Mitochondrial DNA: Inherent Complexities Relevant to Genetic Analyses
Abstract
1. Introduction
2. Mitochondria and mtDNA
2.1. Structure of Human Mitochondrial DNA
2.1.1. The Non-Coding Region and Displacement Loop
2.1.2. Reference Sequences
2.2. Function of Human Mitochondrial DNA
2.2.1. The Functional Role of Human mtDNA
2.2.2. Mitochondrial-Derived Peptides
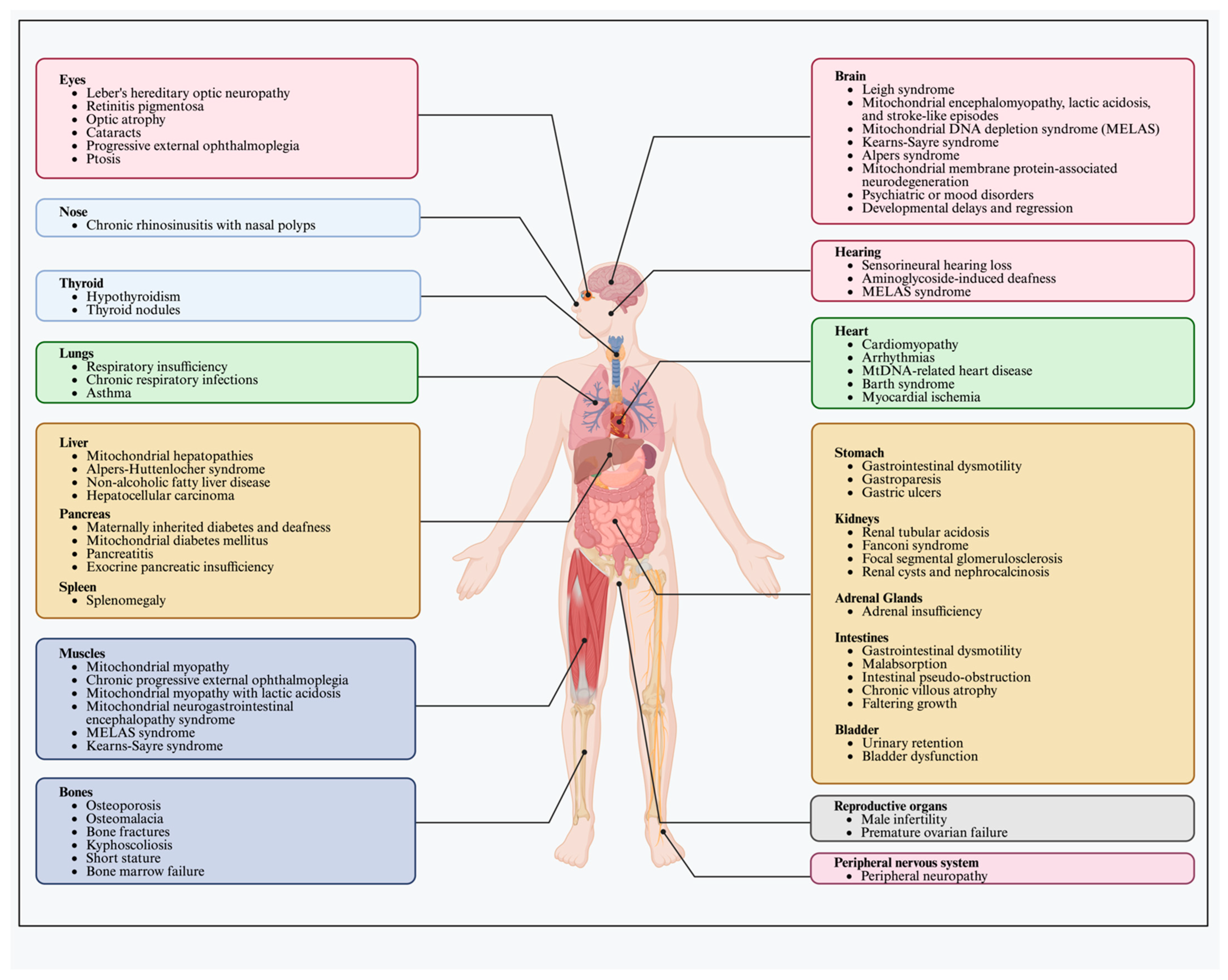
2.3. Human mtDNA-Related Diseases
2.4. Specific Particularities of mtDNA in Relation to Human Population History
2.4.1. Uniparental Inheritance
Developmental
Evolutionary
Paternal Controversy
2.4.2. Hypermutability
2.4.3. Copy Number and Abundance
2.4.4. Cell-Free Circulating mtDNA (ccf-mtDNA)
2.4.5. Heteroplasmy
2.4.6. Haplogroups
2.4.7. Lack of Introns
2.4.8. Sensitivity of mtDNA to Environmental Factors
2.4.9. Epigenetics
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nass, M.M.; Nass, S. Intramitochondrial fibers with DNA characteristics I. Fixation and electron staining reactions. J. Cell Biol. 1963, 19, 593–611. [Google Scholar] [CrossRef]
- Amorim, A.; Fernandes, T.; Taveira, N. Mitochondrial DNA in human identification: A review. PeerJ 2019, 7, e7314. [Google Scholar] [CrossRef]
- Xue, A.; Wu, Y.; Zhu, Z.; Zhang, F.; Kemper, K.E.; Zheng, Z.; Yengo, L.; Lloyd-Jones, L.R.; Sidorenko, J.; Wu, Y. Genome-wide association analyses identify 143 risk variants and putative regulatory mechanisms for type 2 diabetes. Nat. Commun. 2018, 9, 2941. [Google Scholar] [CrossRef]
- Visscher, P.M.; Wray, N.R.; Zhang, Q.; Sklar, P.; McCarthy, M.I.; Brown, M.A.; Yang, J. 10 years of GWAS discovery: Biology, function, and translation. Am. J. Hum. Genet. 2017, 101, 5–22. [Google Scholar] [CrossRef]
- Frayling, T.M.; Timpson, N.J.; Weedon, M.N.; Zeggini, E.; Freathy, R.M.; Lindgren, C.M.; Perry, J.R.; Elliott, K.S.; Lango, H.; Rayner, N.W. A common variant in the FTO gene is associated with body mass index and predisposes to childhood and adult obesity. Science 2007, 316, 889–894. [Google Scholar] [CrossRef]
- Siminovitch, K.A. PTPN22 and autoimmune disease. Nat. Genet. 2004, 36, 1248–1249. [Google Scholar] [CrossRef]
- Wang, K.; Zhang, H.; Kugathasan, S.; Annese, V.; Bradfield, J.P.; Russell, R.K.; Sleiman, P.M.; Imielinski, M.; Glessner, J.; Hou, C. Diverse genome-wide association studies associate the IL12/IL23 pathway with Crohn Disease. Am. J. Hum. Genet. 2009, 84, 399–405. [Google Scholar] [CrossRef]
- Sollis, E.; Mosaku, A.; Abid, A.; Buniello, A.; Cerezo, M.; Gil, L.; Groza, T.; Güneş, O.; Hall, P.; Hayhurst, J. The NHGRI-EBI GWAS Catalog: Knowledgebase and deposition resource. Nucleic Acids Res. 2023, 51, D977–D985. [Google Scholar] [CrossRef]
- Anderson, K.; Cañadas-Garre, M.; Chambers, R.; Maxwell, A.P.; McKnight, A.J. The challenges of chromosome Y analysis and the implications for chronic kidney disease. Front. Genet. 2019, 10, 781. [Google Scholar] [CrossRef]
- Sun, L.; Wang, Z.; Lu, T.; Manolio, T.A.; Paterson, A.D. eXclusionarY: 10 years later, where are the sex chromosomes in GWASs? Am. J. Hum. Genet. 2023, 110, 903–912. [Google Scholar] [CrossRef]
- Parker, K.; Erzurumluoglu, A.M.; Rodriguez, S. The Y chromosome: A complex locus for genetic analyses of complex human traits. Genes 2020, 11, 1273. [Google Scholar] [CrossRef]
- Yonova-Doing, E.; Calabrese, C.; Gomez-Duran, A.; Schon, K.; Wei, W.; Karthikeyan, S.; Chinnery, P.F.; Howson, J.M. An atlas of mitochondrial DNA genotype–phenotype associations in the UK Biobank. Nat. Genet. 2021, 53, 982–993. [Google Scholar] [CrossRef]
- Esser, C.; Ahmadinejad, N.; Wiegand, C.; Rotte, C.; Sebastiani, F.; Gelius-Dietrich, G.; Henze, K.; Kretschmann, E.; Richly, E.; Leister, D. A genome phylogeny for mitochondria among α-proteobacteria and a predominantly eubacterial ancestry of yeast nuclear genes. Mol. Biol. Evol. 2004, 21, 1643–1660. [Google Scholar] [CrossRef]
- Alberts, B.; Johnson, A.; Lewis, J.; Raff, M.; Roberts, K.; Walter, P. The mitochondrion. In Molecular Biology of the Cell, 4th ed.; Garland Science: New York, NY, USA, 2002. [Google Scholar]
- Saxena, R.; de Bakker, P.I.; Singer, K.; Mootha, V.; Burtt, N.; Hirschhorn, J.N.; Gaudet, D.; Isomaa, B.; Daly, M.J.; Groop, L. Comprehensive association testing of common mitochondrial DNA variation in metabolic disease. Am. J. Hum. Genet. 2006, 79, 54–61. [Google Scholar] [CrossRef]
- Mitchell, S.L.; Hall, J.B.; Goodloe, R.J.; Boston, J.; Farber-Eger, E.; Pendergrass, S.A.; Bush, W.S.; Crawford, D.C. Investigating the relationship between mitochondrial genetic variation and cardiovascular-related traits to develop a framework for mitochondrial phenome-wide association studies. BioData Min. 2014, 7, 6. [Google Scholar] [CrossRef]
- Wallace, D.C. A mitochondrial paradigm of metabolic and degenerative diseases, aging, and cancer: A dawn for evolutionary medicine. Annu. Rev. Genet. 2005, 39, 359–407. [Google Scholar] [CrossRef]
- Wallace, D.C. Diseases of the mitochondrial DNA. Annu. Rev. Biochem. 1992, 61, 1175–1212. [Google Scholar] [CrossRef]
- De Moura, M.B.; dos Santos, L.S.; Van Houten, B. Mitochondrial dysfunction in neurodegenerative diseases and cancer. Environ. Mol. Mutagen. 2010, 51, 391–405. [Google Scholar] [CrossRef]
- Smith, E.F.; Shaw, P.J.; De Vos, K.J. The role of mitochondria in amyotrophic lateral sclerosis. Neurosci. Lett. 2019, 710, 132933. [Google Scholar] [CrossRef]
- DiMauro, S. A history of mitochondrial diseases. J. Inherit. Metab. Dis. 2011, 34, 261–276. [Google Scholar] [CrossRef]
- Anderson, S.; Bankier, A.T.; Barrell, B.G.; de Bruijn, M.H.; Coulson, A.R.; Drouin, J.; Eperon, I.C.; Nierlich, D.P.; Roe, B.A.; Sanger, F. Sequence and organization of the human mitochondrial genome. Nature 1981, 290, 457–465. [Google Scholar] [CrossRef]
- Garone, C.; Minczuk, M.; Falkenberg, M. Mitochondrial DNA replication in mammalian cells: Overview of the pathway. Essays Biochem. 2018, 62, 287–296. [Google Scholar] [CrossRef]
- Yasukawa, T.; Kang, D. An overview of mammalian mitochondrial DNA replication mechanisms. J. Biochem. 2018, 164, 183–193. [Google Scholar] [CrossRef]
- Moraes, C.T.; Srivastava, S.; Kirkinezos, I.; Oca-Cossio, J.; Woischnick, M.; Diaz, F. Mitochondrial DNA structure and function. Int. Rev. Neurobiol. 2002, 53, 3–23. [Google Scholar]
- Macreadie, I.G.; Novitski, C.E.; Maxwell, R.J.; John, U.; Ooi, B.-G.; McMullen, G.L.; Lukins, H.; Linnane, A.W.; Nagley, P. Biogenesis of mitochondria: The mitochondrial gene (aap1) coding for mitochondrial ATPase subunit 8 in Saccharomyces cerevisiae. Nucleic Acids Res. 1983, 11, 4435–4451. [Google Scholar] [CrossRef]
- Chomyn, A.; Mariottini, P.; Cleeter, M.W.; Ragan, C.I.; Matsuno-Yagi, A.; Hatefi, Y.; Doolittle, R.F.; Attardi, G. Six unidentified reading frames of human mitochondrial DNA encode components of the respiratory-chain NADH dehydrogenase. Nature 1985, 314, 592–597. [Google Scholar] [CrossRef]
