Ion Exchange in Natural Clinoptilolite: Aspects Related to Its Structure and Applications
Abstract
:1. Introduction
2. Clinoptilolite–Heulandite Series: Classification and Composition Relation
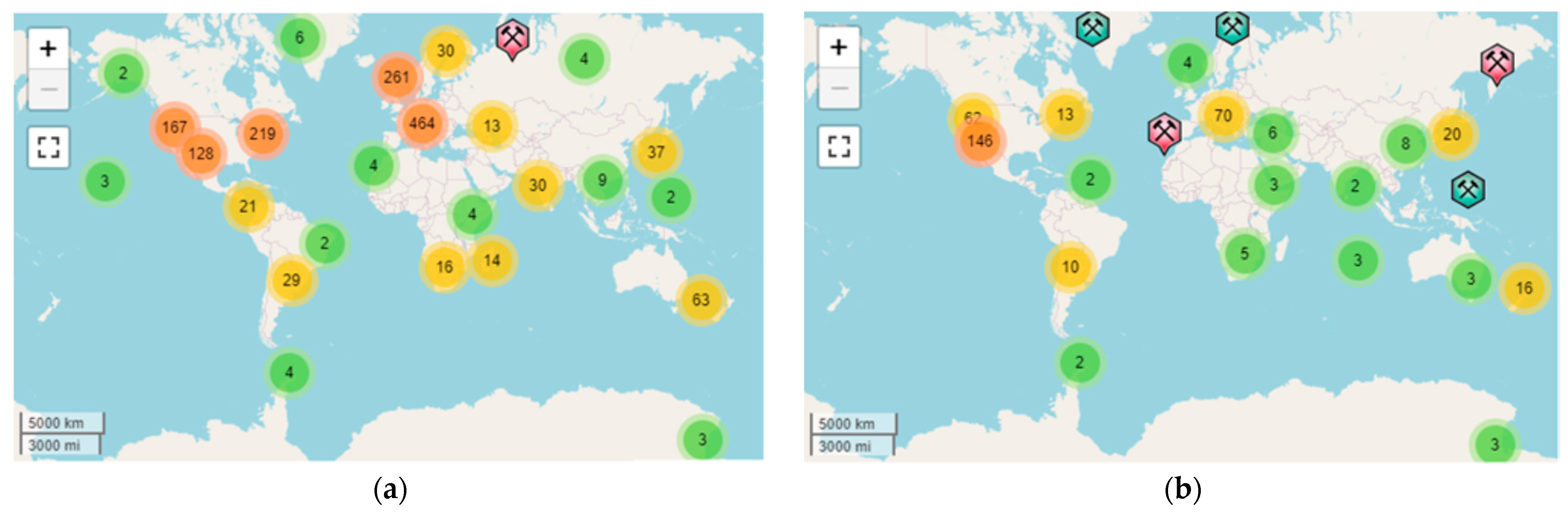
2.1. Main Deposits, Stratification, Impurities
2.2. Composition, Properties, Si/Al, Cationic Pool, Relationship with HEU/CLI Recognition
2.3. Options for Synthetic Clinoptilolite
3. The Ion Exchange Property and the Extra-Framework Cations in Natural Clinoptilolites
3.1. Effect of Initially Present Cations on Ion exchange Properties
3.2. Interplay between Nature and Location of the Exchanged Cation, Its Effects on Adsorption, Zeolite Acidity, etc.
3.3. Strategies for Ion Exchange Modifications
4. Applications of Natural Clinoptilolites Based on Their Ion Exchange Properties
4.1. Removal of Cationic Contaminants from Water
4.1.1. Removal of Heavy Metals
4.1.2. Removal of Dissolved Ammonia
4.1.3. Removal of Contaminants from Organic Solutions
4.2. Storage and Conversion of Undesirable Gas Emissions into the Atmosphere
4.3. Production of Catalysts and Photocatalysts
4.4. Bioactive Materials
4.5. Agricultural Applications
4.6. Zeolites as a Nature-Forming Factor
5. Challenges and Prospects for Innovation
6. Summary
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- EFSA CEF Panel. Scientific Opinion on the safety evaluation of the substance, silver zeolite A (silver zinc sodium ammonium alumino silicate), silver content 2–5 %, for use in food contact materials. EFSA J. 2011, 9, 1999. [Google Scholar] [CrossRef] [Green Version]
- EFSA CEF Panel. Scientific Opinion on the safety evaluation of the active substances iron, sodium chloride, water, silica gel, activated carbon, monosodium glutamate, potassium acid tartrate, powdered cellulose, malic acid, chabazite, hydroxypropyl cellulose, potassium carbonate, sodium thiosulfate, propylene glycol, glycerin, polyethyleneglycol sorbitan monooleate, sodium propionate and clinoptilolite for use in food contact materials. EFSA J. 2013, 11, 3155. [Google Scholar] [CrossRef]
- Services, H. Substances generally recognized as safe. A rule by the Food and Drug Administration on 08/17/2016. Fed. Regist. 2016, 81, 54960–55055. [Google Scholar]
- United States Food and Drug Administration. 2015; Code of Federal Regulations, Title 21. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/cfrsearch.cfm?fr=176.170 (accessed on 11 November 2022).
- United States Food and Drug Administration. 2022; CFR—Code of Federal Regulations Title 21. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/cfrsearch.cfm (accessed on 11 November 2022).
- Eroglu, N. A review: Insecticidal potential of Zeolite (Clinoptilolite), toxicity ratings and general properties of Turkish Zeolites. In Proceedings of the 11th International Working Conference on Stored Product Protection, Chiang Mai, Thailand, 24–28 November 2014; pp. 755–767. [Google Scholar]
- International Agency for Research on Cancer (IARC). Zeolites other than erionite. In IARC Monographs; IARC Publications: Lyon, France, 1997; Volume 68, pp. 307–333. [Google Scholar]
- International Agency for Research on Cancer Agents Classified by the IARC Monographs, Volumes 1–132. [Homepage on the Internet] Lyon: IARC. [Updated August 12, 2022]. Available online: https://monographs.iarc.who.int/agents-classified-by-the-iarc/ (accessed on 11 November 2022).
- International Agency for Research on Cancer Preamble to the IARC Monographs—Scientific Review and Evaluation [Homepage on the Internet] Lyon: IARC. [Updated February 19, 2021]. Available online: https://monographs.iarc.who.int/iarc-monographs-preamble-preamble-to-the-iarc-monographs/ (accessed on 11 November 2022).
- Burnett, C.L. Safety Assessment of Camellia sinensis-Derived Ingredients as Used in cosmetics Status. Draft tentative Report for panel Review. Available online: https://www.cir-safety.org/sites/default/files/Zeolites.pdf (accessed on 11 November 2022).
