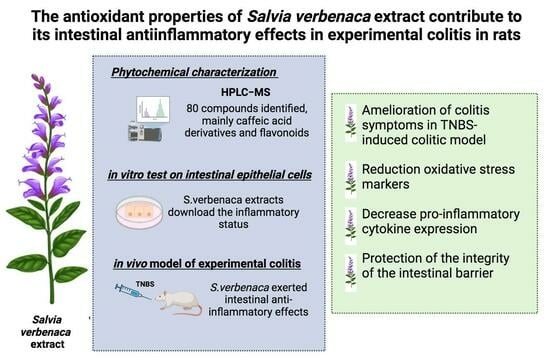
The Antioxidant Properties of Salvia verbenaca Extract Contribute to Its Intestinal Antiinflammatory Effects in Experimental Colitis in Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Plant Material and Preparation of the Extract
2.3. Chemical Composition of S. verbenaca Using High-Performance Liquid Chromatography-Mass Spectrometry Condition
2.4. Total Phenolic Content and Antioxidant Capacity Assays
2.5. In Vitro Studies
2.6. Animals and Experimental Design
2.7. Analysis of Gene Expression by RT-qPCR
2.8. Statistics
3. Results
3.1. Chemical Characterization of S. verbenaca
3.2. Antioxidant Capacity of S. verbenaca
3.3. Immunomodulatory Properties of S. verbenaca in Murine Intestinal Epithelial Cells
3.4. Intestinal Anti-Inflammatory Effect of S. verbenaca in TNBS-Induced Colitis in Rats
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bruner, L.P.; White, A.M.; Proksell, S. Inflammatory Bowel Disease. Prim. Care 2023, 50, 411–427. [Google Scholar] [CrossRef]
- Zhang, Y.Z.; Li, Y.Y. Inflammatory bowel disease: Pathogenesis. World J. Gastroenterol. 2014, 20, 91–99. [Google Scholar] [CrossRef]
- Burisch, J. Crohn’s disease and ulcerative colitis. Occurrence, course and prognosis during the first year of disease in a European population-based inception cohort. Dan. Med. J. 2014, 61, B4778. [Google Scholar] [PubMed]
- Guan, Q.; Zhang, J. Recent Advances: The Imbalance of Cytokines in the Pathogenesis of Inflammatory Bowel Disease. Mediat. Inflamm. 2017, 2017, 4810258. [Google Scholar] [CrossRef]
- Elhag, D.A.; Kumar, M.; Saadaoui, M.; Akobeng, A.K.; Al-Mudahka, F.; Elawad, M.; Al Khodor, S. Inflammatory Bowel Disease Treatments and Predictive Biomarkers of Therapeutic Response. Int. J. Mol. Sci. 2022, 23, 6966. [Google Scholar] [CrossRef]
- Juillerat, P.; Grueber, M.M.; Ruetsch, R.; Santi, G.; Vuillemoz, M.; Michetti, P. Positioning biologics in the treatment of IBD: A practical guide—Which mechanism of action for whom? Curr. Res. Pharmacol. Drug Discov. 2022, 3, 100104. [Google Scholar] [CrossRef]
- Quezada, S.M.; McLean, L.P.; Cross, R.K. Adverse events in IBD therapy: The 2018 update. Expert Rev. Gastroenterol. Hepatol. 2018, 12, 1183–1191. [Google Scholar] [CrossRef] [PubMed]
- Holleran, G.; Scaldaferri, F.; Gasbarrini, A.; Curro, D. Herbal medicinal products for inflammatory bowel disease: A focus on those assessed in double-blind randomised controlled trials. Phytother. Res. 2020, 34, 77–93. [Google Scholar] [CrossRef]
- Algieri, F.; Rodriguez-Nogales, A.; Rodriguez-Cabezas, M.E.; Risco, S.; Ocete, M.A.; Galvez, J. Botanical Drugs as an Emerging Strategy in Inflammatory Bowel Disease: A Review. Mediat. Inflamm. 2015, 2015, 179616. [Google Scholar] [CrossRef] [PubMed]