- Chomyn, A.; Cleeter, M.; Ragan, C.I.; Riley, M.; Doolittle, R.F.; Attardi, G. URF6, last unidentified reading frame of human mtDNA, codes for an NADH dehydrogenase subunit. Science 1986, 234, 614–618. [Google Scholar] [CrossRef]
- Nicholls, T.J.; Minczuk, M. In D-loop: 40 years of mitochondrial 7S DNA. Exp. Gerontol. 2014, 56, 175–181. [Google Scholar] [CrossRef]
- Chen, J.-B.; Yang, Y.-H.; Lee, W.-C.; Liou, C.-W.; Lin, T.-K.; Chung, Y.-H.; Chuang, L.-Y.; Yang, C.-H.; Chang, H.-W. Sequence-based polymorphisms in the mitochondrial D-loop and potential SNP predictors for chronic dialysis. PLoS ONE 2012, 7, e41125. [Google Scholar] [CrossRef]
- Falkenberg, M.; Gaspari, M.; Rantanen, A.; Trifunovic, A.; Larsson, N.-G.; Gustafsson, C.M. Mitochondrial transcription factors B1 and B2 activate transcription of human mtDNA. Nat. Genet. 2002, 31, 289–294. [Google Scholar] [CrossRef]
- Bogenhagen, D.; Clayton, D.A. Mouse L cell mitochondrial DNA molecules are selected randomly for replication throughout the cell cycle. Cell 1977, 11, 719–727. [Google Scholar] [CrossRef]
- Rath, S.; Sharma, R.; Gupta, R.; Ast, T.; Chan, C.; Durham, T.J.; Goodman, R.P.; Grabarek, Z.; Haas, M.E.; Hung, W.H. MitoCarta3. 0: An updated mitochondrial proteome now with sub-organelle localization and pathway annotations. Nucleic Acids Res. 2021, 49, D1541–D1547. [Google Scholar] [CrossRef]
- Gupta, R.; Kanai, M.; Durham, T.J.; Tsuo, K.; McCoy, J.G.; Kotrys, A.V.; Zhou, W.; Chinnery, P.F.; Karczewski, K.J.; Calvo, S.E. Nuclear genetic control of mtDNA copy number and heteroplasmy in humans. Nature 2023, 620, 839–848. [Google Scholar] [CrossRef]
- Jemt, E.; Persson, Ö.; Shi, Y.; Mehmedovic, M.; Uhler, J.P.; Dávila López, M.; Freyer, C.; Gustafsson, C.M.; Samuelsson, T.; Falkenberg, M. Regulation of DNA replication at the end of the mitochondrial D-loop involves the helicase TWINKLE and a conserved sequence element. Nucleic Acids Res. 2015, 43, 9262–9275. [Google Scholar] [CrossRef]
- Tuppen, H.A.; Blakely, E.L.; Turnbull, D.M.; Taylor, R.W. Mitochondrial DNA mutations and human disease. Biochim. Biophys. Acta (BBA)—Bioenerg. 2010, 1797, 113–128. [Google Scholar] [CrossRef]
- Campbell, C.T.; Kolesar, J.E.; Kaufman, B.A. Mitochondrial transcription factor A regulates mitochondrial transcription initiation, DNA packaging, and genome copy number. Biochim. Biophys. Acta (BBA)—Gene Regul. Mech. 2012, 1819, 921–929. [Google Scholar] [CrossRef]
- Ye, C.; Shu, X.O.; Pierce, L.; Wen, W.; Courtney, R.; Gao, Y.-T.; Zheng, W.; Cai, Q. Mutations in the mitochondrial DNA D-loop region and breast cancer risk. Breast Cancer Res. Treat. 2010, 119, 431–436. [Google Scholar] [CrossRef]
- Ding, C.; Li, R.; Wang, P.; Jin, P.; Li, S.; Guo, Z. Identification of sequence polymorphisms in the D-loop region of mitochondrial DNA as a risk factor for lung cancer. Mitochondrial DNA 2012, 23, 251–254. [Google Scholar] [CrossRef]
- Hertweck, K.L.; Dasgupta, S. The landscape of mtDNA modifications in cancer: A tale of two cities. Front. Oncol. 2017, 7, 262. [Google Scholar] [CrossRef]
- Levin, B.C.; Cheng, H.; Reeder, D.J. A human mitochondrial DNA standard reference material for quality control in forensic identification, medical diagnosis, and mutation detection. Genomics 1999, 55, 135–146. [Google Scholar] [CrossRef]
- Syndercombe Court, D. Mitochondrial DNA in forensic use. Emerg. Top. Life Sci. 2021, 5, 415–426. [Google Scholar]
- Lightowlers, R.N.; Chrzanowska-Lightowlers, Z.M.A. Exploring our origins—The importance of OriL in mtDNA maintenance and replication. EMBO Rep. 2012, 13, 1038–1039. [Google Scholar] [CrossRef]
- Chinnery, P.F.; Hudson, G. Mitochondrial genetics. Br. Med. Bull. 2013, 106, 135–159. [Google Scholar] [CrossRef]
- Andrews, R.M.; Kubacka, I.; Chinnery, P.F.; Lightowlers, R.N.; Turnbull, D.M.; Howell, N. Reanalysis and revision of the Cambridge reference sequence for human mitochondrial DNA. Nat. Genet. 1999, 23, 147. [Google Scholar] [CrossRef]
- Behar, D.M.; Van Oven, M.; Rosset, S.; Metspalu, M.; Loogväli, E.-L.; Silva, N.M.; Kivisild, T.; Torroni, A.; Villems, R. A “Copernican” reassessment of the human mitochondrial DNA tree from its root. Am. J. Hum. Genet. 2012, 90, 675–684. [Google Scholar] [CrossRef]
- Bandelt, H.-J.; Kloss-Brandstätter, A.; Richards, M.B.; Yao, Y.-G.; Logan, I. The case for the continuing use of the revised Cambridge Reference Sequence (rCRS) and the standardization of notation in human mitochondrial DNA studies. J. Hum. Genet. 2014, 59, 66–77. [Google Scholar] [CrossRef]
- Mahar, N.S.; Satyam, R.; Sundar, D.; Gupta, I. A systematic comparison of human mitochondrial genome assembly tools. BMC Bioinform. 2023, 24, 341. [Google Scholar] [CrossRef]
- Fernandez-Vizarra, E.; Zeviani, M. Mitochondrial disorders of the OXPHOS system. FEBS Lett. 2021, 595, 1062–1106. [Google Scholar] [CrossRef]
- Sharma, L.K.; Lu, J.; Bai, Y. Mitochondrial respiratory complex I: Structure, function and implication in human diseases. Curr. Med. Chem. 2009, 16, 1266–1277. [Google Scholar] [CrossRef]
- Cecchini, G. Function and structure of complex II of the respiratory chain. Annu. Rev. Biochem. 2003, 72, 77–109. [Google Scholar] [CrossRef]
- Jackson, C.B.; Nuoffer, J.-M.; Hahn, D.; Prokisch, H.; Haberberger, B.; Gautschi, M.; Häberli, A.; Gallati, S.; Schaller, A. Mutations in SDHD lead to autosomal recessive encephalomyopathy and isolated mitochondrial complex II deficiency. J. Med. Genet. 2014, 51, 170–175. [Google Scholar] [CrossRef]
- Hoekstra, A.S.; Bayley, J.-P. The role of complex II in disease. Biochim. Biophys. Acta (BBA)—Bioenerg. 2013, 1827, 543–551. [Google Scholar] [CrossRef]
- Munnich, A.; Rustin, P. Clinical spectrum and diagnosis of mitochondrial disorders. Am. J. Med. Genet. 2001, 106, 4–17. [Google Scholar] [CrossRef]
- Larsson, N.-G.; Clayton, D.A. Molecular genetic aspects of human mitochondrial disorders. Annu. Rev. Genet. 1995, 29, 151–178. [Google Scholar] [CrossRef]
- Ruiz-Pesini, E.; Lott, M.T.; Procaccio, V.; Poole, J.C.; Brandon, M.C.; Mishmar, D.; Yi, C.; Kreuziger, J.; Baldi, P.; Wallace, D.C. An enhanced MITOMAP with a global mtDNA mutational phylogeny. Nucleic Acids Res. 2007, 35 (Suppl. S1), D823–D828. [Google Scholar] [CrossRef]
- Rötig, A. Genetic bases of mitochondrial respiratory chain disorders. Diabetes Metab. 2010, 36, 97–107. [Google Scholar] [CrossRef]
- DiMauro, S.; Tanji, K.; Schon, E.A. The many clinical faces of cytochrome c oxidase deficiency. In Mitochondrial Oxidative Phosphorylation; Springer: Berlin/Heidelberg, Germany, 2012; pp. 341–357. [Google Scholar]
- Rak, M.; Bénit, P.; Chrétien, D.; Bouchereau, J.; Schiff, M.; El-Khoury, R.; Tzagoloff, A.; Rustin, P. Mitochondrial cytochrome c oxidase deficiency. Clin. Sci. 2016, 130, 393–407. [Google Scholar] [CrossRef]