- Rodríguez-Fuentes, G.; Barrios A Iraizoz, M.A.; Perdomo, I.; Cedré, B. Enterex: Anti-diarrheic drug based on purified natural clinoptilolite. Zeolites 1997, 19, 441–448. [Google Scholar] [CrossRef]
- Rodríguez-Fuentes, G.; Denis, A.R.; Barrios Álvarez, M.A.; Colarte, A.I. Antacid drug based on purified natural clinoptilolite. Microporous Mesoporous Mater. 2006, 94, 200–207. [Google Scholar] [CrossRef]
- Rivera, A.; Rodríguez-Fuentes, G.; Altshuler, E. Characterization and neutralizing properties of a natural zeolite/Na2CO3 composite material. Microporous Mesoporous Mater. 1998, 24, 51–58. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez-Fuentes, G.; Rodríguez Iznaga, I.; Boza, A.; Anaisa, P.; Cedré, B.; Bravo-Fariñas, L.; Ruiz, A.; Fernández-Abreu, A.; Sende Odoardo, V. Evaluation of a zinc clinoptilolite (ZZ®) for drinking water treatment. In Aquananotechnology. Global Prospects; Reisner, D.E., Pradeep, T., Eds.; CRC Press: Boca Raton, FL, USA, 2014; p. 26. ISBN 9780429185632. [Google Scholar]
- Colella, C. A critical reconsideration of biomedical and veterinary applications of natural zeolites. Clay Miner. 2011, 46, 295–309. [Google Scholar] [CrossRef]
- Rodríguez-Iznaga, I.; Rodríguez-Fuentes, G.; Petranovskii, V. Ammonium modified natural clinoptilolite to remove manganese, cobalt and nickel ions from wastewater: Favorable conditions to the modification and selectivity to the cations. Microporous Mesoporous Mater. 2018, 255, 200–210. [Google Scholar] [CrossRef]
- Rodríguez-Iznaga, I.; Petranovskii, V.; Rodríguez-Fuentes, G. Ion-exchange of amino- and aqua-complexes of nickel and cobalt in natural clinoptilolite. J. Environ. Chem. Eng. 2014, 2, 1221–1227. [Google Scholar] [CrossRef]
- Rodríguez Iznaga, I.; Petranovskii, V.; Rodríguez Fuentes, G.; Mendoza, C.; Benítez Aguilar, A. Exchange and reduction of Cu2+ ions in clinoptilolite. J. Colloid Interface Sci. 2007, 316, 877–886. [Google Scholar] [CrossRef]
- Rodríguez-Iznaga, I.; Petranovskii, V.; Castillón-Barraza, F.; Concepción-Rosabal, B. Copper-silver bimetallic system on natural clinoptilolite: Thermal reduction of Cu2+ and Ag+ exchanged. J. Nanosci. Nanotechnol. 2011, 11, 5580–5586. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Iznaga, I.; Petranovskii, V.; Chávez-Rivas, F.; Shelyapina, M.G. Bimetallic copper-silver systems supported on natural clinoptilolite: Long-term changes in nanospecies’ composition and stability. Inorganics 2022, 10, 34. [Google Scholar] [CrossRef]
- Derbe, T.; Temesgen, S.; Bitew, M. A short review on synthesis, characterization, and applications of zeolites. Adv. Mater. Sci. Eng. 2021, 2021, 6637898. [Google Scholar] [CrossRef]
- Xu, H.; Wu, P. New progress in zeolite synthesis and catalysis. Natl Sci Rev 2022, 9, nwac045. [Google Scholar] [CrossRef]
- Sharma, V.; Javed, B.; Byrne, H.; Curtin, J.; Tian, F. Zeolites as carriers of nano-fertilizers: From structures and principles to prospects and challenges. Appl. Nano 2022, 3, 163–186. [Google Scholar] [CrossRef]
- Ivankovic, T.; Dikic, J.; Du Roscoat, S.R.; Dekic, S.; Hrenovic, J.; Ganjto, M. Removal of emerging pathogenic bacteria using metal-exchanged natural zeolite bead filter. Water Sci. Technol. 2019, 80, 1085–1098. [Google Scholar] [CrossRef] [Green Version]
- Nizam, M.K.; Azhari, S.; Tamar Jaya, M.A. Modified zeolite as purification material in wastewater treatment: A review. Sci. Res. J. 2021, 18, 177–213. [Google Scholar] [CrossRef]
- US Geological Survey. Mineral Commodity Summaries; Government Printing Office: Washington, DC, USA, 2009. [Google Scholar]
- National Minerals Information Center Zeolites Statistics and Information. Available online: https://www.usgs.gov/centers/national-minerals-information-center/zeolites-statistics-and-information (accessed on 11 November 2022).
- Nesterenko, A.V.; Nesterenko, V.B.; Yablokov, A.V. Radiation protection after the chernobyl catastrophe. Ann. N. Y. Acad. Sci. 2009, 1181, 287–327. [Google Scholar] [CrossRef]
- Dyer, A. Applications of natural zeolites in the treatment of nuclear wastes and fall-out. In Environmental Mineralogy: Microbial Interactions, Anthropogenic Influences, Contaminated Land and Waste Management; Cotter-Howell, J.D., Campbell, L.S., Valsami-Jones, E., Batchelder, M., Eds.; Mineralogical Society of Great Britain and Ireland: London, UK, 2000; Volume 9, pp. 319–368. ISBN 9780903056410. [Google Scholar]
- Belyakov, A. From Chernobyl to Fukushima: An interdisciplinary framework for managing and communicating food security risks after nuclear plant accidents. J. Environ. Stud. Sci. 2015, 5, 404–417. [Google Scholar] [CrossRef]
- Hijikata, T.; Koyama, T.; Aikyo, Y.; Shimura, S.; Kawanishi, M. Strontium adsorption characteristics of natural Zeolites for permeable reactive barrier in Fukushima Daiichi nuclear power station. J. Nucl. Sci. Technol. 2021, 58, 1079–1098. [Google Scholar] [CrossRef]
- Zhang, Y.; Cao, B.; Yin, H.; Meng, L.; Jin, W.; Wang, F.; Xu, J.; Al-Tabbaa, A. Application of zeolites in permeable reactive barriers (PRBs) for in-situ groundwater remediation: A critical review. Chemosphere 2022, 308, 136290. [Google Scholar] [CrossRef]
- Baerlocher, C.; McCusker, L.B. Database of Zeolite Structures. Available online: http://www.iza-structure.org/databases/ (accessed on 11 November 2022).
- Warr, L.N. IMA–CNMNC approved mineral symbols. Miner. Mag. 2021, 85, 291–320. [Google Scholar] [CrossRef]
- Kretz, R. Symbols for rock-forming minerals. Am. Miner. 1983, 68, 277–279. [Google Scholar]
- Iijima, A.; Utada, M. Zeolites in sedimentary rocks, with reference to the depositional environments and zonal iistribution. Sedimentology 1966, 7, 327–357. [Google Scholar] [CrossRef]
- Sheppard, R.A.; Gude, A.J. Diagenesis of Tuffs in the Barstow Formation, Mud Hills, San Bernardino County, California. Physical Properties, Chemistry, and Origin of silicate Minerals Formed in silicic Tuffs of a lacustrine Deposit; United States Government Printing Office: Washington, DC, USA, 1969; Volume 634. [Google Scholar]
- Mumpton, F.A. La roca magica: Uses of natural zeolites in agriculture and industry. Proc. Natl. Acad. Sci. USA 1999, 96, 3463–3470. [Google Scholar] [CrossRef] [Green Version]
- Misaelides, P. Application of natural zeolites in environmental remediation: A short review. Microporous Mesoporous Mater. 2011, 144, 15–18. [Google Scholar] [CrossRef]
- Tankersley, K.B.; Dunning, N.P.; Carr, C.; Lentz, D.L.; Scarborough, V.L. Zeolite water purification at Tikal, an ancient Maya city in Guatemala. Sci. Rep. 2020, 10, 18021. [Google Scholar] [CrossRef] [PubMed]
- Serati-Nouri, H.; Jafari, A.; Roshangar, L.; Dadashpour, M.; Pilehvar-Soltanahmadi, Y.; Zarghami, N. Biomedical applications of zeolite-based materials: A review. Mater. Sci. Eng. C 2020, 116, 111225. [Google Scholar] [CrossRef]
- Pavelić, S.K.; Medica, J.S.; Gumbarević, D.; Filošević, A.; Pržulj, N.; Pavelić, K. Critical review on zeolite clinoptilolite safety and medical applications in vivo. Front. Pharmacol. 2018, 9, 1350. [Google Scholar] [CrossRef]
- Petranovskii, V.; Chaves-Rivas, F.; Hernandez Espinoza, M.A.; Pestryakov, A.; Kolobova, E. Potential uses of natural zeolites for the development of new materials: Short review. MATEC Web Conf. 2016, 85, 01014. [Google Scholar] [CrossRef] [Green Version]
- Market Analysis Report. Zeolite Market Size, Share & Trends Analysis Report by Application (Catalyst, Adsorbent, Detergent Builder), by Product (Natural, Synthetic), By Region (North America, Europe, APAC, CSA, MEA), and Segment Forecasts, 2022–2030. Available online: https://www.grandviewresearch.com/industry-analysis/zeolites-market (accessed on 2 November 2022).
