Three New Asperentin Derivatives from the Algicolous Fungus Aspergillus sp. F00785
Abstract
:1. Introduction

2. Results and Discussion
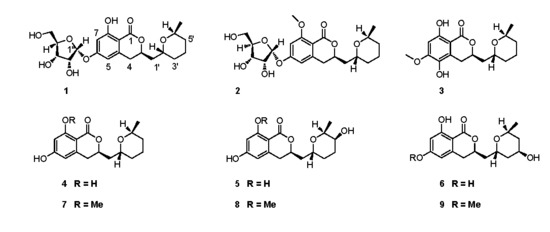
2.1. Structure Elucidation
| No. | 1 | 2 | 3 | 4 | ||||
|---|---|---|---|---|---|---|---|---|
| δH a (J in Hz) | δC b | δH a (J in Hz) | δC b | δH a (J in Hz) | δC b | δH a (J in Hz) | δC b | |
| 1 | 169.6, C | 161.6, C | 170.0, C | 169.9, C | ||||
| 3 | 4.72, m | 76.5, CH | 4.60, m | 74.6, CH | 4.63, m | 76.3, CH | 4.72, m | 76.4, CH |
| 4 | 2.87, m | 33.6, CH2 | 2.92, dd, (16.2, 11.5) | 35.4, CH2 | 3.10, dd, (16.8, 3.4) | 27.3, CH2 | 2.86, m | 33.7, CH2 |
| 2.83, dd, (16.2, 2.9) | 2.62, dd, (16.8, 11.5) | |||||||
| 4a | 141.5, C | 144.0, C | 122.6, C | 141.8, C | ||||
| 5 | 6.42, s | 107.6, CH | 6.56, d, (2.0) | 106.8, CH | 134.3, C | 6.32, s | 106.7, CH | |
| OH-5 | 5.20, s | |||||||
| 6 | 162.5, C | 162.6, C | 153.1, C | 162.9, C | ||||
| OCH3-6 | 3.86, s | 56.2, CH3 | ||||||
| 7 | 6.56, s | 102.9, CH | 6.61, d, (2.0) | 100.1, CH | 6.34, s | 97.8, CH | 6.19, s | 102.0, CH |
| 8 | 163.8, C | 162.9, C | 157.8, C | 164.3, C | ||||
| OCH3-8 | 3.94, s | 56.3, CH3 | ||||||
| 8a | 103.2, C | 106.8, C | 100.7, C | 101.7, C | ||||
| 1′ | 1.98, m; 1.82, m | 39.2, CH2 | 1.92, m; 1.83, m | 39.5, CH2 | 1.95, m; 1.78, m | 39.5, CH2 | 1.99, m; 1.87, m | 39.4, CH2 |
| 2′ | 4.10, m | 66.4, CH | 4.10, m | 66.2, CH | 4.05, m | 66.3, CH | 4.13, m | 66.5, CH |
| 3′ | 1.69, m; 1.35, m | 30.8, CH2 | 1.36, m | 30.9, CH2 | 1.64, m; 1.29, m | 30.8, CH2 | 1.73, m; 1.38, m | 30.9, CH2 |
| 4′ | 1.70, m; 1.62, m | 18.2, CH2 | 1.73, m; 1.64, m | 18.3, CH2 | 1.65, m; 1.56, m | 18.3, CH2 | 1.74, m; 1.65, m | 18.2, CH2 |
| 5′ | 1.68, m; 1.32, m | 31.3, CH2 | 1.72, m | 31.0, CH2 | 1.63, m; 1.26, m | 31.1, CH2 | 1.72, m; 1.36, m | 31.0, CH2 |
| 6′ | 3.94, m | 67.6, CH | 3.96, m | 67.7, CH | 3.89, m | 67.4, CH | 4.02, m | 67.9, CH |
| CH3-6′ | 1.21, d, (4.1) | 19.1, CH3 | 1.22, d, (6.5) | 18.9, CH3 | 1.14, d, (6.5) | 19.2, CH3 | 1.25, d, (6.5) | 18.9, CH3 |
| 1″ | 5.69, d, (3.5) | 100.1, CH | 5.77, d, (4.5) | 100.5, CH | ||||
| 2″ | 4.29, brs | 72.1, CH | 4.34, m | 72.2, CH | ||||
| 3″ | 4.19, brs | 70.1, CH | 4.24, dd, (6.2, 2.7) | 70.4, CH | ||||
| 4″ | 4.21, brs | 86.4, CH | 4.28, dd, (6.2, 2.7) | 86.5, CH | ||||
| 5″ | 3.78, d, (11.6); 3.74, d, (11.6) | 62.3, CH2 | 3.89, dd, (12.1, 3.1); 3.81, dd, (12.1, 3.1) | 62.5, CH2 | ||||
| 8-OH | 11.13, s | 10.86, s | 11.10, s | |||||

2.2. Bioactivity Results

| Compounds | Inhibition Zone (mm) | ||
|---|---|---|---|
| C. gleosporioides Penz | C. gleosporioides (Penz) Sacc. | B. cinerea Pers | |
