Abstract
In the last few decades, natural products from plants have developed immense importance in human health due to their therapeutic multi-functionality. They have also been reported to enhance human gut health, which is another important factor in overall human health. Diabetes Mellitus Type 2 (DM 2) is now a global concern with 6.28% of the world’s population affected by it. Many hypoglycemic drugs currently available in the market are either directly or indirectly based on a number of plant secondary metabolites. In the current study, we aimed to find out the hypoglycemic secondary metabolites from a leaf methanolic extract of Argyreia nervosa (Burm. f.) Bojer (Family: Convolvulaceae). In the in vitro experiment, this extract showed good inhibitory activity against Porcine Pancreatic Alpha-amylase (PPA) with IC50 value of 1.1 mg/mL. The presence of Quercetin and Ursolic acid was identified in the leaf methanolic extract using HPTLC, HPLC, and MS analysis. The calculated IC50 values against PPA, for standard Quercetin and Ursolic acid, were 16.5 µg/mL and 13.2 µg/mL, respectively. The in silico studies used both of these compounds as ligands against PPA (PDB ID: 1OSE) in AutoDock 4.2.6. Significant binding energies of −9.89 kcal/mol and −8.96 kcal/mol were seen for Quercetin and Ursolic acid, respectively, while Acarbose (the drug used as the positive control) had a binding energy of −12.48 kcal/mol. Both Quercetin and Ursolic acid strongly interacted with the pivotal amino acid residues such asGlu233, Asp197, and Asp300, which are present at the active site of PPA, thus upholding our in vitro experimental results. Both the compounds have exhibited beneficial effects on human gut health in DM 2 and related complications. The docking results of them with a few intestinal markers that are significant in gut health will also be discussed.
1. Introduction
Plants have impacted modern medicine by supplying several phytochemicals with therapeutic multi-functionality. In recent years, mankind has faced various disease outbreaks, in which plant-based medicines have proven their supremacy. Today, among the different life-style diseases, Diabetes mellitus Type 2 (DM 2) has received the attention of scientists and researchers worldwide. It is becoming a global concern as about 6.28% of the world’s population is affected by it [1]. DM 2 is a metabolic disorder that is characterized by hyperglycemia due to escalating defects in insulin secretion on the background of insulin resistance. Presently, there are a number of synthetic medicines available to treat this condition. However, these medicines have some side effects on human health. Thus, the hunt for new plant-based anti-hyperglycemic molecules is still on.
One of the most common strategies to treat DM 2 is to inhibit the isoforms of the α-amylase enzyme. This enzyme plays a crucial role in the digestion of food and catalyzes the hydrolysis of starch, increasing blood sugar levels. The association between human pancreatic α-amylase and post-prandial hyperglycemia has been published previously. The delayed starch digestion caused by α-amylase inhibition reduces the rate of absorption of glucose into the blood [2]. Apart from the usual DM 2 complications, recent reports have shown that the human gut microbiota also is negatively affected due to DM 2. Almost 90% of the adult human gut microbiota consists of either Bacteroidetes or Fermicutes. In DM 2 patients, the ratio of Bacteroidetes to Fermicutes decreased and there was a significant drop in the abundance of Bifidobacteria [3].
Plant secondary metabolites belonging to different classes such as phenols, flavonoids, terpenes, glycosides, alkaloids, and saponins have been reported for their α-amylase inhibitory potential. Among them, flavonoids have been extensively studied for their positive effect on the gut microbiota of DM 2 patients [4]. Argyreia nervosa (Burm. f.) Bojer is a medicinal vine that has been utilized for many years due to its therapeutic properties [5]. Our own group has shown that the flavonoids from A. nervosa, namely, Vitexin, Myricetin, Isoquercetin, and Rutin, have strong α-amylase inhibitory potential [6]. However, none of the earlier studies have reported the effect of the secondary metabolites of A. nervosa on human gut microbiota.
Toll-like receptors (TLRs) are the pattern recognition receptors present in the immune cells and intestinal epithelial cells of humans. They play a pivotal role in initiating the inflammatory response toward a broad spectrum of microbial components and controlled activation of both innate and adaptive immunity. Scientific reports have shown an increase in the activity and expression of TLR2 in both type-I and type-II diabetes [7]. It is reported that the microbial consortium present in the intestine is one of the important factors regulating the TLR activation. However, it is also well studied that dietary habits modulate the gut microbiota composition [8].
In the present study, we identified two important pancreatic α-amylase (PPA) inhibitory secondary metabolites, namely, Quercetin and Ursolic acid from A. nervosa that also impact the human gut microbiota. The identification of secondary metabolites was achieved using HPLC and ESI/MS analysis and the PPA inhibitory capacity was demonstrated by in vitro quantitative DNSA assay. Further, the results were validated by the molecular docking studies using the crucial proteins involved in the food digestion (PPA) and human gut microbiota stability (TLR2).
2. Materials and Methods
2.1. Collection and Identification
The leaves of Argyreia nervosa (Burm. f.) Bojer were collected from Savitribai Phule Pune University campus (Latitude 18.549120° N and Longitude 73.828120° E). The collected specimen was deposited and identified at the Botanical Survey of India, Western Regional Centre, Pune-7 with voucher number (BSI/WRC/IDEN.CER./2020/94).
2.2. Preparation of Leaf Extracts
The hot extraction method was followed using Soxhlet apparatus to prepare the leaf extracts. A sequential extraction using six solvents with increasing polarity viz. pet ether, chloroform, ethyl acetate, acetone, and water was carried out. These extracts were concentrated using a rotary evaporator and stored at 4 °C for further use.
2.3. Preliminary Phytochemical Analysis
The preliminary phytochemical tests were performed in triplicates to check the presence of different phytochemicals [9].
2.4. Qualitative Starch-Iodide Assay
The preliminary check of the PPA inhibition potential of the leaf extracts was carried out by performing the method proposed by Zinjarde et al. [10] and detailed in Kulkarni and Kamble [6].
2.5. HPTLC Analysis
2.5.1. Preparation of Standard Stock Solutions
The standard solutions of Quercetin (Sigma-Aldrich, St. Louis, MO, USA, Catalogue No. Q4951) and Ursolic acid (Sigma-Aldrich, USA, Catalogue No. U6753) were prepared at a concentration of 1 mg/mL each in absolute methanol. Ultra-sonication ensured acomplete dissolution.
