Abstract
Subacute sclerosing panencephalitis (SSPE) is a late complication of measles virus infection that occurs in previously healthy children. This disease has no specific cure and is associated with a high degree of disability and mortality. In recent years, there has been an increase in its incidence in relation to a reduction in vaccination adherence, accentuated by the COVID-19 pandemic. In this article, we take stock of the current evidence on SSPE and report our personal clinical experience. We emphasise that, to date, the only effective protection strategy against this disease is vaccination against the measles virus.
1. Introduction
Subacute sclerosing panencephalitis (SSPE) is a rare progressive degenerative disorder of the central nervous system (CNS) caused by persistent measles virus (MeV) infection. Primarily, it affects children and young adolescents and can be fatal [1].
The SSPE burden reflects the epidemiology of natural MeV infection, and it is inversely related to vaccination coverage. According to analyses from the USA and the UK, 4 to 11 cases of SSPE are expected per 100,000 cases of MeV, but the incidence is higher when the infection is contracted under the age of 1 year (18/100,000) [1]. The worldwide incidence of SSPE varies greatly, from approximately 0.2 to 40 cases per million population per year, considering geographical disparity, with a reduction of 82–96% in countries that have achieved increased vaccination coverage, in particular from 2000 to 2016 [2]. However, despite the Global Vaccine Action Plan endorsed by the World Health Organization (WHO), the global elimination of MeV has taken a significant step backward recently, with an increase in cases to 120 per million in 2019, the highest since 2001. Vaccine refusal, an emerging and spreading trend in the population, is considered a significant reason for these changes in population-level susceptibility to MeV [3]. Moreover, the COVID-19 pandemic has further slowed vaccination and surveillance programs [4]. During the international containment measures, reports showed measles–mumps–rubella vaccination counts to be about 19.8% lower in 2020 compared to the same period in 2019 [5].
Given this dangerous scenario, new future outbreaks of MeV infection and related complications are possible if appropriate public health programs are not implemented with immediate effect [4].
Among long-term complications of MeV infection, SSPE has the longest latency, varying from 1 month to 27 years, with a median age at diagnosis of 12 years [6]. A shorter latency is related to an early primary infection, at under 2 years of age, usually associated with family clusters, mainly due to the lack of parental vaccination coverage. Although no gender predisposition has ever been proven, a higher incidence has mainly been reported in boys, with a male-to-female ratio of 2.8:1 [7,8].
After primary infection, persistent virus replication in the CNS leads to progressive destruction of the neurons, which becomes clinically evident several years later [9,10]. Neurons are not the only target of the virus, and numerous neuropathological studies on the brain tissue of SSPE patients have established that, apart from the neurons in the grey matter, many oligodendrocytes are infected in the white matter [11,12,13]. Astrocytes are also infected throughout the CNS, but to a much lesser degree [13].
1.1. Pathogenesis
The pathogenesis of the disease is still under investigation. SSPE is caused by hypermutated MeVs combined with an inadequate cellular response [14].
Although the MeV is serologically monotypic, genetic variability has defined eight clades, including 23 genotypes and a putative new genotype, that are geographically and temporally restricted [15]. Despite the paucity of studies on the molecular epidemiology of SSPE, MeV sequences obtained from brain tissues are homologous to the genotype circulating at the time of primary exposure to MeV [16]. There is no evidence to indicate that wild-type MeV strains differ in terms of either pathogenesis or neurovirulence [17]. Studies on brain tissue of SSPE patients have shown that the MeV in these patients has several genetic mutations that enable the virus to spread and persist in the human brain [18,19,20]. Since human neurons, an important target affected in the disease, do not express the known MeV receptors (signalling lymphocyte activation molecule (SLAM) and nectin 4), how the MeV infects neurons and spreads between them is unknown [21].
MeV recovered from patients with SSPE differs from wild-type viruses in mutations involving the matrix (M), hemagglutinin (H), nucleocapsid (N) and fusion (F) genes [22]. The MeV genome is encapsidated by the N protein and forms the ribonucleoprotein (RNP) complex with the viral RNA-dependent RNA polymerase comprised of the L and P proteins. The two envelope glycoproteins have a role in receptor binding (H protein) and membrane fusion (F protein) [22]. The M gene is highly mutated in almost all cases in the entire open reading frame [18,19]; the N is modified in the carboxyl terminus, and the H has biased hypermutation in a limited region [15,17]. Further, many persisting MeVs have mutations in the F gene, which cause the cytoplasmic tail of the F protein to elongate or shorten [23,24].
Mutations in the M protein impair the formation of new viral particles, helping the replicating virus to persist in the neuronal cells, spread through the synapses and evade the neutralising antibodies [6]. Studies have detected a particular triresidue structural motif at residues 64, 89 and 209, with proline, glutamate and alanine at these positions, respectively, (called PEA) in the primary sequence of the SSPE M proteins, which is absent in vaccine and lab-adapted strains, confirming the theory that only the wild-type virus can cause SSPE [25].
Recent studies have revealed that changes in the ectodomain of the MeV F protein play a key role in the MeV spread in the brain [26,27,28]. Changes in the F protein render it hyperfusogenic, allowing the virus to propagate in the neurons [21,29]. Notably, the F protein, not the receptor-binding H protein, changes during persistence to allow the virus to exhibit tropism for neurons [22]. The changes in the M protein and the cytoplasmic tail of the F protein affect the interaction between the two proteins, increase the surface expression level of the F protein and enhance cell-to-cell fusion [30]. Hyperfusogenic F proteins permit MeVs to enter cells and spread without the need to engage nectin-4 or CD150, known receptors for MeV not present on neural cells [31]. Changes in the H and F proteins can also be associated with persistent infection, with the M protein remaining relatively unaffected [24,32]. Since all three proteins are associated with viral budding from infected cells and putative fusion with uninfected cells, the infection is thought to persist due to defects in these two processes [6,33].
Another hypothesis is that the MeV acts upon another cell receptor heavily concentrated at the synapses to gain entry into a neuron [34]. One of the proposed mechanisms of neuronal spread is via neurokinin-1 [35], while substance P and the fusion inhibitory peptide block viral transmission [22]. CD9 may also play a role in this process because high levels of antibodies against it have been found in the cerebrospinal fluid (CSF) of individuals with SSPE and severe brain atrophy [36].
Some studies have shown that cell adhesion molecule 1 (CADM1) and CADM2 are host factors that enable the MeV to cause membrane fusion in cells lacking the known receptors and to spread between neurons [21,37]. CADM1 and CADM2 interact in cis with the H protein on the same cell membrane, triggering hyperfusogenic F protein-mediated membrane fusion [37]. Recently, a process has been discovered involving transmembrane and cytoplasmic protein transfer that relies on cell–cell contacts established by the nectin adhesive interface. This process has been called nectin-elicited cytoplasm transfer (NECT). NECT can spread MeV infections from epithelial cells to primary neurons, which is possibly the first step of neuropathology [38]. However, the role of CADM1, CADM2 and NECT in favouring the spread of the virus in the brain tissue must be verified.
