Evaluation of the Clinical Effectiveness of the Salmeterol/Fluticasone Fixed-Dose Combination Delivered via the Elpenhaler® Device in Greek Patients with Chronic Obstructive Pulmonary Disease and Comorbidities: The AEOLOS Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Study Design
2.3. Study Outcomes
2.4. Statistical Analysis
3. Results
3.1. Demographic and Other Baseline Characteristics
3.2. Comorbidities in Study Participants
3.3. Improvement in Lung Function at 6 and 12 Months in Patients with and without Comorbidities
3.4. Improvement in Lung Function at 6 and 12 Months According to COPD Severity
3.5. Improvement in Dyspnea (mMRC) in Patients with and without Comorbidities
3.6. Satisfaction from the Elpenhaler® Device as Assessed by the FSI-10 Questionnaire
3.7. Safety Data
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Mathers, C.D.; Loncar, D. Projections of Global Mortality and Burden of Disease from 2002 to 2030. PLoS Med. 2006, 3, e442. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- 2021 GOLD Reports. Available online: https://goldcopd.org/2021-gold-reports/ (accessed on 20 June 2021).
- The Top 10 Causes of Death. Available online: https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death (accessed on 23 June 2021).
- Gershon, A.S.; Dolmage, T.E.; Stephenson, A.; Jackson, B. Chronic Obstructive Pulmonary Disease and Socioeconomic Status: A Systematic Review. COPD 2012, 9, 216–226. [Google Scholar] [CrossRef]
- Aït-Khaled, N.; Enarson, D.A.; Ottmani, S.; El Sony, A.; Eltigani, M.; Sepulveda, R. Chronic Airflow Limitation in Developing Countries: Burden and Priorities. Int. J. Chron. Obstruct. Pulmon. Dis. 2007, 2, 141–150. [Google Scholar]
- Bousquet, J.; Dahl, R.; Khaltaev, N. Global Alliance against Chronic Respiratory Diseases. Eur. Respir. J. 2007, 29, 233–239. [Google Scholar] [CrossRef]
- Nannini, L.J.; Poole, P.; Milan, S.J.; Kesterton, A. Combined Corticosteroid and Long-Acting beta(2)-Agonist in One Inhaler versus Inhaled Corticosteroids Alone for Chronic Obstructive Pulmonary Disease. Cochrane Database Syst. Rev. 2013, 8, CD006826. [Google Scholar] [CrossRef]
- Calverley, P.M.; Anderson, J.A.; Celli, B.; Ferguson, G.T.; Jenkins, C.; Jones, P.W.; Yates, J.C.; Vestbo, J. Salmeterol and Fluticasone Propionate and Survival in Chronic Obstructive Pulmonary Disease. N. Engl. J. Med. 2007, 356, 775–789. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wedzicha, J.A.; Calverley, P.M.; Seemungal, T.A.; Hagan, G.; Ansari, Z.; Stockley, R.A. The Prevention of Chronic Obstructive Pulmonary Disease Exacerbations by Salmeterol/fluticasone Propionate or Tiotropium Bromide. Am. J. Respir. Crit. Care Med. 2008, 177, 19–26. [Google Scholar] [CrossRef] [Green Version]
- OPCRD Data Request—OPC. Available online: http://optimumpatientcare.org/opcrd/ (accessed on 19 June 2021).