2.5.2. Sample Preparation
The dry methanol leaf extract of A. nervosa was accurately weighed to be 5 mg and re-dissolved in 1 mL methanol. This sample was sonicated and filtered through a 0.45 µm membrane filter prior to application on a TLC plate.
2.5.3. Instrumentation and Experimental Conditions
The chromatography was performed according to the method reported with slight modifications [11]. The HPTLC system utilized was created by CAMAG (CAMAG, Muttenz, Switzerland). The mobile phases Toluene: Ethyl acetate: Formic acid (5:4:1 v/v/v) and Toluene: Acetone: Formic acid (7.8:2.2:0.15 v/v/v) were used for the separation of Quercetin and Ursolic acid, respectively. The obtained data were analyzed using WinCATS software (Version 1.4.5) by measuring the peak areas.
2.6. HPLC Analysis
The HPLC analysis was carried out on Waters 2998 HPLC system (Milford, MA, USA) coupled with a photodiode array detector at the Central Instrumentation Facility of S. P. Pune University. The eluted fractions were monitored at 254 nm and were collected at different time points. These collected fractions were checked for their PPA inhibitory potential as mentioned in Section 2.4 and the positive fractions were carried forward for ESI/MS analysis to detect the active compounds.
2.7. ESI-MS Analysis
The electrospray ionization mass spectral measurements were performed on a Bruker Nano-Advance UHPLC LC-MS-MS with a TOF analyzer at the Central Instrumentation Facility of S. P. Pune University. The dry methanolic extract was redissolved in HPLC grade methanol and filtered through 24 µM filter before being injected into the mass spectrometer.
2.8. Quantitative 3,5-Dinitrosalicyclic Acid (DNSA) Assay
To quantify the PPA inhibition, a widely used DNSA assay was performed according to the method proposed by Miller in 1959 [12], and the calculations were performed using the formulae detailed by Kamble et al. [13].
2.9. Statistical Data Analysis
Each experiment was performed in triplicates. The presented data are expressed as mean ± SEM (Standard Error of Mean). The ANOVA was performed for the statistical analysis using PAST version 4.11. The p values ≤ 0.05 were considered as significant.
2.10. In Silico Docking Studies
2.10.1. Protein Preparation
The 3D crystal structures of PPA (PDB ID: 1OSE) and TLR-2 (PDB ID: 5D3I) were retrieved from the Protein Data Bank. Prior to docking, the protein was prepared by removing the pre-existing water and ligand molecules using Discovery Studio Visualizer v20.1.0.19295. The polar hydrogen atoms and Kollman charges were added using AutoDock 4.2.6 software [14]. This processed protein was subsequently saved as PDBQT file and used for docking.
2.10.2. Ligand Preparation
The 3-D structures of the ligand molecules were obtained from the PubChem database in SDF format. Using Discovery Studio Visualizer, these ligands were converted to PDB format.
2.10.3. Docking Studies
The prepared ligands were docked on the receptor proteins PPA and TLR-2 using AutoDock 4.2.6. For PPA, a grid box with spacing 0.375A°centered at (45 A° × 40 A° × 44 A°) was generated at the center of the co-crystallized ligand (Acarbose, positive control) [13]. For TLR-2, the grid box with spacing 0.375 A° centered at (100 A° × 90 A° × 90 A°) was generated around the binding pocket detected with the help of a CASTp online server [15]. The Lamarckian Genetic Algorithm was used for searching the best conformer of ligand molecules. In the docking process, a total of 10 conformers were considered for each compound. Of these 10 conformers, the one with the lowest binding energy and inhibition constant was regarded as having the highest affinity toward PPA and TLR2. Discovery Studio Visualizer was used to visualize the docked conformations of the interacting ligand molecules with selected proteins.
3. Results
3.1. Preliminary Phytochemical Analysis
Although the phytochemical analysis was performed for all the prepared extracts, here, we present the data related to methanol extract as they showed good PPA inhibition. The preliminary phytochemical analysis of the methanol leaf extract of A. nervosa showed the presence of phenols, flavonoids, terpenes, and alkaloids.
3.2. Qualitative Starch-Iodide Assay
The qualitative starch‒iodide assay was performed and analyzed on the basis of the color of the reaction, which is related to the starch‒iodine complex formation. The deep blue color of the reaction suggested that the PPA enzyme was inhibited by the plant secondary metabolites present in the extract keeping the starch undigested. The yellow color of the reaction indicated a complete digestion of the starch by the fully active PPA enzyme. The brownish color of the reaction suggested the partial degradation of the starch. In this assay, chloroform, acetone, and methanol extracts showed good inhibitory activity. However, the methanol extract was carried forward as the deep blue color of the inhibition persisted even after 24 h.
