Development of Piperine-Loaded Solid Self-Nanoemulsifying Drug Delivery System: Optimization, In-Vitro, Ex-Vivo, and In-Vivo Evaluation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
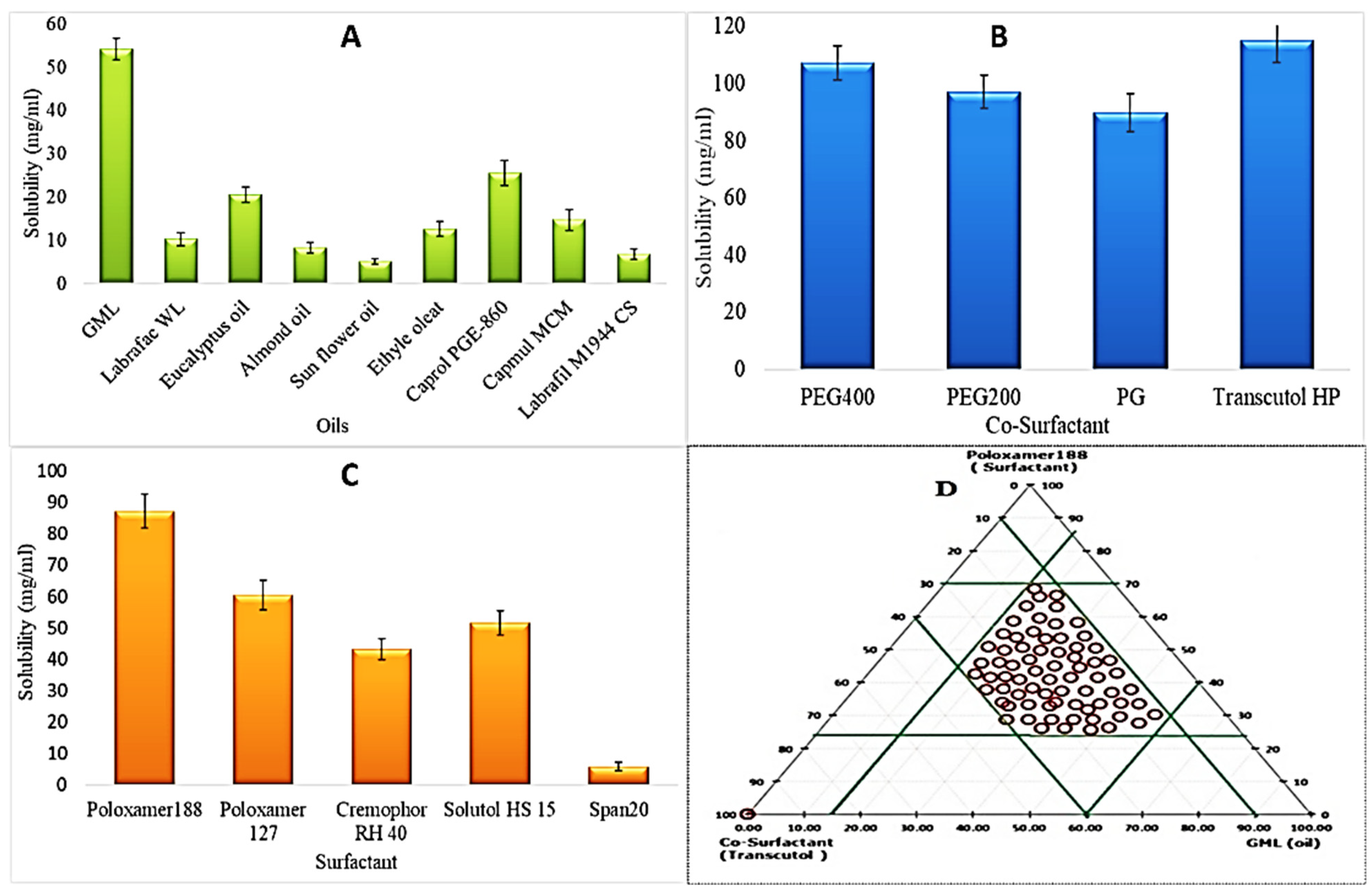
2.2.1. Screening of Formulation Materials
2.2.2. Construction of Pseudo Ternary Phase Diagram
2.3. Optimization
2.4. Development of PE Loaded SNEDDS
2.5. Characterization
2.5.1. Globule Characterization
2.5.2. Self-Nano Emulsification Time
2.5.3. Percentage Transmittance
2.6. Evaluation of Optimized PE-SNEDDS (OF3)
2.7. Drug Release Study
2.8. Permeation Study
2.9. Formulation of Solid PE-SNEDDS (S-PE-SNEDDS)
2.10. Evaluation of S-PE-SNEDDS
2.10.1. Globule Size, PDI, and Zeta Potential
2.10.2. Morphological Study
2.10.3. Thermal Analysis
2.10.4. Drug Content
2.10.5. Drug Release Study
2.10.6. In-Vitro Antioxidant Study
2.10.7. Anti-Microbial Study
2.11. In-Vivo Study
2.11.1. Pharmacodynamic Study
2.11.2. Pharmacokinetic Study
2.11.3. Histological Study
2.12. Stability Study
2.13. Statistical Analysis
3. Results and Discussion
3.1. Screening of Formulation Materials
3.2. Pseudo Ternary Phase Diagram
3.3. Optimization
3.3.1. Effect of Variables over Globule Size (Y1)
3.3.2. Effect of Variables over Percentage Transmittant (Y2)
3.3.3. Effect of Independent Variables over Emulsification Time (Y3)
3.4. Point prediction
3.5. SNEDDS Characterization
3.6. Stability Study
3.7. Viscosity and Refractive Index Measurement
3.8. Drug Release
3.9. Permeation Study
3.10. Development of Solid PE-SNEDDS
3.11. Characterization
3.11.1. Globule Size, PDI, Zeta potential
3.11.2. Scanning Electron Microscopy (SEM)
3.11.3. Thermal Analysis
3.11.4. Drug Release Study
3.11.5. Antioxidant Study
3.11.6. Anti-Microbial Activity
3.12. In Vivo Study
3.12.1. Pharmacodynamic Activity
3.12.2. Pharmacokinetic Study
3.12.3. Histological Examination
3.13. Stability Study
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; de Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension: The Task Force for the Management Of Arterial Hypertension of the European Society of Cardiology and the European Society of Hypertension. J. Hypertens. 2018, 36, 1953–2041. [Google Scholar] [CrossRef] [Green Version]
- Nouri, Z.; Hajialyani, M.; Izadi, Z.; Bahramsoltani, R.; Farzaei, M.H.; Abdollahi, M. Nanophytomedicines for the Prevention of Metabolic Syndrome: A Pharmacological and Biopharmaceutical Review. Front. Bioeng. Biotechnol. 2020, 8, 425. [Google Scholar] [CrossRef] [PubMed]
- Savjani, K.T.; Gajjar, A.K.; Savjani, J.K. Drug Solubility: Importance and Enhancement Techniques. ISRN Pharm. 2012, 2012, 195727. [Google Scholar] [CrossRef] [Green Version]