- Roche, N.; Reddel, H.; Martin, R.; Brusselle, G.; Papi, A.; Thomas, M.; Postma, D.; Thomas, V.; Rand, C.; Chisholm, A.; et al. Quality Standards for Real-World Research. Focus on Observational Database Studies of Comparative Effectiveness. Ann. Am. Thorac. Soc. 2014, 11, S99–S104. [Google Scholar] [CrossRef]
- Bestall, J.C.; Paul, E.A.; Garrod, R.; Garnham, R.; Jones, P.W.; Wedzicha, J.A. Usefulness of the Medical Research Council (MRC) Dyspnoea Scale as a Measure of Disability in Patients with Chronic Obstructive Pulmonary Disease. Thorax 1999, 54, 581–586. [Google Scholar] [CrossRef] [Green Version]
- Zervas, E.; Samitas, K.; Gaga, M. Assessment of Satisfaction with Different Dry Powder Inhalation Devices in Greek Patients with COPD and Asthma: The ANASA Study. Int. J. Chron. Obstruct. Pulmon. Dis. 2016, 11, 1845–1855. [Google Scholar] [CrossRef] [Green Version]
- Tordera, M.P.; Viejo, J.L.; Sanchos, J.; Badia, X.; Cobos, N.; Picado, C.; Sobradillo, V.; del Río, J.M.G.; Duce, F.; Cabrera, L.M. Assessment of Patient Satisfaction and Preferences with Inhalers in Asthma with the FSI-10 Questionnaire. Arch. Bronconeumol. 2008, 44, 346–352. [Google Scholar] [CrossRef]
- Vestbo, J.; Hurd, S.S.; Agustí, A.G.; Jones, P.W.; Vogelmeier, C.; Anzueto, A.; Barnes, P.J.; Fabbri, L.M.; Martinez, F.J.; Nishimura, M.; et al. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease: GOLD Executive Summary. Am. J. Respir. Crit. Care Med. 2013, 187, 347–365. [Google Scholar] [CrossRef]
- Calzetta, L.; Ritondo, B.L.; Matera, M.G.; Cazzola, M.; Rogliani, P. Evaluation of Fluticasone Propionate/salmeterol for the Treatment of COPD: A Systematic Review. Expert Rev. Respir. Med. 2020, 14, 621–635. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Milne, S.; Jaw, J.E.; Yang, C.X.; Xu, F.; Li, X.; Obeidat, M.; Sin, D.D. BMI Is Associated with FEV 1 Decline in Chronic Obstructive Pulmonary Disease: A Meta-Analysis of Clinical Trials. Respir. Res. 2019, 20, 236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whittaker, H.R.; Pimenta, J.M.; Jarvis, D.; Kiddle, S.J.; Quint, J.K. Characteristics Associated with Accelerated Lung Function Decline in a Primary Care Population with Chronic Obstructive Pulmonary Disease. Int. J. Chron. Obstruct. Pulmon. Dis. 2020, 15, 3079–3091. [Google Scholar] [CrossRef] [PubMed]
- Rehman, A.U.; Shah, S.; Abbas, G.; Harun, S.N.; Shakeel, S.; Hussain, R.; Hassali, M.A.A.; Rasool, M.F. Assessment of Risk Factors Responsible for Rapid Deterioration of Lung Function over a Period of One Year in Patients with Chronic Obstructive Pulmonary Disease. Sci. Rep. 2021, 11, 13578. [Google Scholar] [CrossRef] [PubMed]