Once inside the cell, the MeV changes the cell machinery to bypass the immune system and continues reproducing itself inside the cell in a less cytopathic fashion to avoid destroying the host neurons [39].
SSPE may develop only in the case of a particular susceptibility of the host [6]. Impairment of determined immune system functions has been suggested as a leading cause of that susceptibility. Some Japanese studies have demonstrated that mutations in three genes, MxA (an antiviral protein induced by interferon-alpha and interferon-beta), interleukin-4 and interferon-1, are associated with a high risk of SSPE after MeV infection [40,41]. Other studies have suggested a deficient interferon (IFN-α) system in SSPE, with an imbalance in the production of IFN-α in response to stimulations in vitro [42]. Affected individuals showed an imbalance in the high levels of cytokines promoted by Th2 cells, such as IL-4 and IL-1b, and the low levels of cytokines promoted by Th1 cells, such as IFN-α, IL-2, IL-10 and IL-12 [42,43]. This cytokine storm enhances the humoral instead of the cytotoxic response, leading to a predisposition to the self-replication of the virus in the brain [41]. There is also a hypothesis suggesting that SSPE could affect people with transient immunosuppression due to an exposure to another pathogen at the moment of MeV primary infection [44].
Transmission among neurons is mediated by neurokinin synaptic receptors and genetic polymorphism favouring either a humoral response or intraneuronal spreading due to the facilitating polymorphism of the entry molecules [35].
1.2. Clinical Features and Diagnosis
A clinical picture of SSPE is characterised by an insidious and subacute cognitive decline and behavioural alterations, followed by movement disorders, such as myoclonus and ataxia, seizures and visual impairment. After the initial presentation, the disease can progress rapidly, leading to a vegetative state or death within a few years. Theoretically, the clinical course is divided into four stages, according to Jabbour et al. [45], summarised in Table 1.

Table 1.
Clinical stages of SSPE according to Jabbour et al. [45].
Atypical presentations, accounting for 10% of all cases, consist of isolated psychiatric manifestations, gait disturbance as an initial symptom, poorly controlled seizures, isolated extra pyramidal symptoms, visual loss and/or lateralising motor weakness. Usually, these patients do not follow any defined clinical course of the disease.
SSPE can be established according to Dyken’s criteria, described in the Materials and Methods section [46].
An electroencephalogram (EEG) is a valid tool to support the diagnosis. The typical EEG finding consists of periodic, generalised, symmetrical, stereotyped complexes, with high-voltage diphasic waves occurring synchronously throughout the recording and usually clinically associated with myoclonic jerks. These complexes are pathognomonic features of SSPE.
Neuroimaging can provide useful information but is not mandatory for diagnosis. One of the most common features in the magnetic resonance (MR) study of the brain is hyperintensities on T2-weighted images in the cerebral cortex, the periventricular white matter, the basal ganglia and the brainstem, present in the medium stage of the disease. Along with disease progression, diffuse cortical atrophy may be revealed.
The diagnosis can be confirmed by the detection of MeV antigens by a brain biopsy using a reverse-transcriptase polymerase chain reaction for MeV RNA [47].
1.3. Treatment
Currently, there is no specific validated therapy for SSPE. Nevertheless, considering the fatal course of the disease and the possibility of reaching a satisfactory outcome in up to 35% of patients who receive treatment in terms of clinical improvement, clinical stabilisation, slowdown of progression and prolonged survival, it is mandatory to treat patients with SSPE [39].
The most reliable therapies include inosiplex, ribavirin and interferon in different formulations and doses, even if the efficacy of these treatment approaches has not been clearly established (Table 2).

Table 2.
Data on the treatment of SSPE at various paediatric mean ages. See attached file.
Inosine pranobex (IP), a derivative of inosine and the p-acetamido-benzoic acid salt of N,N-dimethylamido-2-propanol, has been used since around 1971 for its immunomodulatory and antiviral properties with a safety profile. It stimulates the proliferation of T-cell lymphocytes, the functioning of natural killer cells, the release of pro-inflammatory cytokines and the inhibition of viral RNA synthesis. The inosiplex dosage was 100 mg/kg/day to a maximum of 3 g/day, taken orally in three divided doses for 6 months [58].
Ribavirin is a broad-spectrum antiviral agent that inhibits a wide range of RNA and DNA viruses. Considering the difficulty in crossing the blood–brain barrier, its efficacy in treating SSPE was discussed, and a new option of intraventricular administration was recently proposed [59].
Other antiviral drugs, such as favipiravir, have been shown to inhibit RNA viruses only in vitro, but this needs further investigation [60].
Interferon therapy (IFN-α) seems to be a promising treatment produced by recombinant DNA techniques, based on the hypothesis that MeV persists on the SNC due to the humoral response instead of a cellular physiological immune response, mediated by IFN-α.
IFN-α therapy also seems to have a positive effect on maintaining the metabolism of the cerebral cortex, as demonstrated through a serial 1⁸FDG-PET study on a 15-year-old girl with stage II subacute sclerosing panencephalitis (SSPE) treated with isoprinosine, IFN-α and ribavirin for 3 years [61].
Despite the lack of clinical studies on the improved results for treatment with IFN-α combined with another drug compared with only inosiplex monotherapy, the combination strategy was suggested [54].
This theory is based on preclinical research giving evidence that the combination of IFN-α with other drugs, such as ribavirin or inosiplex, should be preferred because of a synergic inhibitory effect on the virus’s replication in vitro and in hamster models [62].
In a clinical study in the 1990s, an improvement in the survival curves of patients treated with INF-α compared with control cases was found only in the first 8–9 years. A positive clinical effect of therapy was observed in 72% of the patients in the initial phase of the follow-up. After a period of 6 to 60 months without therapy, the clinical conditions deteriorated in most patients. Therefore, it was suggested that the long-term administration of combined therapy with inosiplex is more effective than its short-term administration [43].
2. Materials and Methods
2.1. Dyken’s Criteria for SSPE Diagnosis
Dyken’s criteria are divided into two major and four minor criteria, as described in Table 3:

Table 3.
SSPE diagnostic criteria.
2.2. Case Descriptions
In the present study, we describe the clinical presentation and the laboratory and instrumental findings for four paediatric cases affected by SSPE, each of whom underwent a long-term combined intrathecal IFN-α therapy. The main aim is to analyse the clinical course of the disease and the therapeutic outcome during a long-term follow-up.
This is a case series of paediatric SSPE patients monitored at the Bambino Gesù Children’s Hospital (Rome, Italy) over 10 years (2011–2021). We retrospectively enrolled 4 patients, all boys, who met Dyken’s criteria for the diagnosis of SSPE. We collected personal information, medical histories and laboratory data from electronic medical records. All patients underwent haematological investigations, including autoimmune workup, MRI, cerebrospinal fluid (CSF) study and EEG at the time of diagnosis. Measles antibodies were measured in the CSF and serum using enzyme immunoassays (ELISA). Then, CSF and serum specimens were sent to the WHO/LabNet National Reference Laboratory (NRL) for measles and rubella, where the level of the neutralising antibody was investigated by the plaque reduction neutralisation test (PRNT80) [63]. Molecular detection of the measles genome was attempted by means of RT-PCR, as previously described [64]. Sequencing of the N-450 region was performed on a measles-positive sample to establish the strain responsible for the infection.
