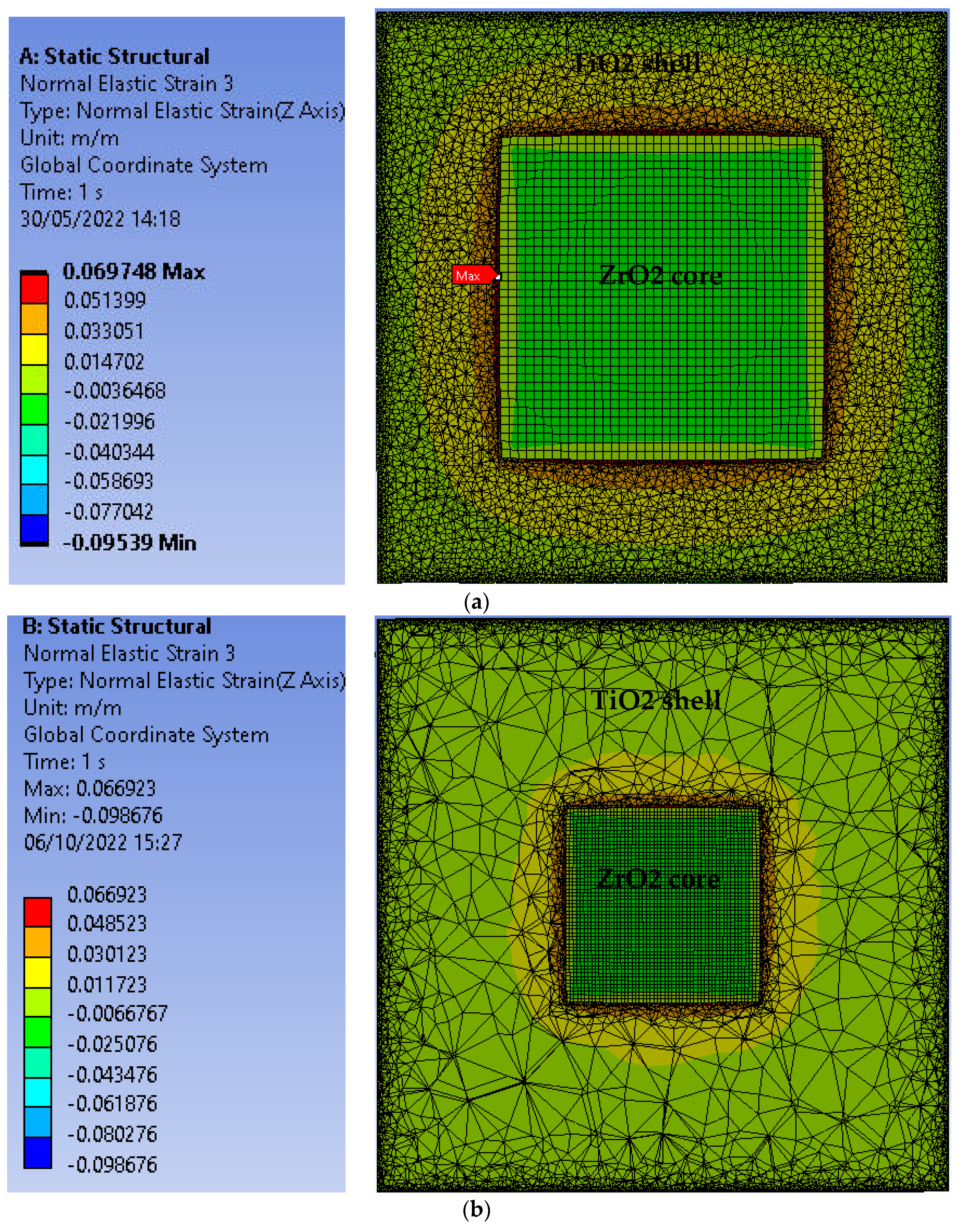
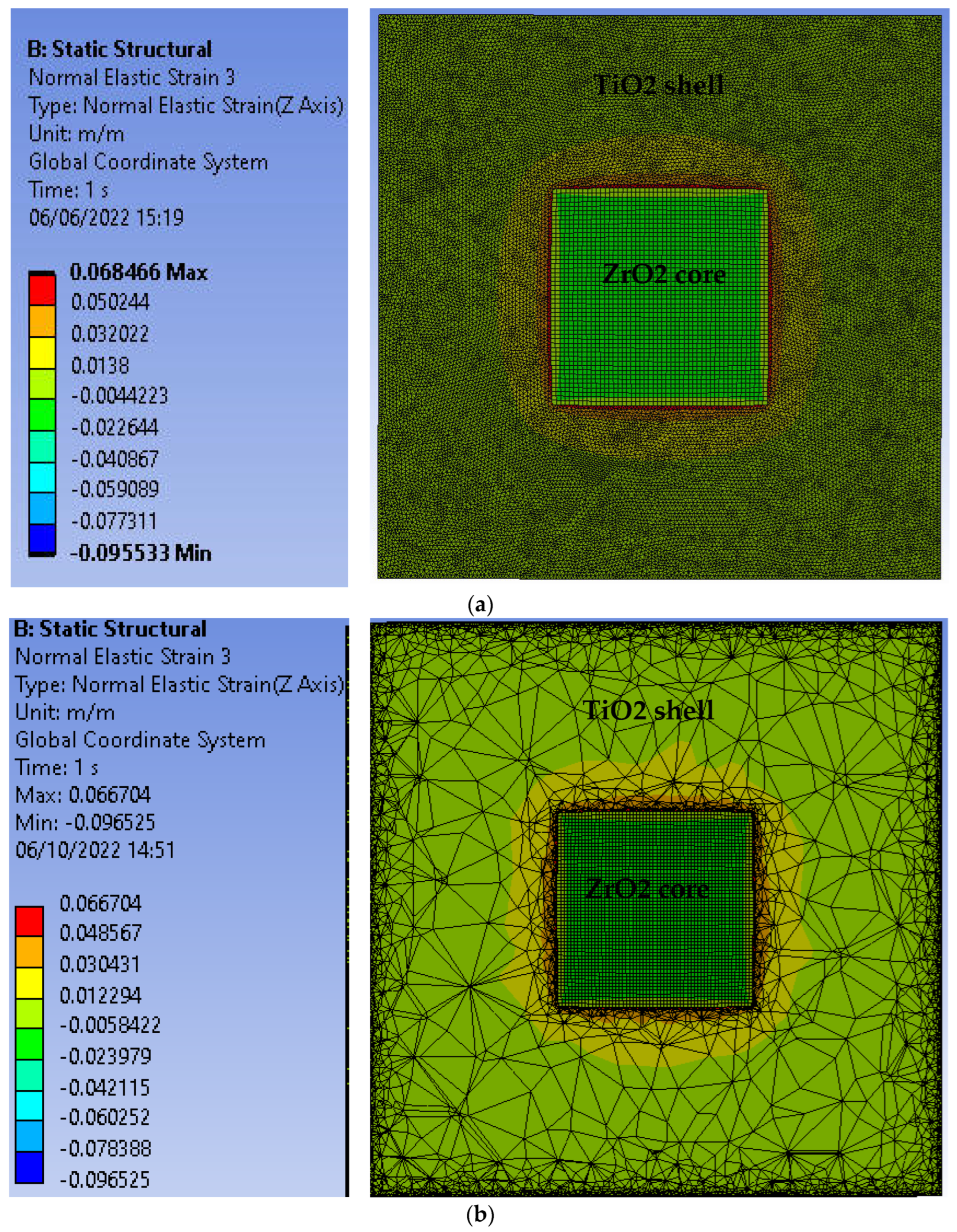
3.1. Strain in Core@shell Nanparticles
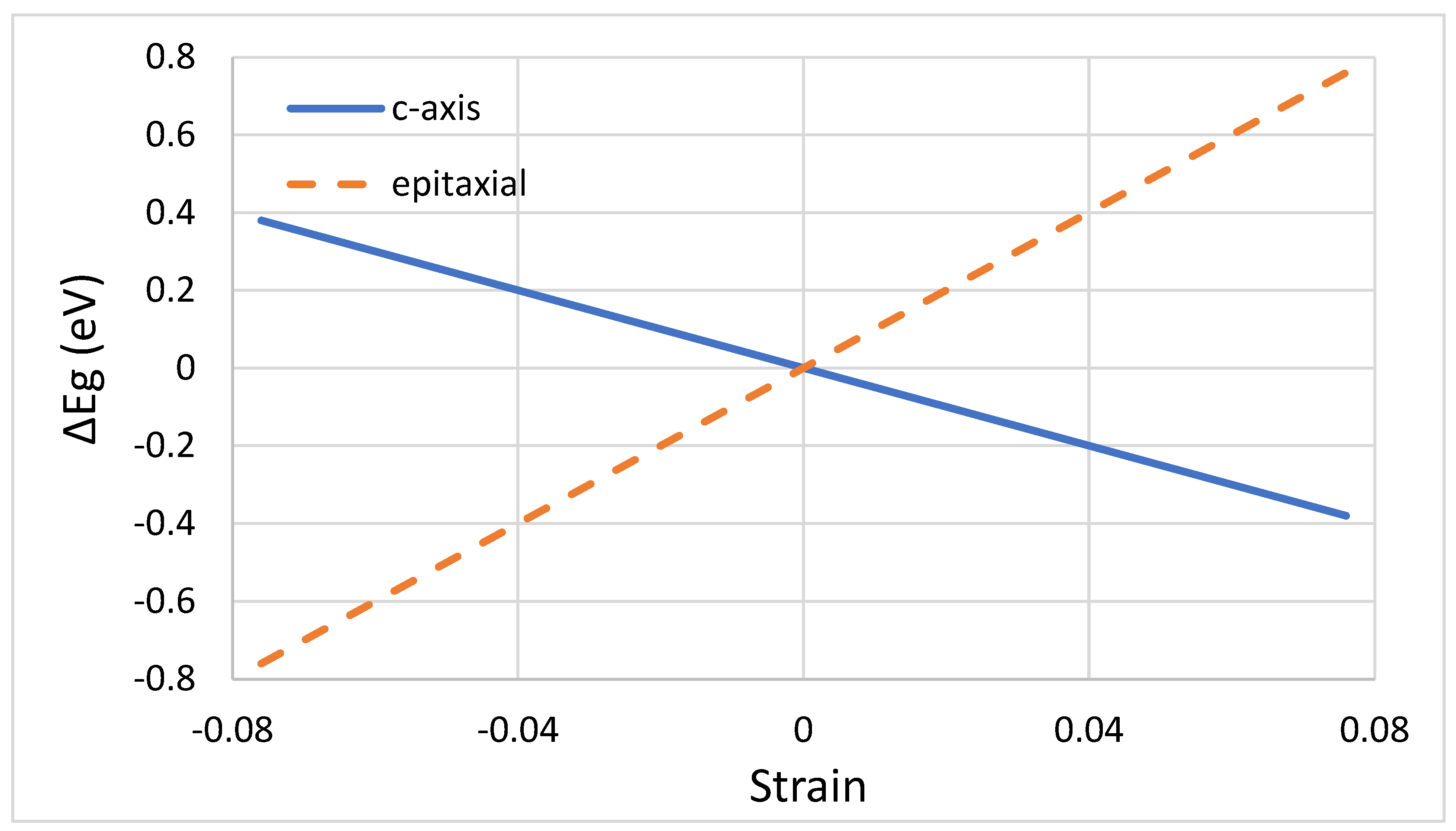
3.2. Electron–Hole Pair Production
3.3. Core@shell Optmization
4. Discussion
5. Conclusions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Reddy, N.L.; Rao, V.N.; Vijayakumar, M.; Santhosh, R.; Anandan, S.; Karthik, M.; Shankar, M.V.; Reddy, K.R.; Shetti, N.P.; Nadagouda, M.N.; et al. A review on frontiers in plasmonic nano-photocatalysts for hydrogen production. Int. J. Hydrogen Energy 2019, 44, 10453–10472. [Google Scholar] [CrossRef]
- Corma, A.; Garcia, H. Photocatalytic reduction of CO2 for fuel production: Possibilities and challenges. J. Catal. 2013, 308, 168–175. [Google Scholar] [CrossRef]
- Tahir, M.; Amin, N.S. Recycling of carbon dioxide to renewable fuels by photocatalysis: Prospects and challenges. Renew. Sustain. Energy Rev. 2013, 25, 560–579. [Google Scholar] [CrossRef]
- Hattori, M.; Einaga, H.; Takeshi, D.; Masaharu, T. Efficient hydrogen production from formic acid using TiO2-supported AgPd@Pd nanocatalysts. J. Mater. Chem. A 2015, 3, 4453–4461. [Google Scholar] [CrossRef]
- Arzate Salgado, S.Y.; Ramirez Zamora, R.M.; Zanella, R.; Peral, J.; Malato, S.; Maldonado, M.I. Photocatalytic hydrogen production in a solar pilot plant using a Au/TiO2 photo catalyst. Int. J. Hydrogen Energy 2016, 41, 11933–11940. [Google Scholar] [CrossRef]
- Sohn, Y.; Huang, W.; Taghipour, F. Recent progress and perspectives in the photocatalytic CO2 reduction of Ti-oxide-based nanomaterials. Appl. Surf. Sci. 2017, 396, 1696–1711. [Google Scholar] [CrossRef]