| (−)-asperentin (4) | 19.7 ± 0.58 | 13.3 ± 3.40 | 1.67 ± 1.11 |
| amphotericin B | 15.7 ± 1.25 | 16.0 ±1.41 | 11.0 ± 0.82 |
3. Experimental Section
3.1. General Experimental Procedures
3.2. Fungi Materials
3.3. Extraction and Purification
3.4. Acid Hydrolysis and Stereochemistry Determination of the Ribofuranose of 6-O-α-d-Ribosyl Asperentin (1)
3.5. Anti-Crop Pathogens Test
4. Conclusion
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pettit, R.K. Culturability and secondary metabolite diversity of extreme microbes: Expanding contribution of deep sea and deep-sea vent microbes to natural product discovery. Mar. Biotechnol. 2011, 13, 1–11. [Google Scholar] [CrossRef]
- Fenical, W. Chemical studies of marine bacteria: Developing a new resource. Chem. Rev. 1993, 93, 1673–1683. [Google Scholar] [CrossRef]
- Schumacher, M.; Kelkel, M.; Dicato, M.; Diederich, M. Gold from the sea: marine compounds as inhibitors of the hallmarks of cancer. Biotechnol. Adv. 2011, 29, 531–547. [Google Scholar] [CrossRef] [PubMed]
- Molinski, T.F.; Dalisay, D.S.; Lievens, S.L.; Saludes, J.P. Drug development from marine natural products. Nat. Rev. Drug Discov. 2009, 8, 69–85. [Google Scholar] [CrossRef] [PubMed]
- Berdy, J. Bioactive microbial metabolites. J. Antibiot. 2005, 58, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.; Vieira, H.; Gaspar, H.; Santos, S. Marketed marine natural products in the pharmaceutical and cosmeceutical industries: Tips for success. Mar. Drugs 2014, 12, 1066–1101. [Google Scholar] [CrossRef] [PubMed]
- D’Incalci, M.; Galmarini, C.M. A review of trabectedin (ET-743): A unique mechanism of action. Mol. Cancer Ther. 2010, 9, 2157–2163. [Google Scholar] [CrossRef] [PubMed]
- Monk, B.J.; Dalton, H.; Benjamin, I.; Tanovic, A. Trabectedin as a new chemotherapy option in the treatment of relapsed platinum sensitive ovarian cancer. Curr. Pharm. Des. 2012, 18, 3754–3769. [Google Scholar] [CrossRef] [PubMed]
- Bauer, A.; Bronstrup, M. Industrial natural product chemistry for drug discovery and development. Nat. Prod. Rep. 2014, 31, 35–60. [Google Scholar] [CrossRef] [PubMed]
- Mayer, A.M.; Glaser, K.B.; Cuevas, C.; Jacobs, R.S.; Kem, W.; Little, R.D.; McIntosh, J.M.; Newman, D.J.; Potts, B.C.; Shuster, D.E. The odyssey of marine pharmaceuticals: A current pipeline perspective. Trends Pharmacol. Sci. 2010, 31, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Mayer, A.M. Marine Pharmaceuticals: The Clinical Pipeline. Available online: http://marinepharmacology.midwestern.edu/clinPipeline.htm (accessed on 16 October 2014).