3.3. HPTLC Analysis
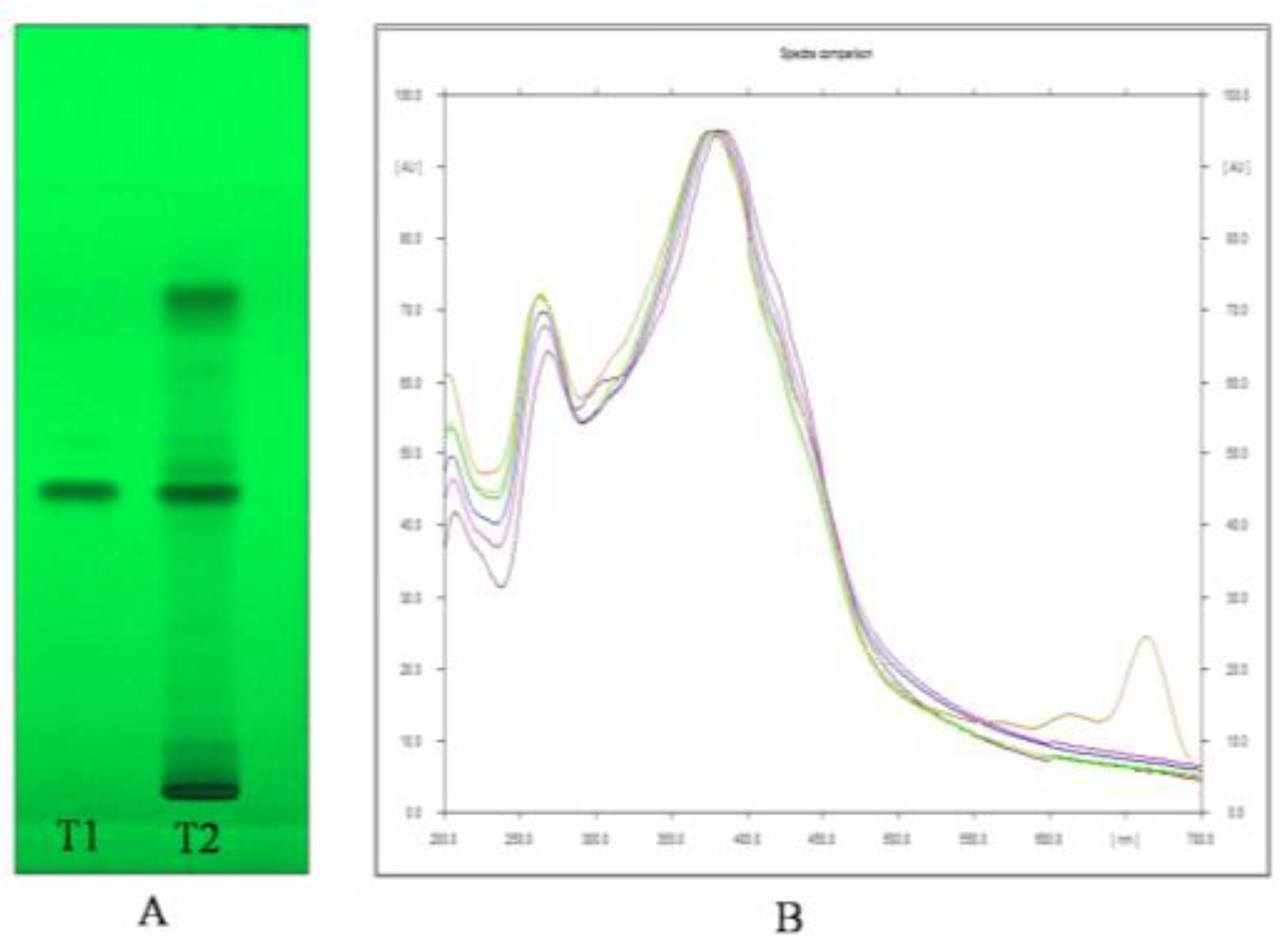
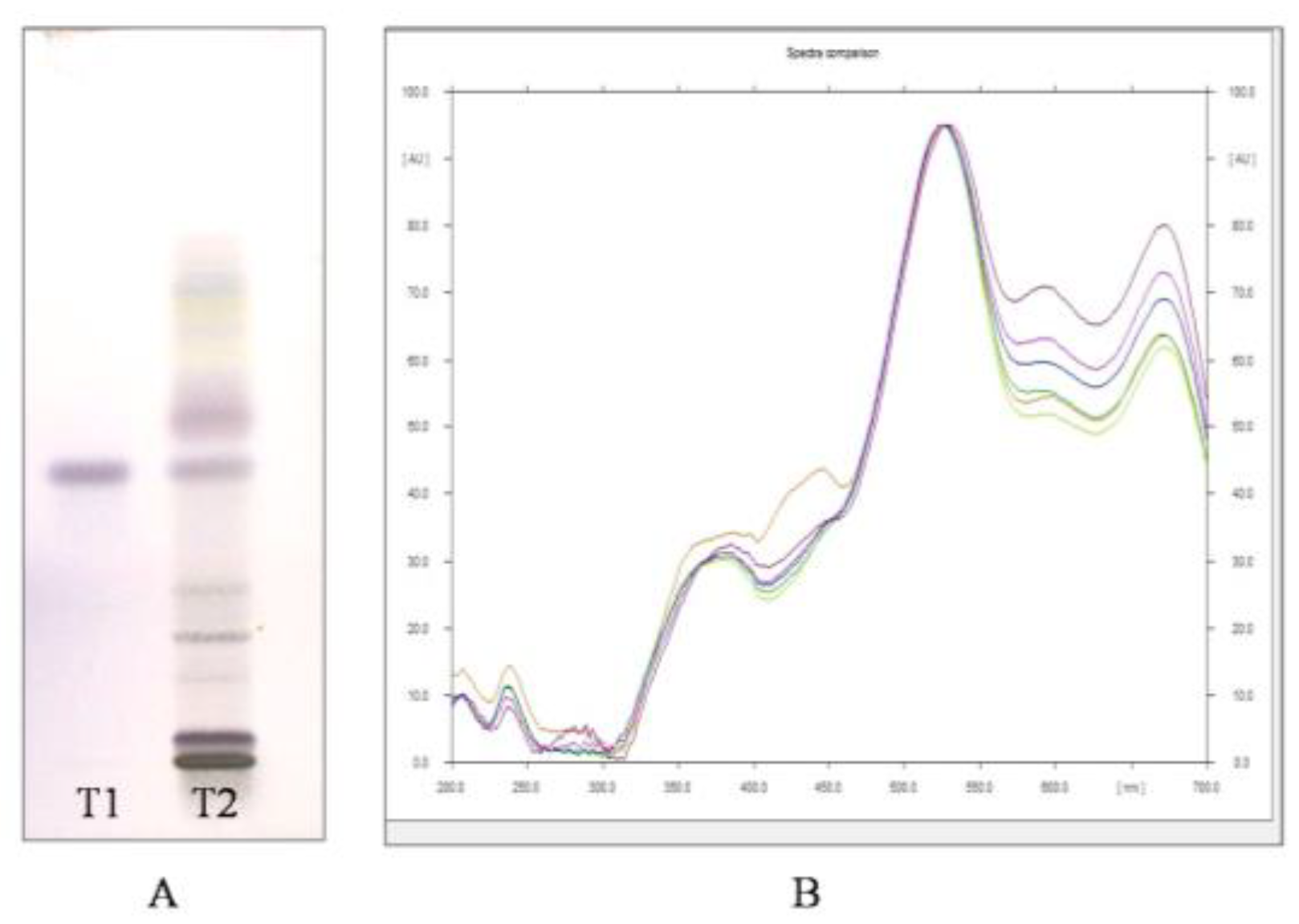
Through HPTLC analysis, it was observed that the methanolic leaf extract of A. nervosa possessed two important plant secondary metabolites, namely, Quercetin and Ursolic acid. Quercetin was separated and detected on the TLC plate (Figure 1A,B) and Ursolic acid was detected and separated on the TLC plate (Figure 2A,B).