- Yang, H.G.; Sun, C.H.; Qiao, S.Z.; Zou, J.; Smith, S.C.; Cheng, H.M.; Lu, G.Q. Anatase TiO2 single crystals with a large percentage of reactive facets. Nature 2008, 453, 638–641. [Google Scholar] [CrossRef] [PubMed]
- Luther, J.M.; Jain, P.K.; Ewers, T.; Alivisatos, A.P. Localized surface plasmon resonances arising from free carriers in doped quantum dots. Nat. Mater. 2011, 10, 361–366. [Google Scholar] [CrossRef]
- Linic, S.; Christopher, P.; Ingram, D.B. Plasmonic-metal nanostructures for efficient conversion of solar to chemical energy. Nat. Mater. 2011, 10, 911–921. [Google Scholar] [CrossRef]
- Wei, Y.; Wu, X.; Zhao, Y.; Wang, L.; Zhao, Z.; Huang, X.; Liu, J.; Li, J. Efficient photocatalysts of TiO2 nanocrystals-supported PtRu alloy nanoparticles for CO2 reduction with H2O: Synergistic effect of Pt-Ru. Appl. Catal. B Environ. 2018, 236, 445–457. [Google Scholar] [CrossRef]
- Shibata, T.; Irie, H.; Hashimoto, K. Enhancement of Photoinduced Highly Hydrophilic Conversion on TiO2 Thin Films by Introducing Tensile Stress. J. Phys. Chem. B 2003, 107, 1066–10698. [Google Scholar] [CrossRef]
- Tavares, C.J.; Marques, S.M.; Lanceros-Mendez, S.; Sencadas, V.; Teixeira, V.; Carneiro, J.O.; Martins, A.J.; Fernades, A.J. Strain analysis of photocatalytic TiO2 thin films on polymer substrates. Thin Sol. Films 2008, 516, 1434–1438. [Google Scholar] [CrossRef]
- Iuga, M.; Steinle-Neumann, G.; Meinhardt, J. Ab-initio simulation of elastic constants for some ceramic materials. Eur. Phys. J. B 2007, 58, 127–133. [Google Scholar] [CrossRef]