- Vezza, T.; Algieri, F.; Rodriguez-Nogales, A.; Garrido-Mesa, J.; Utrilla, M.P.; Talhaoui, N.; Gomez-Caravaca, A.M.; Segura-Carretero, A.; Rodriguez-Cabezas, M.E.; Monteleone, G.; et al. Immunomodulatory properties of Olea europaea leaf extract in intestinal inflammation. Mol. Nutr. Food Res. 2017, 61, 1601066. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Ma, R.; Liu, C.; Liu, H.; Zhu, R.; Guo, S.; Tang, M.; Li, Y.; Niu, J.; Fu, M.; et al. Salvia miltiorrhiza: A Potential Red Light to the Development of Cardiovascular Diseases. Curr. Pharm. Des. 2017, 23, 1077–1097. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Chen, X.P.; Qiu, F.Z. Salviae miltiorrhizae ameliorates cirrhosis and portal hypertension by inhibiting nitric oxide in cirrhotic rats. Hepatobiliary Pancreat. Dis. Int. 2003, 2, 391–396. [Google Scholar] [PubMed]
- Kang, D.G.; Oh, H.; Sohn, E.J.; Hur, T.Y.; Lee, K.C.; Kim, K.J.; Kim, T.Y.; Lee, H.S. Lithospermic acid B isolated from Salvia miltiorrhiza ameliorates ischemia/reperfusion-induced renal injury in rats. Life Sci. 2004, 75, 1801–1816. [Google Scholar] [CrossRef] [PubMed]
- Jalalipour, M.; Yegdaneh, A.; Talebi, A.; Minaiyan, M. Salvia officinalis leaf extracts protect against acute colitis in rats. Res. Pharm. Sci. 2022, 17, 350–359. [Google Scholar] [CrossRef] [PubMed]
- Bonesi, M.; Loizzo, M.R.; Acquaviva, R.; Malfa, G.A.; Aiello, F.; Tundis, R. Anti-inflammatory and Antioxidant Agents from Salvia Genus (Lamiaceae): An Assessment of the Current State of Knowledge. Antiinflamm. Antiallergy Agents Med. Chem. 2017, 16, 70–86. [Google Scholar] [CrossRef]
- Silva, I.; Solas, J.; Pinto, R.; Mateus, V. Chronic Experimental Model of TNBS-Induced Colitis to Study Inflammatory Bowel Disease. Int. J. Mol. Sci. 2022, 23, 4739. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Torre, M.P.; Ferreiro-Vera, C.; Priego-Capote, F.; Perez-Juan, P.M.; Luque de Castro, M.D. Comparison of accelerated methods for the extraction of phenolic compounds from different vine-shoot cultivars. J. Agric. Food Chem. 2012, 60, 3051–3060. [Google Scholar] [CrossRef]
- Cadiz-Gurrea, M.L.; Pinto, D.; Delerue-Matos, C.; Rodrigues, F. Olive Fruit and Leaf Wastes as Bioactive Ingredients for Cosmetics-A Preliminary Study. Antioxidants 2021, 10, 245. [Google Scholar] [CrossRef]
- Ribeiro, B.; Valentao, P.; Baptista, P.; Seabra, R.M.; Andrade, P.B. Phenolic compounds, organic acids profiles and antioxidative properties of beefsteak fungus (Fistulina hepatica). Food Chem. Toxicol. 2007, 45, 1805–1813. [Google Scholar] [CrossRef]
- Camuesco, D.; Peran, L.; Comalada, M.; Nieto, A.; Di Stasi, L.C.; Rodriguez-Cabezas, M.E.; Concha, A.; Zarzuelo, A.; Galvez, J. Preventative effects of lactulose in the trinitrobenzenesulphonic acid model of rat colitis. Inflamm. Bowel Dis. 2005, 11, 265–271. [Google Scholar] [CrossRef]
- Krawisz, J.E.; Sharon, P.; Stenson, W.F. Quantitative assay for acute intestinal inflammation based on myeloperoxidase activity. Assessment of inflammation in rat and hamster models. Gastroenterology 1984, 87, 1344–1350. [Google Scholar] [CrossRef]
- Anderson, M.E. Determination of glutathione and glutathione disulfide in biological samples. Methods Enzymol. 1985, 113, 548–555. [Google Scholar] [CrossRef]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative C(T) method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef]
