The Effect of Plant-Based and Mycoprotein-Based Meat Substitute Consumption on Cardiometabolic Risk Factors: A Systematic Review and Meta-Analysis of Controlled Intervention Trials
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data Sources and Search Strategy
2.2. Study Selection
2.3. Data Extraction and Risk of Bias Assessment
2.4. Intervention
2.5. Outcomes
2.6. Data Synthesis and Analysis
2.7. Certainty of Evidence
3. Results
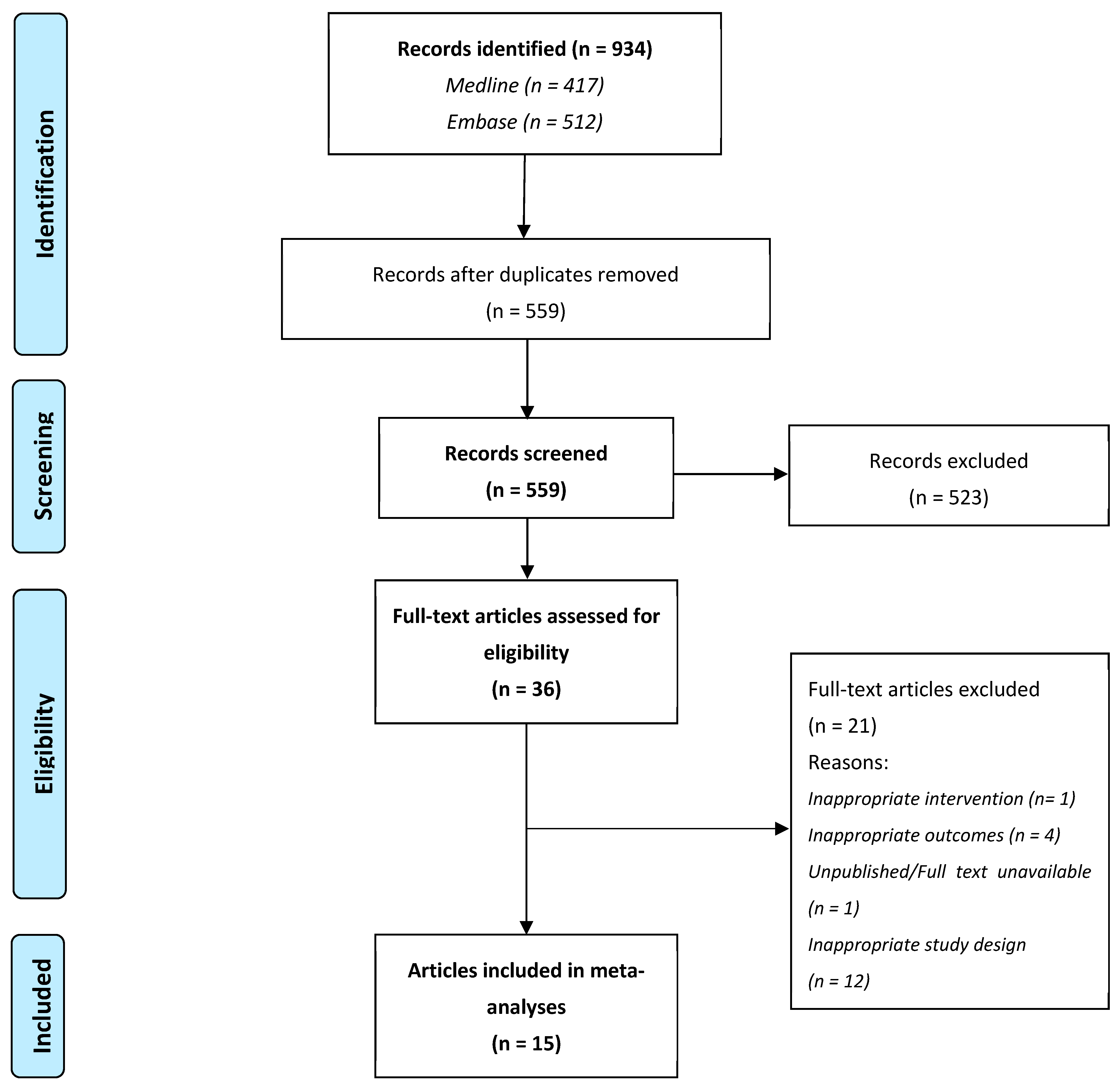
3.1. Study Selection Process
3.2. Study Characteristics and Risk of Bias
3.3. Pooled Effects of Meat Substitutes on Cardiometabolic Risk Factors
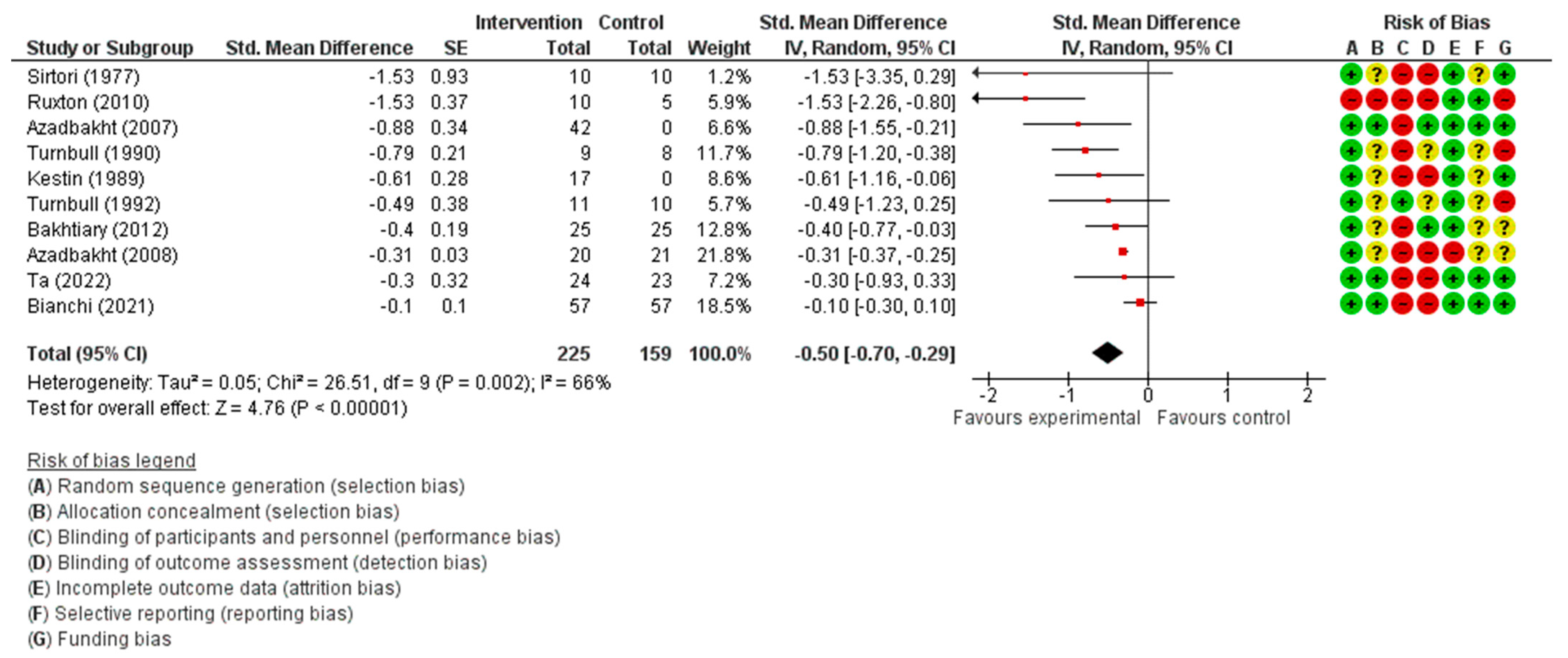
3.3.1. Total Cholesterol Concentration
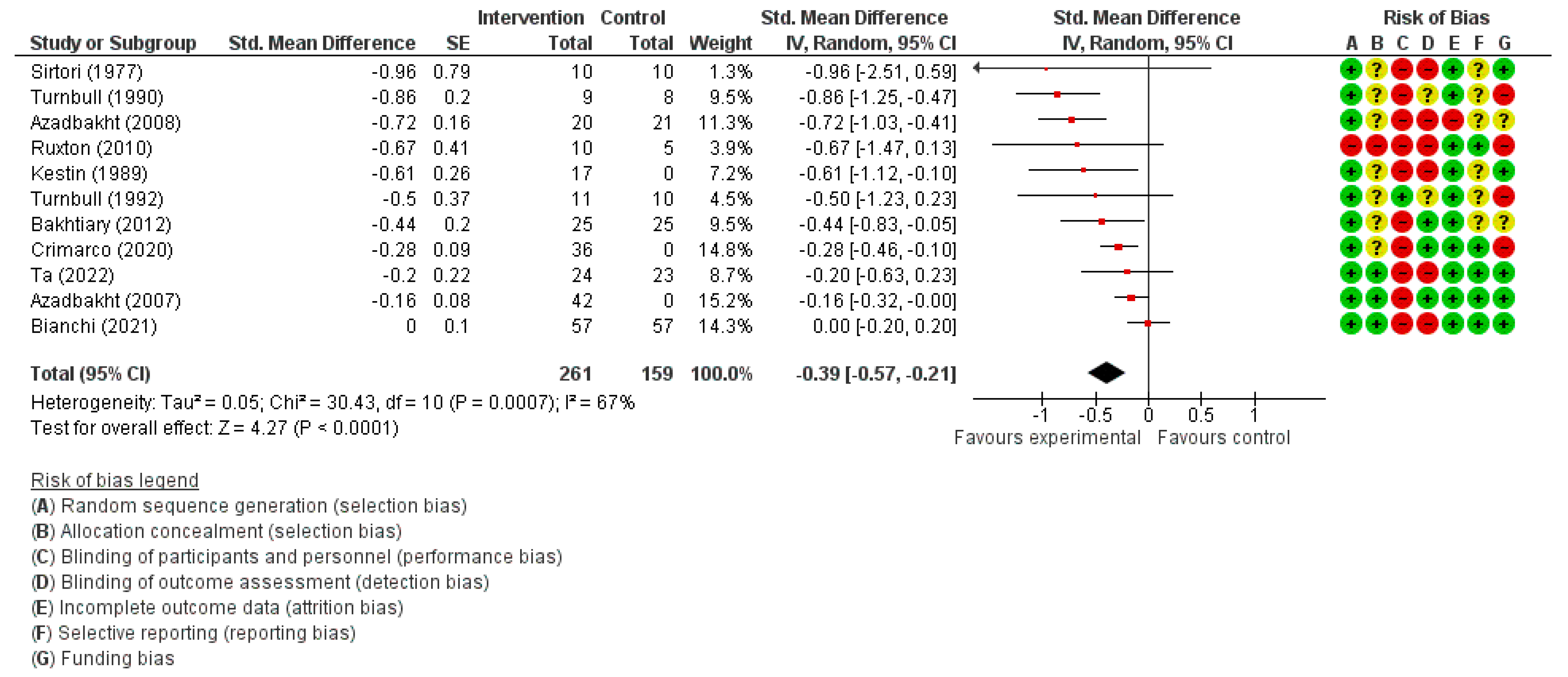
3.3.2. LDL Cholesterol Concentration
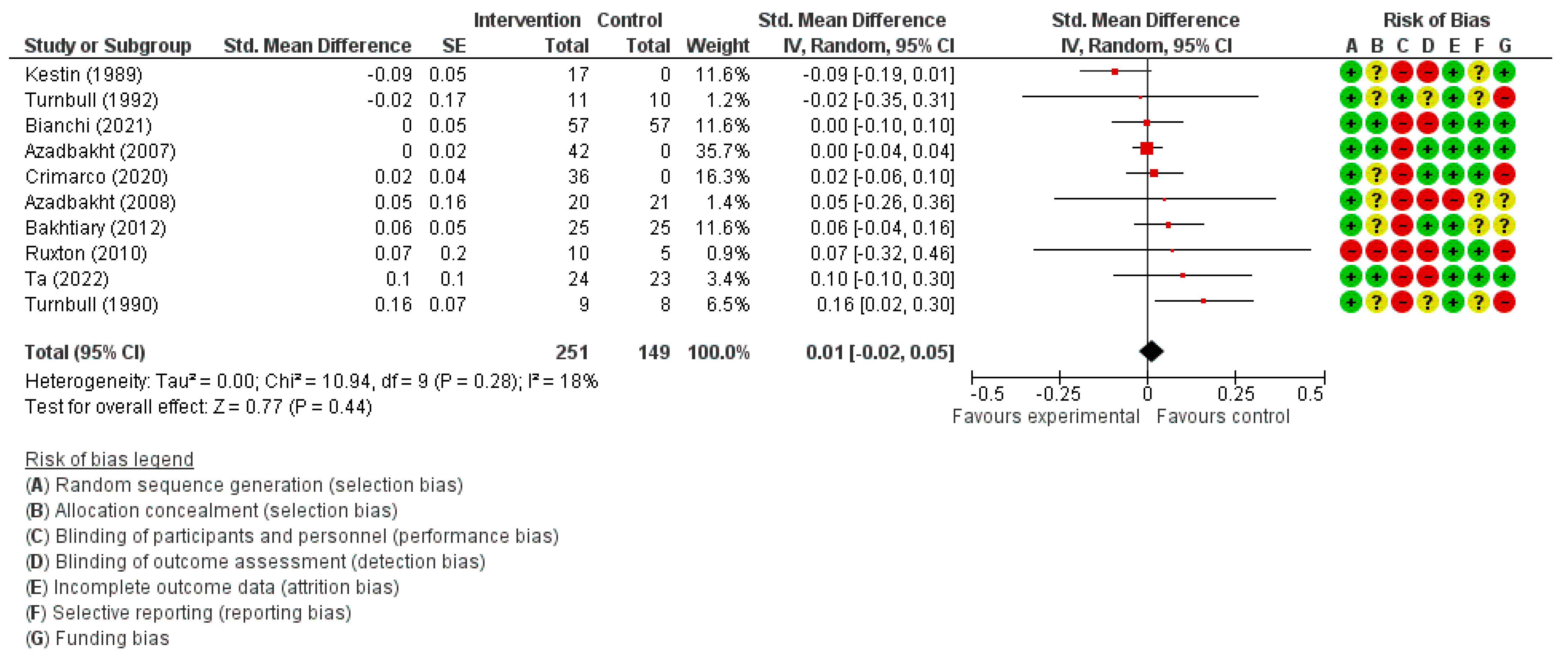
3.3.3. HDL Cholesterol Concentration
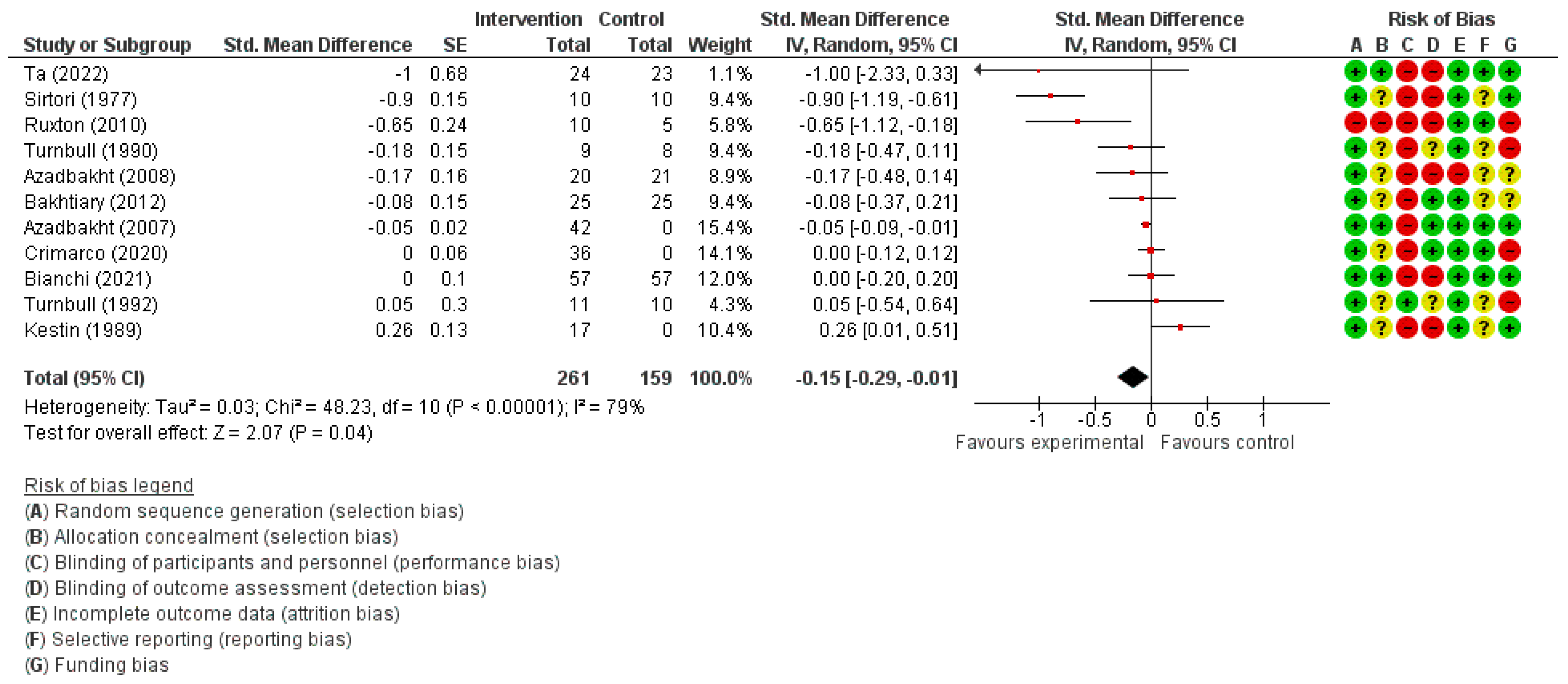
3.3.4. Triglyceride Concentration
3.3.5. Fasting Blood Glucose Concentration
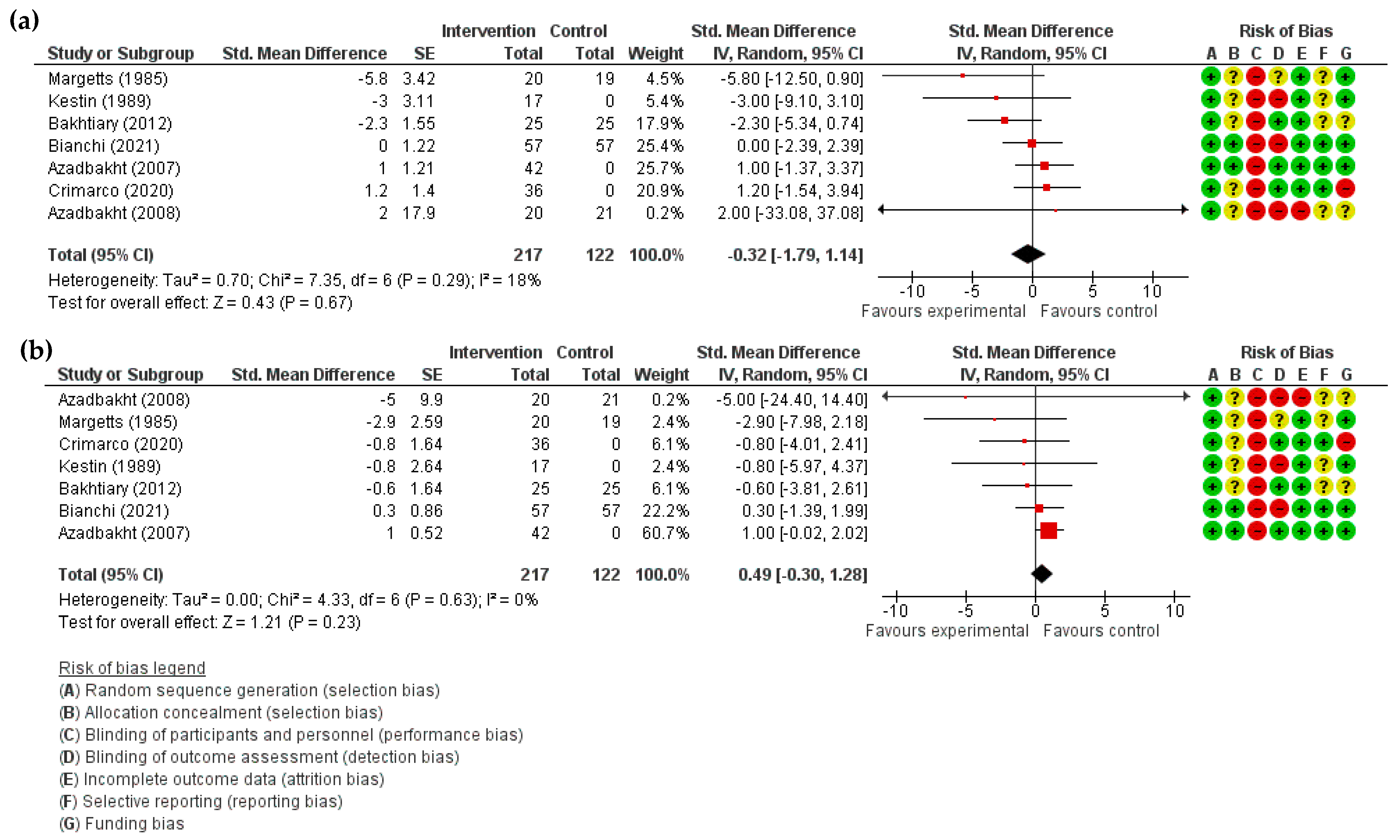
3.3.6. Systolic and Diastolic Blood Pressure
3.3.7. Weight
3.4. Meta-Regression
3.5. Publication Bias
4. Discussion
4.1. Strengths and Limitations
4.2. Potential Mechanisms
4.3. Implications
4.3.1. Public Health
4.3.2. Food Safety
4.3.3. Planetary Health
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Graça, J.; Godinho, C.A.; Truninger, M. Reducing meat consumption and following plant-based diets: Current evidence and future directions to inform integrated transitions. Trends Food Sci. Technol. 2019, 91, 380–390. [Google Scholar] [CrossRef] [Green Version]
- Gibbs, J.; Cappuccio, F.P. Plant-Based Dietary Patterns for Human and Planetary Health. Nutrients 2022, 14, 1614. [Google Scholar] [CrossRef] [PubMed]
- Pohjolainen, P.; Vinnari, M.; Jokinen, P. Consumers′ perceived barriers to following a plant-based diet. Br. Food J. 2015, 117, 1150–1167. [Google Scholar] [CrossRef]
- Bohrer, B.M. An investigation of the formulation and nutritional composition of modern meat analogue products. Food Sci. Hum. Wellness 2019, 8, 320–329. [Google Scholar] [CrossRef]
- Riaz, M.N. Textured soy protein and its uses. Agro Food Industry Hi Tech 2001, 12, 28–31. [Google Scholar]
- Finnigan, T.; Needham, L.; Abbott, C. Mycoprotein: A healthy new protein with a low environmental impact. In Sustainable Protein Sources; Elsevier: Amsterdam, The Netherlands, 2017; pp. 305–325. [Google Scholar]