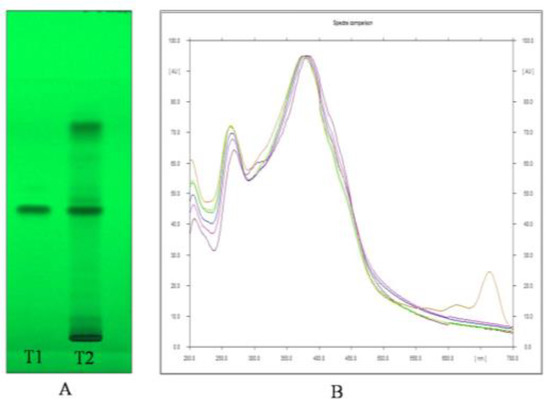
Figure 1.
(A) Image of HPTLC separation of leaf methanolic extract of A. nervosa. Track T1 represents Quercetin standard and track T2 represents leaf methanolic extract of A. nervosa with matching band of quercetin with Rf value of 0.48. (B) Matching identity of Quercetin in standard (Shown in different colours for different standard concentration) and sample track (Orange coloured) by comparing UV-VIS overlay spectra at 380 nm.
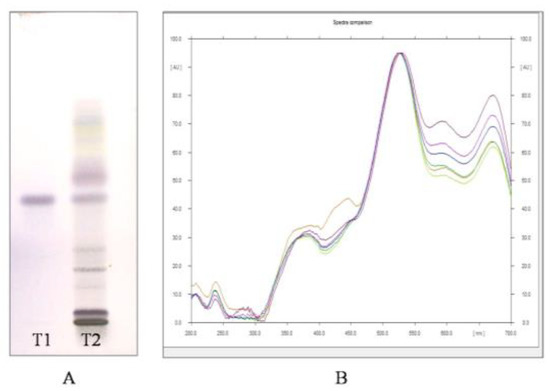
Figure 2.
(A) Image of HPTLC separation of leaf methanolic extract of A. nervosa after derivatization with anisaldehyde-sulphuric acid reagent. Track T1 represents Ursolic acid standard and track T2 represents leaf methanolic extract of A. nervosa with matching band of Ursolic acid with Rf value 0.57. (B) Matching the identity of Ursolic acid in standard (Shown in different colours for different standard concentration) and sample track (Orange coloured) by comparing UV-VIS overlay spectra at 540 nm.
3.4. HPLC Analysis
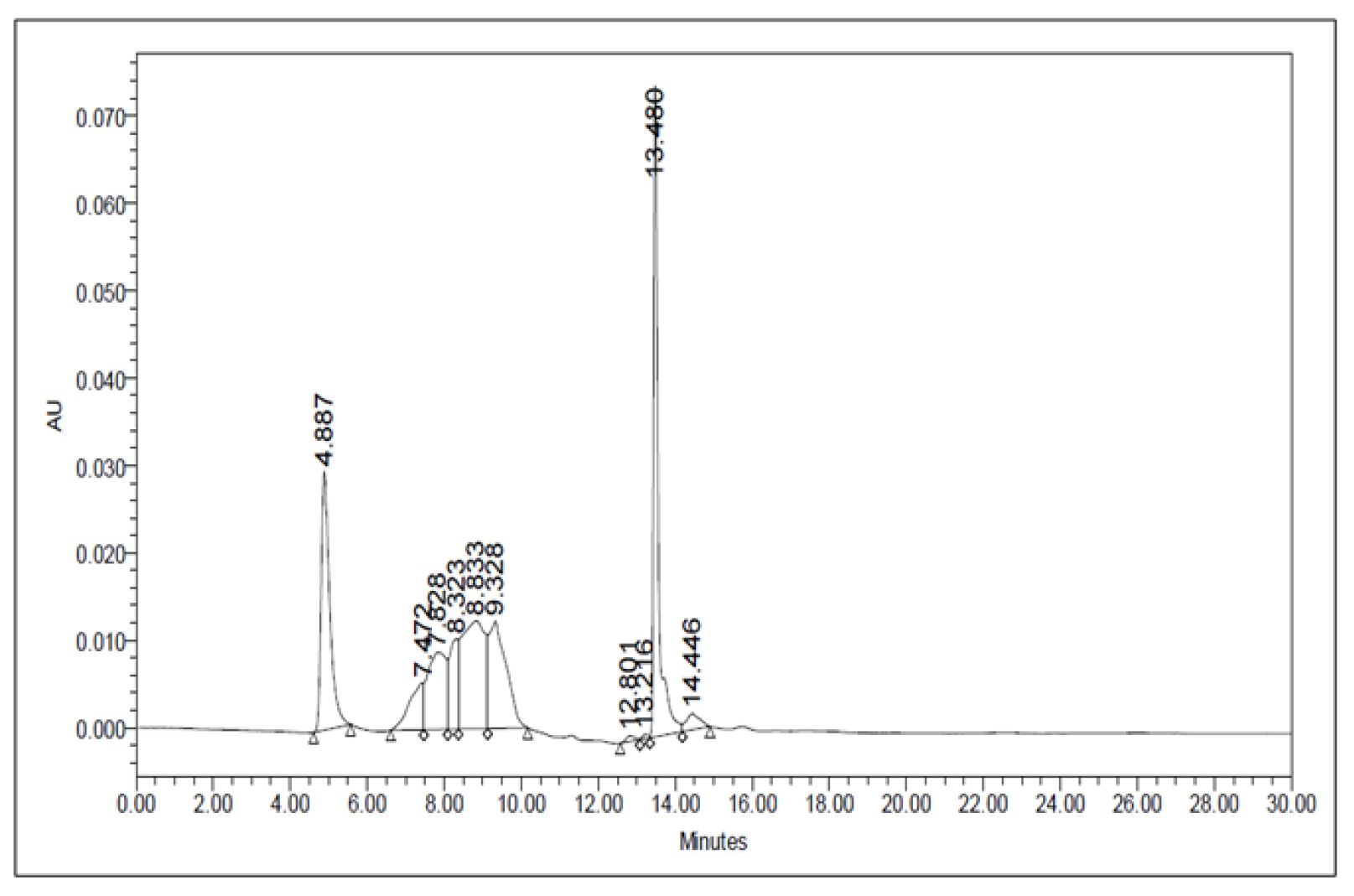
The crude methanolic leaf extract was subjected to the HPLC analysis. The peaks of this sample were collected at three different time points, namely, at 5 min, 6–12 min (5 components), and 14 min (Figure 3). These fractions were subjected to starch‒iodide assay. Among these, the fraction collected at 14 min showed strong PPA inhibitory activity. Hence, this fraction was further analyzed using ESI mass spectrometry.