- Holt, I.; Harding, A.; Petty, R.; Morgan-Hughes, J. A new mitochondrial disease associated with mitochondrial DNA heteroplasmy. Am. J. Hum. Genet. 1990, 46, 428. [Google Scholar]
- Jonckheere, A.I.; Smeitink, J.A.; Rodenburg, R.J. Mitochondrial ATP synthase: Architecture, function and pathology. J. Inherit. Metab. Dis. 2012, 35, 211–225. [Google Scholar] [CrossRef]
- Gruschus, J.M.; Morris, D.L.; Tjandra, N. Evidence of natural selection in the mitochondrial-derived peptides humanin and SHLP6. Sci. Rep. 2023, 13, 14110. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Niikura, T.; Tajima, H.; Yasukawa, T.; Sudo, H.; Ito, Y.; Kita, Y.; Kawasumi, M.; Kouyama, K.; Doyu, M. A rescue factor abolishing neuronal cell death by a wide spectrum of familial Alzheimer’s disease genes and Aβ. Proc. Natl. Acad. Sci. USA 2001, 98, 6336–6341. [Google Scholar] [CrossRef]
- Guo, B.; Zhai, D.; Cabezas, E.; Welsh, K.; Nouraini, S.; Satterthwait, A.C.; Reed, J.C. Humanin peptide suppresses apoptosis by interfering with Bax activation. Nature 2003, 423, 456–461. [Google Scholar] [CrossRef]
- Lee, C.; Yen, K.; Cohen, P. Humanin: A harbinger of mitochondrial-derived peptides? Trends Endocrinol. Metab. 2013, 24, 222–228. [Google Scholar] [CrossRef]
- Ikonen, M.; Liu, B.; Hashimoto, Y.; Ma, L.; Lee, K.-W.; Niikura, T.; Nishimoto, I.; Cohen, P. Interaction between the Alzheimer’s survival peptide humanin and insulin-like growth factor-binding protein 3 regulates cell survival and apoptosis. Proc. Natl. Acad. Sci. USA 2003, 100, 13042–13047. [Google Scholar] [CrossRef]
- Luciano, F.; Zhai, D.; Zhu, X.; Bailly-Maitre, B.; Ricci, J.-E.; Satterthwait, A.C.; Reed, J.C. Cytoprotective peptide humanin binds and inhibits proapoptotic Bcl-2/Bax family protein BimEL. J. Biol. Chem. 2005, 280, 15825–15835. [Google Scholar] [CrossRef]
- Morris, D.L.; Johnson, S.; Bleck, C.K.; Lee, D.-Y.; Tjandra, N. Humanin selectively prevents the activation of pro-apoptotic protein BID by sequestering it into fibers. J. Biol. Chem. 2020, 295, 18226–18238. [Google Scholar] [CrossRef]
- Ying, G.; Iribarren, P.; Zhou, Y.; Gong, W.; Zhang, N.; Yu, Z.-X.; Le, Y.; Cui, Y.; Wang, J.M. Humanin, a newly identified neuroprotective factor, uses the G protein-coupled formylpeptide receptor-like-1 as a functional receptor. J. Immunol. 2004, 172, 7078–7085. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Kurita, M.; Aiso, S.; Nishimoto, I.; Matsuoka, M. Humanin inhibits neuronal cell death by interacting with a cytokine receptor complex or complexes involving CNTF receptor α/WSX-1/gp130. Mol. Biol. Cell 2009, 20, 2864–2873. [Google Scholar] [CrossRef]
- Cobb, L.J.; Lee, C.; Xiao, J.; Yen, K.; Wong, R.G.; Nakamura, H.K.; Mehta, H.H.; Gao, Q.; Ashur, C.; Huffman, D.M. Naturally occurring mitochondrial-derived peptides are age-dependent regulators of apoptosis, insulin sensitivity, and inflammatory markers. Aging 2016, 8, 796. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Zeng, J.; Drew, B.G.; Sallam, T.; Martin-Montalvo, A.; Wan, J.; Kim, S.-J.; Mehta, H.; Hevener, A.L.; de Cabo, R. The mitochondrial-derived peptide MOTS-c promotes metabolic homeostasis and reduces obesity and insulin resistance. Cell Metab. 2015, 21, 443–454. [Google Scholar] [CrossRef] [PubMed]
- Miller, B.; Kim, S.-J.; Kumagai, H.; Yen, K.; Cohen, P. Mitochondria-derived peptides in aging and healthspan. J. Clin. Investig. 2022, 132, e158449. [Google Scholar] [CrossRef] [PubMed]
- Dabravolski, S.A.; Nikiforov, N.G.; Starodubova, A.V.; Popkova, T.V.; Orekhov, A.N. The role of mitochondria-derived peptides in cardiovascular diseases and their potential as therapeutic targets. Int. J. Mol. Sci. 2021, 22, 8770. [Google Scholar] [CrossRef] [PubMed]
- Mavraki, E.; Labrum, R.; Sergeant, K.; Alston, C.L.; Woodward, C.; Smith, C.; Knowles, C.V.; Patel, Y.; Hodsdon, P.; Baines, J.P. Genetic testing for mitochondrial disease: The United Kingdom best practice guidelines. Eur. J. Hum. Genet. 2023, 31, 148–163. [Google Scholar] [CrossRef] [PubMed]
- Gorman, G.S.; Chinnery, P.F.; DiMauro, S.; Hirano, M.; Koga, Y.; McFarland, R.; Suomalainen, A.; Thorburn, D.R.; Zeviani, M.; Turnbull, D.M. Mitochondrial diseases. Nat. Rev. Dis. Primers 2016, 2, 16080. [Google Scholar] [CrossRef] [PubMed]
- Sutovsky, P.; Van Leyen, K.; McCauley, T.; Day, B.N.; Sutovsky, M. Degradation of paternal mitochondria after fertilization: Implications for heteroplasmy, assisted reproductive technologies and mtDNA inheritance. Reprod. Biomed. Online 2004, 8, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Thompson, W.E.; Ramalho-Santos, J.; Sutovsky, P. Ubiquitination of prohibitin in mammalian sperm mitochondria: Possible roles in the regulation of mitochondrial inheritance and sperm quality control. Biol. Reprod. 2003, 69, 254–260. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.; Sato, K. Maternal inheritance of mitochondrial DNA by diverse mechanisms to eliminate paternal mitochondrial DNA. Biochim. Biophys. Acta (BBA)—Mol. Cell Res. 2013, 1833, 1979–1984. [Google Scholar] [CrossRef] [PubMed]
- Cummins, J.M.; Jequier, A.M.; Kan, R. Molecular biology of human male infertility: Links with aging, mitochondrial genetics, and oxidative stress? Mol. Reprod. Dev. 1994, 37, 345–362. [Google Scholar] [CrossRef] [PubMed]
- Shamsi, M.B.; Kumar, R.; Bhatt, A.; Bamezai, R.; Kumar, R.; Gupta, N.P.; Das, T.; Dada, R. Mitochondrial DNA mutations in etiopathogenesis of male infertility. Indian J. Urol. IJU J. Urol. Soc. India 2008, 24, 150. [Google Scholar]
- Song, W.-H.; Ballard, J.W.O.; Yi, Y.-J.; Sutovsky, P. Regulation of mitochondrial genome inheritance by autophagy and ubiquitin-proteasome system: Implications for health, fitness, and fertility. BioMed Res. Int. 2014, 2014, 981867. [Google Scholar] [CrossRef]
- Hoekstra, R.F. Evolutionary origin and consequences of uniparental mitochondrial inheritance. Hum. Reprod. 2000, 15 (Suppl. S2), 102–111. [Google Scholar] [CrossRef] [PubMed]
- Ankel-Simons, F.; Cummins, J.M. Misconceptions about mitochondria and mammalian fertilization: Implications for theories on human evolution. Proc. Natl. Acad. Sci. USA 1996, 93, 13859–13863. [Google Scholar] [CrossRef] [PubMed]
- Steuerwald, N.; Barritt, J.A.; Adler, R.; Malter, H.; Schimmel, T.; Cohen, J.; Brenner, C.A. Quantification of mtDNA in single oocytes, polar bodies and subcellular components by real-time rapid cycle fluorescence monitored PCR. Zygote 2000, 8, 209–215. [Google Scholar] [CrossRef]
- Jansen, R.P.; de Boer, K. The bottleneck: Mitochondrial imperatives in oogenesis and ovarian follicular fate. Mol. Cell. Endocrinol. 1998, 145, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Chinnery, P.F.; Thorburn, D.R.; Samuels, D.C.; White, S.L.; Dahl, H.-H.M.; Turnbull, D.M.; Lightowlers, R.N.; Howell, N. The inheritance of mitochondrial DNA heteroplasmy: Random drift, selection or both? Trends Genet. 2000, 16, 500–505. [Google Scholar] [CrossRef] [PubMed]