- Hutton, B.; Salanti, G.; Caldwell, D.M.; Chaimani, A.; Schmid, C.H.; Cameron, C.; Ioannidis, J.P.; Straus, S.; Thorlund, K.; Jansen, J.P. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: Checklist and explanations. Ann. Intern. Med. 2015, 162, 777–784. [Google Scholar] [CrossRef] [Green Version]
- Sterne, J.A.C.; Gavaghan, D.; Egger, M. Publication and related bias in meta-analysis: Power of statistical tests and prevalence in the literature. J. Clin. Epidemiol. 2000, 53, 1119–1129. [Google Scholar] [CrossRef]
- Duval, S.; Tweedie, R. Trim and fill: A simple funnel-plot–based method of testing and adjusting for publication bias in meta-analysis. Biometrics 2000, 56, 455–463. [Google Scholar] [CrossRef]
- Ruxton, C.H.; McMillan, B. The impact of mycoprotein on blood cholesterol levels: A pilot study. Br. Food J. 2010, 112, 1092–1101. [Google Scholar] [CrossRef]
- Crimarco, A.; Springfield, S.; Petlura, C.; Streaty, T.; Cunanan, K.; Lee, J.; Fielding-Singh, P.; Carter, M.M.; Topf, M.A.; Wastyk, H.C. A randomized crossover trial on the effect of plant-based compared with animal-based meat on trimethylamine-N-oxide and cardiovascular disease risk factors in generally healthy adults: Study With Appetizing Plantfood—Meat Eating Alternative Trial (SWAP-MEAT). Am. J. Clin. Nutr. 2020, 112, 1188–1199. [Google Scholar]
- Kestin, M.; Rouse, I.L.; Correll, R.A.; Nestel, P.J. Cardiovascular disease risk factors in free-living men: Comparison of two prudent diets, one based on lactoovovegetarianism and the other allowing lean meat. Am. J. Clin. Nutr. 1989, 50, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Azadbakht, L.; Kimiagar, M.; Mehrabi, Y.; Esmaillzadeh, A.; Padyab, M.; Hu, F.B.; Willett, W.C. Soy inclusion in the diet improves features of the metabolic syndrome: A randomized crossover study in postmenopausal women. Am. J. Clin. Nutr. 2007, 85, 735–741. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turnbull, W.H.; Leeds, A.R.; Edwards, D.G. Mycoprotein reduces blood lipids in free-living subjects. Am. J. Clin. Nutr. 1992, 55, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Turnbull, W.H.; Leeds, A.R.; Edwards, G.D. Effect of mycoprotein on blood lipids. Am. J. Clin. Nutr. 1990, 52, 646–650. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, F.; Stewart, C.; Astbury, N.M.; Cook, B.; Aveyard, P.; Jebb, S.A. Replacing meat with alternative plant-based products (RE-MAP): A randomized controlled trial of a multicomponent behavioral intervention to reduce meat consumption. Am. J. Clin. Nutr. 2022, 115, 1357–1366. [Google Scholar] [CrossRef] [PubMed]