- Rodrigo, G.J.; Price, D.; Anzueto, A.; Singh, D.; Altman, P.; Bader, G.; Patalano, F.; Fogel, R.; Kostikas, K. LABA/LAMA Combinations versus LAMA Monotherapy or LABA/ICS in COPD: A Systematic Review and Meta-Analysis. Int. J. Chron. Obstruct. Pulmon. Dis. 2017, 12, 907–922. [Google Scholar] [CrossRef] [Green Version]
- Mahler, D.A.; Wells, C.K. Evaluation of Clinical Methods for Rating Dyspnea. Chest 1988, 93, 580–586. [Google Scholar] [CrossRef] [Green Version]
- American Thoracic Society. Surveillance for Respiratory Hazards in the Occupational Setting. Am. Rev. Respir. Dis. 1982, 126, 952–956. [Google Scholar]
- LIZARS, J. Standardized Questionaries on Respiratory Symptoms. Br. Med. J. 1960, 2, 1665. [Google Scholar]
- Bausewein, C.; Farquhar, M.; Booth, S.; Gysels, M.; Higginson, I.J. Measurement of Breathlessness in Advanced Disease: A Systematic Review. Respir. Med. 2007, 101, 399–410. [Google Scholar] [CrossRef] [PubMed]
- Coleta, K.D.; Silveira, L.V.; Lima, D.F.; Rampinelli, E.A.; Godoy, I.; Godoy, I. Predictors of First-Year Survival in Patients with Advanced COPD Treated Using Long-Term Oxygen Therapy. Respir. Med. 2008, 102, 512–518. [Google Scholar] [CrossRef] [Green Version]
- Oga, T.; Nishimura, K.; Tsukino, M.; Hajiro, T.; Mishima, M. Dyspnoea with Activities of Daily Living versus Peak Dyspnoea during Exercise in Male Patients with COPD. Respir. Med. 2006, 100, 965–971. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sundh, J.; Janson, C.; Lisspers, K.; Ställberg, B.; Montgomery, S. The Dyspnoea, Obstruction, Smoking, Exacerbation (DOSE) Index Is Predictive of Mortality in COPD. Prim. Care Respir. J. 2012, 21, 295–301. [Google Scholar] [CrossRef] [Green Version]
- Nishimura, K.; Izumi, T.; Tsukino, M.; Oga, T. Dyspnea Is a Better Predictor of 5-Year Survival than Airway Obstruction in Patients with COPD. Chest 2002, 121, 1434–1440. [Google Scholar] [CrossRef] [Green Version]
- Casanova, C.; Marin, J.M.; Martinez-Gonzalez, C.; de Lucas-Ramos, P.; Mir-Viladrich, I.; Cosio, B.; Peces-Barba, G.; Solanes-García, I.; Agüero, R.; Feu-Collado, N.; et al. Differential Effect of Modified Medical Research Council Dyspnea, COPD Assessment Test, and Clinical COPD Questionnaire for Symptoms Evaluation Within the New GOLD Staging and Mortality in COPD. Chest 2015, 148, 159–168. [Google Scholar] [CrossRef]
- Lewthwaite, H.; Jensen, D.; Ekström, M. How to Assess Breathlessness in Chronic Obstructive Pulmonary Disease. Int. J. Chron. Obstruct. Pulmon. Dis. 2021, 16, 1581–1598. [Google Scholar] [CrossRef]