All the patients were treated with combined intrathecal IFN-α therapy and this was followed up by a neurological examination, EEG and MRI at specific intervals after the initial reports. Restaging was made according to standardised classification at 6 months after diagnosis and during the last medical check-up.
3. Results
Case Series
We presented four male children, three of whom were exposed to measles before the first dose of vaccine, between 14 days and 11 months of age, with a confirmed maternal epidemiological link. Only one of them was exposed to the virus at 3 years of age, when he had already received the first dose of the measles vaccine, without a clear link of infection. In particular, the parents reported an episode of fever with a mild skin rash lasting 3 days.
In the same patient, it was hypothesised that a single dose of the vaccine did not result in sufficient immunological protection against primary infection, although immunological congenital defects were excluded with specific investigations (white blood cell count; lymphocyte subpopulation count; immunoglobulin Ig G, Ig M and IgG subclasses dosage and study of the lymphocytic phenotype B; and specific vaccine response to Haemophilus and Pneumoccoccus vaccinatiom). No information about the response to the first dose of the measles-containing vaccine (MMR) before the onset of neurological symptoms was found in the medical records, but the patient showed anti-measles IgG antibodies in his blood and liquor at the onset of SSPE symptoms.
Clinical, instrumental and laboratory findings are provided in Table 4.

Table 4.
Clinical, instrumental and laboratory findings in children with SSPE at diagnosis.
None of the patients received infusion of polyvalent immunoglobulin for post-exposure prophylaxis. None received a dose of vaccine prior to infection with MMR. Three of the patients had a similar latency time between exposure and SSPE onset, with a mean standard deviation (SD) age of 4.1 years ± 0.42 (range 46.30–55 months). The older infected boy had an increased latency of 12 years.
Clinical presentations at the onset ranged from stage 2A to stage 3 and included a wide spectrum of insidious symptoms, from abnormal behaviour and recurrent falls to visible motor and cognitive impairment. Children were referred to our hospital with a clear neurological symptomatology, and all of them presented myoclonus and atonic seizures. In addition, ballistic movements of the lower limbs and atypical clinical history with poorly controlled seizures were observed in patient B. Only one of them, patient C at 6 years of age, presented a rapid global neurological deterioration with spastic quadriplegia during an infective febrile episode. The same patient showed the better response to therapy in clinical terms, improving from the clinical stage 3 to stage 2D.
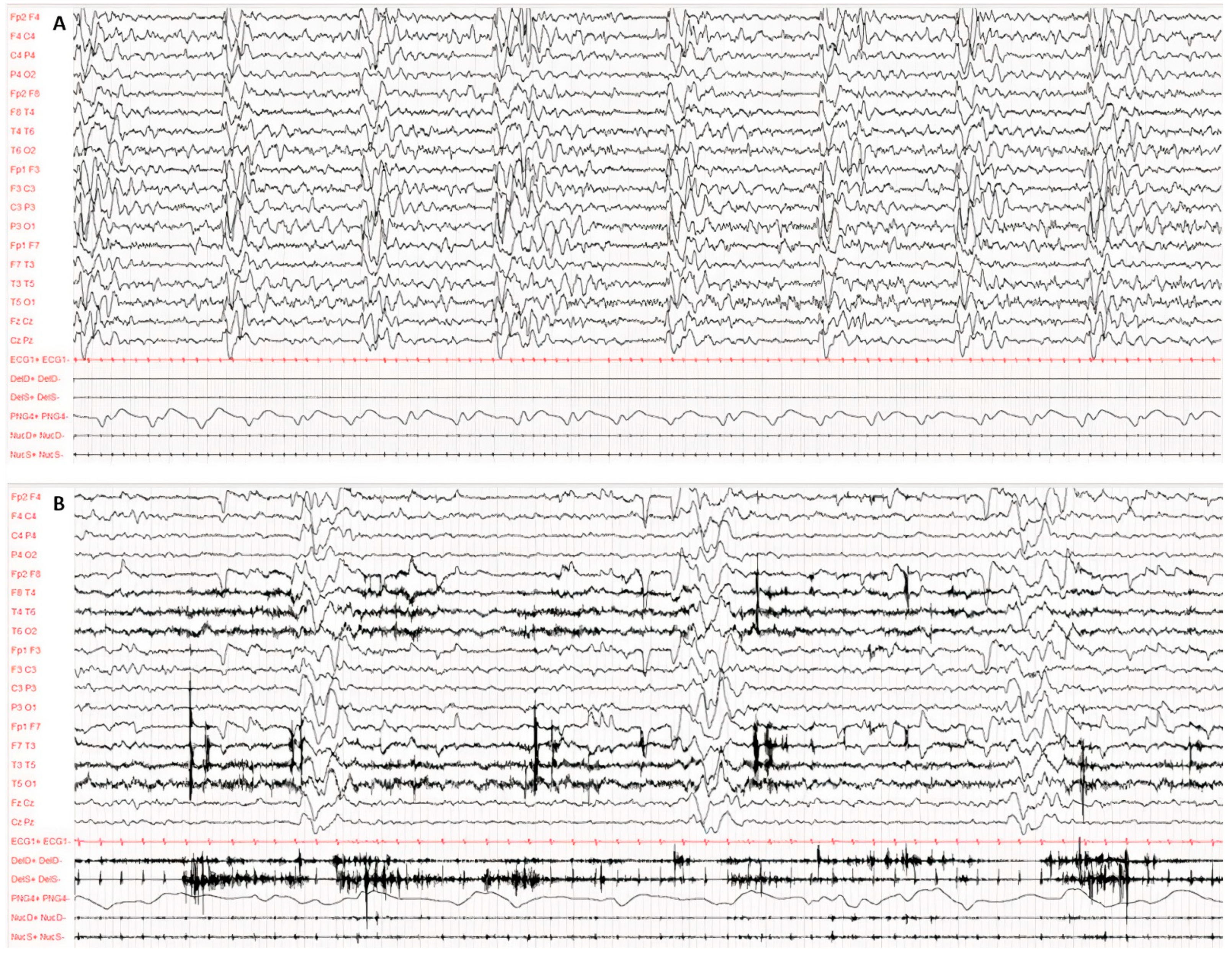
Diagnosis was made according to Dyken’s criteria. All patients fulfilled 3 out of the 4 criteria, in particular, clinical history, typical EEG and elevated CSF globulin levels, with the exception of the invasive brain immunopathology assay, which was not performed. Figure 1 and Figure 2 present the typical EEG findings for SSPE.