- Hernández Espinosa, J.M.Á. El ABC de las Zeolitas Naturales en México; Instituto de Ciencias-BUAP. Publica por Gandhi: Puebla, Mexico, 2020; ISBN 978-607-8738-00-7. [Google Scholar]
- Pabiś-Mazgaj, E.; Gawenda, T.; Pichniarczyk, P.; Stempkowska, A. Mineral composition and structural characterization of the clinoptilolite powders obtained from zeolite-rich tuffs. Minerals 2021, 11, 1030. [Google Scholar] [CrossRef]
- Zvereva, I.; Kremnev, R.; Sirotov, V.; Rodríguez-Iznaga, I.; Hernández, M.-A.; Petranovskii, V. Thermal analysis and porosimetry of natural zeolites from mexican and cuban deposits. Appl. Solid State Chem. 2017, 1, 35–41. [Google Scholar] [CrossRef]
- Zvereva, I.A.; Shelyapina, M.G.; Chislov, M.; Novakowski, V.; Malygina, E.; Rodríguez-Iznaga, I.; Hernández, M.A.; Petranovskii, V. A comparative analysis of natural zeolites from various Cuban and Mexican deposits: Structure, composition, thermal properties and hierarchical porosity. J. Therm. Anal. Calorim. 2022, 147, 6147–6159. [Google Scholar] [CrossRef]
- The Hudson Institute of Mineralogy. Available online: https://www.mindat.org/a/hudsoninstituteofmineralogy (accessed on 11 November 2022).
- Heulandite. Available online: https://www.mindat.org/min-1889.html (accessed on 11 November 2022).
- Clinoptilolite. Available online: https://www.mindat.org/min-1082.html (accessed on 11 November 2022).
- Stocker, K.; Ellersdorfer, M.; Lehner, M.; Raith, J.G. Characterization and utilization of natural zeolites in technical applications. Berg-Und Hüttenmännische Mon. 2017, 162, 142–147. [Google Scholar] [CrossRef]
- Mumpton, F.A. Clinoptilolite redefined. Am. Miner. J. Earth Planet Mater. 1960, 45, 351–369. [Google Scholar]
- Iijima, A. Geology of natural zeolites and zeolitic rocks. Pure Appl. Chem. 1980, 52, 2115–2130. [Google Scholar] [CrossRef]
- Marantos, I.; Christidis, G.G.; Ulmanu, M. Zeolite formation and deposits. In Handbook of Natural Zeolites; Inglezakis, V.J., Zorpas, A.A., Eds.; Bentham Science Publishers: Sharjah, United Arab Emirates, 2012; pp. 28–51. ISBN 9781608054466. [Google Scholar]
- Koyama, K.; Takeuchi, Y. Clinoptilolite: The distribution of potassium atoms and its role in thermal stability. Z. Krist. 1977, 145, 216–239. [Google Scholar]
- Baerlocher, C.; McCusker, L.B.L.B.; Olson, D.H. Atlas of Zeolite Framework Types, 6th ed.; Elsevier B.V.: Amsterdam, The Netherlands; London, UK; New York, NY, USA; Oxford, UK; Paris, France; Shannon, Ireland; Tokyo, Japan, 2007. [Google Scholar]
- Wise, W.S. Handbook of natural zeolites. In International Zeolite Association; Collela, C., Ed.; De Frede Editore, Natural Zeolites Commission: Napoli, Italy, 2013; p. 354. [Google Scholar]
- IZA Commission on Natural Zeolites. Available online: http://www.iza-online.org/natural/Datasheets/Clinoptilolite/clinoptilolite.htm (accessed on 11 November 2022).
- IZA Commission on Natural Zeolites. Available online: http://www.iza-online.org/natural/Datasheets/Heulandite/heulandite.htm (accessed on 11 November 2022).
- Zhukov, Y.M.; Shelyapina, M.G.; Zvereva, I.A.; Efimov, A.Y.; Petranovskii, V. Microwave assisted versus convention Cu2+ exchange in mordenite. Microporous Mesoporous Mater. 2018, 259, 220–228. [Google Scholar] [CrossRef]
- Shelyapina, M.G.; Krylova, E.A.; Zhukov, Y.M.; Zvereva, I.A.; Rodriguez-Iznaga, I.; Petranovskii, V.; Fuentes-Moyado, S. Comprehensive analysis of the copper exchange implemented in ammonia and protonated forms of mordenite using microwave and conventional methods. Molecules 2019, 24, 4216. [Google Scholar] [CrossRef] [Green Version]
- Coombs, D.; Alberti, A.; Armbruster, T.; Artioli, G.; Colella, C.; Galli, E.; Grice, J.; Liebau, F.; Mandarino, J.; Minato, H.; et al. Recommended nomenclature for zeolite minerals: Report of the subcommittee on zeolites of the international mineralogical association, commission on new minerals and mineral names. Can Miner. 1997, 35, 1571–1606. [Google Scholar]
- Boles, J.R. Composition, optical properties, cell dimensions, and thermal stability of some heulandite group zeolites. Am. Miner. 1972, 57, 1463–1493. [Google Scholar]
- Alietti, A. Polymorphism and crystal-chemistry of heulandites and clinoptilolites. Am. Miner. 1972, 57, 1437–1462. [Google Scholar]
- Bish, D.L.; Boak, J.M. Clinoptilolite-heulandite nomenclature. Rev. Miner. Geochem. 2001, 45, 206–216. [Google Scholar] [CrossRef]
- Mason, B.; Sand, L.B. Clinoptilolite from Patagonia: The relationship between clinoptilolite and heulandite. Am. Miner. 1960, 45, 341–350. [Google Scholar]
- Bish, D. Effects of composition on the dehydration behavior of clinoptilolite and heulandite. In Properties and Utilization of Natural Zeolites; Kallo, D., Sherry, H., Eds.; Akademiai Kiado: Budapest, Hungary, 1988; pp. 565–576. [Google Scholar]
- Ward, R.L.; McKague, H.L. Clinoptilolite and heulandite structural differences as revealed by multinuclear nuclear magnetic resonance spectroscopy. J. Phys. Chem. 1994, 98, 1232–1237. [Google Scholar] [CrossRef]
- Drebushchak, V.A.; Naumov, V.N.; Nogteva, V.V.; Belitsky, I.A.; Paukov, I.E. Low-temperature heat capacity of heulandite: Comparison with clinoptilolite. Thermochim. Acta 2000, 348, 33–40. [Google Scholar] [CrossRef]
- Kantiranis, N.; Chrissafis, C.; Filippidis, A.; Paraskevopoulos, K.M. Thermal distinction of HEU-type mineral phases contained in Greek zeolite-rich volcaniclastic tuffs. Eur. J. Miner. 2006, 18, 509–516. [Google Scholar] [CrossRef]
- Seryotkin, Y.V. High-pressure behavior of HEU-type zeolites: X-ray diffraction study of clinoptilolite-Na. Microporous Mesoporous Mater. 2016, 235, 20–31. [Google Scholar] [CrossRef]
- Palmer, J.L.; Gunter, M.E. Optical properties of natural and cation-exchanged heulandite group zeolites. Am. Miner. 2000, 85, 225–230. [Google Scholar] [CrossRef]
- Bish, D.L.; Carey, J.W. Thermal behavior of natural zeolites. Rev. Miner. Geochem. 2001, 45, 403–452. [Google Scholar] [CrossRef]
- IZA Database Zeolite Structure, Materials with Framework Type HEU. Available online: https://asia.iza-structure.org/IZA-SC/material_rm.php?STC=HEU (accessed on 18 November 2022).