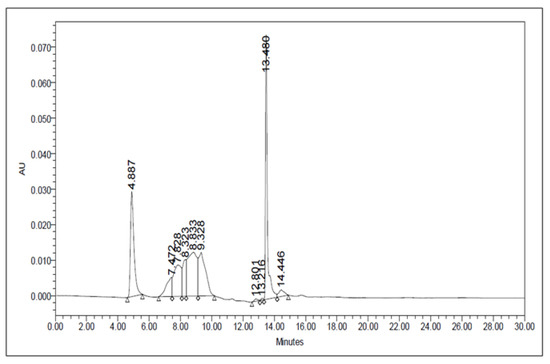
Figure 3.
HPLC chromatogram of A. nervosa leaf methanol extract.
3.5. ESI-MS Analysis
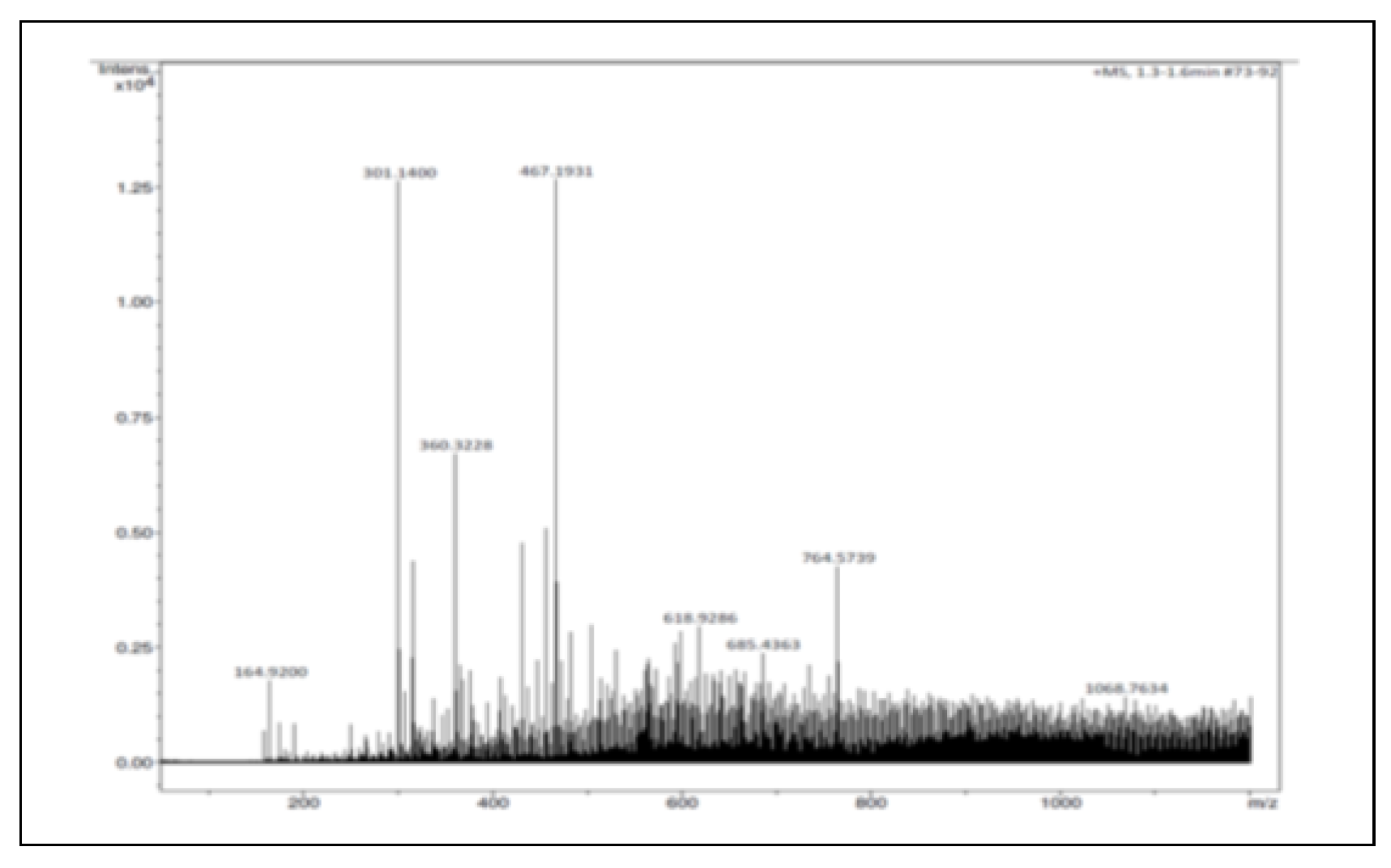
The ElectroSpray Ionization-Mass Spectrometry (ESI-MS) analysis indicated the presence of two abundant peaks of molar mass 301.1400 and 467.1931, which are assigned to Quercetin and Ursolic acid, respectively (Figure 4).
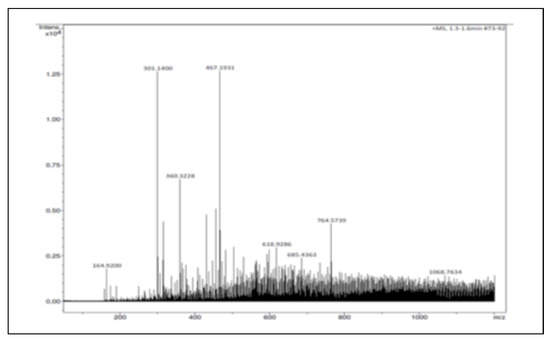
Figure 4.
ESI-Mass spectrum of the methanolic extract fraction eluted at 14 min usingHPLC.
3.6. Quantitative 3,5-Dinitrosalicyclic Acid (DNSA) Assay
As the methanol extract showed good results for the qualitative starch‒iodide assay, to quantify the result, we performed a DNSA assay. To demonstrate that this inhibitory activity was due to the presence of the secondary metabolites Quercetin and Ursolic acid, a separate DNSA assay was also performed for the standards alone. The percent inhibition of the PPA increased linearly as the concentration of the methanol extract increased. The calculated IC50 value of the leaf methanol extract was 1.1 mg/mL. The individual IC50 values for Quercetin and Ursolic acid were 16.5 µg/mL and 13.2 µg/mL, respectively.