- Afroz, N.; Gutzwiller, F.S.; Mackay, A.J.; Naujoks, C.; Patalano, F.; Kostikas, K. Patient-Reported Outcomes (PROs) in COPD Clinical Trials: Trends and Gaps. Int. J. Chron. Obstruct. Pulmon. Dis. 2020, 15, 1789–1800. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.; Lareau, S.; Mahler, D.A. Measuring the Effects of COPD on the Patient. Respir. Med. 2005, 99 (Suppl. B), S11–S18. [Google Scholar] [CrossRef] [Green Version]
- Chrystyn, H.; Small, M.; Milligan, G.; Higgins, V.; Gil, E.G.; Estruch, J. Impact of Patients’ Satisfaction with Their Inhalers on Treatment Compliance and Health Status in COPD. Respir. Med. 2014, 108, 358–365. [Google Scholar] [CrossRef] [Green Version]
- Laube, B.L.; Janssens, H.M.; de Jongh, F.H.; Devadason, S.G.; Dhand, R.; Diot, P.; Everard, M.L.; Horvath, I.; Navalesi, P.; Voshaar, T.; et al. What the Pulmonary Specialist Should Know about the New Inhalation Therapies. Eur. Respir. J. 2011, 37, 1308–1417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Molimard, M.; Colthorpe, P. Inhaler Devices for Chronic Obstructive Pulmonary Disease: Insights from Patients and Healthcare Practitioners. J. Aerosol. Med. Pulm. Drug Deliv. 2015, 28, 219–228. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van der Palen, J.; van der Valk, P.; Goosens, M.; Groothuis-Oudshoorn, K.; Brusse-Keizer, M. A Randomised Cross-over Trial Investigating the Ease of Use and Preference of Two Dry Powder Inhalers in Patients with Asthma or Chronic Obstructive Pulmonary Disease. Expert Opin. Drug Deliv. 2013, 10, 1171–1178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miravitlles, M.; Montero-Caballero, J.; Richard, F.; Santos, S.; García-Rivero, J.L.; Ortega, F.; Ribera, X. A Cross-Sectional Study to Assess Inhalation Device Handling and Patient Satisfaction in COPD. Int. J. Chron. Obstruct. Pulmon. Dis. 2016, 407, 407–415. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ozturk, C.; Kaya, A.; Bilgin, C.; Yucesoy, L.; Ikidag, B.; Demirel, M.; Baslilar, S.; Saylan, B.; Senol, T.; Aganoglu, S.; et al. Evaluation of Inhaler Technique and Patient Satisfaction With Fixed-Combination Budesonide/Formoterol Dry-Powder Inhaler in Chronic Obstructive Pulmonary Disease (COPD): Data on Real-Life Clinical Practice in Turkey. Chest 2013, 144, 726A. [Google Scholar] [CrossRef]
- Darbà, J.; Ramírez, G.; Sicras, A.; Francoli, P.; Torvinen, S.; la Rosa, R.S. The Importance of Inhaler Devices: The Choice of Inhaler Device May Lead to Suboptimal Adherence in COPD Patients. Int. J. Chron. Obstruct. Pulmon. Dis. 2015, 10, 2335–2345. [Google Scholar] [CrossRef] [Green Version]