- Marchington, D.; Hartshorne, G.; Barlow, D.; Poulton, J. Homopolymeric tract heteroplasmy in mtDNA from tissues and single oocytes: Support for a genetic bottleneck. Am. J. Hum. Genet. 1997, 60, 408. [Google Scholar] [PubMed]
- Lynch, M. Mutation accumulation in transfer RNAs: Molecular evidence for Muller’s ratchet in mitochondrial genomes. Mol. Biol. Evol. 1996, 13, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Otten, A.B.; Smeets, H.J. Evolutionary defined role of the mitochondrial DNA in fertility, disease and ageing. Hum. Reprod. Update 2015, 21, 671–689. [Google Scholar] [CrossRef] [PubMed]
- Muller, H.J. The relation of recombination to mutational advance. Mutat. Res./Fundam. Mol. Mech. Mutagen. 1964, 1, 2–9. [Google Scholar] [CrossRef]
- Sutovsky, P. Ubiquitin-dependent proteolysis in mammalian spermatogenesis, fertilization, and sperm quality control: Killing three birds with one stone. Microsc. Res. Technol. 2003, 61, 88–102. [Google Scholar] [CrossRef]
- Schwartz, M.; Vissing, J. Paternal inheritance of mitochondrial DNA. N. Engl. J. Med. 2002, 347, 576–580. [Google Scholar] [CrossRef] [PubMed]
- Egger, J.; Wilson, J. Mitochondrial inheritance in a mitochondrially mediated disease. N. Engl. J. Med. 1983, 309, 142–146. [Google Scholar] [CrossRef]
- Schwartz, M.; Vissing, J. No evidence for paternal inheritance of mtDNA in patients with sporadic mtDNA mutations. J. Neurol. Sci. 2004, 218, 99–101. [Google Scholar] [CrossRef]
- Taylor, R.W.; McDonnell, M.T.; Blakely, E.L.; Chinnery, P.F.; Taylor, G.A.; Howell, N.; Zeviani, M.; Briem, E.; Carrara, F.; Turnbull, D.M. Genotypes from patients indicate no paternal mitochondrial DNA contribution. Ann. Neurol. Off. J. Am. Neurol. Assoc. Child Neurol. Soc. 2003, 54, 521–524. [Google Scholar] [CrossRef]
- Filosto, M.; Mancuso, M.; Vives-Bauza, C.; Vilà, M.R.; Shanske, S.; Hirano, M.; Andreu, A.L.; DiMauro, S. Lack of paternal inheritance of muscle mitochondrial DNA in sporadic mitochondrial myopathies. Ann. Neurol. Off. J. Am. Neurol. Assoc. Child Neurol. Soc. 2003, 54, 524–526. [Google Scholar] [CrossRef] [PubMed]
- Bandelt, H.-J.; Kong, Q.-P.; Parson, W.; Salas, A. More evidence for non-maternal inheritance of mitochondrial DNA? J. Med. Genet. 2005, 42, 957–960. [Google Scholar] [CrossRef]
- Luo, S.; Valencia, C.A.; Zhang, J.; Lee, N.-C.; Slone, J.; Gui, B.; Wang, X.; Li, Z.; Dell, S.; Brown, J. Biparental inheritance of mitochondrial DNA in humans. Proc. Natl. Acad. Sci. USA 2018, 115, 13039–13044. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Pagnamenta, A.T.; Gleadall, N.; Sanchis-Juan, A.; Stephens, J.; Broxholme, J.; Tuna, S.; Odhams, C.A.; Fratter, C.; Turro, E. Nuclear-mitochondrial DNA segments resemble paternally inherited mitochondrial DNA in humans. Nat. Commun. 2020, 11, 1740. [Google Scholar] [CrossRef]
- Lutz-Bonengel, S.; Niederstätter, H.; Naue, J.; Koziel, R.; Yang, F.; Sänger, T.; Huber, G.; Berger, C.; Pflugradt, R.; Strobl, C. Evidence for multi-copy Mega-NUMT s in the human genome. Nucleic Acids Res. 2021, 49, 1517–1531. [Google Scholar] [CrossRef]
- Saccone, C.; Gissi, C.; Lanave, C.; Larizza, A.; Pesole, G.; Reyes, A. Evolution of the mitochondrial genetic system: An overview. Gene 2000, 261, 153–159. [Google Scholar] [CrossRef]
- Mambo, E.; Gao, X.; Cohen, Y.; Guo, Z.; Talalay, P.; Sidransky, D. Electrophile and oxidant damage of mitochondrial DNA leading to rapid evolution of homoplasmic mutations. Proc. Natl. Acad. Sci. USA 2003, 100, 1838–1843. [Google Scholar] [CrossRef] [PubMed]
- Mattiazzi, M.; Vijayvergiya, C.; Gajewski, C.D.; DeVivo, D.C.; Lenaz, G.; Wiedmann, M.; Manfredi, G. The mtDNA T8993G (NARP) mutation results in an impairment of oxidative phosphorylation that can be improved by antioxidants. Hum. Mol. Genet. 2004, 13, 869–879. [Google Scholar] [CrossRef] [PubMed]
- Nijtmans, L.G.; Henderson, N.S.; Attardi, G.; Holt, I.J. Impaired ATP synthase assembly associated with a mutation in the human ATP synthase subunit 6 gene. J. Biol. Chem. 2001, 276, 6755–6762. [Google Scholar] [CrossRef] [PubMed]
- Song, S.; Pursell, Z.F.; Copeland, W.C.; Longley, M.J.; Kunkel, T.A.; Mathews, C.K. DNA precursor asymmetries in mammalian tissue mitochondria and possible contribution to mutagenesis through reduced replication fidelity. Proc. Natl. Acad. Sci. USA 2005, 102, 4990–4995. [Google Scholar] [CrossRef] [PubMed]
- Anderson, A.P.; Luo, X.; Russell, W.; Yin, Y.W. Oxidative damage diminishes mitochondrial DNA polymerase replication fidelity. Nucleic Acids Res. 2020, 48, 817–829. [Google Scholar] [CrossRef] [PubMed]
- Bebenek, K.; Kunkel, T.A. Functions of DNA polymerases. Adv. Protein Chem. 2004, 69, 137–165. [Google Scholar]
- Chinnery, P.F.; Elliott, H.R.; Hudson, G.; Samuels, D.C.; Relton, C.L. Epigenetics, epidemiology and mitochondrial DNA diseases. Int. J. Epidemiol. 2012, 41, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Kukat, C.; Wurm, C.A.; Spåhr, H.; Falkenberg, M.; Larsson, N.-G.; Jakobs, S. Super-resolution microscopy reveals that mammalian mitochondrial nucleoids have a uniform size and frequently contain a single copy of mtDNA. Proc. Natl. Acad. Sci. USA 2011, 108, 13534–13539. [Google Scholar] [CrossRef] [PubMed]
- Elson, J.L.; Majamaa, K.; Howell, N.; Chinnery, P.F. Associating mitochondrial DNA variation with complex traits. Am. J. Hum. Genet. 2007, 80, 378–382. [Google Scholar] [CrossRef][Green Version]
- Guyatt, A.L.; Brennan, R.R.; Burrows, K.; Guthrie, P.A.; Ascione, R.; Ring, S.M.; Gaunt, T.R.; Pyle, A.; Cordell, H.J.; Lawlor, D.A. A genome-wide association study of mitochondrial DNA copy number in two population-based cohorts. Hum. Genom. 2019, 13, 6. [Google Scholar] [CrossRef]
- Filograna, R.; Mennuni, M.; Alsina, D.; Larsson, N.G. Mitochondrial DNA copy number in human disease: The more the better? FEBS Lett. 2021, 595, 976–1002. [Google Scholar] [CrossRef]
- Elson, J.; Samuels, D.; Turnbull, D.; Chinnery, P. Random intracellular drift explains the clonal expansion of mitochondrial DNA mutations with age. Am. J. Hum. Genet. 2001, 68, 802–806. [Google Scholar] [CrossRef]
- Wei, W.; Keogh, M.J.; Wilson, I.; Coxhead, J.; Ryan, S.; Rollinson, S.; Griffin, H.; Kurzawa-Akanbi, M.; Santibanez-Koref, M.; Talbot, K. Mitochondrial DNA point mutations and relative copy number in 1363 disease and control human brains. Acta Neuropathol. Commun. 2017, 5, 13. [Google Scholar] [CrossRef]
- Yamamoto, K.; Sakaue, S.; Matsuda, K.; Murakami, Y.; Kamatani, Y.; Ozono, K.; Momozawa, Y.; Okada, Y. Genetic and phenotypic landscape of the mitochondrial genome in the Japanese population. Commun. Biol. 2020, 3, 104. [Google Scholar] [CrossRef]
- Gentiluomo, M.; Giaccherini, M.; Gào, X.; Guo, F.; Stocker, H.; Schöttker, B.; Brenner, H.; Canzian, F.; Campa, D. Genome-wide association study of mitochondrial copy number. Hum. Mol. Genet. 2022, 31, 1346–1355. [Google Scholar] [CrossRef] [PubMed]