3.7. In Silico Docking Studies
3.7.1. With PPA
The results of molecular docking using AutoDock 4.2.6 are shown in Table 1, thus indicating the docking scores and interaction details for the identified compounds: Quercetin and Ursolic acid bound with PPA. The binding energies of the ligand molecules is presented in kcal/mol. These studies were performed in comparison with the positive control Acarbose. The interactions between the protein‒ligand complexes were analyzed by Discovery Studio Visualizer. The crucial amino acids Glu233, Asp197, and Asp300 in α-amylase were found to be involved in bond formation between the protein and ligand molecule underpinning the theory of α-amylase inhibition.

Table 1.
Binding energies of the ligands and Acarbose with PPA. (The residues shown in bold are catalytic residues present at active site of PPA).
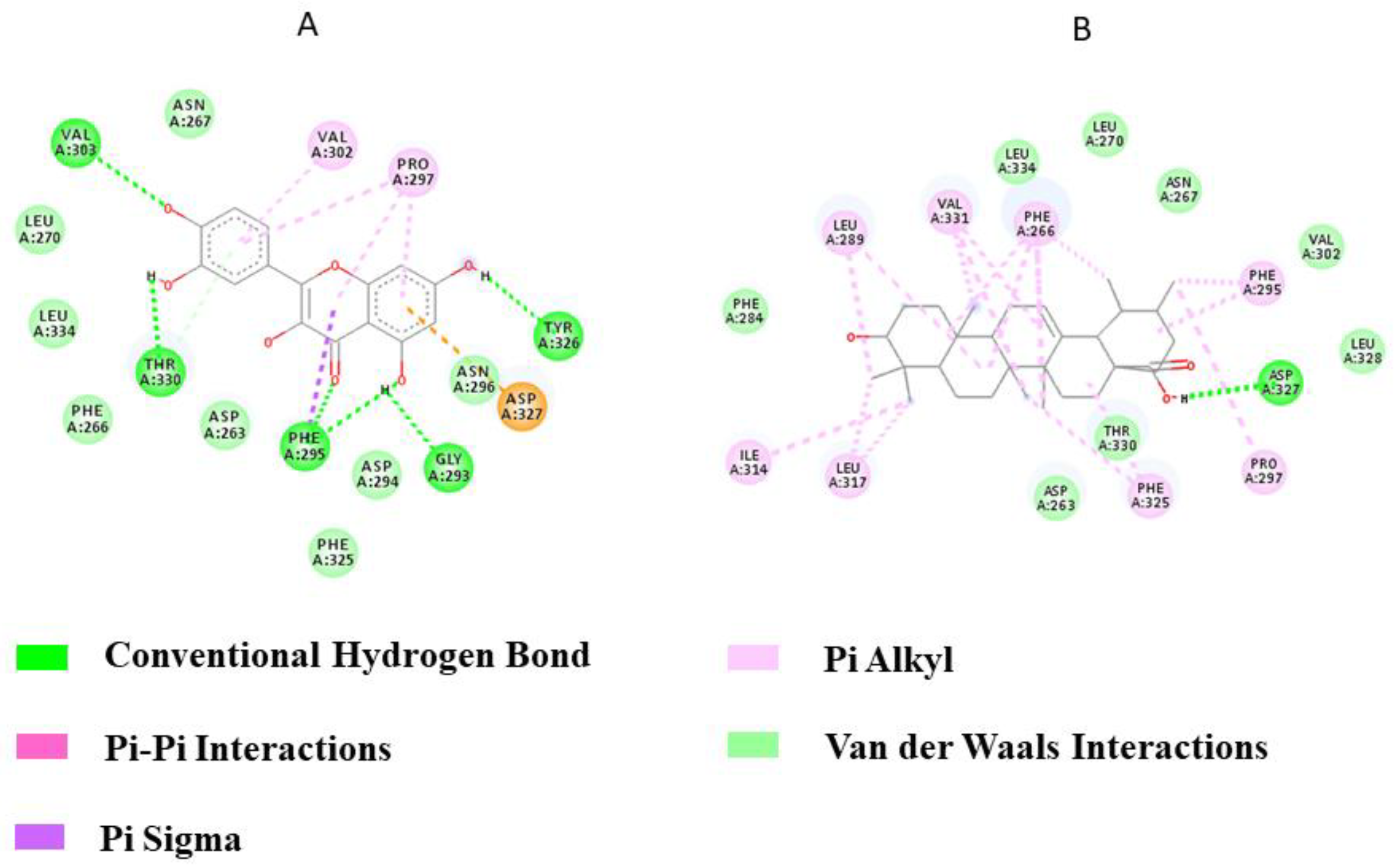
3.7.2. With TLR-2
Quercetin interacted with TLR2 via five hydrogen bonds through amino acid residues VAL303, THR330, PHE295, GLY293, and TYR326 while Ursolic acid bound to TLR2 via a single amino acid residue, ASP327 (Figure 5). The binding energies were −11.55 kcal/mol and −11.85 kcal/mol for Quercetin and Ursolic acid, respectively. From the docking analysis, it can be concluded that the flavanol Quercetin is a strong candidate for the inhibition of TLR2 compared to the terpenoid Ursolic acid, as it interacted with TLR2 via a greater number of hydrogen bonds.
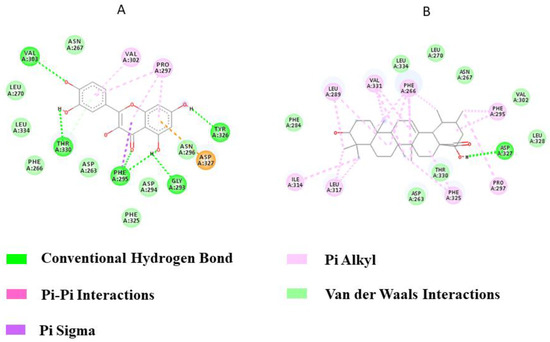
Figure 5.
2D representation of TLR2 Protein‒Ligand interactions: (A) Quercetin, (B) Ursolic acid.