- Tang, J.L.; Sun, J.; He, Z.G. Self-Emulsifying Drug Delivery Systems: Strategy for Improving Oral Delivery of Poorly Soluble Drugs. Curr. Drug ther. 2007, 2, 85–93. [Google Scholar] [CrossRef] [Green Version]
- Abdel-Daim, M.M.; Sayed, A.A.; Abdeen, A.; Aleya, L.; Ali, D.; Alkahtane, A.A.; Alarifi, S.; Alkahtani, S. Piperine enhances the antioxidant and anti-inflammatory activities of thymoquinone against microcystin-LR-induced hepatotoxicity and neurotoxicity in mice. Oxidative Med. Cell. Longev. 2019, 5, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Intasar, S.; Taqvi, H.; Shah, A.J.; Gilani, A.H. Blood Pressure Lowering and Vasomodulator Effects of Piperine. J. Cardiovasc. Pharmacol. 2008, 52, 452–458. [Google Scholar]
- Labib, M.; Sohrab, M. Protective effecct of black seed oil from Nigella sativa against murine cytomegalovirus infection. Int. J. Immunopharmacol. 2000, 22, 729–740. [Google Scholar]
- Stojanović-Radić, Z.; Pejčić, M.; Dimitrijević, M.; Aleksić, A.; Anil Kumar, N.V.; Salehi, B.; Cho, W.C.; Sharifi-Rad, J. Piperine-A Major Principle of Black Pepper: A review of its bioactivity and studies. Appl. Sci. 2019, 20, 4270. [Google Scholar] [CrossRef] [Green Version]
- Shao, B.; Cui, C.; Ji, H.; Tang, J.; Wang, Z.; Liu, H.; Qin, M.; Li, X.; Wu, L. Enhanced oral bioavailability of piperine by self-emulsifying drug delivery systems: In vitro, in vivo and in situ intestinal permeability studies. Drug Delivery. 2015, 22, 740–747. [Google Scholar] [CrossRef] [Green Version]
- Zafar, F.; Jahan, N.; Rahman, K.U.; Bhatti, H.N. Increased Oral Bioavailability of Piperine from an Optimized Piper nigrum Nanosuspension. Planta Med. 2019, 85, 249–257. [Google Scholar] [CrossRef] [Green Version]
- Ray, L.; Karthik, R.; Srivastava, V.; Singh, S.P.; Pant, A.B.; Goyal, N.; Gupta, K.C. Efficient antileishmanial activity of amphotericin B and piperine entrapped in enteric coated guar gum nanoparticles. Drug Deliv. Transl. Res. 2021, 11, 118–130. [Google Scholar] [CrossRef] [PubMed]
- Zhou, K.; Yan, Y.; Chen, D.; Huang, L.; Li, C.; Meng, K.; Wang, S.; Algharib, S.A.; Yuan, Z.; Xie, S. Solid Lipid Nanoparticles for Duodenum Targeted Oral Delivery of Tilmicosin. Pharmaceutics 2020, 12, 731. [Google Scholar] [CrossRef]
- Shah, N.V.; Seth, A.K.; Balaraman, R.; Aundhia, C.J.; Maheshwari, R.A.; Parmar, G.R. Nanostructured lipid carriers for oral bioavailability enhancement of raloxifene: Design and in vivo study. J. Adv. Res. 2016, 7, 423–434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jain, S.; Kumar, D.; Swarnakar, N.K.; Thanki, K. Biomaterials Polyelectrolyte stabilized multilayered liposomes for oral delivery of paclitaxel. Biomaterials. 2012, 33, 6758–6768. [Google Scholar] [CrossRef] [PubMed]