- Breck, D.W.; Skeels, G.W. Silicon Substituted Zeolite Compositions and Process for Preparing Same. U.S. Patent 4,503,023, 5 March 1985. [Google Scholar]
- Khodabandeh, S.; Davis, M.E. Synthesis of a heulandite-type zeolite by hydrothermal conversion of zeolite P1. Chem Commun 1996, Volume, 1205–1206. [Google Scholar] [CrossRef] [Green Version]
- Ambrozova, P.; Kynicky, J.; Urubek, T.; Nguyen, V.D. Synthesis and modification of clinoptilolite. Molecules 2017, 22, 1107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chávez-Rivas, F.; Rodríguez-Fuentes, G.; Berlier, G.; Rodríguez-Iznaga, I.; Petranovskii, V.; Zamorano-Ulloa, R.; Coluccia, S. Evidence for controlled insertion of Fe ions in the framework of clinoptilolite natural zeolites. Microporous Mesoporous Mater. 2013, 167, 76–81. [Google Scholar] [CrossRef]
- Kirov, G.; Dimova, L.; Stanimirova, T. Gallery character of porous space and local extra-framework configurations in the HEU-type structure. Microporous Mesoporous Mater. 2020, 293, 109792. [Google Scholar] [CrossRef]
- Lehto, J.; Harjula, R. Experimentation in ion exchange studies—The problem of getting reliable and comparable results. React. Funct. Polym. 1995, 27, 121–146. [Google Scholar] [CrossRef]
- Recommendations on ion exchange nomenclature. Pure Appl. Chem. 1972, 29, 617–624. [CrossRef] [Green Version]
- Inglezakis, V.J. The concept of “capacity” in zeolite ion-exchange systems. J. Colloid Interface Sci. 2005, 281, 68–79. [Google Scholar] [CrossRef]
- Caputo, D.; Pepe, F. Experiments and data processing of ion exchange equilibria involving Italian natural zeolites: A review. Microporous Mesoporous Mater. 2007, 105, 222–231. [Google Scholar] [CrossRef]
- Colella, C. Ion exchange equilibria in zeolite minerals. Min. Depos. 1996, 31, 554–562. [Google Scholar] [CrossRef]
- Eisenman, G. Cation selective glass electrodes and their mode of operation. Biophys. J. 1962, 2, 259–323. [Google Scholar] [CrossRef] [Green Version]
- Sherry, H.S. The ion-exchange properties of the zeolites. In Ion Exchange. A Series of Advances; Marinsky, J.A., Ed.; Marcel Dekker: New York, NY, USA, 1969; pp. 89–133. [Google Scholar]
- Barrer, R.M. Cation-exchange equilibria in zeolites and feldspathoids. In Natural Zeolites: Occurrence, Properties, Use; Sand, L.B., Mumpton, F.A., Eds.; Pergamon Press: New York, NY, USA, 1978; pp. 385–395. [Google Scholar]
- Rodríguez-Iznaga, I.; Gómez, A.; Rodríguez-Fuentes, G.; Benítez-Aguilar, A.; Serrano-Ballan, J. Natural clinoptilolite as an exchanger of Ni2+ and NH4+ ions under hydrothermal conditions and high ammonia concentration. Microporous Mesoporous Mater. 2002, 53, 71–80. [Google Scholar] [CrossRef]
- Rodríguez-Iznaga, I.; Rodríguez-Fuentes, G.; Benítez-Aguilar, A. Role of carbonate ions in the ion-exchange Ni2+ = 2NH4+ in natural clinoptilolite. Microporous Mesoporous Mater. 2000, 41, 129–136. [Google Scholar] [CrossRef]
- Garcia-Basabe, Y.; Gomez, A.; Rodriguez-Iznaga, I.; Montero, A.; Vlaic, G.; Lausi, A.; Ruiz-Salvador, A.R. Locating extra-framework cations in low-silica zeolites by a combinatorial approach of the direct space method and rietveld refinement: Application to Ni2+ and Co2+ enriched clinoptilolite. J. Phys. Chem. C 2010, 114, 5964–5974. [Google Scholar] [CrossRef]
- Garcia-Basabe, Y.; Ruiz-Salvador, A.R.; Maurin, G.; De Menorval, L.C.; Rodriguez-Iznaga, I.; Gomez, A. Location of extra-framework Co2+, Ni2+, Cu2+ and Zn2+ cations in natural and dealuminated clinoptilolite. Microporous Mesoporous Mater. 2012, 155, 233–239. [Google Scholar] [CrossRef]
- Travieso, N.; Labre, G.; Gonzales, J.A.; Arcoya, A.; Sedane, X. Propiedades de tamiz molecular de formas intercambiadas de clinoptilolita natural. In 3era Conferencia Internacional Sobre Ocurrencia, Propiedades y Usos de las Zeolitas Naturales; Rodrıguez-Fuentes, G., Gonzalez, J., Eds.; International Conference Center: Habana, Cuba, 1991; pp. 150–156. [Google Scholar]
- Inglezakis, V.J.; Loizidou, M.M.; Grigoropoulou, H.P. Ion exchange studies on natural and modified zeolites and the concept of exchange site accessibility. J. Colloid Interface Sci. 2004, 275, 570–576. [Google Scholar] [CrossRef] [PubMed]
- Nightingale, E.R. Phenomenological theory of ion solvation. Effective radii of hydrated ions. J. Phys. Chem. 1959, 63, 1381–1387. [Google Scholar] [CrossRef]
- Cotton, F.A.; Wilkinson, G. Advanced Inorganic Chemistry, 5th ed.; Wiley: New York, NY, USA, 1988. [Google Scholar]
- Huheey, J.E.; Keiter, E.A.; Keiter, R. Inorganic Chemistry: Principles of Structure and Reactivity, 4th ed.; Harper Collins College Publishers: New York, NY, USA, 1993. [Google Scholar]
- de las Pozas, C.; Lopez-Cordero, R.; Diaz-Aguila, C.; Cora, M.; Roque-Malherbe, R. Location and reducibility of Ni ions in HEU-Type zeolites. J. Solid State Chem. 1995, 114, 108–111. [Google Scholar] [CrossRef]
- Gorimbo, J.; Taenzana, B.; Kapanji, K.; Jewell, L.L. Equilibrium ion exchange studies of Ni2+ on homoionic forms of clinoptilolite. S. Afr. J. Sci. 2014, 110. [Google Scholar] [CrossRef]
- Armbruster, T.; Simoncic, P.; Döbelin, N.; Malsy, A.; Yang, P. Cu2+-acetate and Cu2+-ammine exchanged heulandite: A structural comparison. Microporous Mesoporous Mater. 2003, 57, 121–131. [Google Scholar] [CrossRef]
- Petrov, O.E. Cation exchange in clinoptilolite: An X-ray powder diffraction analysis. In Natural Zeolites ’93: Occurrence, Properties, Use; Ming, D.W., Mumpton, F.A., Eds.; International Committee on Natural Zeolites: Brockport, New York, NY, USA, 1995; p. 221. [Google Scholar]