- Grekas, N.; Athanassiou, A.; Iskos, C.; Panagiotakos, D.; Papataxiarchou, A.; Porichi, O. Reliability of the FSI-10 Questionnaire for the Assessment of the Usability of Drug Inhalers in Greek Patients. Available online: https://www.mednet.gr/archives/2011-2/257abs.html (accessed on 20 June 2021).
- Atkinson, M.J.; Sinha, A.; Hass, S.L.; Colman, S.S.; Kumar, R.N.; Brod, M.; Rowland, C.R. Validation of a General Measure of Treatment Satisfaction, the Treatment Satisfaction Questionnaire for Medication (TSQM), Using a National Panel Study of Chronic Disease. Health Qual. Life Outcomes 2004, 2, 12. [Google Scholar] [CrossRef] [Green Version]
- Shikiar, R.; Rentz, A.M. Satisfaction with Medication: An Overview of Conceptual, Methodologic, and Regulatory Issues. Value Health 2004, 7, 204–215. [Google Scholar] [CrossRef] [Green Version]
- Wing, K.; Williamson, E.; Carpenter, J.R.; Wise, L.; Schneeweiss, S.; Smeeth, L.; Quint, J.K.; Douglas, I. Real World Effects of COPD Medications: A Cohort Study with Validation against Results from Randomised Controlled Trials. Eur. Respir. J. 2021, 57, 2001586. [Google Scholar] [CrossRef]
- Roche, N.; Pribil, C.; Van Ganse, E.; Serrier, P.; Housset, B.; Poirier, D.; Texier, N.; Schück, S.; Boucot, I. Real-Life Use of Fluticasone Propionate/salmeterol in Patients with Chronic Obstructive Pulmonary Disease: A French Observational Study. BMC Pulm. Med. 2014, 14, 56. [Google Scholar] [CrossRef] [Green Version]
- Martinez, C.H.; Mannino, D.M.; Divo, M.J. Defining COPD-Related Comorbidities, 2004–2014. Chronic ObstrPulm Dis. 2014, 1, 51–63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mannino, D.M.; Higuchi, K.; Yu, T.C.; Zhou, H.; Li, Y.; Tian, H.; Suh, K. Economic Burden of COPD in the Presence of Comorbidities. Chest 2015, 148, 138–150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sin, D.D.; Man, S.F.P. Why Are Patients with Chronic Obstructive Pulmonary Disease at Increased Risk of Cardiovascular Diseases? The Potential Role of Systemic Inflammation in Chronic Obstructive Pulmonary Disease. Circulation 2003, 12, 15–16. [Google Scholar] [CrossRef]
- Man, S.F.P.; Paul Man, S.F.; Leipsic, J.A.; Man, J.P.; Sin, D.D. Is Atherosclerotic Heart Disease in COPD a Distinct Phenotype? Chest 2011, 140, 569–571. [Google Scholar] [CrossRef] [PubMed]
- Niewoehner, D.E.; Lokhnygina, Y.; Rice, K.; Kuschner, W.G.; Sharafkhaneh, A.; Sarosi, G.A.; Krumpe, P.; Pieper, K.; Kesten, S. Risk Indexes for Exacerbations and Hospitalizations Due to COPD. Chest 2007, 131, 20–28. [Google Scholar] [CrossRef]