- Kisi, E.H.; Howard, C.J. Elastic constants of tetragonal zirconia measured by a new powder diffraction technique. J. Am. Ceram. Soc. 1998, 81, 1682–1684. [Google Scholar] [CrossRef]
- Yin, W.-J.; Chen, S.; Yang, J.-H.; Gong, X.-G.; Yan, Y.; Wei, S.-H. Effective band gap narrowing of anatase TiO2 by strain along a soft crystal direction. Appl. Phys. Lett. 2010, 96, 221901. [Google Scholar] [CrossRef]
- Burmajdad, A.; Nazeer, A.A.; Al Sagheer, F.; Nahar, S.; Zaki, M.I. Controlled Synthesis of ZrO2 Nanoparticles with Tailored Size, Morphology and Crystal Phases via Organic/Inorganic Hybrid Films. Sci. Rep. 2018, 8, 3695. [Google Scholar] [CrossRef]
- Wei, Z.; Kowalski, E.; Ohanti, B. Enhanced photocatalytic activity by particle morphology: Preparation, characterization, and photocatalytic activities of octahedral anatase titania particles. Chem. Lett. 2014, 43, 346–348. [Google Scholar] [CrossRef]
- Pellegrino, F.; Isopescu, R.; Pellutiè, L.; Sordello, F.; Rossi, A.M.; Ortel, E.; Martra, G.; Hodoroaba, V.-D.; Maurino, V. Machine learning approach for elucidating and predicting the role of synthesis parameters on the shape and size of TiO2 nanoparticles. Sci. Rep. 2020, 10, 18910. [Google Scholar] [CrossRef]
- Pellegrino, F.; Ortel, E.; Mileke, J.; Schmidt, R.; Maurino, V.; Hodoroaba, V.-D. Controlled particle size and shape distribution: A feasibility study toward reference materials for quality assurance of nonspherical nanoparticle characterization. Adv. Eng. Mater. 2021, 24, 2101347. [Google Scholar] [CrossRef]