4. Discussion
Plant-derived natural products have been known for their diverse and versatile therapeutic properties. These have been explored for their efficacy against many diseases such as cancer, arthritis, malaria, Alzheimer’s, and diabetes [16].
In the present study, the leaf methanolic extract of A. nervosa was explored for its α-amylase inhibitory activity. It showed the presence of major classes of secondary metabolites viz. phenols, flavonoids, terpenes, and alkaloids. In the qualitative starch‒iodide assay, the methanol extract showed persistent PPA inhibition. To find out the active PPA inhibitory molecules in the methanolic extract, HPTLC and HPLC analyses were performed. This unveiled the presence of two important secondary metabolites, namely, Quercetin and Ursolic acid. The presence of these secondary metabolites was validated using the ESI-MS analysis.
Quercetin, a well-known flavonoid, is known for its α-amylase inhibitory activity. A study performed in rat pancreas reported that Quercetin inhibited the α-amylase with an IC50 value of 0.061 µM [17]. Ursolic acid, a terpenoid, is known for its strong α-amylase inhibitory property. The in vitro study carried out by Wang et.al proved that Ursolic acid possessed α-amylase inhibitory potency with an IC50 value of 0.482 ± 0.12 mg/mL [18]. We found the IC50 values of Quercetin and Ursolic acid to be 16.5 µg/mL and 13.2 µg/mL (Section 3.6).
The molecular docking analysis showed that both Quercetin and Ursolic acid have a binding affinity toward PPA, with binding energies of −9.89 kcal/mol and −8.96 kcal/mol, respectively. The positive control, Acarbose, had a binding energy of −12.48 kcal/mol. Thus, both the secondary metabolites have exceptionally good PPA inhibition potency. The molecular docking and molecular dynamics studies of Quercetin and Ursolic acid from Morinda citrifolia fruits against human pancreatic α-amylase demonstrated that both phytochemicals were good inhibitors with binding energies of −2.16 kcal/mol and −8.58 kcal/mol, respectively [19].
It needs to be noted that, although both Quercetin and Ursolic acid have shown strong PPA inhibitory potential with low IC50 values individually in our experiments, our methanolic extract containing these two active phytochemicals has shown a high IC50 value, indicating the possibility for the presence of certain additional molecules that are interfering with the PPA inhibition. Thus, the crude extract warrants further purification.
A healthy gut microbiota is an essential aspect of the overall health of the human body. Studies have shown that gut microbiota plays a pivotal role in physiological processes in the body such as the regulation of fat storage, immunity, biological barriers, nutritional absorption, growth, and development [20]. Plant-derived phenols, flavonoids, terpenoids, and alkaloids have beneficial effects on human gut microbiota.
A study conducted on STZ-induced diabetic peripheral neuropathy (DPN) rat models reported that these rats showed gut dysbiosis compared to control rats, as observed by fecal microbiome16S rRNA gene sequencing. Quercetin was found to be bactericidal against potential pathogenic bacteria such as Oxalobacter and Klebsiella, that are positively correlated with DPN phenotypes and high ROS production levels. On the other hand, Quercetin enriched the abundance of probiotic genera like Actinobacteria that were negatively correlated with DPN phenotypes and ROS production levels [21].
Terpenoids also display anti-inflammatory, anti-viral, anti-cancer, anti-microbial, and anti-diabetic activities. However, so far, very few terpenoids have been investigated for their effects on gut microbiota. In the gut, terpenes are metabolized in the liver as well as biotransformed by intestinal microorganisms such as Fusobacterium nucleatum, Bacteroides fragilis, and Peptostreptococcus anaerobius through the processes of hydrolysis and reduction. It is widely known that about 30% of diabetic people suffer from liver malfunction. Wan et al. reported the repairing effect of Ursolic acid, a tri-terpenoid, on bacterial dysbiosis in liver fibrosis in mice. The potential beneficial bacteria, such as Firmicutes, Lactobacillus, and Bifidobacterium were seen to be increased in the Ursolic acid group [22].
A time-dependent high glucose dose is known to induce a marked increase in Toll-Like Receptors TLR2 and TLR4 mRNA and protein expression in human monocytes. TLR2 and TLR4 mediate inflammation in activated cells via NF-ḱB signaling. Thus, targeting these inflammatory receptors by the action of plant secondary metabolites may be an advantageous strategy to ameliorate the development of DM2 and complications related to it. Especially, TLR2 is known to be responsible for production of several molecules, including ROS and pro-inflammatory cytokines that contribute to the worsening of type II diabetes and related complications. Considering these facts, the down-regulation of TLR2 can be used as a novel approach to treat type II diabetes [23].
5. Conclusions
The intent of the present study was to find out the hypoglycemic secondary metabolites from the leaf methanolic extract of Argyreia nervosa (Burm. f.) Bojer. The leaf methanolic extract contained Quercetin and Ursolic acid. They exhibited great α-amylase inhibitory activity in both in vitro and in silico experiments. For the first time, we have shown excellent in silico docking of both these molecules on the active site of TLR-2, a bridging molecule of inflammation and gut microbiota composition. Our current work proposed the multi-functional role of Quercetin and Ursolic acid as α-amylase inhibitors as well as human gut health enhancers. Further experimental studies would be carried out in this regard.
Author Contributions
Conceptualization, A.A.K. (Anjali A. Kulkarni); methodology, A.D.K. and A.A.K. (Anjali A. Kulkarni); software, A.D.K.; validation, A.D.K. and A.A.K. (Anjali A. Kulkarni); formal analysis, A.D.K.; investigation, A.D.K. and A.A.K. (Anjali A. Kulkarni); resources, A.D.K. and A.A.K.(Anjali A. Kulkarni); data curation, A.D.K., A.A.K. (Anupa A. Kumbhar) and A.A.K. (Anjali A. Kulkarni); writing—original draft preparation, A.D.K. and A.A.K. (Anjali A. Kulkarni); writing—review and editing, R.P.K., A.A.K. (Anupa A. Kumbhar), and A.A.K. (Anjali A. Kulkarni); visualization, R.P.K., A.A.K.(Anupa A. Kumbhar) and A.A.K. (Anjali A. Kulkarni); supervision, A.A.K. (Anjali A. Kulkarni); project administration, A.A.K. (Anjali A. Kulkarni); funding acquisition A.D.K. and A.A.K. (Anjali A. Kulkarni). All authors have read and agreed to the published version of the manuscript.