- Dimowa, L.; Tzvetanova, Y. Powder XRD study of changes of Cd2+ modified clinoptilolite at different stages of the ion exchange process. Minerals 2021, 11, 1130. [Google Scholar] [CrossRef]
- Dimowa, L.T.; Petrov, O.E.; Tarasov, M.P.; Kadiyski, M.K. Structural study of Tl-exchanged natural clinoptilolite using Rietveld refinement. Bulg. Chem. Commun. 2017, 49, 46–52. [Google Scholar]
- Dimowa, L.; Tzvetanova, Y.; Petrov, O.; Piroeva, I.; Ublekov, F. Powder XRD structural study of Ba2+ modified clinoptilolite at different stages of the ion exchange process conducted at two temperature regimes—Room temperature and 90 °C. Minerals 2020, 10, 938. [Google Scholar] [CrossRef]
- Dimova, L.; Petrov, O.; Kadiyski, M.; Lihareva, N.; Stoyanova-Ivanova, A.; Mikli, V. Preparation and Rietveld refinement of Ag-exchanged clinoptilolite. Clay Miner. 2011, 46, 205–212. [Google Scholar] [CrossRef]
- Dimowa, L.T.; Petrov, O.E.; Djourelov, N.I.; Shivachev, B.L. Structural study of Zn-exchanged natural clinoptilolite using powder XRD and positron annihilation data. Clay Miner. 2015, 50, 41–54. [Google Scholar] [CrossRef]
- Cerjan Stefanović, Š.; Zabukovec Logar, N.; Margeta, K.; Novak Tušar, N.; Arčon, I.; Maver, K.; Kovač, J.; Kaučič, V. Structural investigation of Zn2+ sorption on clinoptilolite tuff from the Vranjska Banja deposit in Serbia. Microporous Mesoporous Mater. 2007, 105, 251–259. [Google Scholar] [CrossRef]
- Dimowa, L.T.; Petrov, S.L.; Shivachev, B.L. Natural and Zn exchanged clinoptilolite: In situ high temperature XRD study of structural behavior and cation positions. Bulg. Chem. Commun. 2013, 45, 466–473. [Google Scholar]
- Attanoos, R.L.; Churg, A.; Galateau-Salle, F.; Gibbs, A.R.; Roggli, V.L. Malignant mesothelioma and its non-asbestos causes. Arch. Pathol. Lab. Med. 2018, 142, 753–760. [Google Scholar] [CrossRef] [Green Version]
- Emri, S.A. The cappadocia mesothelioma epidemic: Its influence in Turkey and abroad. Ann. Transl. Med. 2017, 5, 239. [Google Scholar] [CrossRef] [Green Version]
- Ackley, M.W.; Rege, S.U.; Saxena, H. Application of natural zeolites in the purification and separation of gases. Microporous Mesoporous Mater. 2003, 61, 25–42. [Google Scholar] [CrossRef]
- Maesen, T.L.M.; Beerdsen, E.; Calero, S.; Dubbeldam, D.; Smit, B. Understanding cage effects in the n-alkane conversion on zeolites. J. Catal. 2006, 237, 278–290. [Google Scholar] [CrossRef]
- Pavelić, K.; Hadžija, M.; Bedrica, L.; Pavelić, J.; Crossed D signikić, I.; Katić, M.; Kralj, M.; Bosnar, M.H.; Kapitanović, S.; Poljak-Blaži, M.; et al. Natural zeolite clinoptilolite: New adjuvant in anticancer therapy. J. Mol. Med. 2000, 78, 708–720. [Google Scholar] [CrossRef] [PubMed]
- Briffa, J.; Sinagra, E.; Blundell, R. Heavy metal pollution in the environment and their toxicological effects on humans. Heliyon 2020, 6, e04691. [Google Scholar] [CrossRef] [PubMed]
- Suganthi, M.; Ramesh, N. Treatment of water using natural zeolite as membrane filter. J. Environ. Prot. Ecol. 2022, 23, 520–530. [Google Scholar]
- Qasem, N.A.A.; Mohammed, R.H.; Lawal, D.U. Removal of heavy metal ions from wastewater: A comprehensive and critical review. npj Clean Water 2021, 4, 36. [Google Scholar] [CrossRef]
- Baldwin, D.R.; Marshall, W.J. Heavy metal poisoning and its laboratory investigation. Ann. Clin. Biochem. 1999, 36, 267–300. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Li, L.; Yu, J. Applications of zeolites in sustainable chemistry. Chem 2017, 3, 928–949. [Google Scholar] [CrossRef] [Green Version]
- Yuna, Z. Review of the natural, modified, and synthetic zeolites for heavy metals removal from wastewater. Environ. Eng. Sci. 2016, 33, 443–454. [Google Scholar] [CrossRef]
- Arancibia-Miranda, N.; Baltazar, S.E.; García, A.; Muñoz-Lira, D.; Sepúlveda, P.; Rubio, M.A.; Altbir, D. Nanoscale zero valent supported by Zeolite and Montmorillonite: Template effect of the removal of lead ion from an aqueous solution. J. Hazard. Mater. 2016, 301, 371–380. [Google Scholar] [CrossRef]
- Senila, M.; Neag, E.; Cadar, O.; Kovacs, E.D.; Aschilean, I.; Kovacs, M.H. Simultaneous removal of heavy metals (Cu, Cd, Cr, Ni, Zn and Pb) from aqueous solutions using thermally treated Romanian zeolitic volcanic tuff. Molecules 2022, 27, 3938. [Google Scholar] [CrossRef]
- Katsou, E.; Malamis, S.; Tzanoudaki, M.; Haralambous, K.J.; Loizidou, M. Regeneration of natural zeolite polluted by lead and zinc in wastewater treatment systems. J. Hazard. Mater. 2011, 189, 773–786. [Google Scholar] [CrossRef]
- Shaheen, S.M.; Derbalah, A.S.; Moghanm, F.S. Removal of heavy metals from aqueous solution by zeolite in competitive sorption system. Int. J. Environ. Sci. Dev. 2012, 3, 362–367. [Google Scholar] [CrossRef]
- Panayotova, M.; Velikov, B. Influence of zeolite transformation in a homoionic form on the removal of some heavy metal ions from wastewater. J. Environ. Sci. Health-Part A Toxic/Hazard. Subst. Environ. Eng. 2003, 38, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Vassileva, P.; Voikova, D. Investigation on natural and pretreated Bulgarian clinoptilolite for ammonium ions removal from aqueous solutions. J. Hazard. Mater. 2009, 170, 948–953. [Google Scholar] [CrossRef] [PubMed]
- Taneva, N. Removal of ammonium and phosphates from aqueous solutions by activated and modified Bulgarian clinoptilolite. J. Chem. Eng. Mater. Sci. 2012, 3, 79–85. [Google Scholar] [CrossRef]