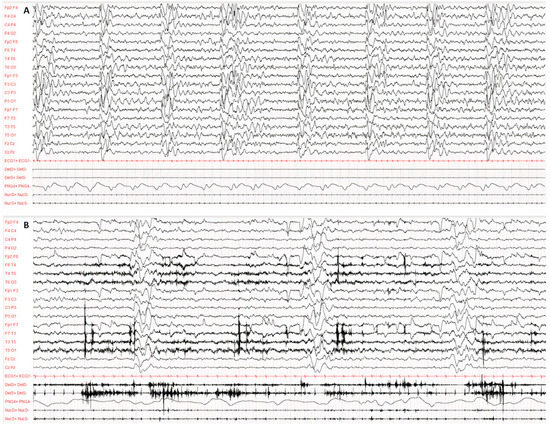
Figure 1.
Typical EEG at the onset of symptoms, performed according to the international 10–20 system with digital acquisition and polygraphy. EEG recording of 60 s awake (A) and during sleep (B). Pathological background activity is globally slowed down, disorganised and undifferentiated both in wakefulness and in sleep. Continuous periodic (about 5–6 s) and polyphasic complexes consisting of several high-voltage, irregular and bilateral asynchronous delta waves. The polygraphy recorded atonic seizures of limbs, head and face concomitantly with the periodic complexes.
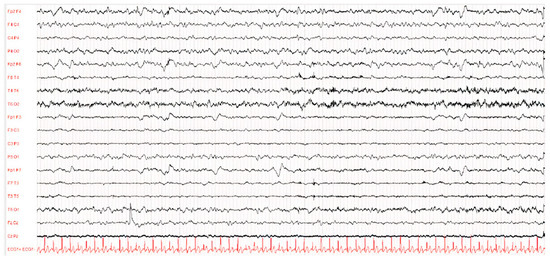
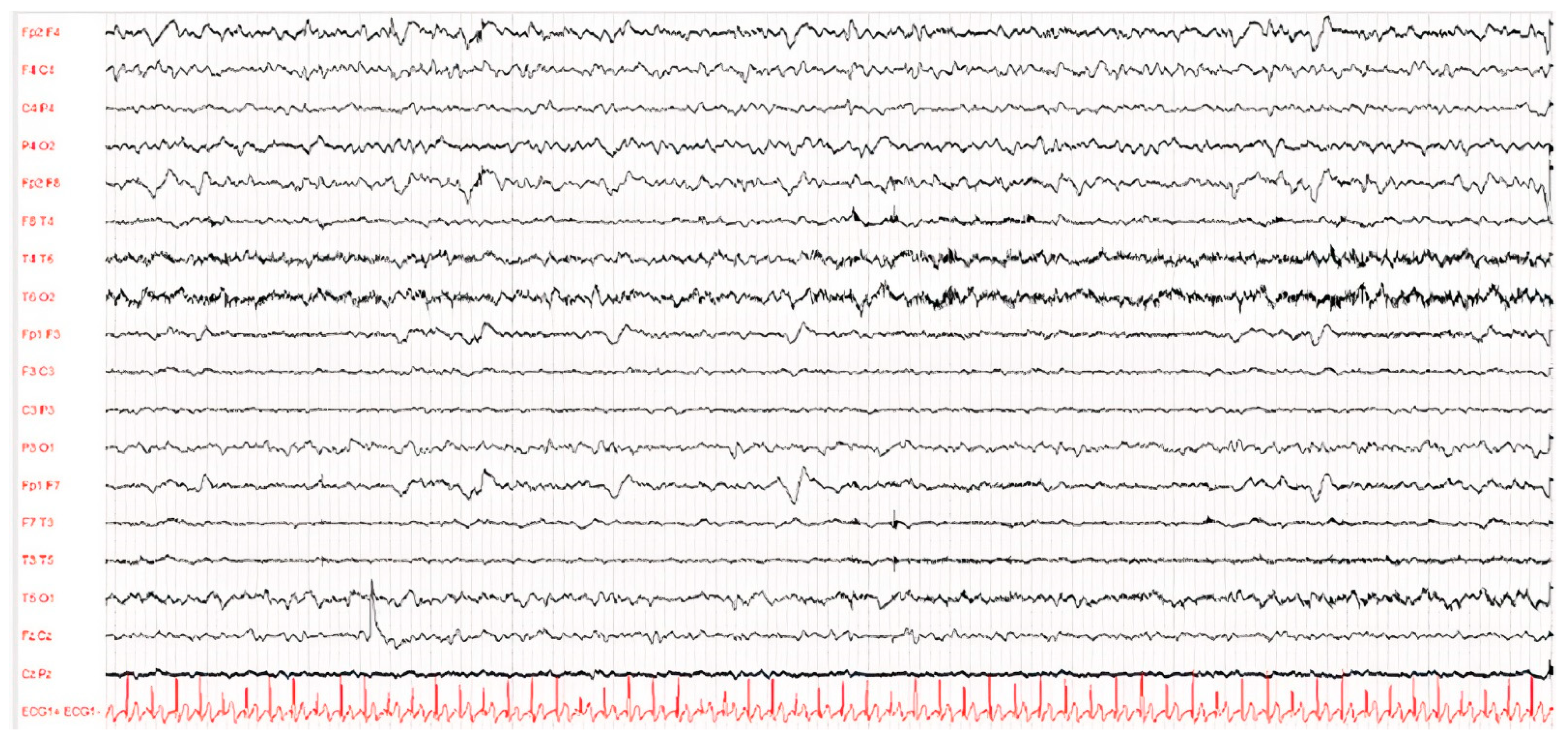
Figure 2.
EEG of patient D after 1 year of illness. The waking EEG trace showed globally disorganised, undifferentiated and asymmetrical brain activity due to the presence of hypovolted activity in the left frontal–central–temporal regions.
The EEG showed a typical periodic pattern in all patients and was a main exam used to guide studies by other laboratories (Figure 1 and Figure 2).