Funding
A.D.K. would like to acknowledge CSIR-UGC for providing JRF fellowship UGC-Ref. No.: 788/(CSIR-UGC NET DEC.2018). A.A.K. (Anjali A. Kulkarni) would like to acknowledge DST-NRF Indo Korea grant (INT/KOREA/P31) for partial funding support.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Khan, M.A.B.; Hashim, M.J.; King, J.K.; Govender, R.D.; Mustafa, H.; Al Kaabi, J. Epidemiology of type 2 diabetes–global burden of disease and forecasted trends. J. Epidemiol. Glob. 2020, 10, 107. [Google Scholar] [CrossRef] [PubMed]
- Tarling, C.A.; Woods, K.; Zhang, R.; Brastianos, H.C.; Brayer, G.D.; Andersen, R.J.; Withers, S.G. The search for novel human pancreatic α-amylase inhibitors: High-throughput screening of terrestrial and marine natural product extracts. ChemBioChem 2008, 9, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Nie, Q.; Chen, H.; Hu, J.; Fan, S.; Nie, S. Dietary compounds and traditional Chinese medicine ameliorate type 2 diabetes by modulating gut microbiota. Crit. Rev. Food Sci. Nutr. 2019, 59, 848–863. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Luo, Y.; Hu, Z.; Qin, D.; Luo, F. Targeting gut microbiota in type 2 diabetes mellitus: Potential roles of dietary flavonoids. Food Biosci. 2022, 45, 101500. [Google Scholar] [CrossRef]
- Galani, V.J.; Patel, B.G.; Patel, N.B. Argyreia speciosa (Linn. f.) sweet: A comprehensive review. Pharmacogn. Rev. 2010, 4, 172. [Google Scholar] [CrossRef]
- Kulkarni, A.A.; Kamble, A.D. Flavonoids from Argyreia nervosa (Burm. f.) Bojer: A ready arsenal against pests as well as Diabetes. Biol. Life Sci. Forum 2020, 4, 56. [Google Scholar] [CrossRef]
- Jialal, I.; Kaur, H. The role of toll-like receptors in diabetes-induced inflammation: Implications for vascular complications. Curr. Diabetes Rep. 2012, 12, 172–179. [Google Scholar] [CrossRef]
- Tremaroli, V.; Bäckhed, F. Functional interactions between the gut microbiota and host metabolism. Nature 2012, 489, 242–249. [Google Scholar] [CrossRef]
- Trease, G.E.; Evans, W.C. Pharmacognosy, 15th ed.; Saunders Publishers: London, UK, 2002; pp. 42–44, 221–229, 246–249, 304–306, 331–332, 391–393. [Google Scholar]
- Zinjarde, S.S.; Bhargava, S.Y.; Kumar, A.R. Potent α-amylase inhibitory activity of Indian Ayurvedic medicinal plants. BMC Complement. Med. Ther. 2011, 11, 5. [Google Scholar] [CrossRef]
- Rout, K.K.; Singh, R.K.; Barik, D.P.; Mishra, S.K. Thin-Layer chromatographic separation and validated HPTLC method for quantification of Ursolic acid in various Ocimum species. J. Food Drug Anal. 2012, 20, 22. [Google Scholar]
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Kamble, R.P.; Ghosh, P.; Kulkarni, A.A. Identification of α-amylase inhibitory compounds from leaves of Careya arborea Roxb. and in silico docking studies. S. Afr. J. Bot. 2022, 151, 493–503. [Google Scholar] [CrossRef]
- Rizvi, S.M.D.; Shakil, S.; Haneef, M. A simple click by click protocol to perform docking: AutoDock 4.2 made easy for non-bioinformaticians. EXCLI J. 2013, 12, 831. [Google Scholar] [PubMed]
- Tian, W.; Chen, C.; Lei, X.; Zhao, J.; Liang, J. CASTp 3.0: Computed atlas of surface topography of proteins. Nucleic Acids Res. 2018, 46, W363–W367. [Google Scholar] [CrossRef] [PubMed]
- Ota, A.; Ulrih, N.P. An overview of herbal products and secondary metabolites used for management of type two diabetes. Front. Pharmacol. 2017, 8, 436. [Google Scholar] [CrossRef]
- Oboh, G.; Ademosun, A.O.; Ayeni, P.O.; Omojokun, O.S.; Bello, F. Comparative effect of quercetin and rutin on α-amylase, α-glucosidase, and some pro-oxidant-induced lipid peroxidation in rat pancreas. Comp. Clin. Path. 2015, 24, 1103–1110. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, J.; Yan, Y.; Liu, D.; Wang, C.; Wang, H. Inhibition of glycosidase by ursolic acid: In vitro, in vivo and in silico study. J. Sci. Food Agric. 2020, 100, 986–994. [Google Scholar] [CrossRef]
- Lolok, N.; Sumiwi, S.A.; Muhtadi, A.; Susilawati, Y.; Hendriani, R.; Ramadhan, D.S.F.; Levita, J.; Sahidin, I. Molecular docking and molecular dynamics studies of bioactive compounds contained in noni fruit (Morinda citrifolia L.) against human pancreatic α-amylase. J. Biomol. Struct. Dyn. 2022, 40, 7091–7098. [Google Scholar] [CrossRef]
- Guo, P.; Wu, C.M. Gut microbiota brings a novel way to illuminate mechanisms of natural products in vivo. Chin. Herb. Med. 2017, 9, 301–306. [Google Scholar] [CrossRef]
- Xie, J.; Song, W.; Liang, X.; Zhang, Q.; Shi, Y.; Liu, W.; Shi, X. Protective effect of quercetin on streptozotocin-induced diabetic peripheral neuropathy rats through modulating gut microbiota and reactive oxygen species level. Biomed. Pharmacother. 2020, 127, 110147. [Google Scholar] [CrossRef] [PubMed]
- Wan, S.Z.; Liu, C.; Huang, C.K.; Luo, F.Y.; Zhu, X. Ursolic acid improves intestinal damage and bacterial dysbiosis in liver fibrosis mice. Front. Pharmacol. 2019, 10, 1321. [Google Scholar] [CrossRef] [PubMed]
- Sepehri, Z.; Kiani, Z.; Nasiri, A.A.; Kohan, F. Toll-like receptor 2 and type 2 diabetes. Cell. Mol. Biol. Lett. 2016, 21, 2. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).