- Sirtori, C.; Agradi, E.; Conti, F.; Mantero, O.; Gatti, E. Soybean-protein diet in the treatment of type-II hyperlipoproteinaemia. Lancet 1977, 309, 275–277. [Google Scholar] [CrossRef] [PubMed]
- Margetts, B.M.; Beilin, L.J.; Armstrong, B.K.; Vandongen, R. Vegetarian diet in the treatment of mild hypertension: A randomized controlled trial. J. Hypertens. Suppl. 1985, 3, S429–S431. [Google Scholar] [CrossRef]
- Azadbakht, L.; Atabak, S.; Esmaillzadeh, A. Soy protein intake, cardiorenal indices, and C-reactive protein in type 2 diabetes with nephropathy: A longitudinal randomized clinical trial. Diabetes Care 2008, 31, 648–654. [Google Scholar] [CrossRef] [Green Version]
- Bakhtiary, A.; Yassin, Z.; Hanachi, P.; Rahmat, A.; Ahmad, Z.; Jalali, F. Effects of soy on metabolic biomarkers of cardiovascular disease in elderly women with metabolic syndrome. Arch. Iran Med. 2012, 15, 462–468. [Google Scholar]
- Ta, N.T.; Ngo, H.T.T.; Nguyen, P.M.; Truong, T.T.; Nguyen, G.H.; Dinh, H.T.D.; Nguyen, L.T.; Le, H.T.; Nguyen, K.C.; Yamamoto, S. Effectiveness of Textured Soybean Protein on Blood Biochemistry in Vietnamese Type 2 Diabetes Mellitus Patients. J. Nutr. Sci. Vitaminol. 2022, 68, 32–38. [Google Scholar] [CrossRef]
- Yokoyama, Y.; Levin, S.M.; Barnard, N.D. Association between plant-based diets and plasma lipids: A systematic review and meta-analysis. Nutr. Rev. 2017, 75, 683–698. [Google Scholar] [CrossRef] [Green Version]
- Gibbs, J.; Gaskin, E.; Ji, C.; Miller, M.A.; Cappuccio, F.P. The effect of plant-based dietary patterns on blood pressure: A systematic review and meta-analysis of controlled intervention trials. J. Hypertens. 2021, 39, 23–37. [Google Scholar] [CrossRef] [PubMed]
- Barnard, N.D.; Levin, S.M.; Yokoyama, Y. A systematic review and meta-analysis of changes in body weight in clinical trials of vegetarian diets. J. Acad. Nutr. Diet. 2015, 115, 954–969. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, Y.; Barnard, N.D.; Levin, S.M.; Watanabe, M. Vegetarian diets and glycemic control in diabetes: A systematic review and meta-analysis. Cardiovasc. Diagn. Ther. 2014, 4, 373. [Google Scholar] [PubMed]