- Larsson, N.-G.; Wang, J.; Wilhelmsson, H.; Oldfors, A.; Rustin, P.; Lewandoski, M.; Barsh, G.S.; Clayton, D.A. Mitochondrial transcription factor A is necessary for mtDNA maintance and embryogenesis in mice. Nat. Genet. 1998, 18, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Matsushima, Y.; Matsumura, K.; Ishii, S.; Inagaki, H.; Suzuki, T.; Matsuda, Y.; Beck, K.; Kitagawa, Y. Functional domains of chicken mitochondrial transcription factor A for the maintenance of mitochondrial DNA copy number in lymphoma cell line DT40. J. Biol. Chem. 2003, 278, 31149–31158. [Google Scholar] [CrossRef]
- Ekstrand, M.I.; Falkenberg, M.; Rantanen, A.; Park, C.B.; Gaspari, M.; Hultenby, K.; Rustin, P.; Gustafsson, C.M.; Larsson, N.-G. Mitochondrial transcription factor A regulates mtDNA copy number in mammals. Hum. Mol. Genet. 2004, 13, 935–944. [Google Scholar] [CrossRef]
- Kanki, T.; Ohgaki, K.; Gaspari, M.; Gustafsson, C.M.; Fukuoh, A.; Sasaki, N.; Hamasaki, N.; Kang, D. Architectural role of mitochondrial transcription factor A in maintenance of human mitochondrial DNA. Mol. Cell. Biol. 2004, 24, 9823–9834. [Google Scholar] [CrossRef]
- Curran, J.E.; Johnson, M.P.; Dyer, T.D.; Göring, H.H.; Kent, J.W.; Charlesworth, J.C.; Borg, A.J.; Jowett, J.B.; Cole, S.A.; MacCluer, J.W. Genetic determinants of mitochondrial content. Hum. Mol. Genet. 2007, 16, 1504–1514. [Google Scholar] [CrossRef][Green Version]
- Cai, N.; Li, Y.; Chang, S.; Liang, J.; Lin, C.; Zhang, X.; Liang, L.; Hu, J.; Chan, W.; Kendler, K.S. Genetic control over mtDNA and its relationship to major depressive disorder. Curr. Biol. 2015, 25, 3170–3177. [Google Scholar] [CrossRef] [PubMed]
- Longchamps, R.; Yang, S.; Castellani, C.; Shi, W.; Lane, J.; Grove, M.; Bartz, T.; Sarnowski, C.; Liu, C.; Burrows, K. Genome-wide analysis of mitochondrial DNA copy number reveals loci implicated in nucleotide metabolism, platelet activation, and megakaryocyte proliferation. Hum. Genet. 2022, 141, 127–146. [Google Scholar] [CrossRef] [PubMed]
- Nishino, I.; Spinazzola, A.; Hirano, M. Thymidine phosphorylase gene mutations in MNGIE, a human mitochondrial disorder. Science 1999, 283, 689–692. [Google Scholar] [CrossRef] [PubMed]
- Kaukonen, J.; Juselius, J.K.; Tiranti, V.; Kyttälä, A.; Zeviani, M.; Comi, G.P.; Keränen, S.; Peltonen, L.; Suomalainen, A. Role of adenine nucleotide translocator 1 in mtDNA maintenance. Science 2000, 289, 782–785. [Google Scholar] [CrossRef] [PubMed]
- Moraes, C.T. What regulates mitochondrial DNA copy number in animal cells? TRENDS Genet. 2001, 17, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.; Johnson, J.; Gough, D.J.; Donoghue, J.; Cagnone, G.; Vaghjiani, V.; Brown, K.A.; Johns, T.G.; St John, J.C. Mitochondrial DNA copy number is regulated by DNA methylation and demethylation of POLGA in stem and cancer cells and their differentiated progeny. Cell Death Dis. 2015, 6, e1664. [Google Scholar] [CrossRef] [PubMed]
- Guha, M.; Avadhani, N.G. Mitochondrial retrograde signaling at the crossroads of tumor bioenergetics, genetics and epigenetics. Mitochondrion 2013, 13, 577–591. [Google Scholar] [CrossRef]
- Castellani, C.A.; Longchamps, R.J.; Sun, J.; Guallar, E.; Arking, D.E. Thinking outside the nucleus: Mitochondrial DNA copy number in health and disease. Mitochondrion 2020, 53, 214–223. [Google Scholar] [CrossRef]
- Jeng, J.Y.; Yeh, T.S.; Lee, J.W.; Lin, S.H.; Fong, T.H.; Hsieh, R.H. Maintenance of mitochondrial DNA copy number and expression are essential for preservation of mitochondrial function and cell growth. J. Cell. Biochem. 2008, 103, 347–357. [Google Scholar] [CrossRef]
- Zhang, Z.; Yang, D.; Zhou, B.; Luan, Y.; Yao, Q.; Liu, Y.; Yang, S.; Jia, J.; Xu, Y.; Bie, X. Decrease of MtDNA copy number affects mitochondrial function and involves in the pathological consequences of ischaemic stroke. J. Cell. Mol. Med. 2022, 26, 4157–4168. [Google Scholar] [CrossRef]
- Ashar, F.N.; Zhang, Y.; Longchamps, R.J.; Lane, J.; Moes, A.; Grove, M.L.; Mychaleckyj, J.C.; Taylor, K.D.; Coresh, J.; Rotter, J.I. Association of mitochondrial DNA copy number with cardiovascular disease. JAMA Cardiol. 2017, 2, 1247–1255. [Google Scholar] [CrossRef] [PubMed]
- Tin, A.; Grams, M.E.; Ashar, F.N.; Lane, J.A.; Rosenberg, A.Z.; Grove, M.L.; Boerwinkle, E.; Selvin, E.; Coresh, J.; Pankratz, N. Association between mitochondrial DNA copy number in peripheral blood and incident CKD in the atherosclerosis risk in communities study. J. Am. Soc. Nephrol. 2016, 27, 2467–2473. [Google Scholar] [CrossRef]
- Guo, W.; Jiang, L.; Bhasin, S.; Khan, S.M.; Swerdlow, R.H. DNA extraction procedures meaningfully influence qPCR-based mtDNA copy number determination. Mitochondrion 2009, 9, 261–265. [Google Scholar] [CrossRef] [PubMed]
- Longchamps, R.J.; Castellani, C.A.; Yang, S.Y.; Newcomb, C.E.; Sumpter, J.A.; Lane, J.; Grove, M.L.; Guallar, E.; Pankratz, N.; Taylor, K.D. Evaluation of mitochondrial DNA copy number estimation techniques. PLoS ONE 2020, 15, e0228166. [Google Scholar] [CrossRef] [PubMed]
- Garg, P.; Jadhav, B.; Lee, W.; Rodriguez, O.L.; Martin-Trujillo, A.; Sharp, A.J. A phenome-wide association study identifies effects of copy-number variation of VNTRs and multicopy genes on multiple human traits. Am. J. Hum. Genet. 2022, 109, 1065–1076. [Google Scholar] [CrossRef]
- Aguirre, M.; Rivas, M.A.; Priest, J. Phenome-wide burden of copy-number variation in the UK biobank. Am. J. Hum. Genet. 2019, 105, 373–383. [Google Scholar] [CrossRef]
- Chong, M.; Mohammadi-Shemirani, P.; Perrot, N.; Nelson, W.; Morton, R.; Narula, S.; Lali, R.; Khan, I.; Khan, M.; Judge, C. GWAS and ExWAS of blood mitochondrial DNA copy number identifies 71 loci and highlights a potential causal role in dementia. Elife 2022, 11, e70382. [Google Scholar] [CrossRef] [PubMed]
- Chouchani, E.T.; Methner, C.; Nadtochiy, S.M.; Logan, A.; Pell, V.R.; Ding, S.; James, A.M.; Cochemé, H.M.; Reinhold, J.; Lilley, K.S. Cardioprotection by S-nitrosation of a cysteine switch on mitochondrial complex I. Nat. Med. 2013, 19, 753–759. [Google Scholar] [CrossRef] [PubMed]
- Schon, E.A.; DiMauro, S.; Hirano, M. Human mitochondrial DNA: Roles of inherited and somatic mutations. Nat. Rev. Genet. 2012, 13, 878–890. [Google Scholar] [CrossRef]
- Sanglard, L.P.; Kuehn, L.A.; Snelling, W.M.; Spangler, M.L. Influence of environmental factors and genetic variation on mitochondrial DNA copy number. J. Anim. Sci. 2022, 100, skac059. [Google Scholar] [CrossRef]
- Hägg, S.; Jylhävä, J.; Wang, Y.; Czene, K.; Grassmann, F. Deciphering the genetic and epidemiological landscape of mitochondrial DNA abundance. Hum. Genet. 2021, 140, 849–861. [Google Scholar] [CrossRef]