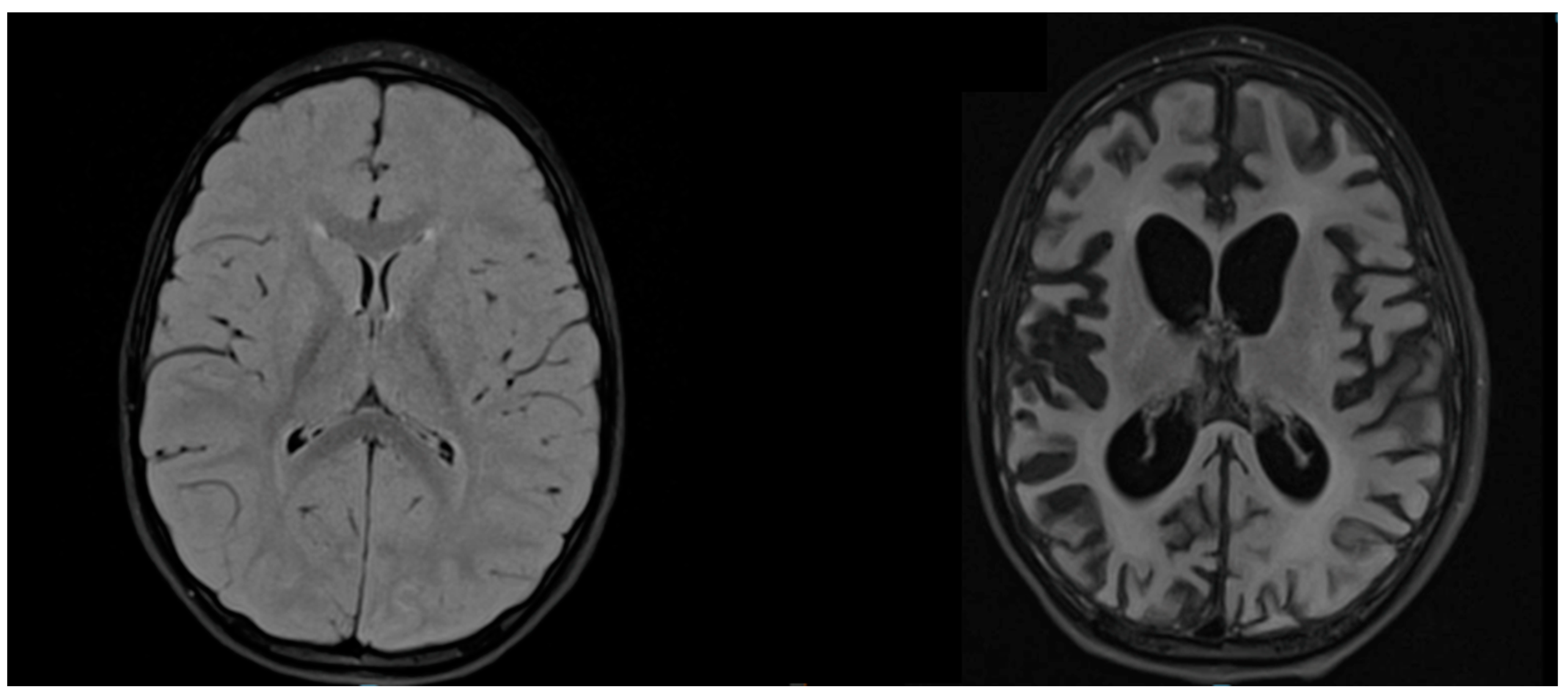
T2-weighted sequences of brain MRI showed hyperintensities in all patients, but in different areas, apparently not related to clinical severity (Figure 3).
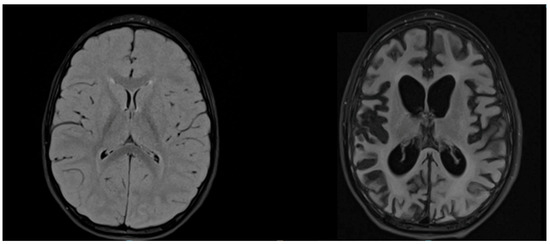
Figure 3.
MRI of patient D. Axial T2-weighted FLAIR MRIs at the time of diagnosis (age 3.9 years) and after 1 year of illness (age 4.9 years) show, respectively, signal alteration of the bilateral posterior parieto-occipital region and severe cerebral atrophy.
Blood examinations at diagnosis showed a normal blood cell count and normal markers of inflammation. All patients had high levels of anti-measles IgG in the CSF and in blood, while patient D also presented anti-measles IgM in blood and in the CSF.
Normally, IgG is not produced intrathecally in any significant amount and most of the IgG found in the CSF is derived peripherally and crosses the blood–brain barrier (BBB). The presence of IgG in the CSF is, therefore, not synonymous with CNS inflammation. However, in a range of neuroinflammatory conditions, most notably MS, a small number of B cell clones in the CNS produce oligoclonal IgG.
Although the serological profile shows a previous measles infection in the absence of vaccination, the high measles IgG titre suggests reinfection or reactivation of the virus.
As shown in Table 5, all the patients had high levels of neutralising antibodies against measles, in both serum and the CSF, quantified by PNRT80 assay.

Table 5.
Laboratory findings.
No measles genome was detected in the blood or CSF specimens, apart from patient C. In this case, the genome was detected in the CSF sample and sequenced for genetic characterisation. The strain responsible for the previous measles infection was found to belong to the MeV genotype B3 and to be 99.8% similar to strains circulating in Italy in 2011 and closely related to the strain named MVi/Harare.ZWE/38.09/1 (MeaNS ID 11292), the year of the suspected exposure. The genome sequence was submitted to the Measles Nucleotide Surveillance database (MeaNS ID 151625).
Brain biopsy was not required for diagnosis in any of our patients as they met the remaining major or minor criteria.
The treatment regimen consisted of IFN-α intrathecal infusions through a Rackham reservoir, started at 100,000 units/m2 and increased to 1 million units/m2 of body surface area (BSA) per day within 5 days; after allowing the patients 2 days’ rest, we administered a dosage of 1 million units/m2 of BSA weekly for 6 months, followed by monthly infusions (Table 6). Patient B was treated with a continuous intrathecal infusion pump after a 5-day regimen loading dose and 6 months of monthly infusion. Three patients were also given oral inosiplex 100 mg/kg daily, while one of them, patient D, received oral ribavirin 20 mg/kg daily for 7 days and an infusion of IVIG and vitamin A 50,000 UI two times a day. Symptomatic neurological therapy was added to control seizures and myoclonus. Patient D received vitamin A supplementation despite the absence of signs suggestive of vitamin deficiency (e.g., xerophthalmia, Bitot’s spots and corneal ulceration), in accordance with the most recent guidelines that suggest this approach in the case of severe and potentially life-threatening complications of MeV infection [14].

Table 6.
Treatment details and follow-up in children with SSPE.
IFN-α therapy was administered for a period ranging from 6 months to 3 years, while adjuvant therapy was continued from 3 months to 3.6 years. Patient D had been recently diagnosed, and after 9 months of therapy he discontinued treatment due to poor family compliance.
As far as side effects related to IFN-α therapy are concerned, we registered fever, lethargy and vomiting during the period 24–48 h after the first IFN-α infusion in 3 out of 4 patients. The fever responded to paracetamol and rapidly disappeared, while vomiting was mild and resolved spontaneously. A side effect related to the Rackham device was documented only in patient D, who was admitted to our department after 3 months of therapy because of a cerebral abscess in the same area as the intraventricular device, which was promptly removed and repositioned. A methicillin-resistant Staphylococcus aureus strain was isolated in the patient, but he improved in terms of clinical conditions with the administration of a wide-spectrum intravenous antibiotic therapy. Three months later, the same therapy regimen was re-administered because of a rapid neurological deterioration.
In the last follow-up, at the age of 12 years, and after 4 years without therapy, patient C was diagnosed with SARS-CoV-2 infection, Omicron variant. His clinical conditions were good, with fever for 4 days, oxygen saturation always at about 99% and no other symptoms. He was administered one dose of monoclonal antibody Sotrovimab 250 mg.