- Cross, R.K.; Wilson, K.T. Nitric oxide in inflammatory bowel disease. Inflamm. Bowel Dis. 2003, 9, 179–189. [Google Scholar] [CrossRef]
- Ko, J.K.; Auyeung, K.K. Inflammatory bowel disease: Etiology, pathogenesis and current therapy. Curr. Pharm. Des. 2014, 20, 1082–1096. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zhao, W.; Lan, P.; Mou, X. The microbiome in inflammatory bowel diseases: From pathogenesis to therapy. Protein Cell 2021, 12, 331–345. [Google Scholar] [CrossRef]
- Cai, Z.; Wang, S.; Li, J. Treatment of Inflammatory Bowel Disease: A Comprehensive Review. Front. Med. 2021, 8, 765474. [Google Scholar] [CrossRef] [PubMed]
- Ganji-Arjenaki, M.; Rafieian-Kopaei, M. Phytotherapies in inflammatory bowel disease. J. Res. Med. Sci. 2019, 24, 42. [Google Scholar] [CrossRef]
- Lin, Z.; Wu, H.; Fu, Y.; Dai, S. Application of Herbaceous Medications for Inflammatory Bowel Disease as a Complementary and Alternative Therapy. Inflamm. Bowel Dis. 2019, 25, 1886–1895. [Google Scholar] [CrossRef] [PubMed]
- Tian, T.; Wang, Z.; Zhang, J. Pathomechanisms of Oxidative Stress in Inflammatory Bowel Disease and Potential Antioxidant Therapies. Oxid. Med. Cell Longev. 2017, 2017, 4535194. [Google Scholar] [CrossRef] [PubMed]
- Balmus, I.M.; Ciobica, A.; Trifan, A.; Stanciu, C. The implications of oxidative stress and antioxidant therapies in Inflammatory Bowel Disease: Clinical aspects and animal models. Saudi J. Gastroenterol. 2016, 22, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Aratani, Y. Myeloperoxidase: Its role for host defense, inflammation, and neutrophil function. Arch. Biochem. Biophys. 2018, 640, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Danese, S.; Gasbarrini, A. Chemokines in inflammatory bowel disease. J. Clin. Pathol. 2005, 58, 1025–1027. [Google Scholar] [CrossRef] [PubMed]
- Danese, S.; Semeraro, S.; Marini, M.; Roberto, I.; Armuzzi, A.; Papa, A.; Gasbarrini, A. Adhesion molecules in inflammatory bowel disease: Therapeutic implications for gut inflammation. Dig. Liver Dis. 2005, 37, 811–818. [Google Scholar] [CrossRef] [PubMed]
- Ohtsuka, Y.; Lee, J.; Stamm, D.S.; Sanderson, I.R. MIP-2 secreted by epithelial cells increases neutrophil and lymphocyte recruitment in the mouse intestine. Gut 2001, 49, 526–533. [Google Scholar] [CrossRef] [PubMed]
- Holmes, E.W.; Yong, S.L.; Eiznhamer, D.; Keshavarzian, A. Glutathione content of colonic mucosa: Evidence for oxidative damage in active ulcerative colitis. Dig. Dis. Sci. 1998, 43, 1088–1095. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, M.; Pohin, M.; Powrie, F. Cytokine Networks in the Pathophysiology of Inflammatory Bowel Disease. Immunity 2019, 50, 992–1006. [Google Scholar] [CrossRef]
- Papadakis, K.A.; Targan, S.R. Role of cytokines in the pathogenesis of inflammatory bowel disease. Annu. Rev. Med. 2000, 51, 289–298. [Google Scholar] [CrossRef]
- Neurath, M.F. Cytokines in inflammatory bowel disease. Nat. Rev. Immunol. 2014, 14, 329–342. [Google Scholar] [CrossRef]