- Chopra, S.S. Industry Funding of Clinical Trials: Benefit or Bias? JAMA 2003, 290, 113–114. [Google Scholar] [CrossRef] [Green Version]
- Miller, W.G.; Myers, G.L.; Sakurabayashi, I.; Bachmann, L.M.; Caudill, S.P.; Dziekonski, A.; Edwards, S.; Kimberly, M.M.; Korzun, W.J.; Leary, E.T. Seven direct methods for measuring HDL and LDL cholesterol compared with ultracentrifugation reference measurement procedures. Clin. Chem. 2010, 56, 977–986. [Google Scholar] [CrossRef] [Green Version]
- Derbyshire, E.; Ayoob, K.-T. Mycoprotein: Nutritional and health properties. Nutr. Today 2019, 54, 7–15. [Google Scholar] [CrossRef]
- Curtain, F.; Grafenauer, S. Plant-based meat substitutes in the flexitarian age: An audit of products on supermarket shelves. Nutrients 2019, 11, 2603. [Google Scholar] [CrossRef] [Green Version]
- Islam, M.A.; Amin, M.N.; Siddiqui, S.A.; Hossain, M.P.; Sultana, F.; Kabir, M.R. Trans fatty acids and lipid profile: A serious risk factor to cardiovascular disease, cancer and diabetes. Diabetes Metab. Syndr. Clin. Res. Rev. 2019, 13, 1643–1647. [Google Scholar] [CrossRef]
- Siri-Tarino, P.W.; Sun, Q.; Hu, F.B.; Krauss, R.M. Saturated fat, carbohydrate, and cardiovascular disease. Am. J. Clin. Nutr. 2010, 91, 502–509. [Google Scholar] [CrossRef] [Green Version]
- Hegsted, D. Serum-cholesterol response to dietary cholesterol: A re-evaluation. Am. J. Clin. Nutr. 1986, 44, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Ferdowsian, H.R.; Barnard, N.D. Effects of plant-based diets on plasma lipids. Am. J. Cardiol. 2009, 104, 947–956. [Google Scholar] [CrossRef] [PubMed]
- Brown, L.; Rosner, B.; Willett, W.W.; Sacks, F.M. Cholesterol-lowering effects of dietary fiber: A meta-analysis. Am. J. Clin. Nutr. 1999, 69, 30–42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nishina, P.M.; Freedland, R.A. The effects of dietary fiber feeding on cholesterol metabolism in rats. J. Nutr. 1990, 120, 800–805. [Google Scholar] [CrossRef]
- Anderson, J.W.; Johnstone, B.M.; Cook-Newell, M.E. Meta-analysis of the effects of soy protein intake on serum lipids. N. Engl. J. Med. 1995, 333, 276–282. [Google Scholar] [CrossRef]