- Taffarel, S.R.; Rubio, J. On the removal of Mn2+ ions by adsorption onto natural and activated Chilean zeolites. Miner. Eng. 2009, 22, 336–343. [Google Scholar] [CrossRef]
- Rajic, N.; Stojakovic, D.; Jovanovic, M.; Logar, N.Z.; Mazaj, M.; Kaucic, V. Removal of nickel(II) ions from aqueous solutions using the natural clinoptilolite and preparation of nano-NiO on the exhausted clinoptilolite. Appl. Surf. Sci. 2010, 257, 1524–1532. [Google Scholar] [CrossRef]
- Figueiredo, H.; Quintelas, C. Tailored zeolites for the removal of metal oxyanions: Overcoming intrinsic limitations of zeolites. J. Hazard. Mater. 2014, 274, 287–299. [Google Scholar] [CrossRef] [Green Version]
- Elizondo-Villarreal, N.; Obregón-Guerra, R.; García-Méndez, M.; Sánchez-Espinoza, A.P.; Alcorta-García, M.A.; Torres-Barrera, R.O.; Coello, V.; Castaño, V.M. Nanomodification of a natural clinoptilolite zeolite. Rev. Adv. Mater. Sci. 2016, 47, 74–78. [Google Scholar]
- Du, G.; Li, Z.; Liao, L.; Hanson, R.; Leick, S.; Hoeppner, N.; Jiang, W.T. Cr(VI) retention and transport through Fe(III)-coated natural zeolite. J. Hazard. Mater. 2012, 221–222, 118–123. [Google Scholar] [CrossRef]
- Lv, G.; Li, Z.; Jiang, W.T.; Ackley, C.; Fenske, N.; Demarco, N. Removal of Cr(VI) from water using Fe(II)-modified natural zeolite. Chem. Eng. Res. Des. 2014, 92, 384–390. [Google Scholar] [CrossRef]
- Salgado-Gómez, N.; Macedo-Miranda, M.G.; Olguín, M.T. Chromium VI adsorption from sodium chromate and potassium dichromate aqueous systems by hexadecyltrimethylammonium-modified zeolite-rich tuff. Appl. Clay Sci. 2014, 95, 197–204. [Google Scholar] [CrossRef]
- Inglezakis, V.J.; Zorpas, A.A.; Loizidou, M.D.; Grigoropoulou, H.P. Simultaneous removal of metals Cu2+, Fe3+ and Cr3+ with anions SO42- and HPO42- using clinoptilolite. Microporous Mesoporous Mater. 2003, 61, 167–171. [Google Scholar] [CrossRef]
- European Environment Agency. Available online: https://www.eea.europa.eu/data-and-maps/explore-interactive-maps/wise-soe-ammonium-in-rivers (accessed on 15 November 2022).
- Molins-Legua, C.; Meseguer-Lloret, S.; Moliner-Martinez, Y.; Campíns-Falcó, P. A guide for selecting the most appropriate method for ammonium determination in water analysis. TrAC—Trends Anal. Chem. 2006, 25, 282–290. [Google Scholar] [CrossRef]
- Berenzen, N.; Schulz, R.; Liess, M. Effects of chronic ammonium and nitrite contamination on the macroinvertebrate community in running water microcosms. Water Res. 2001, 35, 3478–3482. [Google Scholar] [CrossRef]
- Saiapina, O.; Vronska, Y.; Dzyadevych, S.; Jaffrezic-Renault, N. Clinoptilolite-based Conductometric Sensors for Detection of Ammonium in Aqueous Solutions. Electroanalysis 2020, 32, 1993–2001. [Google Scholar] [CrossRef]
- Valdés, M.G.; Pérez-Cordoves, A.I.; Díaz-García, M.E. Zeolites and zeolite-based materials in analytical chemistry. TrAC—Trends Anal. Chem. 2006, 25, 24–30. [Google Scholar] [CrossRef]
- Walcarius, A. Zeolite-modified electrodes in electroanalytical chemistry. Anal. Chim. Acta 1999, 384, 1–16. [Google Scholar] [CrossRef]
- Saiapina, O.Y.; Pyeshkova, V.M.; Soldatkin, O.O.; Melnik, V.G.; Kurç, B.A.; Walcarius, A.; Dzyadevych, S.V.; Jaffrezic-Renault, N. Conductometric enzyme biosensors based on natural zeolite clinoptilolite for urea determination. Mater. Sci. Eng. C 2011, 31, 1490–1497. [Google Scholar] [CrossRef]
- Guida, S.; Potter, C.; Jefferson, B.; Soares, A. Preparation and evaluation of zeolites for ammonium removal from municipal wastewater through ion exchange process. Sci. Rep. 2020, 10, 12426. [Google Scholar] [CrossRef]
- Inglezakis, V.J.; Loizidou, M.D. Ion exchange of some heavy metal ions from polar organicsolvents into zeolite. Desalination 2007, 211, 238–248. [Google Scholar] [CrossRef]
- Shelyapina, M.G.; Rodríguez-Iznaga, I.; Petranovskii, V. Materials for CO2, SOx, and NOx emission reduction. In Handbook of Nanomaterials and Nanocomposites for Energy and Environmental Applications; Kharissova, O.V., Martínez, L.M.T., Kharisov, B.I., Eds.; Springer: Cham, Switzerland, 2020; pp. 1–30. ISBN 978-3-030-11155-7. [Google Scholar]
- Bai, R.; Song, X.; Yan, W.; Yu, J. Low-energy adsorptive separation by zeolites. Natl. Sci. Rev. 2022, 9, nwac064. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Yu, Y.; Li, Y.; Min, X.; Zhang, J.; Sun, T. Methane separation and capture from nitrogen rich gases by selective adsorption in microporous materials: A review. Sep. Purif. Technol. 2022, 283, 120206. [Google Scholar] [CrossRef]
- Hoang, H.G.; Thuy, B.T.P.; Lin, C.; Vo, D.-V.N.; Tran, H.T.; Bahari, M.B.; Le, V.G.; Vu, C.T. The nitrogen cycle and mitigation strategies for nitrogen loss during organic waste composting: A review. Chemosphere 2022, 300, 134514. [Google Scholar] [CrossRef]
- Manu, M.K.; Wang, C.; Li, D.; Varjani, S.; Wong, J.W.C. Impact of zeolite amendment on composting of food waste digestate. J. Clean Prod. 2022, 371, 133408. [Google Scholar] [CrossRef]
- Mehrabadi, Z.; Faghihian, H. Clinoptilolite modified with TiO2 for simultaneous elimination of two herbicides; 2,4-D and MCPA by UV and sunlight-assisted photocatalytic degradation. Mater. Res. Bull. 2019, 119, 110569. [Google Scholar] [CrossRef]
- Nikazar, M.; Gholivand, K.; Mahanpoor, K. Using TiO2 supported on Clinoptilolite as a catalyst for photocatalytic degradation of azo dye Disperse yellow 23 in water. Kinet. Catal. 2007, 48, 214–220. [Google Scholar] [CrossRef]