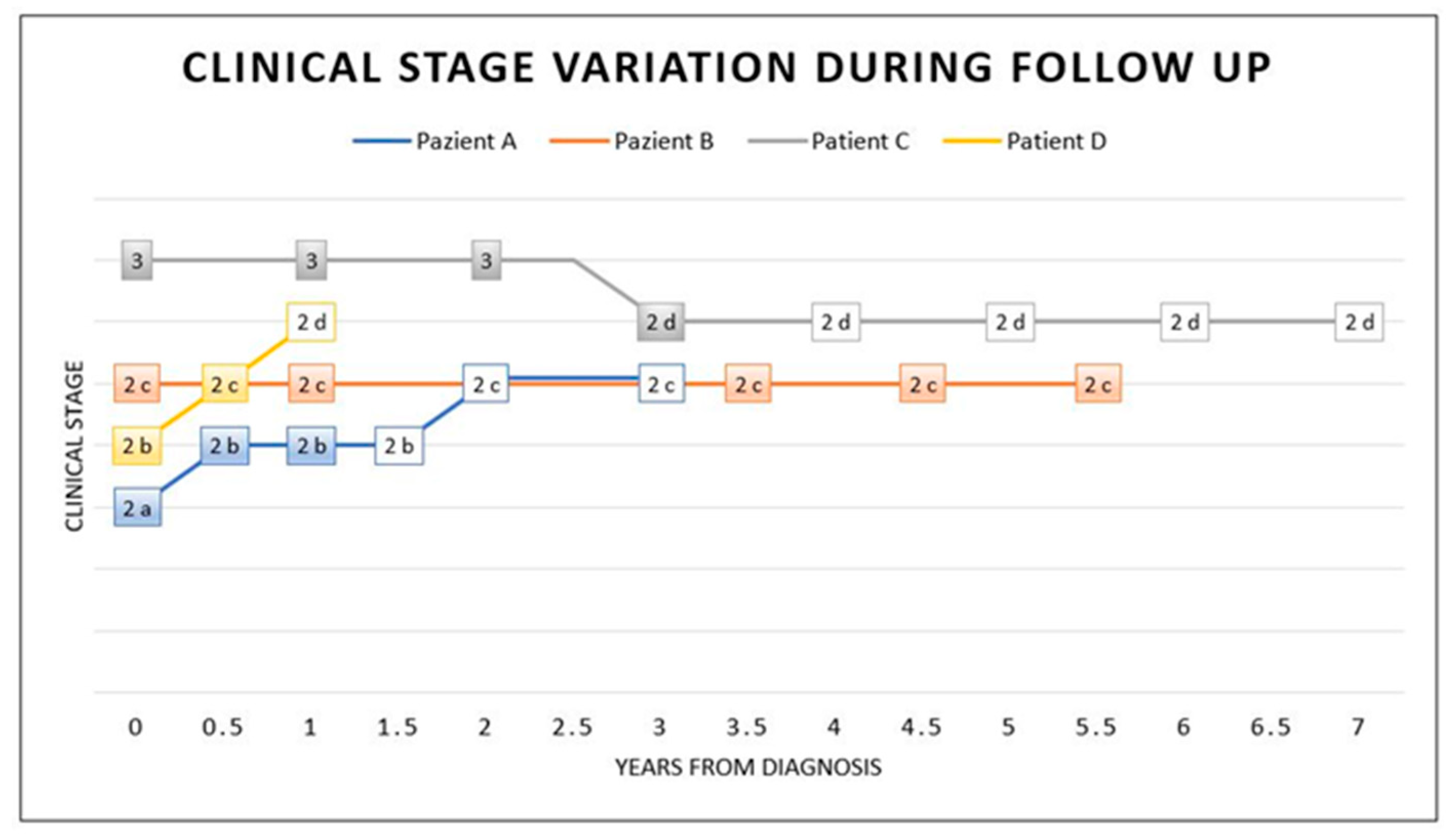
The clinical response to therapy was different in our patients: patient A showed a slow progression, patient B stabilised, patient C improved and patient D showed a progression, probably related to the cerebral abscess, which delayed therapy (Figure 4).
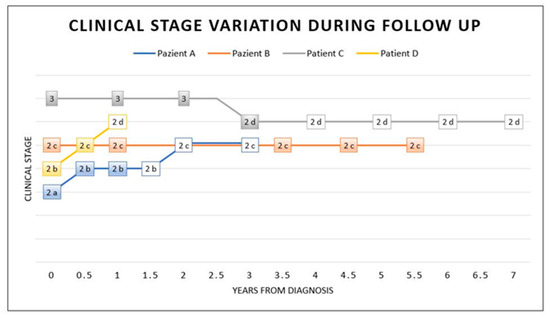
Figure 4.
Stage variation during and after treatment. This graphic shows the clinical stage variation at diagnosis (time 0), during IFN-α therapy (coloured box) and after therapy (white box). Intrathecal IFN-α therapy was considered the main treatment in this graphic (coloured box). As illustrated, at time 0, a diagnosis was made and coincided with the start of the treatment. Patient A took therapy for 1 year. He presented mild clinical progression in the following year, with relative stabilisation in stage 2c, even after 2 years of therapy discontinuation. Patients undergoing IFN-α therapy for a long time (5.5 years for patient B and 3 years for patient C) showed clinical stabilisation (patient B) or a mild improvement with a subsequent stabilisation even after 4 years without therapy (patient C). Patient D took therapy for a short time, with a discontinuation, and he showed clinical progression.
4. Discussion
The neurological sequelae of primary measles encephalitis (PME), acute post-infectious measles encephalomyelitis (APME), measles inclusion body encephalitis (MIBE) and SSPE post MeV are less common than other MeV-related complications and can lead to severe disability or death.
Diagnosis of MeV-related SSPE is based on the characteristic symptomatology, specific electroencephalographic changes and elevated titres of MeV antibodies in the serum and the CSF.
We presented a group of SSPE patients who underwent long-term treatment with INF-α intrathecal 1,000,000 U/m2 once a week and isoprinosine 100 mg/kg/day orally for a mean period of 24 months.
This treatment scheme is the one suggested by the International Consortium on SSPE [54].
Our case series reports an improved response with treatment for a longer duration with the combination of intrathecal INF-α and oral isoprinosine. However, we did not find different results compared to those of the trial by Gascon et al. [54]. In the work of the international consensus, compared to treatment with isoprinosine alone, the combined therapy used for 6 months did not show significant efficacy in terms of morbidity outcomes (improvement, stabilisation, worsening after treatment stopped, deterioration). In the first 6 months of follow-up of our case series, we observed two good clinical responses (patients B and C), confirmed in the last control, in terms of clinical stabilisation (1 year and 4 years without therapy, respectively). These patients were treated for a longer period, of about 3 years, without therapy discontinuation.