- Damasceno, N.R.T.; Apolinário, E.; Flauzino, F.D.; Fernandes, I.; Abdalla, D.S.P. Soy isoflavones reduce electronegative low-density lipoprotein (LDL−) and anti-LDL− autoantibodies in experimental atherosclerosis. Eur. J. Nutr. 2007, 46, 125–132. [Google Scholar] [CrossRef]
- Rimbach, G.; Boesch-Saadatmandi, C.; Frank, J.; Fuchs, D.; Wenzel, U.; Daniel, H.; Hall, W.L.; Weinberg, P.D. Dietary isoflavones in the prevention of cardiovascular disease–A molecular perspective. Food Chem. Toxicol. 2008, 46, 1308–1319. [Google Scholar] [CrossRef]
- Assessment, R. Major lipids, apolipoproteins, and risk of vascular disease. JAMA 2009, 302, 1993–2000. [Google Scholar]
- Collaboration, N.R.F. Repositioning of the global epicentre of non-optimal cholesterol. Nature 2020, 582, 73. [Google Scholar]
- Law, M.R.; Wald, N.J.; Rudnicka, A. Quantifying effect of statins on low density lipoprotein cholesterol, ischaemic heart disease, and stroke: Systematic review and meta-analysis. BMJ 2003, 326, 1423. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- JBS 2: Joint British Societies’ guidelines on prevention of cardiovascular disease in clinical practice. Heart 2005, 91, v1–v52. [CrossRef] [Green Version]
- Santo, R.E.; Kim, B.F.; Goldman, S.E.; Dutkiewicz, J.; Biehl, E.; Bloem, M.W.; Neff, R.A.; Nachman, K.E. Considering plant-based meat substitutes and cell-based meats: A public health and food systems perspective. Front. Sustain. Food Syst. 2020, 4, 134. [Google Scholar] [CrossRef]
- Bao, W.; Rong, Y.; Rong, S.; Liu, L. Dietary iron intake, body iron stores, and the risk of type 2 diabetes: A systematic review and meta-analysis. BMC Med. 2012, 10, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fang, X.; An, P.; Wang, H.; Wang, X.; Shen, X.; Li, X.; Min, J.; Liu, S.; Wang, F. Dietary intake of heme iron and risk of cardiovascular disease: A dose–response meta-analysis of prospective cohort studies. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Fonseca-Nunes, A.; Jakszyn, P.; Agudo, A. Iron and cancer risk—A systematic review and meta-analysis of the epidemiological evidence. Cancer Epidemiol. Prev. Biomark. 2014, 23, 12–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- David, S.; Levi, C.S.; Fahoum, L.; Ungar, Y.; Meyron-Holtz, E.G.; Shpigelman, A.; Lesmes, U. Revisiting the carrageenan controversy: Do we really understand the digestive fate and safety of carrageenan in our foods? Food Funct. 2018, 9, 1344–1352. [Google Scholar] [CrossRef]