- Slebioda, T.J.; Kmiec, Z. Tumour necrosis factor superfamily members in the pathogenesis of inflammatory bowel disease. Mediat. Inflamm. 2014, 2014, 325129. [Google Scholar] [CrossRef]
- da Cunha, F.M.; Duma, D.; Assreuy, J.; Buzzi, F.C.; Niero, R.; Campos, M.M.; Calixto, J.B. Caffeic acid derivatives: In vitro and in vivo anti-inflammatory properties. Free Radic. Res. 2004, 38, 1241–1253. [Google Scholar] [CrossRef] [PubMed]
- Vezza, T.; Rodriguez-Nogales, A.; Algieri, F.; Utrilla, M.P.; Rodriguez-Cabezas, M.E.; Galvez, J. Flavonoids in Inflammatory Bowel Disease: A Review. Nutrients 2016, 8, 211. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.; Liu, W.; Mu, Y.P.; Zhang, H.; Wang, X.N.; Zhao, C.Q.; Chen, J.M.; Liu, P. Pharmacological Effects of Salvianolic Acid B Against Oxidative Damage. Front. Pharmacol. 2020, 11, 572373. [Google Scholar] [CrossRef] [PubMed]
- Oz, H.S.; Chen, T.; de Villiers, W.J. Green Tea Polyphenols and Sulfasalazine have Parallel Anti-Inflammatory Properties in Colitis Models. Front. Immunol. 2013, 4, 132. [Google Scholar] [CrossRef] [PubMed]
- Azuma, T.; Shigeshiro, M.; Kodama, M.; Tanabe, S.; Suzuki, T. Supplemental naringenin prevents intestinal barrier defects and inflammation in colitic mice. J. Nutr. 2013, 143, 827–834. [Google Scholar] [CrossRef] [PubMed]
- Camuesco, D.; Comalada, M.; Rodriguez-Cabezas, M.E.; Nieto, A.; Lorente, M.D.; Concha, A.; Zarzuelo, A.; Galvez, J. The intestinal anti-inflammatory effect of quercitrin is associated with an inhibition in iNOS expression. Br. J. Pharmacol. 2004, 143, 908–918. [Google Scholar] [CrossRef] [PubMed]
- Dou, W.; Zhang, J.; Ren, G.; Ding, L.; Sun, A.; Deng, C.; Wu, X.; Wei, X.; Mani, S.; Wang, Z. Mangiferin attenuates the symptoms of dextran sulfate sodium-induced colitis in mice via NF-kappaB and MAPK signaling inactivation. Int. Immunopharmacol. 2014, 23, 170–178. [Google Scholar] [CrossRef]
- Comalada, M.; Camuesco, D.; Sierra, S.; Ballester, I.; Xaus, J.; Galvez, J.; Zarzuelo, A. In vivo quercitrin anti-inflammatory effect involves release of quercetin, which inhibits inflammation through down-regulation of the NF-kappaB pathway. Eur. J. Immunol. 2005, 35, 584–592. [Google Scholar] [CrossRef]
- Yu, F.Y.; Huang, S.G.; Zhang, H.Y.; Ye, H.; Chi, H.G.; Zou, Y.; Lv, R.X.; Zheng, X.B. Effects of baicalin in CD4 + CD29 + T cell subsets of ulcerative colitis patients. World J. Gastroenterol. 2014, 20, 15299–15309. [Google Scholar] [CrossRef]
- Schreiber, S.; Rosenstiel, P.; Hampe, J.; Nikolaus, S.; Groessner, B.; Schottelius, A.; Kuhbacher, T.; Hamling, J.; Folsch, U.R.; Seegert, D. Activation of signal transducer and activator of transcription (STAT) 1 in human chronic inflammatory bowel disease. Gut 2002, 51, 379–385. [Google Scholar] [CrossRef]
- Pedersen, J.; Coskun, M.; Soendergaard, C.; Salem, M.; Nielsen, O.H. Inflammatory pathways of importance for management of inflammatory bowel disease. World J. Gastroenterol. 2014, 20, 64–77. [Google Scholar] [CrossRef]