- Harerimana, N.V.; Paliwali, D.; Romero-Molina, C.; Bennett, D.A.; Pa, J.; Goate, A.; Swerdlow, R.H.; Andrews, S.J. The role of mitochondrial genome abundance in Alzheimer’s disease. Alzheimer’s Dement. 2023, 19, 2069–2083. [Google Scholar] [CrossRef] [PubMed]
- Alvarado-Vásquez, N. Circulating Cell-Free Mitochondrial DNA as the Probable Inducer of Early Endothelial Dysfunction in the Prediabetic Patient; Elsevier: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Liu, J.; Cai, X.; Xie, L.; Tang, Y.; Cheng, J.; Wang, J.; Wang, L.; Gong, J. Circulating Cell Free Mitochondrial DNA is a Biomarker in the Development of Coronary Heart Disease in the Patients with Type 2 Diabetes. Clin. Lab. 2015, 61, 661–667. [Google Scholar] [CrossRef]
- Varhaug, K.N.; Vedeler, C.A.; Myhr, K.-M.; Aarseth, J.H.; Tzoulis, C.; Bindoff, L.A. Increased levels of cell-free mitochondrial DNA in the cerebrospinal fluid of patients with multiple sclerosis. Mitochondrion 2017, 34, 32–35. [Google Scholar] [CrossRef] [PubMed]
- Sudakov, N.; Popkova, T.; Katyshev, A.; Goldberg, O.; Nikiforov, S.; Pushkarev, B.; Klimenkov, I.; Lepekhova, S.; Apartsin, K.; Nevinsky, G. Level of blood cell-free circulating mitochondrial DNA as a novel biomarker of acute myocardial ischemia. Biochemistry 2015, 80, 1387–1392. [Google Scholar] [CrossRef]
- Dantham, S.; Srivastava, A.K.; Gulati, S.; Rajeswari, M.R. Plasma circulating cell-free mitochondrial DNA in the assessment of Friedreich’s ataxia. J. Neurol. Sci. 2016, 365, 82–88. [Google Scholar] [CrossRef]
- Berry, W.; Algar, E.; Kumar, B.; Desmond, C.; Swan, M.; Jenkins, B.J.; Croagh, D. Endoscopic ultrasound-guided fine-needle aspirate-derived preclinical pancreatic cancer models reveal panitumumab sensitivity in KRAS wild-type tumors. Int. J. Cancer 2017, 140, 2331–2343. [Google Scholar] [CrossRef]
- Lu, H.; Busch, J.; Jung, M.; Rabenhorst, S.; Ralla, B.; Kilic, E.; Mergemeier, S.; Budach, N.; Fendler, A.; Jung, K. Diagnostic and prognostic potential of circulating cell-free genomic and mitochondrial DNA fragments in clear cell renal cell carcinoma patients. Clin. Chim. Acta 2016, 452, 109–119. [Google Scholar] [CrossRef]
- Kohler, C.; Radpour, R.; Barekati, Z.; Asadollahi, R.; Bitzer, J.; Wight, E.; Bürki, N.; Diesch, C.; Holzgreve, W.; Zhong, X.Y. Levels of plasma circulating cell free nuclear and mitochondrial DNA as potential biomarkers for breast tumors. Mol. Cancer 2009, 8, 105. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, E.H.; Fawzy, A.; Ahmad, O.K.; Ali, A.M. Plasma circulating cell-free nuclear and mitochondrial DNA as potential biomarkers in the peripheral blood of breast cancer patients. Asian Pac. J. Cancer Prev. 2016, 16, 8299–8305. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Itagaki, K.; Hauser, C.J. Mitochondrial DNA is released by shock and activates neutrophils via p38 map kinase. Shock 2010, 34, 55–59. [Google Scholar] [CrossRef]
- Bisharyan, Y.; Clark, T.G. Calcium-dependent mitochondrial extrusion in ciliated protozoa. Mitochondrion 2011, 11, 909–918. [Google Scholar] [CrossRef]
- Rehman, A.; Kumari, R.; Kamthan, A.; Tiwari, R.; Srivastava, R.K.; van der Westhuizen, F.H.; Mishra, P.K. Cell-free circulating mitochondrial DNA: An emerging biomarker for airborne particulate matter associated with cardiovascular diseases. Free. Radic. Biol. Med. 2023, 195, 103–120. [Google Scholar] [CrossRef]
- Podlesniy, P.; Figueiro-Silva, J.; Llado, A.; Antonell, A.; Sanchez-Valle, R.; Alcolea, D.; Lleo, A.; Molinuevo, J.L.; Serra, N.; Trullas, R. Low cerebrospinal fluid concentration of mitochondrial DNA in preclinical Alzheimer disease. Ann. Neurol. 2013, 74, 655–668. [Google Scholar] [CrossRef]
- Gambardella, S.; Limanaqi, F.; Ferese, R.; Biagioni, F.; Campopiano, R.; Centonze, D.; Fornai, F. ccf-mtDNA as a potential link between the brain and immune system in neuro-immunological disorders. Front. Immunol. 2019, 10, 1064. [Google Scholar] [CrossRef]
- Stewart, J.B.; Chinnery, P.F. The dynamics of mitochondrial DNA heteroplasmy: Implications for human health and disease. Nat. Rev. Genet. 2015, 16, 530–542. [Google Scholar] [CrossRef]
- Szczepanowska, K.; Trifunovic, A. Different faces of mitochondrial DNA mutators. Biochim. Biophys. Acta (BBA)—Bioenerg. 2015, 1847, 1362–1372. [Google Scholar] [CrossRef] [PubMed]
- Durham, S.E.; Samuels, D.C.; Cree, L.M.; Chinnery, P.F. Normal levels of wild-type mitochondrial DNA maintain cytochrome c oxidase activity for two pathogenic mitochondrial DNA mutations but not for m. 3243A→G. Am. J. Hum. Genet. 2007, 81, 189–195. [Google Scholar] [CrossRef]
- Walker, M.A.; Lareau, C.A.; Ludwig, L.S.; Karaa, A.; Sankaran, V.G.; Regev, A.; Mootha, V.K. Purifying selection against pathogenic mitochondrial DNA in human T cells. N. Engl. J. Med. 2020, 383, 1556–1563. [Google Scholar] [CrossRef]
- Li, M.; Schröder, R.; Ni, S.; Madea, B.; Stoneking, M. Extensive tissue-related and allele-related mtDNA heteroplasmy suggests positive selection for somatic mutations. Proc. Natl. Acad. Sci. USA 2015, 112, 2491–2496. [Google Scholar] [CrossRef] [PubMed]
- Albayrak, L.; Khanipov, K.; Pimenova, M.; Golovko, G.; Rojas, M.; Pavlidis, I.; Chumakov, S.; Aguilar, G.; Chávez, A.; Widger, W.R. The ability of human nuclear DNA to cause false positive low-abundance heteroplasmy calls varies across the mitochondrial genome. BMC Genom. 2016, 17, 1017. [Google Scholar] [CrossRef] [PubMed]
- Maude, H.; Davidson, M.; Charitakis, N.; Diaz, L.; Bowers, W.H.; Gradovich, E.; Andrew, T.; Huntley, D. NUMT confounding biases mitochondrial heteroplasmy calls in favor of the reference allele. Front. Cell Dev. Biol. 2019, 7, 201. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.B.; Chinnery, P.F. Extreme heterogeneity of human mitochondrial DNA from organelles to populations. Nat. Rev. Genet. 2021, 22, 106–118. [Google Scholar] [CrossRef]
- Wei, W.; Tuna, S.; Keogh, M.J.; Smith, K.R.; Aitman, T.J.; Beales, P.L.; Bennett, D.L.; Gale, D.P.; Bitner-Glindzicz, M.A.; Black, G.C. Germline selection shapes human mitochondrial DNA diversity. Science 2019, 364, eaau6520. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Schroeder, R.; Ko, A.; Stoneking, M. Fidelity of capture-enrichment for mtDNA genome sequencing: Influence of NUMTs. Nucleic Acids Res. 2012, 40, e137. [Google Scholar] [CrossRef] [PubMed]
- Calvignac, S.; Konecny, L.; Malard, F.; Douady, C.J. Preventing the pollution of mitochondrial datasets with nuclear mitochondrial paralogs (numts). Mitochondrion 2011, 11, 246–254. [Google Scholar] [CrossRef]
- Rossignol, R.; Faustin, B.; Rocher, C.; Malgat, M.; Mazat, J.-P.; Letellier, T. Mitochondrial threshold effects. Biochem. J. 2003, 370, 751–762. [Google Scholar] [CrossRef] [PubMed]
- Wallace, D.C.; Chalkia, D. Mitochondrial DNA genetics and the heteroplasmy conundrum in evolution and disease. Cold Spring Harb. Perspect. Biol. 2013, 5, a021220. [Google Scholar] [CrossRef] [PubMed]