- Besada, V.; Andrade, J.M.; Schultze, F.; González, J.J. Heavy metals in edible seaweeds commercialised for human consumption. J. Mar. Syst. 2009, 75, 305–313. [Google Scholar] [CrossRef]
- Lim, S.S.; Vos, T.; Flaxman, A.D.; Danaei, G.; Shibuya, K.; Adair-Rohani, H.; Amann, M.; Anderson, H.R.; Andrews, K.G.; Aryee, M.; et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990-2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2224–2260. [Google Scholar] [CrossRef] [Green Version]
- Voon, P.T.; Ng, T.K.W.; Lee, V.K.M.; Nesaretnam, K. Diets high in palmitic acid (16: 0), lauric and myristic acids (12: 0+ 14: 0), or oleic acid (18: 1) do not alter postprandial or fasting plasma homocysteine and inflammatory markers in healthy Malaysian adults. Am. J. Clin. Nutr. 2011, 94, 1451–1457. [Google Scholar] [CrossRef] [Green Version]
- Hadi, J.; Brightwell, G. Safety of Alternative Proteins: Technological, environmental and regulatory aspects of cultured meat, plant-based meat, insect protein and single-cell protein. Foods 2021, 10, 1226. [Google Scholar] [CrossRef]
- Finnigan, T.J.A.; Wall, B.T.; Wilde, P.J.; Stephens, F.B.; Taylor, S.L.; Freedman, M.R. Mycoprotein: The future of nutritious nonmeat protein, a symposium review. Curr. Dev. Nutr. 2019, 3, nzz021. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verma, A.K.; Kumar, S.; Das, M.; Dwivedi, P.D. A comprehensive review of legume allergy. Clin. Rev. Allergy Immunol. 2013, 45, 30–46. [Google Scholar] [CrossRef] [PubMed]
- Inomata, N. Wheat allergy. Curr. Opin. Allergy Clin. Immunol. 2009, 9, 238–243. [Google Scholar] [CrossRef]
- UNEP. Assessing the Environmental Impacts of Consumption and Production: Priority Products and Materials; UNEP: Nairobi, Kenya, 2010; p. 80. [Google Scholar]
- Stehfest, E.; Bouwman, L.; Van Vuuren, D.P.; Den Elzen, M.G.; Eickhout, B.; Kabat, P. Climate benefits of changing diet. Clim. Chang. 2009, 95, 83–102. [Google Scholar] [CrossRef]
- Poore, J.; Nemecek, T. Reducing food’s environmental impacts through producers and consumers. Science 2018, 360, 987–992. [Google Scholar] [CrossRef] [Green Version]
- Pimentel, D.; Berger, B.; Filiberto, D.; Newton, M.; Wolfe, B.; Karabinakis, E.; Clark, S.; Poon, E.; Abbett, E.; Nandagopal, S. Water resources: Agricultural and environmental issues. BioScience 2004, 54, 909–918. [Google Scholar] [CrossRef] [Green Version]