- Hossen, I.; Hua, W.; Ting, L.; Mehmood, A.; Jingyi, S.; Duoxia, X.; Yanping, C.; Hongqing, W.; Zhipeng, G.; Kaiqi, Z.; et al. Phytochemicals and inflammatory bowel disease: A review. Crit. Rev. Food Sci. Nutr. 2020, 60, 1321–1345. [Google Scholar] [CrossRef] [PubMed]
- Lechuga, S.; Ivanov, A.I. Disruption of the epithelial barrier during intestinal inflammation: Quest for new molecules and mechanisms. Biochim. Biophys. Acta Mol. Cell Res. 2017, 1864, 1183–1194. [Google Scholar] [CrossRef] [PubMed]
- Chelakkot, C.; Ghim, J.; Ryu, S.H. Mechanisms regulating intestinal barrier integrity and its pathological implications. Exp. Mol. Med. 2018, 50, 1–9. [Google Scholar] [CrossRef]
- Martini, E.; Krug, S.M.; Siegmund, B.; Neurath, M.F.; Becker, C. Mend Your Fences: The Epithelial Barrier and its Relationship With Mucosal Immunity in Inflammatory Bowel Disease. Cell Mol. Gastroenterol. Hepatol. 2017, 4, 33–46. [Google Scholar] [CrossRef] [PubMed]
- Feng, P.P.; Fang, X.S.; Zhao, S.H.; Fu, J.Y.; Zhang, H.T.; Yi, Y.L.; Li, C.Y.; Jiang, C.L.; Chen, D.P. Salvianolic acid B decreases interleukin-1beta-induced colitis recurrence in mice. Chin. Med. J. 2020, 133, 1436–1444. [Google Scholar] [CrossRef] [PubMed]
- Einerhand, A.W.; Renes, I.B.; Makkink, M.K.; van der Sluis, M.; Buller, H.A.; Dekker, J. Role of mucins in inflammatory bowel disease: Important lessons from experimental models. Eur. J. Gastroenterol. Hepatol. 2002, 14, 757–765. [Google Scholar] [CrossRef]
- Van der Sluis, M.; De Koning, B.A.; De Bruijn, A.C.; Velcich, A.; Meijerink, J.P.; Van Goudoever, J.B.; Buller, H.A.; Dekker, J.; Van Seuningen, I.; Renes, I.B.; et al. Muc2-deficient mice spontaneously develop colitis, indicating that MUC2 is critical for colonic protection. Gastroenterology 2006, 131, 117–129. [Google Scholar] [CrossRef]
- Wang, Y.; Srinivasan, K.; Siddiqui, M.R.; George, S.P.; Tomar, A.; Khurana, S. A novel role for villin in intestinal epithelial cell survival and homeostasis. J. Biol. Chem. 2008, 283, 9454–9464. [Google Scholar] [CrossRef]
- Galvez, J.; Coelho, G.; Crespo, M.E.; Cruz, T.; Rodriguez-Cabezas, M.E.; Concha, A.; Gonzalez, M.; Zarzuelo, A. Intestinal anti-inflammatory activity of morin on chronic experimental colitis in the rat. Aliment. Pharmacol. Ther. 2001, 15, 2027–2039. [Google Scholar] [CrossRef]
- Sanchez de Medina, F.; Galvez, J.; Romero, J.A.; Zarzuelo, A. Effect of quercitrin on acute and chronic experimental colitis in the rat. J. Pharmacol. Exp. Ther. 1996, 278, 771–779. [Google Scholar] [PubMed]







| Score | Criteria |
|---|---|
| 0 | No damage |
| 1 | Hyperemia, no ulcers |
| 2 | Linear ulcer with no significant inflammation |
| 3 | Linear ulcer with inflammation at one site |
| 4 | Two or more sites of ulceration/inflammation |
| 5 | Two or more major sites of ulceration and inflammation or one site of ulceration/inflammation extending along the length of the colon |
| 6–10 | If damage covers along the length of the colon, the score is increased by 1 for each additional centimeter of involvement |
| Gene | Organism | Sequence 5′–3′ | Annealing T °C | Accesion Number |
|---|---|---|---|---|