- Sene, R.A.; Sharifnia, S.; Moradi, G.R. On the impact evaluation of various chemical treatments of support on the photocatalytic properties and hydrogen evolution of sonochemically synthesized TiO2/Clinoptilolite. Int. J. Hydrog. Energy 2018, 43, 695–707. [Google Scholar] [CrossRef]
- Nezamzadeh-Ejhieh, A.; Moazzeni, N. Sunlight photodecolorization of a mixture of Methyl Orange and Bromocresol Green by CuS incorporated in a clinoptilolite zeolite as a heterogeneous catalyst. J. Ind. Eng. Chem. 2013, 19, 1433–1442. [Google Scholar] [CrossRef]
- Petranovskii, V.; Pestryakov, A.N.; Kazantseva, L.K.; Cruz, J.; Kryazhov, A.N. Formation of catalytically active copper and nickel nanoparticles in natural zeolites. Stud. Surf. Sci. Catal. 2008, 174, 513–516. [Google Scholar] [CrossRef]
- Petranovskii, V.P.; Pestryakov, A.N.; Kazantseva, L.K.; Castillon Barraza, F.; Farias, M.H. Formation of catalytically active copper nanoparticles in natural zeolites for complete oxidation of hydrocarbons. Int. J. Mod. Phys. B 2005, 19, 2333–2338. [Google Scholar] [CrossRef]
- Azimi, S.C.; Shirini, F.; Pendashteh, A.R. Advanced oxidation process as a green technology for dyes removal from wastewater: A review. Iran. J. Chem. Chem. Eng. 2021, 40, 1467–1489. [Google Scholar] [CrossRef]
- Sodha, V.; Shahabuddin, S.; Gaur, R.; Ahmad, I.; Bandyopadhyay, R.; Sridewi, N. Comprehensive review on zeolite-based nanocomposites for treatment of effluents from wastewater. Nanomaterials 2022, 12, 3199. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Aguiñaga, E.A.; Elizalde-González, M.P.; Sabinas-Hernández, S.A. Unpredicted photocatalytic activity of clinoptilolite-mordenite natural zeolite. RSC Adv. 2020, 10, 39251–39260. [Google Scholar] [CrossRef] [PubMed]
- Sydorchuk, V.; Vasylechko, V.; Khyzhun, O.; Gryshchouk, G.; Khalameida, S.; Vasylechko, L. Effect of high-energy milling on the structure, some physicochemical and photocatalytic properties of clinoptilolite. Appl. Catal. A Gen. 2021, 610, 117930. [Google Scholar] [CrossRef]
- Saramok, M.; Szymaszek, A.; Inger, M.; Antoniak-Jurak, K.; Samojeden, B.; Motak, M. Modified zeolite catalyst for a NOx selective catalytic reduction process in nitric acid plants. Catalysts 2021, 11, 450. [Google Scholar] [CrossRef]
- Sobuś, N.; Król, M.; Piotrowski, M.; Michorczyk, B.; Czekaj, I.; Kornaus, K.; Trenczek-Zając, A.; Komarek, S. Conversion of dihydroxyacetone to carboxylic acids on pretreated clinoptilolite modified with iron, copper, and cobalt. Catal. Commun. 2022, 171, 106509. [Google Scholar] [CrossRef]
- Mačala, J.; Pandová, I. Natural zeolite-clinoptilolite—Raw material serviceable in the reduction of toxical components at combustion engines noxious gases. Gospod. Surowcami Miner. 2007, 23, 19–26. [Google Scholar]
- Moreno-Tost, R.; Santamaría-González, J.; Rodríguez-Castellón, E.; Jiménez-López, A.; Autié, M.A.; Glacial, M.C.; Castro, G.A.; Guerra, M. Selective catalytic reduction of nitric oxide by ammonia over Ag and Zn-exchanged Cuban natural zeolites. Z. Für Anorg. Und Allg. Chem. 2005, 631, 2253–2257. [Google Scholar] [CrossRef]
- Ates, A.; Reitzmann, A.; Hardacre, C.; Yalcin, H. Abatement of nitrous oxide over natural and iron modified natural zeolites. Appl. Catal. A Gen. 2011, 407, 67–75. [Google Scholar] [CrossRef]
- Godelitsas, A.; Armbruster, T. HEU-type zeolites modified by transition elements and lead. Microporous Mesoporous Mater. 2003, 61, 3–24. [Google Scholar] [CrossRef]
- Cerri, G.; de Gennaro, M.; Bonferoni, M.C.; Caramella, C. Zeolites in biomedical application: Zn-exchanged clinoptilolite-rich rock as active carrier for antibiotics in anti-acne topical therapy. Appl. Clay Sci. 2004, 27, 141–150. [Google Scholar] [CrossRef]
- Park, H.J.; Lim, H.M. Antimicrobial properties of Ag-exchanged natural and synthetic zeolites: A short review. Curr. Green Chem. 2015, 2, 354–361. [Google Scholar] [CrossRef]
- Guerra, R.; Lima, E.; Viniegra, M.; Guzmán, A.; Lara, V. Growth of Escherichia coli and Salmonella typhi inhibited by fractal silver nanoparticles supported on zeolites. Microporous Mesoporous Mater. 2012, 147, 267–273. [Google Scholar] [CrossRef]
- De Gennaro, M.; Cerri, G.; Caramella, C.M.; Bonferoni, M.C. Pharmaceutical Zeolite-Based Compositions Containing Zinc and Erythromycin, to be Used in the Treatment of Acne. International Patent WO2002100420A2, 19 December 2002. [Google Scholar]
- Vergara-Figueroa, J.; Alejandro-Martín, S.; Pesenti, H.; Cerda, F.; Fernández-Pérez, A.; Gacitúa, W. Obtaining nanoparticles of chilean natural zeolite and its ion exchange with copper salt (Cu2+) for antibacterial applications. Materials 2019, 12, 2202. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guidelines for Drinking-Water Quality: 4th Edition, Incorporating the 1st Addendum; World Health Organization: Geneva, Switzerland, 2017; ISBN 978-92-4-154995-0.
- de Campos Bernardi, A.C.; Polidoro, J.C.; de Melo Monte, M.B.; Pereira, E.I.; de Oliveira, C.R.; Ramesh, K. Enhancing nutrient use efficiency using zeolites minerals—A review. Adv. Chem. Eng. Sci. 2016, 6, 295–304. [Google Scholar] [CrossRef]
- Pal, D.K.; Wani, S.P.; Sahrawat, K.L. Zeolitic soils of the Deccan basalt areas in India: Their pedology and edaphology. Curr. Sci. 2013, 105, 309–318. [Google Scholar]
- Brito-Rojas, A.D.; Coutín-Correa, D.P.; Rodríguez-Fuentes, G. Volcanogenic Sedimentary Zeolite Deposits in Cuba. Report to the Commission on Natural Zeolite of the International Zeolite Association (IZA). Available online: http://www.iza-online.org/natural/Catalog/Cuba.pdf (accessed on 19 November 2022).