- Ye, M.; Zhang, Q.; Hu, Y.; Ge, J.; Lu, Z.; He, L.; Chen, Z.; Yin, Y. Magnetically recoverable core–shell nanocomposites with enhanced photocatalytic activity. Chem. Eur. J. 2010, 16, 6243–6250. [Google Scholar] [CrossRef]
- Hines, M.A.; Guyot-Sionnest, P. Synthesis and characterization of strongly luminescing ZnS-capped CdSe nanocrystals. J. Phys. Ceram. 1996, 100, 468–471. [Google Scholar] [CrossRef]
- Yuan, Q.; Wang, T.; Yu, P.; Zhang, H.; Zhang, H.; Ji, W. A review on the electroluminescence properties of quantum-dot light-emitting diodes. Org. Electron. 2021, 90, 106086. [Google Scholar] [CrossRef]
- Sahu, A.; Kumar, D. Core-shell quantum dots: A review on classification, materials, application and theoretical modelling. J. Alloys Comp. 2022, 924, 166508. [Google Scholar] [CrossRef]
- Wagner, A.M.; Knipe, J.M.; Orive, G.; Peppas, N.A. Quantum dots in biomedical applications. Acta Biomater. 2019, 94, 44–63. [Google Scholar] [CrossRef] [PubMed]
- Chandan, H.R.; Schiffman, J.D.; Balakrichna, R.G. Quantum dots as fluorescent probes: Synthesis, surface chemistry, energy transfer mechanisms, and applications. Sens. Actuators B Chem. 2018, 258, 1191–1214. [Google Scholar]
- Xu, Y.; Zhou, Z.; Zou, M.; Liu, Y.; Zheng, Y.; Yang, Y.; Lan, S.; Lan, J.; Nan, C.-W.; Lin, Y.-H. Multi-field driven hybrid catalysts for CO2 reduction: Progress, mechanism and perspective. Mater. Today 2022, 54, 225–246. [Google Scholar] [CrossRef]