| Gapdh | Mouse/Rat | FW: CCATCACCATCTTCCAGGAG RV: CCTGCTTCACCACCTTCTTG | 60 | NM_001289726.1 |
| Il6 | Mouse | FW: TAGTCCTTCCTACCCCAATTTCC RV: TTGGTCCTTAGCCACTCCTTCC | 60 | NM_031168.2 |
| Icam-1 | Mouse | FW: CAGTCCGCTGTGCTTTGAGA RV: CGGAAACGAATACACGGTGAT | 62 | NC_000075.7 |
| Mip-2 | Mouse | FW: CAGTGAGCTGCGCTGTCCAATG RV: CAGTTAGCCTTGCCTTTGTTCAG | 57 | NC_000071.7 |
| Muc2 | Mouse | FW: GCAGTCCTCAGTGGCACCTC RV: CACCGTGGGGCTACTGGAGAG | 60 | NC_000073.7 |
| Tnfα | Mouse | FW: AACTAGTGGTGCCAGCCGAT RV: CTTCACAGAGCAATGACTCC | 60 | NM_001278601.1 |
| Cinc-1 | Rat | FW: GGCAGGGATTCACTTCAAGA RV: GCCATCGGTGCAATCTATCT | 60 | NC_051349.1 |
| Icam-1 | Rat | FW: AGGTATCCATCCATCCCACA RV: AGTGTCTCATTGCCACGGAG | 60 | NC_051343.1 |
| Il1β | Rat | FW: GATCTTTGAAGAAGAGCCCG RV: AACTATGTCCCGACCATTGC | 59 | NC_051338.1 |
| Il6 | Rat | FW: CTTCCCTACTTCACAAGTC RV: CTCCATTAGGAGAGCATTG | 60 | NC_051339.1 |
| Il12a | Rat | FW: ACGCTACCTCCTCTTCTTG RV: ATGTCGTCCGTGGTCTTC | 60 | NC_051337.1 |
| Il17 | Rat | FW: TGGACTCTGAGCCGCAATGAGG RV: GACGCATGGCGGACAATAGAGG | 60 | NC_051344.1 |
| Il23 | Rat | FW: ATCCAGTGTGGTGATGGTTGTG RV: TGTCCGAGTCCAGCAGGTG | 60 | NC_051342.1 |
| iNos | Rat | FW: AAGAGACGCACAGGCAGAGG RV: AGCAGGCACACGCAATGAT | 60 | NC_051345.1 |
| Mcp-1 | Rat | FW: CACTATGCAGGTCTCTGTCACG RV: CTGGTCACTTCTACAGAAGTGC | 60 | NC_051345.1 |
| Muc-2 | Rat | FW: ACCACCATTACCACCACCTCAG RV: CGATCACCACCATTGCCACTG | 60 | NC_051336.1 |
| Muc-3 | Rat | FW: CACAAAGGCAAGAGTCCAGA RV: ACTGTGCTTGGTGCTGAATG | 60 | NC_051347.1 |
| Villin | Rat | FW: TGTGGAACTGGCAGGGAG RV: GGGGTGGGTCTTGAGGTATT | 59 | NC_051344.1 |
| Peak | RT | Measured m/z | Molecular Formula | Proposed Compounds | Main Fragments |
|---|---|---|---|---|---|
| 1 | 1.01 | 195.0538 | C6H12O7 | Gluconic acid | NF |
| 2 | 1.14 | 133.0165 | C4H6O5 | Malic acid | NF |
| 3 | 1.28 | 191.0226 | C6H8O7 | Citric acid | NF |
| 4 | 1.58 | 96.9610 | - | Sulfate | NF |
| 5 | 1.73 | 96.9610 | - | Sulfate | NF |
| 6 | 2.20 | 191.0559 | C7H12O6 | Quinic acid | NF |
| 7 | 2.39 | 395.0965 | C18H20O10 | Acetylisobiflorin | 179 |
| 8 | 2.74 | 315.0723 | C13H16O9 | Gentisoyl glucoside | 152, 108 |
| 9 | 2.85 | 551.1058 | C24H24O15 | Phloroscorbinol hexaacetate | NF |
| 10 | 3.19 | 153.0188 | C7H6O4 | Dihydroxybenzoic acid | 109 |
| 11 | 3.38 | 181.0502 | C9H10O4 | Dihydrocaffeic acid isomer 1 | 135 |
| 12 | 3.67 | 175.0608 | C7H12O5 | Isopropylmalic acid | NF |
| 13 | 4.35 | 391.0667 | C18H16O10 | Pentahydroxy trimethoxy flavone isomer 1 | 347, 303 |
| 14 | 4.55 | 325.0927 | C15H18O8 | p-Coumaric acid glucoside | 163 |
| 15 | 4.72 | 391.0661 | C18H16O10 | Pentahydroxy trimethoxy flavone isomer 2 | 347, 303 |
| 16 | 5.66 | 475.0886 | C22H20O12 | Chrysoeriol glucuronide | 299, 227 |
| 17 | 6.22 | 475.1833 | C21H32O12 | Cistanoside E | 179, 135 |
| 18 | 6.32 | 181.0504 | C9H10O4 | Dihydrocaffeic acid isomer 2 | NF |
| 19 | 6.80 | 179.0346 | C9H8O4 | Caffeic acid | 135 |
| 20 | 7.34 | 593.1158 | C30H26O13 | O-coumaroyl orientin | NF |
| 21 | 7.65 | 639.1199 | C27H28O18 | Quercetin glucosyl-glucuronide isomer 1 | 463, 300 |