Furthermore, the clinical picture showed stability in children who had stopped treatment. Overall, in the last follow-up, ranging from 1 year to 7 years after starting therapy, the best result we detected was a slowdown in disease progression in three out of four patients. Only one patient showed a rapid clinical progression, with neurological deterioration, probably related to the complication of a cerebral abscess causing a discontinuation in therapy. Nevertheless, even when we found a positive response in terms of slow progression or life extension, the quality of life remained low due to the high rate of neurological and neurovegetative morbidity that characterises the disease.
In the recent literature, there is one case report describing the longest follow-up, of 13 years, in a female patient aged 13 years at diagnosis and treated with oral inosiplex for 5 years and intraventricular INF-α for 13 years. The complex positive effect in terms of clinical improvement was mentioned, without many side effects except for one episode of bacterial meningitis [66]. A more detailed study analysed 18 patients treated with at least 12 months of intraventricular INF-α and oral inosiplex, reporting clinical stabilisation or improvement in 44% of the patients [57].
Taking into account the studies available, IFN-α therapy was conducted for a period no longer than 3–12 months, improving clinical conditions in 10% to 36% of the cases. The therapy was administrated in different ways, such as intra-muscular, subcutaneous and intravenous. The follow-up ranged from 6 months to 3 years, with the exception of the above-mentioned case report [54,56,59,67].
Compared with previous case reports on the use of intrathecal INF-α and orally isoprinosine, our study showed that longer treatment duration does not offer improved results except in terms of an increase in survival and disease stabilisation.
Regarding other therapy schemes, a previous study described the treatment of three patients with combined oral isoprinosine and intrathecal ribavirin plus INF-α. The treatment lasted more than 5 years. In this case, the authors found a slowdown in progression in two patients and stability in one patient.
Even early treatment does not lead to a reduction in the stage of the disease. In fact, our patients who started treatment within a few weeks of diagnosis showed final staging levels associated with high disability and morbidity. Already highlighted by other authors, this often leads to younger children with a rapidly progressive disease course from onset [57].
The data in the literature are conflicting regarding the effects of the various treatment protocols used [50,51,53]. In particular, controlled studies are scarce and highlight how the long-term use of antivirals or immunomodulators can be considered in patients with SSPE to obtain benefits in terms of stabilising the disease or increasing survival [51,53,55].
With the exception of data from the international consortium on SSPE, the available data in the literature are contrasting and based on small case series, with different combinations of drugs used and different treatment durations. In particular, little information is available for the paediatric population about a protracted therapy with IFN-α in terms of efficacy and side effects.
WHO recognises vitamin A deficiency as a risk factor for MeV [68]. The effect of vitamin A in MeV may be mediated by the integrity of epithelial cells and the reinforcement of the immune system [69]. However, the role of vitamin A in the pathogenesis of SSPE needs to be clarified, as there are no studies demonstrating efficacy in the treatment of the disease. Only one of our patients (patient D) underwent treatment with vitamin A according to the WHO scheme but without presenting positive changes in the disease stroke compared to the others.
SSPE is a serious disease that, to date, does not have a specific cure capable of leading to a regression of the symptoms or preventing associated disability [70,71]. As of now, vaccination is the only valid weapon to prevent the disease and it should be considered as both an individual and a social duty. In addition, individual patients may have different backgrounds, modalities of onset and clinical courses. This diversity contributes to making diagnosis and treatment of SSPE patients difficult.
Since the MeV vaccine is approved from 12 months of life, the first year is the period the patient is most at risk of a possible infection because of the absence of immunity. In fact, three out of the four cases we presented contracted MeV infection during this time window, when relatives’ vaccination coverage is a matter of major concern. In these cases, SSPE develops with a shorter latency, in line with data in the literature [6]. Moreover, there is a lack of evidence and standardised guidelines about postexposure prophylaxis in children without evidence of immunity. None of our cases underwent immune globulin administration after exposure, which may prevent or modify the clinical course of MeV according to observational studies and meta-analyses [43,44,45].
A recent literature review [72] analysed the current positions as set out in the national guidelines of the United States, Australia, New Zealand, Canada and the United Kingdom, concluding that intravenous immunoglobulin (IVIG) or intra-muscular immunoglobulin (IMIG) could be administered to infants aged less than 6 months as early as possible after exposure.
A systematic review [73] analysed the role of passive immunisation within 7 days of exposure in non-vaccinated people to prevent MeV infection and concluded that treated people had 83% less risk of primary infection than non-treated people. Interestingly, there is no systematic analysis in the literature of the efficacy of post-exposure prophylaxis to prevent long-term complications from MeV, such as SSPE [74,75].
Endo et al. [76], assessing the efficacy of postexposure prophylaxis against MeV with immunoglobulin, found a titre-dependent effect, with higher anti-MeV titres providing the greatest protection. Children who did not develop the disease had received a mean dose of 10.9 IU/kg compared with 5.7 IU/kg for children in whose case post-exposure prophylaxis with IG intramuscular failed.
In none of the SSPE series present in the literature we have data on whether post-exposure prophylaxis prevents SSPE. Data in this area would help to determine whether this could be a valid strategy to reduce the risk of short- and long-term complications associated with MeV. This is an important issue, especially in the COVID-19 era, if we consider that during the pandemic and the lockdown periods, worldwide, there has been a reduction in vaccination campaign compliance, with an increase in the cases of MeV in the paediatric population and therefore a risk that in the near future more SSPE cases will be seen [4,5]
Diagnosis of SSPE is challenging and relies on the detection of increased concentrations of MeV IgG antibodies in the cerebrospinal fluid in a clinically compatible context. The results from serological testing alone are not convincing proof of intrathecal production of MeV IgG. MeV viral copies in the blood or the CSF may be absent [14].
Intrathecal antibody synthesis against the MeV causative antigen must be detected to confirm a diagnosis of SSPE [77]. In this view, the methodology of the exam is relevant because qualitative methods, such as the antibody index (AI), can help detect the intrathecal antibody for a causative antigen as well as a part of a polyspecific immune response. The quantitation of the intrathecal antibody fraction in the CSF with ELISA helps to discriminate both cases [78].
Just the presence of anti-MeV IgG in blood is by no means the basis for a diagnosis because it could be due to a previous vaccination, as, for example, in our patient B. Characteristically, a child with SSPE is one who was always healthy, who contracted or had exposure to a case of MeV and who after a few years suddenly and progressively developed a neurological deterioration with atonic epileptic seizures. In these children, the EEG with typical periodic complexes already adds a strong suspicion of SSPE. In these patients, it is mandatory to perform a lumbar puncture to determine anti-MeV Ig in the CSF. A possible aim of future SPPE research would be to investigate whether children who had contracted MeV prior to vaccination could then be screened for the risk of developing SSPE by studying the susceptibility genes (MxA, interleukin-4 and interferon genes, INF-α) we discussed above.
The lack of a blood marker also supports the hypothesis that SSPE is associated with an inflammation state localised only in the CNS, giving a reason for intrathecal target therapy. Results from the genetic analysis of the MeV genome in one of the reported cases confirm that the strain identified was similar to the strains that circulated at the moment of infection.
To the best of our knowledge, this is the first case report on SSPE with long therapy courses and the longest follow-up.