- Arzani, G.; Haeri, A.; Daeihamed, M.; Bakhtiari-Kaboutaraki, H.; Dadashzadeh, S. Niosomal carriers enhance oral bioavailability of carvedilol: Effects of bile salt-enriched vesicles and carrier surface charge. Int. J. Nanomed. 2015, 10, 4797–4813. [Google Scholar]
- Kontogiannidou, E.; Meikopoulos, T.; Gika, H.; Panteris, E.; Vizirianakis, I.S.; Müllertz, A.; Fatouros, D.G. In Vitro Evaluation of Self-Nano-Emulsifying Drug Delivery Systems (SNEDDS) Containing Room Temperature Ionic Liquids (RTILs) for the Oral Delivery of Amphotericin B. Pharmaceutics 2020, 12, 699. [Google Scholar] [CrossRef]
- Liu, C.; Lv, L.; Guo, W.; Mo, L.; Huang, Y.; Li, G.; Huang, X. Self-Nanoemulsifying Drug Delivery System of Tetrandrine for Improved Bioavailability: Physicochemical Characterization and Pharmacokinetic Study. Biomed. Res. Int. 2018, 27, 6763057. [Google Scholar] [CrossRef]
- Verma, S.; Singh, S.K.; Verma, P.R.P. Fabrication of lipidic nanocarriers of loratadine for facilitated intestinal permeation using multivariate design approach. Drug. Dev. Ind. Pharm. 2016, 42, 288–306. [Google Scholar] [CrossRef]
- Alghananim, A.; Ozalp, Y.; Mesut, B.; Serakinci, N.; Qzsoy, Y.; Gungor, S. A Solid Ultra Fine Self-Nanoemulsifying Drug Delivery System (S-SNEDDS) of Deferasirox for Improved Solubility: Optimization, Characterization, and In Vitro Cytotoxicity Studies. Pharmaceuticals 2020, 13, 162. [Google Scholar] [CrossRef]
- Kassem, A.M.; Ibrahim, H.M.; Samy, A.M. Development and Optimization of Atorvastatin Calcium loaded Self–nanoemulsifying Drug Delivery System (SNEDDS) for Enhancing Oral Bioavailability: In vitro and in vivo Evaluation. J Microencapsul. 2017, 34, 319–333. [Google Scholar] [CrossRef]
- Verma, R.; Kaushi, A.; Almeer, R.; Rahman, M.H.; Abdel-Daim, M.M.; Kaushik, D. Improved Pharmacodynamic Potential of Rosuvastatin by Self-Nanoemulsifying Drug Delivery System: An in vitro and in vivo Evaluation. Int. J. Nanomed. 2021, 16, 905–924. [Google Scholar] [CrossRef]
- Cho, H.Y.; Kang, J.H.; Ngo, L.; Tran, P.; Lee, Y.B. Preparation and Evaluation of Solid-Self-Emulsifying Drug Delivery System Containing Paclitaxel for Lymphatic Delivery. J. Nanomater. 2016, 2016, 18–20. [Google Scholar] [CrossRef]
- Santosh, M.K.; Shaila, D.; Rajyalakshmi, I.; Rao, I.S. RP-HPLC Method for Determination of Piperine from Piper longum Linn. and Piper nigrum Linn. E-J. Chem. 2005, 2, 131–135. [Google Scholar] [CrossRef] [Green Version]