- Heinke, J.; Lannerstad, M.; Gerten, D.; Havlík, P.; Herrero, M.; Notenbaert, A.M.O.; Hoff, H.; Müller, C. Water use in global livestock production—Opportunities and constraints for increasing water productivity. Water Resour. Res. 2020, 56, e2019WR026995. [Google Scholar] [CrossRef]


| Author | Country | Year | Design | Duration (Weeks) | N | Mean Age or Age Range (Years) | Men (%) | Intervention | Control | Quantity of Substitute Per Day | Population |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Sirtori [17] | Italy | 1977 | RCT, O, P | 6 | 20 | 40–68 | 50 | Plant-based | Omnivorous | Textured soy protein in place of animal protein | Type-II hyperlipoproteinemia |
| Margetts [18] | Aus | 1985 | RCT, O, P | 12 | 39 | 49.9 | 71.8 | Plant-based | Omnivorous | LOV diet with substitutes derived from wheat gluten, soy, and peanut protein | Mild hypertension |
| Kestin [12] | Aus | 1989 | RCT, O, C | 6 | 17 | 44 | 100 | Plant-based | Omnivorous | LOV diet with substitutes derived from wheat gluten, soy, and peanut protein | Healthy |
| Turnbull [15] | UK | 1990 | RCT, O, P | 3 | 17 | 19–48 | 29.4 | Mycoprotein | Omnivorous | 191 g wet weight, 40 g dry weight | TC > 5.2 mmol/L |
| Turnbull [14] | UK | 1992 | RCT, B, P | 8 | 21 | 21–61 | 66.7 | Mycoprotein | Omnivorous diet | 130 g wet weight, 26.9 g dry weight | TC > 5.2 mmol/L |
| Azadbakht [13] | Iran | 2007 | RCT, O, C | 8 | 42 | NR | 0 | Plant-based | Omnivorous diet | 30 g textured soy protein | Postmenopausal women |
| Azadbakht [19] | Iran | 2008 | RCT, O, P | 208 | 41 | 62.1 | 43.9 | Plant-based | Omnivorous | 15.5 g textured soy protein | Type 2 diabetics with nephropathy |
| Ruxton [10] | UK | 2010 | CT, O, P | 6 | 15 | 39.2 | NR | Mycoprotein | Omnivorous diet | 88 g wet weight, 21 g dry weight | TC > 4.19 mmol/L |
| Bakhtiary [20] | Iran | 2012 | RCT, O, P | 12 | 50 | 64.4 | 0 | Plant-based | Omnivorous | 35 g textured soy protein | Elderly women with MetS |
| Crimarco [11] | US | 2020 | RCT, O, C | 8 | 36 | 50 | 33 | Plant-based | Omnivorous | ≥2 servings of Beyond Meat | Healthy |
| Bianchi [16] | UK | 2021 | RCT, O, P | 4 | 114 | 35 | 33 | Mycoprotein + plant-based | Omnivorous | Participants were provided with a range of substitutes every fortnight | Healthy |