- Chang, C.L.; Robinson, S.C.; Mills, G.D.; Sullivan, G.D.; Karalus, N.C.; McLachlan, J.D.; Hancox, R.J. Biochemical Markers of Cardiac Dysfunction Predict Mortality in Acute Exacerbations of COPD. Thorax 2011, 66, 764–768. [Google Scholar] [CrossRef] [Green Version]
- Calverley, P.M.A.; Anderson, J.A.; Celli, B.; Ferguson, G.T.; Jenkins, C.; Jones, P.W.; Crim, C.; Willits, L.R.; Yates, J.C.; Vestbo, J.; et al. Cardiovascular Events in Patients with COPD: TORCH Study Results. Thorax 2010, 65, 719–725. [Google Scholar] [CrossRef] [Green Version]
- Magnussen, H.; Disse, B.; Rodriguez-Roisin, R.; Kirsten, A.; Watz, H.; Tetzlaff, K.; Towse, L.; Finnigan, H.; Dahl, R.; Decramer, M.; et al. Withdrawal of Inhaled Glucocorticoids and Exacerbations of COPD. N. Engl. J. Med. 2014, 371, 1285–1294. [Google Scholar] [CrossRef] [Green Version]
- Marquis, K.; Maltais, F.; Duguay, V.; Bezeau, A.-M.; LeBlanc, P.; Jobin, J.; Poirier, P. The Metabolic Syndrome in Patients with Chronic Obstructive Pulmonary Disease. J. Cardiopulm. Rehabil. 2005, 25, 226–232. [Google Scholar] [CrossRef]
- Díez-Manglano, J.; Working Group on COPD; Spanish Society of Internal Medicine; Barquero-Romero, J.; Almagro, P.; Cabrera, F.J.; García, F.L.; Montero, L.; Soriano, J.B. COPD Patients with and without Metabolic Syndrome: Clinical and Functional Differences. Intern. Emerg. Med. 2014, 9, 419–425. [Google Scholar] [CrossRef]




| Demographics | (Ν, %) |
|---|---|
| Age—years | N = 1016 |
| Mean ± SD | 69.5 ± 9.5 |
| Gender—no. (%) | N = 1016 |
| Female | 264 (26) |
| BMI—kg/m2 | N = 1016 |
| Mean ± SD | 28.6 ± 5.3 |
| Years with COPD | N = 1016 |
| Mean ± SD | 7.3 ± 6.0 |
| mMRC dyspnea scale—no. (%) | N = 1016 |
| Stage 0 | 7 (0.7) |
| Stage 1 | 190 (18.7) |
| Stage 2 | 457 (45) |
| Stage 3 | 321 (31.6) |
| Stage 4 | 41 (4) |
| Classification according to GOLD spirometric stages—no. (%) | N = 1016 |
| GOLD 1 (Mild) | 0 (0) |
| GOLD 2 (Moderate) | 471 (46.4) |
| GOLD 3 (Severe) | 483 (47.5) |
| GOLD 4 (Extremely severe) | 62 (6.1) |
| Classification according to GOLD 2020 Groups—no. (%) | N = 1016 |
| Group A | 56 (5.5) |
| Group B | 286 (28.1) |
| Group C | 326 (32.1) |
| Group D | 348 (34.3) |
| Previous COPD treatment | (N, %) |
| No. (%) of patients with no past COPD treatment | 199 (19.6) |
| No. (%) of patients with at least one past COPD treatment | 817 (80.4) |
| Previous COPD maintenance treatment *—no. (%) | N = 817 |
| LABA | 66 (8.1) |
| LAMA | 244 (29.8) |
| LABA + LAMA | 322 (39.4) |
| Roflumilast | 29 (3.5) |