- Shanmugam, S.; Baskaran, R.; Balakrishnan, P.; Thapa, P.; Yong, C.S.; Yoo, B.K. Solid self-nanoemulsifying drug delivery system (S-SNEDDS) containing phosphatidylcholine for enhanced bioavailability of highly lipophilic bioactive carotenoid lutein. Eur. J. Pharm. Biopharm. 2011, 79, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Beg, S.; Swain, S.; Singh, H.P.; Patra, C.N.; Rao, M.E.B. Development, Optimization, and Characterization of Solid Self-Nanoemulsifying Drug Delivery Systems of Valsartan Using Porous Carriers. AAPS PharmSciTech 2012, 13, 1416–1427. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caddeo, C.; Pucci, L.; Gabriele, M.; Carbone, C.; Fernàndez-Busquets, X.; Valenti, D.; Pons, R.; Vassallo, A.; Fadda, A.M.; Manconi, M. Stability, biocompatibility and antioxidant activity of PEG-modified liposomes containing resveratrol. Int. J. Pharm. 2018, 538, 40–47. [Google Scholar] [CrossRef]
- Imam, S.S.; Alshehri, S.; Alzahrani, T.A.; Hussain, A.; Altamimi, M.A. Formulation and Evaluation of Supramolecular Food-Grade Piperine HP β CD and TPGS Complex: Dissolution, Physicochemical Characterization, Molecular Docking, In Vitro Antioxidant Activity, and Antimicrobial Assessment. Molecules 2020, 25, 4716. [Google Scholar] [CrossRef]
- Khemakhem, I.; Abdelhedi, O.; Trigui, I.; Ayadi, M.A.; Bouaziz, M. Structural, antioxidant and antibacterial activities of polysaccharides extracted from olive leaves. Int. J. Biol. 2018, 106, 425–432. [Google Scholar] [CrossRef]
- Shazly, G.A. Corrigendum to “Ciprofloxacin Controlled-Solid Lipid Nanoparticles: Characterization, In Vitro Release, and Antibacterial Activity Assessment”. Biomed Res. Int. 2017, 2017, 6761452. [Google Scholar] [CrossRef] [Green Version]
- Di, X.; Wang, X.; Liu, Y. Effect of piperine on the bioavailability and pharmacokinetics of emodin in rats. J. Pharm. Biomed. Anal. 2015, 115, 144–149. [Google Scholar] [CrossRef]
- Ren, T.; Hu, M.; Cheng, Y.; Shek, T.L.; Xiao, M.; Ho, N.J.; Zhang, C.; Leung, S.S.Y.; Zuo, Z. Piperine-loaded nanoparticles with enhanced dissolution and oral bioavailability for epilepsy control. Eur. J. Pharm. Sci. 2019, 137, 104988. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Ye, X.; Shang, X.; Peng, X.; Bao, Q.; Liu, M.; Guo, M.; Li, F. Enhanced oral bioavailability of silybin by a supersaturable self-emulsifiyng drug delivery system (SSEDDS). Colloids Surf. A Physicochem. Eng. Asp. 2012, 396, 22–28. [Google Scholar] [CrossRef]