- Fatmah Siregar, A.F.; Sipahutar, I.A.; Masunaga, T. Beneficial effect of silicon application and intermittent irrigation on improving rice productivity in Indonesia. Indones. J. Agron. 2020, 48, 15–21. [Google Scholar] [CrossRef]
- Alhousari, F.; Greger, M. Silicon and mechanisms of plant resistance to insect pests. Plants 2018, 7, 33. [Google Scholar] [CrossRef] [Green Version]
- Bakhat, H.F.; Bibi, N.; Zia, Z.; Abbas, S.; Hammad, H.M.; Fahad, S.; Ashraf, M.R.; Shah, G.M.; Rabbani, F.; Saeed, S. Silicon mitigates biotic stresses in crop plants: A review. Crop Prot. 2018, 104, 21–34. [Google Scholar] [CrossRef]
- Eugercios Silva, A.R.; Álvarez-Cobelas, M.; Montero González, E. Impactos del nitrógeno agrícola en los ecosistemas acuáticos. Ecosistemas 2017, 26, 37–44. [Google Scholar] [CrossRef] [Green Version]
- Latifah, O.; Ahmed, O.H.; Majid, N.M.A. Enhancing nitrogen availability from urea using clinoptilolite zeolite. Geoderma 2017, 306, 152–159. [Google Scholar] [CrossRef]
- Souza, I.M.S.; Gurgel, G.C.S.; Medeiros, A.M.; Zonta, E.; Ruiz, J.A.C.; Paskocimas, C.A.; Motta, F.V.; Bomio, M.R.D. The use of clinoptilolite as carrier of nitrogened fertilizer with controlled release. J. Environ. Chem. Eng. 2018, 6, 4171–4177. [Google Scholar] [CrossRef]
- Omar, L.; Ahmed, O.H.; Jalloh, M.B.; Muhamad, A.M.N. Soil nitrogen fractions, nitrogen use efficiency and yield of Zea mays L. Grown on a tropical acid soil treated with composts and clinoptilolite zeolite. Appl. Sci. 2020, 10, 4139. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, Y.; Chen, H.; Wu, Q.; Chi, D. Assessing the performance of clinoptilolite for controlling and releasing ammonium in agricultural applications. Energy Rep. 2021, 7, 887–895. [Google Scholar] [CrossRef]
- International Zeolite. Available online: https://internationalzeolite.com/nerea/ (accessed on 5 November 2022).
- Mihok, F.; Macko, J.; Oriňak, A.; Oriňaková, R.; Kovaľ, K.; Sisáková, K.; Petruš, O.; Kostecká, Z. Controlled nitrogen release fertilizer based on zeolite clinoptilolite: Study of preparation process and release properties using molecular dynamics. Curr. Res. Green Sustain. Chem. 2020, 3, 100030. [Google Scholar] [CrossRef]
- Tzanakakis, V.A.; Monokrousos, N.; Chatzistathis, T. Effects of clinoptilolite zeolite and vermiculite on nitrification and nitrogen and phosphorus acquiring enzymes in a nitrogen applied agricultural soil. J. Soil Sci. Plant Nutr. 2021, 21, 2791–2802. [Google Scholar] [CrossRef]
- Noviello, M.; Gattullo, C.E.; Faccia, M.; Paradiso, V.M.; Gambacorta, G. Application of natural and synthetic zeolites in the oenological field. Food Res. Int. 2021, 150 Pt A, 110737. [Google Scholar] [CrossRef]
- Ríos Reyes, C.A.; Ríos Gutiéttez, M.P.; Joya Neira, S. The importance of minerals in medical geology: Impacts of the environment on health. Arch. Med. 2020, 21, 182–202. [Google Scholar] [CrossRef]
- International Medical Geology Association. Available online: https://medicalgeology.org/ (accessed on 15 November 2022).
- Gomes, C.D.S.F.; Silva, J.B.P. Minerals and clay minerals in medical geology. Appl. Clay Sci. 2007, 36, 4–21. [Google Scholar] [CrossRef]
- Farkhutdinov, I.; Farkhutdinova, L.; Zlobina, A.; Farkhutdinov, A.; Volfson, I.; Matveenko, I. Historical aspects of medical geology. Earth Sci. Hist. 2020, 39, 172–183. [Google Scholar] [CrossRef]
- Badora, A. The influence of zeolites on quality indicators of soil-plant connection and food safety. In Zeolites—Useful Minerals; Belviso, C., Ed.; IntechOpen: London, UK, 2016; pp. 203–215. [Google Scholar]
- Zhukov, Y.M.; Efimov, A.Y.; Shelyapina, M.G.; Petranovskii, V.; Zhizhin, E.V.; Burovikhina, A.; Zvereva, I.A. Effect of preparation method on the valence state and encirclement of copper exchange ions in mordenites. Microporous Mesoporous Mater. 2016, 224, 415–419. [Google Scholar] [CrossRef]
- Akdeniz, Y.; Ülkü, S. Microwave effect on ion-exchange and structure of clinoptilolite. J. Porous Mater. 2007, 14, 55–60. [Google Scholar] [CrossRef] [Green Version]
- Erten-Kaya, Y.; Cakicioglu-Ozkan, F. Effect of ultrasound on the kinetics of cation exchange in NaX zeolite. Ultrason. Sonochem. 2012, 19, 701–706. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Safari, S.; Khoshbin, R.; Karimzadeh, R. Beneficial use of ultrasound irradiation in synthesis of beta-clinoptilolite composite used in heavy oil upgrading process. RSC Adv. 2019, 9, 16797–16811. [Google Scholar] [CrossRef] [PubMed]
- Znak, Z.; Zin, O.; Mashtaler, A.; Korniy, S.; Sukhatskiy, Y.; Gogate, P.R.; Mnykh, R.; Thanekar, P. Improved modification of clinoptilolite with silver using ultrasonic radiation. Ultrason. Sonochem. 2021, 73, 105496. [Google Scholar] [CrossRef] [PubMed]








| Mineral Classification | Ref. [64] | Ref. [65] | ||
|---|---|---|---|---|
| Si/Al | ∑M++/∑M+ | Si/Al | ∑M++/∑M+ | |
| Heulandite | 2.90 ÷ 3.52 | 10.31 ÷ 1.61 | 2.85 ÷ 3.73 | 3.69 ÷ 0.88 |
| Intermediate | 3.57 ÷ 4.31 | 2.20 ÷ 0.43 | 3.45 ÷ 4.35 | 2.06 ÷ 0.73 |
| Clinoptilolite | 4.12 ÷ 5.10 | 0.60 ÷ 0.09 | 5.10 ÷ 5.20 | 0.37 ÷ 0.16 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Iznaga, I.; Shelyapina, M.G.; Petranovskii, V. Ion Exchange in Natural Clinoptilolite: Aspects Related to Its Structure and Applications. Minerals 2022, 12, 1628. https://doi.org/10.3390/min12121628
Rodríguez-Iznaga I, Shelyapina MG, Petranovskii V. Ion Exchange in Natural Clinoptilolite: Aspects Related to Its Structure and Applications. Minerals. 2022; 12(12):1628. https://doi.org/10.3390/min12121628
Chicago/Turabian StyleRodríguez-Iznaga, Inocente, Marina G. Shelyapina, and Vitalii Petranovskii. 2022. "Ion Exchange in Natural Clinoptilolite: Aspects Related to Its Structure and Applications" Minerals 12, no. 12: 1628. https://doi.org/10.3390/min12121628