- Wallace, D.C. Bioenergetics in human evolution and disease: Implications for the origins of biological complexity and the missing genetic variation of common diseases. Philos. Trans. R. Soc. B Biol. Sci. 2013, 368, 20120267. [Google Scholar] [CrossRef]
- Dür, A.; Huber, N.; Parson, W. Fine-tuning phylogenetic alignment and haplogrouping of mtDNA sequences. Int. J. Mol. Sci. 2021, 22, 5747. [Google Scholar] [CrossRef]
- Schönherr, S.; Weissensteiner, H.; Kronenberg, F.; Forer, L. Haplogrep 3-an interactive haplogroup classification and analysis platform. Nucleic Acids Res. 2023, 51, W263–W268. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, K.N.; Aubrey, E.; Howe, L.D.; Stergiakouli, E.; Rodriguez, S.; Kearney, P.M.; O’Keeffe, L.M. Mitochondrial DNA haplogroups and trajectories of cardiometabolic risk factors during childhood and adolescence: A prospective cohort study. PLoS ONE 2023, 18, e0284226. [Google Scholar] [CrossRef] [PubMed]
- Chinnery, P.F.; Gomez-Duran, A. Oldies but goldies mtDNA population variants and neurodegenerative diseases. Front. Neurosci. 2018, 12, 682. [Google Scholar] [CrossRef] [PubMed]
- Brandon, M.C.; Ruiz-Pesini, E.; Mishmar, D.; Procaccio, V.; Lott, M.T.; Nguyen, K.C.; Spolim, S.; Patil, U.; Baldi, P.; Wallace, D.C. MITOMASTER: A bioinformatics tool for the analysis of mitochondrial DNA sequences. Hum. Mutat. 2009, 30, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Yao, Y.-G. An update to MitoTool: Using a new scoring system for faster mtDNA haplogroup determination. Mitochondrion 2013, 13, 360–363. [Google Scholar] [CrossRef] [PubMed]
- Van Oven, M.; Kayser, M. Updated comprehensive phylogenetic tree of global human mitochondrial DNA variation. Hum. Mutat. 2009, 30, E386–E394. [Google Scholar] [CrossRef] [PubMed]
- Vianello, D.; Sevini, F.; Castellani, G.; Lomartire, L.; Capri, M.; Franceschi, C. HAPLOFIND: A New Method for High-Throughput mt DNA Haplogroup Assignment. Hum. Mutat. 2013, 34, 1189–1194. [Google Scholar] [CrossRef] [PubMed]
- Mishmar, D.; Ruiz-Pesini, E.; Golik, P.; Macaulay, V.; Clark, A.G.; Hosseini, S.; Brandon, M.; Easley, K.; Chen, E.; Brown, M.D. Natural selection shaped regional mtDNA variation in humans. Proc. Natl. Acad. Sci. USA 2003, 100, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Ingman, M.; Kaessmann, H.; Pääbo, S.; Gyllensten, U. Mitochondrial genome variation and the origin of modern humans. Nature 2000, 408, 708–713. [Google Scholar] [CrossRef]
- López, S.; Van Dorp, L.; Hellenthal, G. Human dispersal out of Africa: A lasting debate. Evol. Bioinform. 2015, 11, EBO-S33489. [Google Scholar] [CrossRef]
- Wallace, D.C. Mitochondrial DNA variation in human radiation and disease. Cell 2015, 163, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Wallace, D., Ruiz-Pesini, E., Mishmar, D., Eds.; mtDNA variation, climatic adaptation, degenerative diseases, and longevity. Cold Spring Harb. Symp. Quant. Biol. 2003, 68, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Pesini, E.; Mishmar, D.; Brandon, M.; Procaccio, V.; Wallace, D.C. Effects of purifying and adaptive selection on regional variation in human mtDNA. Science 2004, 303, 223–226. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Pesini, E.; Wallace, D.C. Evidence for adaptive selection acting on the tRNA and rRNA genes of human mitochondrial DNA. Hum. Mutat. 2006, 27, 1072–1081. [Google Scholar] [CrossRef] [PubMed]
- Wallace, D.C. Mitochondrial DNA mutations in disease and aging. Environ. Mol. Mutagen. 2010, 51, 440–450. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-S.; Torroni, A.; Excoffier, L.; Santachiara-Benerecetti, A.S.; Wallace, D.C. Analysis of mtDNA variation in African populations reveals the most ancient of all human continent-specific haplogroups. Am. J. Hum. Genet. 1995, 57, 133. [Google Scholar]
- Watson, E.; Forster, P.; Richards, M.; Bandelt, H.-J. Mitochondrial footprints of human expansions in Africa. Am. J. Hum. Genet. 1997, 61, 691–704. [Google Scholar] [CrossRef] [PubMed]
- Torroni, A.; Miller, J.A.; Moore, L.G.; Zamudio, S.; Zhuang, J.; Droma, T.; Wallace, D.C. Mitochondrial DNA analysis in Tibet: Implications for the origin of the Tibetan population and its adaptation to high altitude. Am. J. Phys. Anthropol. 1994, 93, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Kivisild, T.; Tolk, H.-V.; Parik, J.; Wang, Y.; Papiha, S.S.; Bandelt, H.-J.; Villems, R. The emerging limbs and twigs of the East Asian mtDNA tree. Mol. Biol. Evol. 2002, 19, 1737–1751. [Google Scholar] [CrossRef]
- Ballinger, S.W.; Schurr, T.G.; Torroni, A.; Gan, Y.-Y.; Hodge, J.A.; Hassan, K.; Chen, K.H.; Wallace, D.C. Southeast Asian mitochondrial DNA analysis reveals genetic continuity of ancient mongoloid migrations. Genetics 1992, 130, 139–152. [Google Scholar] [CrossRef]
- Comas, D.; Plaza, S.; Wells, R.S.; Yuldaseva, N.; Lao, O.; Calafell, F.; Bertranpetit, J. Admixture, migrations, and dispersals in Central Asia: Evidence from maternal DNA lineages. Eur. J. Hum. Genet. 2004, 12, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Macaulay, V.; Richards, M.; Hickey, E.; Vega, E.; Cruciani, F.; Guida, V.; Scozzari, R.; Bonné-Tamir, B.; Sykes, B.; Torroni, A. The emerging tree of West Eurasian mtDNAs: A synthesis of control-region sequences and RFLPs. Am. J. Hum. Genet. 1999, 64, 232–249. [Google Scholar] [CrossRef] [PubMed]
- Torroni, A.; Huoponen, K.; Francalacci, P.; Petrozzi, M.; Morelli, L.; Scozzari, R.; Obinu, D.; Savontaus, M.-L.; Wallace, D.C. Classification of European mtDNAs from an analysis of three European populations. Genetics 1996, 144, 1835–1850. [Google Scholar] [CrossRef]
- Torroni, A.; Schurr, T.G.; Cabell, M.F.; Brown, M.D.; Neel, J.V.; Larsen, M.; Smith, D.G.; Vullo, C.M.; Wallace, D.C. Asian affinities and continental radiation of the four founding Native American mtDNAs. Am. J. Hum. Genet. 1993, 53, 563. [Google Scholar] [PubMed]
- Johnson, D.C.; Shrestha, S.; Wiener, H.W.; Makowsky, R.; Kurundkar, A.; Wilson, C.M.; Aissani, B. Mitochondrial DNA diversity in the African American population. Mitochondrial DNA 2015, 26, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.D.; Torroni, A.; Reckord, C.L.; Wallace, D.C. Phylogenetic analysis of Leber’s hereditary optic neuropathy mitochondrial DNA’s indicates multiple independent occurrences of the common mutations. Hum. Mutat. 1995, 6, 311–325. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.D.; Sun, F.; Wallace, D.C. Clustering of Caucasian Leber hereditary optic neuropathy patients containing the 11778 or 14484 mutations on an mtDNA lineage. Am. J. Hum. Genet. 1997, 60, 381. [Google Scholar] [PubMed]