- Kim, D.S.; Cho, J.H.; Park, J.H.; Kim, J.S.; Song, E.S.; Kwon, J.; Giri, B.R.; Jin, S.G.; Kim, K.S.; Choi, H.G.; et al. Self-microemulsifying drug delivery system (SMEDDS) for improved oral delivery and photostability of methotrexate. Int. J. Nanomed. 2019, 14, 4949–4960. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kamble, R.N.; Mehta, P.P.; Kumar, A. Efavirenz Self-Nano-Emulsifying Drug Delivery System: In Vitro and In Vivo Evaluation. AAPS PharmSciTech 2016, 17, 1240–1247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, R.; Khursheed, R.; Kumar, R.; Awasthi, A.; Sharma, N.; Gowthamarajan, K.; Wadhwani, A. Self-nanoemulsifying drug delivery system of fisetin: Formulation, optimization, characterization and cytotoxicity assessment. J. Drug Deliv. Sci. Technol. 2019, 54, 101252. [Google Scholar] [CrossRef]
- Patel, J.; Dhingani, A.; Tilala, J.; Raval, M.; Sheth, N. Formulation and Development of Self-Nanoemulsifying Granules of Olmesartan Medoxomil for Bioavailability Enhancement. Part. Sci. Technol. 2014, 12, 37–41. [Google Scholar] [CrossRef]
- Balakumar, K.; Vijaya, C.; Tamil, N.; Hari, R.; Abdu, S. Self nanoemulsifying drug delivery system (SNEDDS) of Rosuvastatin calcium: Design, formulation, bioavailability and pharmacokinetic evaluation. Colloids Surf. B Biointerfaces 2013, 112, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Alhasani, K.F.; Kazi, M.; Ibrahim, M.A.; Shahba, A.A.; Alanazi, F.K. Self-nanoemulsifying ramipril tablets: A novel delivery system for the enhancement of drug dissolution and stability. Int. J. Nanomed. 2019, 14, 5435–5448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huguet-Casquero, A.; Moreno-Sastre, M.; López-Méndez, T.B.; Gainza, E.; Pedraz, J.L. Encapsulation of oleuropein in nanostructured lipid carriers: Biocompatibility and antioxidant efficacy in lung epithelial cells. Pharmaceutics 2020, 12, 429. [Google Scholar] [CrossRef]
- Cui, H.; Wu, J.; Li, C.; Lin, L. Improving anti-listeria activity of cheese packaging via nanofiber containing nisin-loaded nanoparticles. LWT—Food Sci. Technol. 2017, 81, 233–242. [Google Scholar] [CrossRef]
- Cui, H.; Zhou, H.; Lin, L. The specific antibacterial effect of the Salvia oil nanoliposomes against Staphylococcus aureus biofilms on milk container. Food Control. 2016, 61, 92–98. [Google Scholar] [CrossRef]
- Mgbeahuruike, E.E.; Stålnacke, M.; Vuorela, H.; Holm, Y. Antibiotics Antimicrobial and Synergistic Effects of Commercial Piperine and Piperlongumine in Combination with antimicrobials. Antibiotics 2019, 8, 55. [Google Scholar] [CrossRef] [PubMed] [Green Version]











| Formulation Variables | Level (Coded Value) | ||