| Ta [21] | Vietnam | 2022 | RCT, O, P | 4 | 47 | 60.9 | 38.3 | Plant-based | Omnivorous | 40 g textured soy protein | Type 2 diabetics |
| GRADE | ||||||||
|---|---|---|---|---|---|---|---|---|
| Outcomes | Effect (95% CI) | N (No. Studies) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Certainty of Evidence |
| Total Cholesterol (mmol/L) | SMD 0.50 lower (0.70 lower to 0.29 lower) | 384 (10) | ⊕⊕⊕◯ MODERATE | |||||
| LDL Cholesterol (mmol/L) | SMD 0.39 lower (0.57 lower to 0.21 lower) | 420 (11) | ⊕⊕◯◯ LOW | |||||
| HDL Cholesterol (mmol/L) | SMD 0.01 higher (0.02 lower to 0.05 higher) | 400 (10) | ⊕⊕⊕◯ MODERATE | |||||
| Triglycerides (mmol/L) | SMD 0.15 lower (0.29 lower to 0.01 lower) | 420 (11) | ⊕⊕◯◯ LOW | |||||
| Fasting Blood Glucose (mmol/L) | SMD 0.08 lower (0.23 lower to 0.08 higher) | 231 (6) | ⊕◯◯◯ VERY LOW | |||||
| Systolic Blood Pressure (mmHg) | SMD 0.32 lower (1.79 lower to 1.14 higher) | 339 (7) | ⊕⊕◯◯ LOW | |||||
| Diastolic Blood Pressure (mmHg) | SMD 0.49 higher (0.30 lower to 1.28 higher) | 339 (7) | ⊕⊕◯◯ LOW | |||||
| Weight (Kg) | SMD 0.12 lower (1.52 lower to 1.27 higher) | 294 (5) | ⊕⊕◯◯ LOW | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gibbs, J.; Leung, G.-K. The Effect of Plant-Based and Mycoprotein-Based Meat Substitute Consumption on Cardiometabolic Risk Factors: A Systematic Review and Meta-Analysis of Controlled Intervention Trials. Dietetics 2023, 2, 104-122. https://doi.org/10.3390/dietetics2010009
Gibbs J, Leung G-K. The Effect of Plant-Based and Mycoprotein-Based Meat Substitute Consumption on Cardiometabolic Risk Factors: A Systematic Review and Meta-Analysis of Controlled Intervention Trials. Dietetics. 2023; 2(1):104-122. https://doi.org/10.3390/dietetics2010009
Chicago/Turabian StyleGibbs, Joshua, and Gah-Kai Leung. 2023. "The Effect of Plant-Based and Mycoprotein-Based Meat Substitute Consumption on Cardiometabolic Risk Factors: A Systematic Review and Meta-Analysis of Controlled Intervention Trials" Dietetics 2, no. 1: 104-122. https://doi.org/10.3390/dietetics2010009
APA StyleGibbs, J., & Leung, G.-K. (2023). The Effect of Plant-Based and Mycoprotein-Based Meat Substitute Consumption on Cardiometabolic Risk Factors: A Systematic Review and Meta-Analysis of Controlled Intervention Trials. Dietetics, 2(1), 104-122. https://doi.org/10.3390/dietetics2010009