| Theophylline | 18 (2.2) |
| Other treatment ** | 174 (21.3) |
| Comorbidities | (Ν, %) |
|---|---|
| No. (%) of patients with no comorbidities | 238 (23.4) |
| No. (%) of patients with at least one comorbidity | 778 (76.6) |
| Number of comorbidities per patient—no. (%) | |
| 1 | 256 (32.9) |
| 2 | 233 (29.9) |
| >2 | 289 (37.2) |
| Number of patients per comorbidity category—no. (%) * | N = 778 |
| Cardiovascular diseases | 653 (83.9) |
| Coronary artery disease | 175 (22.5) |
| Arterial hypertension | 557 (71.6) |
| Cardiac failure | 122 (15.7) |
| Peripheral arterial disease | 41 (5.3) |
| Pulmonary embolism | 9 (1.2) |
| Other | 88 (11.3) |
| Metabolic diseases | 373 (47.9) |
| Diabetes Mellitus Type ΙΙ | 169 (21.7) |
| Dyslipidemia | 265 (34.1) |
| Other | 33 (4.2) |
| Psychiatric diseases | 160 (20.6) |
| Depression | 83 (10.7) |
| Anxiety disorder | 88 (11.3) |
| Other | 11 (1.4) |
| Malignancies | 56 (7.2) |
| Lung | 13 (1.7) |
| Lymphoma | 0 (0) |
| Breast | 9 (1.2) |
| Other | 32 (4.1) |
| Other diseases | 132 (17) |
| Osteoporosis | 47 (6) |
| Other | 84 (10.8) |
| No. (%) of patients with cardiovascular and metabolic diseases | 711 (91.4) |
| Spirometry | Baseline (Day 0) N = 1016 | Visit 1 (6 Months) N = 792 | Visit 2 (12 Months) N = 746 |
|---|---|---|---|
| FEV1 predicted L— | |||
| Mean ± SD | 1.35 ± 0.37 | 1.50 ± 0.42 | 1.56 ± 0.45 |
| FEV1% predicted—% | |||
| Mean ± SD | 48.43 ± 8.61 | 54.17 ± 10.87 | 56.47 ± 11.82 |
| Change of FEV1 (L) from baseline (Mean ± SD, p-value) | - | 0.15 ± 0.22, <0.0001 | 0.21 ± 0.25, <0.0001 |
| Change of FEV1 (L) between 6 and 12 months * (Mean ± SD, p-value) | - | - | 0.06 ± 0.19, <0.0001 |
| FVC predicted L— | |||
| Mean ± SD | 2.48 ± 0.72 | 2.59 ± 0.72 | 2.67 ± 0.76 |
| FVC % predicted | |||
| Mean ± SD | 68.61 ± 14.42 | 71.61 ± 14.07 | 74.19 ± 15.42 |
| Change of FVC (L) from baseline (Mean ± SD, p-value) | - | 0.11 ± 0.35, <0.0001 | 0.19 ± 0.41, <0.0001 |
| Change of FVC (L) between 6 and 12 months * (Mean ± SD, p-value) | - | - | 0.09 ± 0.38, <0.0001 |
| FEV1/FVC ratio | |||
| Mean ± SD | 0.58 ± 0.08 | 0.62 ± 0.10 | 0.62 ± 0.11 |
| Change of FEV1/FVC from baseline (Mean ± SD, p-value) | - | 0.036 ± 0.07, <0.0001 | 0.039 ± 0.07, <0.0001 |
| Change of FEV1/FVC between 6 and 12 months * (Mean ± SD, p-value) | - | - | 0.002 ± 0.08, 0.595 |
| Spirometry | ΔFEV1 0–6 Months (Mean ± SD) | ΔFEV1 0–12 Months (Mean ± SD) | ΔFEV1 6–12 Months (Mean ± SD) |
|---|---|---|---|
| GOLD 2020 Groups | |||
| (* p-value) | 0.015 | <0.001 | 0.014 |
| Group A | 0.07 ± 0.23 | 0.19 ± 0.25 | 0.12 ± 0.18 |