|---|---|---|---|
| Low (−1) | Medium (0) | High (+1) | |
| Oil (%) | 15 | 37.5 | 60 |
| Surfactant (%) | 25 | 47.5 | 70 |
| Co-surfactant (%) | 10 | 25 | 40 |
| Responses | Goal | ||
| Globule size (nm) | Minimum | ||
| Transmittance (%) | Maximum | ||
| Emulsification time (Second) | Minimum | ||
| Code | Oil Concentration (%) | Conc of Surfactant (%) | Conc of Co-Surfactant (%) | Globule Size (nm) | Transmittant (%) | Emulsification Time (S) | |||
|---|---|---|---|---|---|---|---|---|---|
| Actual Value * | Predicted Value | Actual Value * | Predicted Value | Actual Value * | Predicted Value | ||||
| F1 | 15 | 25 | 25 | 62.03 ± 3.54 | 63.05 | 97.28 ± 1.23 | 97.17 | 59 ± 2 | 60 |
| F2 | 60 | 25 | 25 | 200.34 ± 4.23 | 201.70 | 85.23 ± 2.42 | 85.22 | 144 ± 3 | 143 |
| F3 | 15 | 70 | 25 | 30.52 ± 5.12 | 28.30 | 98.00 ± 2.12 | 98.01 | 46 ± 2 | 48 |
| F4 | 60 | 70 | 25 | 117.91 ± 3.65 | 116.89 | 96.15 ± 3.65 | 96.26 | 81 ± 2 | 80 |
| F5 | 15 | 47.5 | 10 | 65.54 ± 5.61 | 66.14 | 93.30 ± 1.42 | 93.37 | 62 ± 3 | 63 |
| F6 | 60 | 47.5 | 10 | 178.78 ± 5.43 | 179.24 | 89.10 ± 1.23 | 89.07 | 135 ± 2 | 136 |
| F7 | 15 | 47.5 | 40 | 25.16 ± 6.17 | 24.70 | 99.89 ± 2.46 | 99.92 | 33 ± 3 | 32 |
| F8 | 60 | 47.5 | 40 | 140.54 ± 5.32 | 138.86 | 90.57 ± 1.76 | 90.50 | 75 ± 2 | 77 |
| F9 | 37.5 | 25 | 10 | 170.76 ± 7.43 | 167.84 | 88.54 ± 1.27 | 88.58 | 106 ± 2 | 107 |
| F10 | 37.5 | 70 | 10 | 95.23 ± 6.76 | 95.56 | 96.23 ± 1.65 | 96.15 | 42 ± 2 | 43 |
| F11 | 37.5 | 25 | 40 | 115.43 ± 3.65 | 114.44 | 94.12 ± 1.61 | 94.20 | 35 ± 2 | 35 |
| F12 | 37.5 | 70 | 40 | 65.65 ± 3.87 | 67.16 | 98.56 ± 1.46 | 98.52 | 23 ± 3 | 24 |
| F13 * | 37.5 | 47.5 | 25 | 109.21 ± 6.32 | 103.12 | 97.56 ± 2.43 | 97.79 | 62 ± 2 | 62 |
| F14 * | 37.5 | 47.5 | 25 | 101.54 ± 5.32 | 103.54 | 97.98 ± 1.43 | 97.79 | 61 ± 3 | 62 |
| F15 * | 37.5 | 47.5 | 25 | 104.24 ± 3.54 | 103.76 | 97.76 ± 1.65 | 97.79 | 63 ± 2 | 62 |
| F16 * | 37.5 | 47.5 | 25 | 100.76 ± 5.36 | 103.63 | 97.76 ± 1.67 | 97.79 | 63 ± 3 | 62 |
| F17 * | 37.5 | 47.5 | 25 | 101.71 ± 4.58 | 103.21 | 97.89 ± 1.87 | 97.79 | 62 ± 2 | 62 |
| Statistical Term | Globule Size (Y1) | % Transmittant (Y2) | Emulsification Time (Y3) |
|---|---|---|---|
| R2 | 0.9980 | 0.9995 | 0.9996 |
| Adjusted R2 | 0.9954 | 0.9988 | 0.9991 |
| Model F-Value | 388.10 | 1476.83 | 1936.34 |
| Model p-value | <0.0001 | <0.0001 | <0.0001 |
| Lack of fit F-value | 0.51 | 0.67 | 0.39 |
| Lack of fit p-value | 0.6945 * | 0.6129 * | 0.7693 * |
| Adequate Precision | 70.594 | 130.299 | 169.586 |
| Code | Composition of SNEDDS | Actual Value | Predicted Value | ||||