| 22 | 7.89 | 405.0834 | C19H18O10 | Glucopyranosyl-trihydroxy-9H-xanthen-9-one | 273, 317, 387 |
| 23 | 8.12 | 639.1157 | C27H28O18 | Quercetin glucosyl-glucuronide isomer 2 | 299 |
| 24 | 8.20 | 589.0826 | C19H26O21 | Unknown 1 | |
| 25 | 8.28 | 783.1637 | C33H36O22 | Quercetin glucoside derivative | 607, 505, 463, 300, 545 |
| 26 | 8.48 | 353.0876 | C16H18O9 | Caffeoyl quinic acid | 191, 179 |
| 27 | 8.63 | 347.0777 | C17H16O8 | Dihydroxy tetramethoxyxanthone | NF |
| 28 | 8.78 | 655.1288 | C31H28O16 | Acetylated flavonol glycoside isomer 1 | 285, 461, 447 |
| 29 | 8.86 | 329.0774 | C14H18O9 | Flavonol derivative | 285 |
| 30 | 8.92 | 655.1292 | C31H28O16 | Acetylated flavonol glycoside isomer 2 | 285, 461 |
| 31 | 8.96 | 655.1299 | C31H28O16 | Acetylated flavonol glycoside isomer 3 | 285, 461, 447 |
| 32 | 9.02 | 637.1058 | C27H26O18 | Luteolin diglucuronide | 351, 285 |
| 33 | 9.10 | 571.1082 | C27H24O14 | Yunnaneic acid E isomer 1 | 197, 285, 135, 527, 241, 329, 439 |
| 34 | 9.23 | 571.1083 | C27H24O14 | Yunnaneic acid E isomer 2 | 197, 135, 285, 347, 527, 241, 483, 439 |
| 35 | 9.29 | 539.1189 | C27H24O12 | Yunnaneic acid D | 297, 179, 197, 161, 135, 359 |
| 36 | 9.35 | 415.1953 | C20H32O9 | Yunnaneic acid derivative 1 | 329, 179, 161, 297 |
| 37 | 9.47 | 597.1263 | C29H26O14 | Yunnaneic acid F | 197, 135, 329, 179 |
| 38 | 9.63 | 309.0616 | C14H14O8 | Feruloylmalic acid | 193, 134 |
| 39 | 9.71 | 555.1157 | C27H24O13 | Salvianolic acid K isomer 1 | 197, 135, 329, 179 |
| 40 | 9.76 | 585.1273 | C28H26O14 | Naringenin digalloylglucopyranoside isomer 1 | 271 |
| 41 | 9.79 | 585.1244 | C28H26O14 | Naringenin digalloylglucopyranoside isomer 2 | 271 |
| 42 | 9.84 | 585.1221 | C28H26O14 | Naringenin digalloylglucopyranoside isomer 3 | 271 |
| 43 | 9.87 | 461.0740 | C21H18O12 | Luteolin glucuronide | 285 |
| 44 | 9.95 | 361.0938 | C18H18O8 | Crotepoxide | 273, 241 |
| 45 | 10.03 | 527.1208 | C26H24O12 | Yunnaneic acid derivative 2 isomer 1 | 135, 197, 179, 285 |
| 46 | 10.15 | 719.1629 | C36H32O16 | Sagerinic acid | 359, 161, 197, 179 |
| 47 | 10.24 | 527.1198 | C26H24O12 | Yunnaneic acid derivative 2 isomer 2 | 285, 197, 135, 241, 179, 439 |
| 48 | 10.33 | 717.1479 | C33H34O18 | Salvianolic acid L | 359, 161, 197 |
| 49 | 10.49 | 511.1293 | C26H24O11 | Salvianolic acid A hydrate | 269, 197, 135, 179 |
| 50 | 10.58 | 343.0924 | C22H16O4 | Unknown 2 | |
| 51 | 10.64 | 555.1153 | C27H24O13 | Salvianolic acid K isomer 2 | 359, 161, 135, 493, 401, 537 |
| 52 | 10.74 | 711.3987 | C37H60O13 | Hydroxytormentic acid derivative isomer 1 | 503 |
| 53 | 10.87 | 533.1302 | C25H26O13 | Yunnaneic acid derivative 3 isomer 1 | 197 |
| 54 | 10.93 | 541.1365 | C27H26O12 | Yunnaneic acid derivative 4 | 197, 509, 135, 179 |
| 55 | 10.95 | 533.1315 | C25H26O13 | Yunnaneic acid derivative 3 isomer 2 | 197 |
| 56 | 10.99 | 537.1053 | C27H22O12 | Lithospermic acid A | 295, 161, 359, 135, 197, 493 |
| 57 | 11.06 | 651.2319 | C31H40O15 | Martynoside | 175, 475 |
| 58 | 11.17 | 373.0936 | C19H18O8 | Methyl rosmarinate isomer 1 | 197, 161 |