- Wei, Z.; Endo-Kmura, M.; Wang, K.; Colbeau-Justin, C.; Kowalski, E. Influence of semiconductor morphology on photocatalytic activity of plasmonic photocatalysts: Titanate nanowires and octahedral anatase nanoparticles. Nanomaterials 2019, 9, 1447. [Google Scholar] [CrossRef]

| Material | C11 (GPa) | C12 (GPa) | C13 (GPa) | C33 (GPa) | C44 (GPa) | C66 (GPa) |
|---|---|---|---|---|---|---|
| a-TiO2 | 320 1 | 151 1 | 143 1 | 190 1 | 54 1 | 60 1 |
| t-ZrO2 | 327 2 | 100 2 | 62 2 | 264 2 | 59 2 | 64 2 |
| Core Diameter (nm) | Shell Thickness (nm) | Average Increase in Shell Electron–Hole Pair Production | TiO2 Volume Fraction | Overall Increase in Electron–Hole Pair Production |
|---|---|---|---|---|
| 10 | 4 | 25% | 83% | 21% |
| 10 | 7 | 24% | 93% | 22% |
| 10 | 10 | 22% | 96% | 21% |
| 12 | 5 | 25% | 84% | 21% |
| 12 | 10 | 23% | 95% | 22% |
| 12 | 12 | 22% | 96% | 21% |
| 12 | 10 (half) 1 | 10% | 90% | 9% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Swadener, J.G. Strain Engineering of ZrO2@TiO2 Core@shell Nanoparticle Photocatalysts. Solar 2023, 3, 15-24. https://doi.org/10.3390/solar3010002
Swadener JG. Strain Engineering of ZrO2@TiO2 Core@shell Nanoparticle Photocatalysts. Solar. 2023; 3(1):15-24. https://doi.org/10.3390/solar3010002
Chicago/Turabian StyleSwadener, John G. 2023. "Strain Engineering of ZrO2@TiO2 Core@shell Nanoparticle Photocatalysts" Solar 3, no. 1: 15-24. https://doi.org/10.3390/solar3010002
APA StyleSwadener, J. G. (2023). Strain Engineering of ZrO2@TiO2 Core@shell Nanoparticle Photocatalysts. Solar, 3(1), 15-24. https://doi.org/10.3390/solar3010002