|---|---|---|---|---|---|---|---|
| Oil | Globule Size (nm) | Transmittant (%) | Emulsification Time (Sec) | Globule Size (nm) | Transmittant (%) | Emulsification Time (Sec) | |
| OF1 | 37.50:47.50:25.00 | 104.23 ± 2.54 | 97.87 ± 2.12 | 61 ± 1 | 103 | 97.79 | 62 |
| OF2 | 30.00:46.28:25.00 | 86.54 ± 3.65 | 98.65 ± 2.17 | 55 ± 2 | 84.93 | 98.55 | 56 |
| OF3 | 25.00:46.28:25.00 | 70.34 ± 3.27 | 99.02 ± 2.02 | 53 ± 2 | 71.21 | 98.87 | 54 |
| Ingredients | Concentration (mg) | Composition (%) |
|---|---|---|
| Piperine | 20 | 7.51 |
| GML | 25 | 9.39 |
| Poloxamer 188 | 46.28 | 17.38 |
| Transcutol HP | 25 | 9.39 |
| Avicel | 150 mg | 56.33 |
| S.No. | Kinetic Model | Plot | R2 |
|---|---|---|---|
| 1 | Zero order | % cumulative amount of drug released versus time | 0.9891 |
| 2 | First order | Log % cumulative drug remaining versus time | 0.8142 |
| 3 | Higuchi’s model | % cumulative amount of drug released versus square root of time | 0.9631 |
| 4 | Korsmeyer-Peppas model | Log of fraction of drug released/ Log cumulative % drug released versus log time | 0.9352 n = 0.2096 |
| Parameters | Pure PE | S-PE-SNEDDS |
|---|---|---|
| Cmax (µg/mL) | 149.18 ± 25.29 | 585.14 ± 39.47 |
| Tmax (h) | 1 | 2 |
| Ke (h−1) | 0.11 ± 0.009 | 0.0958 ± 0.006 |
| T1/2 (h) | 5.81 ± 0.57 | 7.23 ± 0.39 |
| AUC0–12 (µg·h/mL) | 667.14 ± 27.39 | 3718.99 ± 83.72 |
| AUC0–α (µg·h/mL) | 866.14 ± 43.91 | 5177.43 ± 157.36 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zafar, A.; Imam, S.S.; Alruwaili, N.K.; Alsaidan, O.A.; Elkomy, M.H.; Ghoneim, M.M.; Alshehri, S.; Ali, A.M.A.; Alharbi, K.S.; Yasir, M.; et al. Development of Piperine-Loaded Solid Self-Nanoemulsifying Drug Delivery System: Optimization, In-Vitro, Ex-Vivo, and In-Vivo Evaluation. Nanomaterials 2021, 11, 2920. https://doi.org/10.3390/nano11112920
Zafar A, Imam SS, Alruwaili NK, Alsaidan OA, Elkomy MH, Ghoneim MM, Alshehri S, Ali AMA, Alharbi KS, Yasir M, et al. Development of Piperine-Loaded Solid Self-Nanoemulsifying Drug Delivery System: Optimization, In-Vitro, Ex-Vivo, and In-Vivo Evaluation. Nanomaterials. 2021; 11(11):2920. https://doi.org/10.3390/nano11112920
Chicago/Turabian StyleZafar, Ameeduzzafar, Syed Sarim Imam, Nabil K. Alruwaili, Omar Awad Alsaidan, Mohammed H. Elkomy, Mohammed M. Ghoneim, Sultan Alshehri, Ahmed Mahmoud Abdelhaleem Ali, Khalid Saad Alharbi, Mohd Yasir, and et al. 2021. "Development of Piperine-Loaded Solid Self-Nanoemulsifying Drug Delivery System: Optimization, In-Vitro, Ex-Vivo, and In-Vivo Evaluation" Nanomaterials 11, no. 11: 2920. https://doi.org/10.3390/nano11112920