- Brown, M.D.; Starikovskaya, E.; Derbeneva, O.; Hosseini, S.; Allen, J.C.; Mikhailovskaya, I.E.; Sukernik, R.I.; Wallace, D.C. The role of mtDNA background in disease expression: A new primary LHON mutation associated with Western Eurasian haplogroup J. Hum. Genet. 2002, 110, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Veronese, N.; Stubbs, B.; Koyanagi, A.; Vaona, A.; Demurtas, J.; Schofield, P.; Maggi, S. Mitochondrial genetic haplogroups and cardiovascular diseases: Data from the Osteoarthritis Initiative. PLoS ONE 2019, 14, e0213656. [Google Scholar] [CrossRef]
- Sun, D.; Wei, Y.; Zheng, H.-X.; Jin, L.; Wang, J. Contribution of mitochondrial DNA variation to chronic disease in East Asian populations. Front. Mol. Biosci. 2019, 6, 128. [Google Scholar] [CrossRef]
- Antonyova, V.; Kejík, Z.; Brogyanyi, T.; Kaplánek, R.; Pajkova, M.; Talianova, V.; Hromadka, R.; Masařík, M.; Sýkora, D.; Mikšátková, L. Role of mtDNA disturbances in the pathogenesis of Alzheimer’s and Parkinson’s disease. DNA Repair 2020, 91, 102871. [Google Scholar] [CrossRef]
- Marom, S.; Friger, M.; Mishmar, D. MtDNA meta-analysis reveals both phenotype specificity and allele heterogeneity: A model for differential association. Sci. Rep. 2017, 7, 43449. [Google Scholar] [CrossRef]
- Elson, J.L.; Andrews, R.M.; Chinnery, P.F.; Lightowlers, R.N.; Turnbull, D.M.; Howell, N. Analysis of European mtDNAs for recombination. Am. J. Hum. Genet. 2001, 68, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Taanman, J.-W. The mitochondrial genome: Structure, transcription, translation and replication. Biochim. Biophys. Acta (BBA)—Bioenerg. 1999, 1410, 103–123. [Google Scholar] [CrossRef]
- Zardoya, R. Recent advances in understanding mitochondrial genome diversity. F1000Research 2020, 9, 270. [Google Scholar] [CrossRef]
- Habbane, M.; Montoya, J.; Rhouda, T.; Sbaoui, Y.; Radallah, D.; Emperador, S. Human mitochondrial DNA: Particularities and diseases. Biomedicines 2021, 9, 1364. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Sun, L.; Chen, X.; Zhang, D. Oxidative stress, mitochondrial damage and neurodegenerative diseases. Neural Regen. Res. 2013, 8, 2003. [Google Scholar] [PubMed]
- Byun, H.-M.; Baccarelli, A.A. Environmental exposure and mitochondrial epigenetics: Study design and analytical challenges. Hum. Genet. 2014, 133, 247–257. [Google Scholar] [CrossRef]
- Peters, A.; Nawrot, T.S.; Baccarelli, A.A. Hallmarks of environmental insults. Cell 2021, 184, 1455–1468. [Google Scholar] [CrossRef]
- Marroqui, L.; Tudurí, E.; Alonso-Magdalena, P.; Quesada, I.; Nadal, Á.; Dos Santos, R.S. Mitochondria as target of endocrine-disrupting chemicals: Implications for type 2 diabetes. J. Endocrinol. 2018, 239, R27–R45. [Google Scholar] [CrossRef]
- Meyer, J.N.; Leung, M.C.; Rooney, J.P.; Sendoel, A.; Hengartner, M.O.; Kisby, G.E.; Bess, A.S. Mitochondria as a target of environmental toxicants. Toxicol. Sci. 2013, 134, 1–17. [Google Scholar] [CrossRef]
- Rovira-Llopis, S.; Bañuls, C.; Diaz-Morales, N.; Hernandez-Mijares, A.; Rocha, M.; Victor, V.M. Mitochondrial dynamics in type 2 diabetes: Pathophysiological implications. Redox Biol. 2017, 11, 637–645. [Google Scholar] [CrossRef]
- Yoon, Y.; Galloway, C.A.; Jhun, B.S.; Yu, T. Mitochondrial dynamics in diabetes. Antioxid. Redox Signal. 2011, 14, 439–457. [Google Scholar] [CrossRef]
- Alonso-Magdalena, P.; Rivera, F.J.; Guerrero-Bosagna, C. Bisphenol-A and metabolic diseases: Epigenetic, developmental and transgenerational basis. Environ. Epigenet. 2016, 2, dvw022. [Google Scholar] [CrossRef]
- Wallace, D.C.; Fan, W. Energetics, epigenetics, mitochondrial genetics. Mitochondrion 2010, 10, 12–31. [Google Scholar] [CrossRef] [PubMed]
- Morin, A.; Letouzé, E.; Gimenez-Roqueplo, A.P.; Favier, J. Oncometabolites-driven tumorigenesis: From genetics to targeted therapy. Int. J. Cancer 2014, 135, 2237–2248. [Google Scholar] [CrossRef]
- Stoccoro, A.; Coppedè, F. Mitochondrial DNA methylation and human diseases. Int. J. Mol. Sci. 2021, 22, 4594. [Google Scholar] [CrossRef] [PubMed]
- FC Lopes, A. Mitochondrial metabolism and DNA methylation: A review of the interaction between two genomes. Clin. Epigenetics 2020, 12, 182. [Google Scholar]
- Sirard, M.-A. Distribution and dynamics of mitochondrial DNA methylation in oocytes, embryos and granulosa cells. Sci. Rep. 2019, 9, 11937. [Google Scholar] [CrossRef] [PubMed]
- Low, H.C.; Chilian, W.M.; Ratnam, W.; Karupaiah, T.; Md Noh, M.F.; Mansor, F.; Ng, Z.X.; Pung, Y.F. Changes in mitochondrial epigenome in type 2 diabetes mellitus. Br. J. Biomed. Sci. 2023, 80, 10884. [Google Scholar] [CrossRef] [PubMed]
- Nass, M.M. Differential methylation of mitochondrial and nuclear DNA in cultured mouse, hamster and virus-transformed hamster cells in vivo and in vitro methylation. J. Mol. Biol. 1973, 80, 155–175. [Google Scholar] [CrossRef] [PubMed]
- Castegna, A.; Iacobazzi, V.; Infantino, V. The mitochondrial side of epigenetics. Physiol. Genom. 2015, 47, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Minocherhomji, S.; Tollefsbol, T.O.; Singh, K.K. Mitochondrial regulation of epigenetics and its role in human diseases. Epigenetics 2012, 7, 326–334. [Google Scholar] [CrossRef] [PubMed]
- Rakyan, V.K.; Down, T.A.; Balding, D.J.; Beck, S. Epigenome-wide association studies for common human diseases. Nat. Rev. Genet. 2011, 12, 529–541. [Google Scholar] [CrossRef]
- Flanagan, J.M. Epigenome-wide association studies (EWAS): Past, present, and future. Cancer Epigenetics: Risk Assessment, Diagnosis, Treatment and Prognosis; Springer: Berlin/Heidelberg, Germany, 2015; pp. 51–63. [Google Scholar]

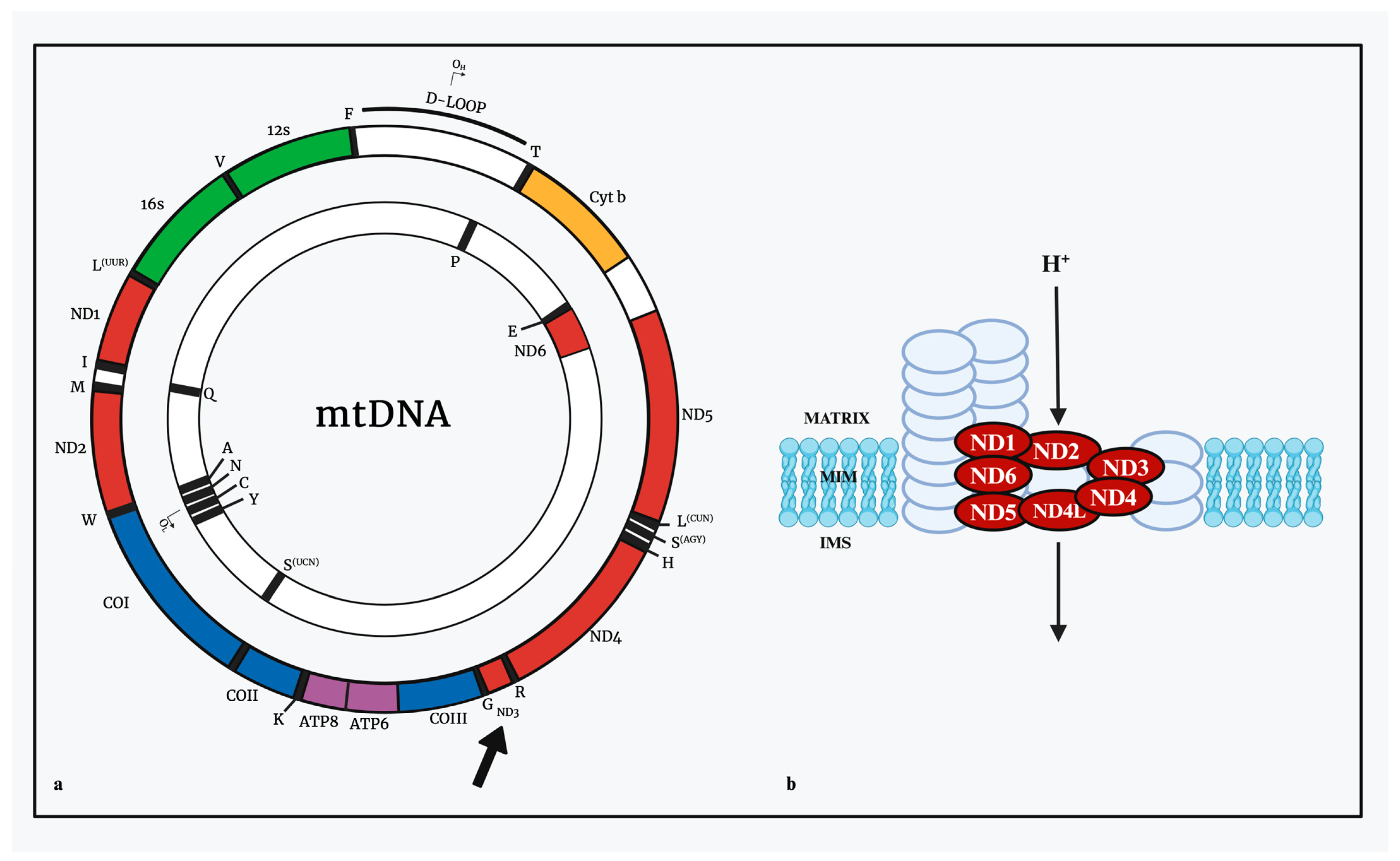

| GENES ENCODING | HEAVY STRAND | LIGHT STRAND |
|---|---|---|
| rRNA | 12s (MT-RNR1) | |
| 16s (MT-RNR2) | ||
| tRNA | MT-TR | MT-TA |
| MT-TD | MT-TC | |
| MT-TG | MT-TE | |
| MT-TH | MT-TN | |
| MT-TI | MT-TP | |
| MT-TLI (CUN) | MT-TQ | |
| MT-TL2 (UUR) | MT-TS1 (UCN) | |
| MT-TK | MT-TY | |
| MT-TM | ||
| MT-TF | ||
| MT-TS2 (AGY) | ||
| MT-TT | ||
| MT-TW | ||
| MT-TV | ||
| POLYPEPTIDES | COMPLEX I | COMPLEX I |
| MT-ND1 | MT-ND6 | |
| MT-ND2 | ||
| MT-ND3 | ||
| MT-ND4 | ||
| MT-ND4L | ||
| MT-ND5 | ||
| COMPLEX III | ||
| MT-CYB | ||
| COMPLEX IV | ||
| MT-CO1 | ||
| MT-CO2 | ||
| MT-CO3 | ||
| COMPLEX V | ||
| MT-ATP6 | ||
| MT-ATP8 | ||
| TOTAL GENES | 28 | 9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferreira, T.; Rodriguez, S. Mitochondrial DNA: Inherent Complexities Relevant to Genetic Analyses. Genes 2024, 15, 617. https://doi.org/10.3390/genes15050617
Ferreira T, Rodriguez S. Mitochondrial DNA: Inherent Complexities Relevant to Genetic Analyses. Genes. 2024; 15(5):617. https://doi.org/10.3390/genes15050617
Chicago/Turabian StyleFerreira, Tomas, and Santiago Rodriguez. 2024. "Mitochondrial DNA: Inherent Complexities Relevant to Genetic Analyses" Genes 15, no. 5: 617. https://doi.org/10.3390/genes15050617
APA StyleFerreira, T., & Rodriguez, S. (2024). Mitochondrial DNA: Inherent Complexities Relevant to Genetic Analyses. Genes, 15(5), 617. https://doi.org/10.3390/genes15050617