| 59 | 11.21 | 373.0917 | C19H18O8 | Methyl rosmarinate isomer 2 | 135, 175, 197 |
| 60 | 11.38 | 711.3966 | C37H60O13 | Hydroxytormentic acid derivative isomer 2 | 503 |
| 61 | 11.43 | 481.1125 | C25H22O10 | Silybin | 301, 283 |
| 62 | 11.62 | 493.1144 | C26H22O10 | Salvianolic acid A | 295 |
| 63 | 11.71 | 479.0992 | C25H20O10 | Dehydrosilybin | 299, 281 |
| 64 | 11.76 | 491.0985 | C26H20O10 | Salvianolic acid C | 293 |
| 65 | 11.99 | 327.2151 | C18H32O5 | Trihydroxyoctadecadienoic acid | NF |
| 66 | 12.19 | 695.4023 | C37H60O12 | Unknown 3 | |
| 67 | 12.47 | 329.2330 | C18H34O5 | Pinellic acid | NF |
| 68 | 12.57 | 523.1242 | C27H24O11 | Salvianolic acid derivative | 135, 179, 491, 359 |
| 69 | 12.77 | 345.1717 | C20H26O5 | Rosmanol isomer 1 | NF |
| 70 | 13.56 | 307.1909 | C18H28O4 | Unknown 4 | |
| 71 | 14.20 | 345.1702 | C20H26O5 | Rosmanol isomer 2 | 301, 283 |
| 72 | 14.32 | 311.2206 | C18H32O4 | Octadecenedioic acid | NF |
| 73 | 14.74 | 331.1918 | C20H28O4 | Carnosic acid | 287, 244 |
| 74 | 14.99 | 721.3674 | C34H58O16 | Palmitoleic-linolenicglucoside | 397, 277, 415, 235 |
| 75 | 15.80 | 293.2116 | C18H30O3 | Hydroxylinolenic acid isomer 1 | NF |
| 76 | 15.82 | 293.2119 | C18H30O3 | Hydroxylinolenic acid isomer 2 | NF |
| 77 | 16.39 | 295.2276 | C18H32O3 | Hydroxylinoleic acid | NF |
| 78 | 16.68 | 455.3168 | C29H44O4 | Diosgenin acetate | NF |
| 79 | 17.22 | 471.3479 | C30H48O4 | Maslinic acid isomer 1 | NF |
| 80 | 17.36 | 471.3485 | C30H48O4 | Maslinic acid isomer 2 | NF |
| Method | Value |
|---|---|
| Folin–Ciocalteu (mg GAE/g d.e.) | 166 ± 4 |
| FRAP (mmol eq. FeSO4/g d.e.) | 2.6 ± 0.1 |
| TEAC (mmol eq. Trolox/g d.e.) | 0.73 ± 0.02 |
| ORAC (mmol eq. Trolox/g d.e.) | 0.92 ± 0.03 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vezza, T.; Molina-Tijeras, J.A.; Rodríguez-Nogales, A.; Garrido-Mesa, J.; Cádiz-Gurrea, M.d.l.L.; Segura-Carretero, A.; González-Tejero, M.R.; Rodríguez-Cabezas, M.E.; Gálvez, J.; Algieri, F. The Antioxidant Properties of Salvia verbenaca Extract Contribute to Its Intestinal Antiinflammatory Effects in Experimental Colitis in Rats. Antioxidants 2023, 12, 2071. https://doi.org/10.3390/antiox12122071
Vezza T, Molina-Tijeras JA, Rodríguez-Nogales A, Garrido-Mesa J, Cádiz-Gurrea MdlL, Segura-Carretero A, González-Tejero MR, Rodríguez-Cabezas ME, Gálvez J, Algieri F. The Antioxidant Properties of Salvia verbenaca Extract Contribute to Its Intestinal Antiinflammatory Effects in Experimental Colitis in Rats. Antioxidants. 2023; 12(12):2071. https://doi.org/10.3390/antiox12122071
Chicago/Turabian StyleVezza, Teresa, Jose Alberto Molina-Tijeras, Alba Rodríguez-Nogales, Jose Garrido-Mesa, María de la Luz Cádiz-Gurrea, Antonio Segura-Carretero, María Reyes González-Tejero, María Elena Rodríguez-Cabezas, Julio Gálvez, and Francesca Algieri. 2023. "The Antioxidant Properties of Salvia verbenaca Extract Contribute to Its Intestinal Antiinflammatory Effects in Experimental Colitis in Rats" Antioxidants 12, no. 12: 2071. https://doi.org/10.3390/antiox12122071