| Group B | 0.17 ± 0.19 | 0.25 ± 0.24 | 0.07 ± 0.15 |
| Group C | 0.19 ± 0.22 | 0.26 ± 0.27 | 0.07 ± 0.23 |
| Group D | 0.13 ± 0.20 | 0.17 ± 0.23 | 0.03 ± 0.18 |
| Variables | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| β | 95% CI | * p | β | 95% CI | * p | |
| Age | −0.030 | −0.003, 0.001 | 0.418 | 0.16 | −0.002, 0.002 | 0.689 |
| Gender (female) | −0.026 | −0.055, 0.026 | 0.481 | −0.016 | −0.050, 0.032 | 0.656 |
| BMI | 0.085 | 0.001, 0.007 | 0.020 | 0.083 | 0.001, 0.007 | 0.024 |
| mMRC | −0.087 | −0.051, −0.005 | 0.018 | −0.072 | −0.050, 0.000 | 0.047 |
| Comorbidities | −0.089 | −0.098, −0.010 | 0.015 | −0.078 | −0.092, −0.003 | 0.035 |
| GOLD 2020 Groups | −0.093 | −0.046, −0.006 | 0.011 | −0.099 | −0.050, −0.006 | 0.013 |
| Baseline FEV1 | 0.018 | −0.037, 0.061 | 0.627 | −0.016 | −0.063, 0.041 | 0.682 |
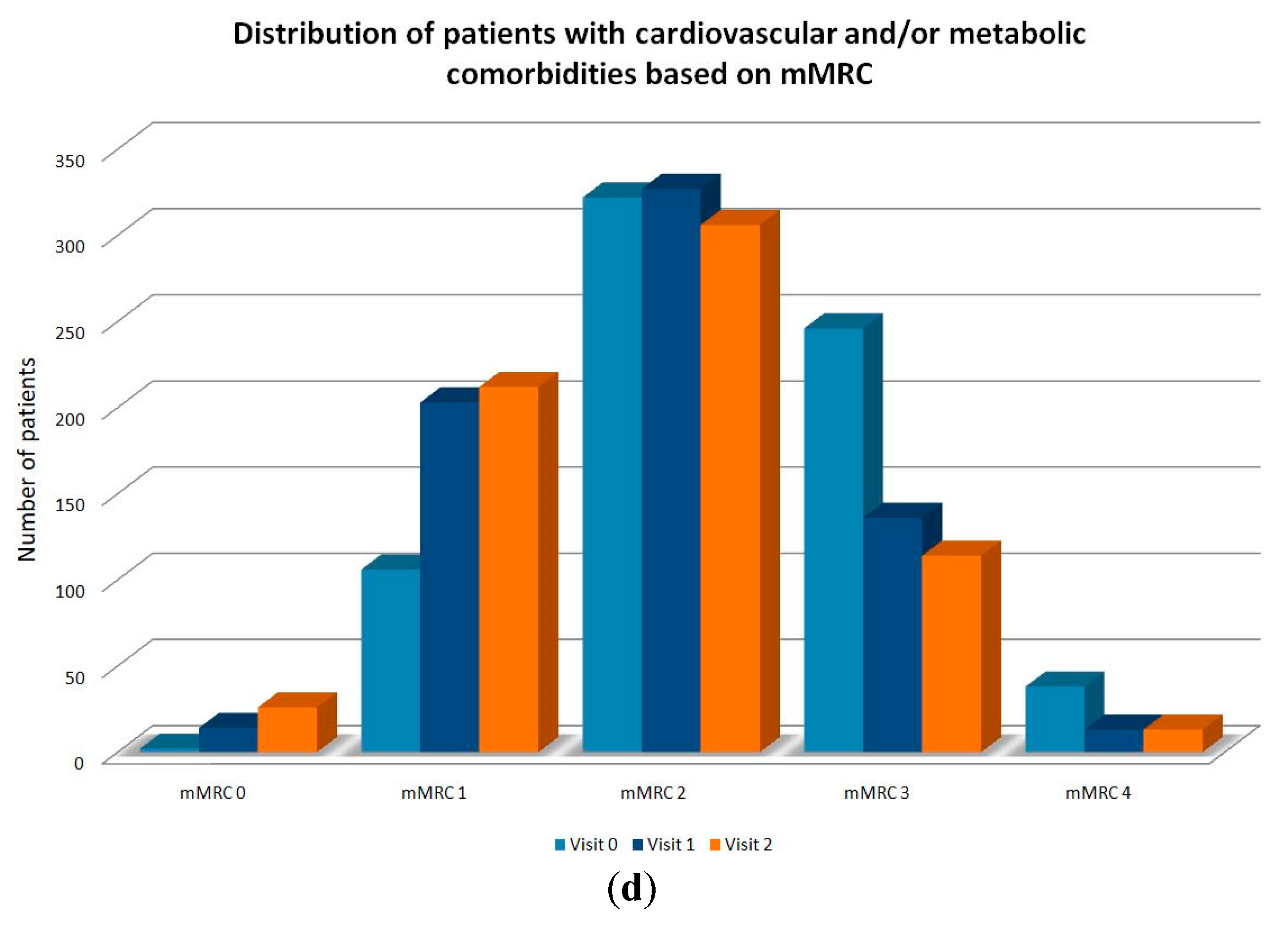
| mMRC Dyspnea Scale | Baseline (Day 0) N = 1016, % | Visit 1 (6 Months) N = 992, % | Visit 2 (12 Months) N = 966, % |
|---|---|---|---|
| Stage 0 | 7 (0.7) | 24 (2.4) | 67 (6.9) |
| Stage 1 | 190 (18.7) | 315 (31.8) | 299 (31) |
| Stage 2 | 457 (45) | 466 (47) | 434 (44.9) |
| Stage 3 | 321 (31.6) | 172 (17.3) | 148 (15.3) |
| Stage 4 | 41 (4) | 15 (1.5) | 18 (1.9) |
| Difference in mMRC scale between visits * (p-value) | <0.0001 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Steiropoulos, P.; Tryfon, S.; Kyriakopoulos, C.; Bartziokas, K.; Kostikas, K. Evaluation of the Clinical Effectiveness of the Salmeterol/Fluticasone Fixed-Dose Combination Delivered via the Elpenhaler® Device in Greek Patients with Chronic Obstructive Pulmonary Disease and Comorbidities: The AEOLOS Study. J. Pers. Med. 2021, 11, 1159. https://doi.org/10.3390/jpm11111159
Steiropoulos P, Tryfon S, Kyriakopoulos C, Bartziokas K, Kostikas K. Evaluation of the Clinical Effectiveness of the Salmeterol/Fluticasone Fixed-Dose Combination Delivered via the Elpenhaler® Device in Greek Patients with Chronic Obstructive Pulmonary Disease and Comorbidities: The AEOLOS Study. Journal of Personalized Medicine. 2021; 11(11):1159. https://doi.org/10.3390/jpm11111159
Chicago/Turabian StyleSteiropoulos, Paschalis, Stavros Tryfon, Christos Kyriakopoulos, Konstantinos Bartziokas, and Konstantinos Kostikas. 2021. "Evaluation of the Clinical Effectiveness of the Salmeterol/Fluticasone Fixed-Dose Combination Delivered via the Elpenhaler® Device in Greek Patients with Chronic Obstructive Pulmonary Disease and Comorbidities: The AEOLOS Study" Journal of Personalized Medicine 11, no. 11: 1159. https://doi.org/10.3390/jpm11111159