- Aguilar, A.; Ingemansson, T.; Magnien, E. Extremophile microorganisms as cell factories: Support from the European Union. Extremophiles 1998, 2, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Kamekura, M. Diversity of extremely halophilic bacteria. Extremophiles 1998, 2, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Koch, A.L. Genetic Response of Microbes to Extreme Challenges. J. Theor. Biol. 1993, 160, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Mejanelle, L.; Lopez, J.F.; Gunde-Cimerman, N.; Grimalt, J.O. Ergosterol biosynthesis in novel melanized fungi from hypersaline environments. J. Lipid Res. 2001, 42, 352–358. [Google Scholar] [PubMed]
- Springer, J.P.; Cutler, H.G.; Crumley, F.G.; Cox, R.H.; Davis, E.E.; Thean, J.E. Plant-growth regulatory effects and stereochemistry of cladosporin. J. Agric. Food Chem. 1981, 29, 853–855. [Google Scholar] [CrossRef]
- Grove, J.F. New metabolic products of Aspergillus flavus. I. Asperentin, its methyl ethers, and 5′-hydroxyasperentin. J. Chem. Soc. Perkin Trans. 1 1972, 19, 2400–2406. [Google Scholar] [CrossRef] [PubMed]
- Scott, P.M.; Van Walbeek, W.; MacLean, W.M. Cladosporin, a new antifungal metabolite from Cladosporium cladosporioides. J. Antibiot. 1971, 24, 747–755. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, X.; Lee, U.; Kang, J.S.; Choi, H.D.; Son, B.W. A new radical scavenging anthracene glycoside, asperflavinribofuranoside, and polyketides from a marine isolate of the fungus Microsporum. Chem. Pharm. Bull. 2006, 54, 882–883. [Google Scholar] [CrossRef] [PubMed]
- Zinner, H. Die Acetate der d-Ribose. Chem. Ber. 1953, 86, 817–824. [Google Scholar] [CrossRef]
- Pretsch, E.B.P.; Affolter, C. Structure Determination of Organic Compounds, Tables of Spectral Data, 3rd ed.; Springer-Verlag Berlin: Heidelberg, Germany, 2000; p. 152. [Google Scholar]
- Grove, J.F. New metabolic products of Aspergillus flavus. IV. 4′-Hydroxyasperentin and 5′-hydroxyasperentin 8-methyl ether. J. Chem. Soc. Perkin. 1 1973, 22, 2704–2706. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Zhou, Z.Y.; Jiang, M.Y.; Wang, F.; Liu, J.K. A new isoprenyl phenyl ether riboside from the culture of basidiomycete Laccaria amethystea. J. Asian Nat. Prod. Res. 2010, 12, 723–726. [Google Scholar] [CrossRef] [PubMed]
- Li, D.L.; Li, X.M.; Wang, B.G. Natural anthraquinone derivatives from a marine mangrove plant-derived endophytic fungus Eurotium rubrum: Structural elucidation and DPPH radical scavenging activity. J. Microbiol. Biotechnol. 2009, 19, 675–680. [Google Scholar] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, Q.; Guo, K.; Li, X.-Y.; Zheng, X.-Y.; Kong, X.-J.; Zheng, Z.-H.; Xu, Q.-Y.; Deng, X. Three New Asperentin Derivatives from the Algicolous Fungus Aspergillus sp. F00785. Mar. Drugs 2014, 12, 5993-6002. https://doi.org/10.3390/md12125993
Tang Q, Guo K, Li X-Y, Zheng X-Y, Kong X-J, Zheng Z-H, Xu Q-Y, Deng X. Three New Asperentin Derivatives from the Algicolous Fungus Aspergillus sp. F00785. Marine Drugs. 2014; 12(12):5993-6002. https://doi.org/10.3390/md12125993
Chicago/Turabian StyleTang, Qian, Kai Guo, Xiao-Yang Li, Xiu-Ying Zheng, Xiang-Jian Kong, Zhong-Hui Zheng, Qing-Yan Xu, and Xianming Deng. 2014. "Three New Asperentin Derivatives from the Algicolous Fungus Aspergillus sp. F00785" Marine Drugs 12, no. 12: 5993-6002. https://doi.org/10.3390/md12125993
APA StyleTang, Q., Guo, K., Li, X.-Y., Zheng, X.-Y., Kong, X.-J., Zheng, Z.-H., Xu, Q.-Y., & Deng, X. (2014). Three New Asperentin Derivatives from the Algicolous Fungus Aspergillus sp. F00785. Marine Drugs, 12(12), 5993-6002. https://doi.org/10.3390/md12125993