5. Conclusions
In conclusion, many steps are still needed to achieve the goal of eradicating measles. Considering the insufficient data on post-exposure prophylaxis efficacy and the limitations of available therapies, the only concrete strategy against measles and related complications is prevention through vaccination. The COVID-19 pandemic has led to dangerous immunity gaps resulting from suspended awareness campaigns and delayed immunisation activities, leading to medical concern over the risk of new measles outbreaks in the coming years. The relentless and fatal course of SSPE that we have highlighted in this study underscores the importance of vaccination programs and demands urgent action to reverse a pending measles catastrophe that will affect children and public health.
Author Contributions
L.P., diagnosis, clinical and neuroradiological evaluations of patients, conceptualization, writing—original draft, supervision, validation; M.E.A. and L.S., writing—original draft preparation, writing—review and editing; A.C., F.G., C.E.M. and C.G.N., visualization; M.B., A.M., P.B., E.D., M.K., S.G., writing—review and editing; F.U., G.S., M.A.N.F., G.M., R.M., formal analysis; F.V. and M.V., supervision and validation; F.M., writing—original draft preparation, supervision and validation. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and the Bambino Gesù Children’s Hospital Ethical committee.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available upon reasonable request from the corresponding author.
Acknowledgments
The authors wish to thank Roberto Giuseppetti of the WHO/LabNet National Reference Laboratory at the ISS for technical support and Alessia Caratelli, Valentina Cecchetti and Daniela Casale at the DMI of ISS for technical and administrative support.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Campbell, H.; Andrews, N.; Brown, K.E.; Miller, E. Review of the effect of measles vaccination on the epidemiology of SSPE. Int. J. Epidemiol. 2007, 36, 1334–1348. [Google Scholar] [CrossRef] [PubMed]
- Alexander, J.P.; Patel, M.; Goodson, J.; Alexander, J. Progress Toward Regional Measles Elimination—Worldwide, 2000–2019. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 1700–1705. [Google Scholar] [CrossRef]
- Phadke, V.K.; Bednarczyk, R.A.; Omer, S.B.; Omer, S. Vaccine Refusal and Measles Outbreaks in the US. JAMA 2020, 324, 1344–1345. [Google Scholar] [CrossRef] [PubMed]
- Durrheim, D.N.; Andrus, J.K.; Tabassum, S.; Bashour, H.; Githanga, D.; Pfaff, G. A dangerous measles future looms beyond the COVID-19 pandemic. Nat. Med. 2021, 27, 360–361. [Google Scholar] [CrossRef]
- White, J.M.; McDonald, H.; Tessier, E.; White, J. Early impact of the coronavirus disease (COVID-19) pandemic and physical distancing measures on routine childhood vaccinations in England, January to April 2020. Eurosurveillance 2020, 25, 2000848. [Google Scholar] [CrossRef]
- Garg, R.K.; Mahadevan, A.; Malhotra, H.S.; Rizvi, I.; Kumar, N.; Uniyal, R. Subacute sclerosing panencephalitis. Rev. Med. Virol. 2019, 29, e2058. [Google Scholar] [CrossRef]
- Miller, C.; Farrington, C.P.; Harbert, K. The epidemiology of subacute sclerosing panencephalitis in England and Wales 1970–1989. Int. J. Epidemiol. 1992, 21, 998–1006. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jafri, S.K.; Kumar, R.; Ibrahim, S. Subacute sclerosing panencephalitis—Current perspectives. Pediatr. Health Med. Ther. 2018, 9, 67–71. [Google Scholar] [CrossRef]
- Lawrence, D.M.; Patterson, C.E.; Gales, T.L.; D’Orazio, J.L.; Vaughn, M.M.; Rall, G.F. Measles virus spread between neurons requires cell contact but not CD46 expression, syncytium formation, or extracellular virus production. J. Virol. 2000, 74, 1908–1918. [Google Scholar] [CrossRef] [PubMed]
- Magurano, F.; Marella, G.L.; Marchi, A.; Filia, A.; Marsella, L.T.; Potenza, S.; Massa, R.; Bucci, P.; Baggieri, M.; Nicoletti, L. A case of fulminant subacute sclerosing panencephalitis presenting with acute myoclonic-astatic epilepsy. Ann. Ist. Super. Sanita 2017, 53, 167–169. [Google Scholar] [CrossRef] [PubMed]
- Budka, H.; Lassmann, H.; Popow-Kraupp, T. Measles virus antigen in panencephalitis. An immunomorphological study stressing dendritic involvement in SSPE. Acta Neuropathol. 1982, 56, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Allen, I.V.; McQuaid, S.; McMahon, J.; Kirk, J.; McConnell, R. The significance of measles virus antigen and genome distribution in the CNS in SSPE for mechanisms of viral spread and demyelination. J. Neuropathol. Exp. Neurol. 1996, 55, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Plumb, J.; Duprex, W.P.; Stewart Cameron, C.H.; Richter-Landsberg, C.; Talbot, P.; McQuaid, S. Infection of human oligodendroglioma cells by a recombinant measles virus expressing enhanced green fluorescent protein. J. Neurovirol. 2002, 8, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Hübschen, J.M.; Gouandjika-Vasilache, I.; Dina, J. Measles. Lancet 2022, 399, 678–690. [Google Scholar] [CrossRef]
- World Health Organization. Update of the nomenclature for describing the genetic characteristics of wild-type measles viruses: New genotypes and reference strains. Wkly. Epidemiol. Rec. 2003, 78, 229–232. [Google Scholar]
- Rima, B.K.; Earle, J.A.P.; Yeo, R.P.; Herlihy, L.; Baczko, K.; Ter Meulen, V.; Carabana, J.; Caballero, M.; Celma, M.L.; Fernandez-Munoz, R. Temporal and geographical distribution of measles virus genotypes. J. Gen. Virol. 1995, 76, 1173–1180. [Google Scholar] [CrossRef]
- Barrero, P.R.; Grippo, J.; Viegas, M.; Mistchenko, A.S. Wild-type Measles Virus in Brain Tissue of Children with Subacute Sclerosing Panencephalitis, Argentina. Emerg. Infect. Dis. 2003, 9, 1333. [Google Scholar] [CrossRef]
- Baczko, K.; Lampe, J.; Liebert, U.G.; Brinckmann, U.; ter Meulen, V.; Pardowitz, I.; Budka, H.; Cosby, S.L.; Isserte, S.; Rima, B.K. Clonal expansion of hypermutated measles virus in a SSPE brain. Virology 1993, 197, 188–195. [Google Scholar] [CrossRef]
- Baricevic, M.; Forcic, D.; Santak, M.; Mazuran, R. A comparison of complete untranslated regions of measles virus genomes derived from wild-type viruses and SSPE brain tissues. Virus Genes 2007, 35, 17–27. [Google Scholar] [CrossRef]
- Barrero, P.R.; Zandomeni, R.O.; Mistchenko, A.S. Measles virus circulation in Argentina: 1991–1999. Arch. Virol. 2001, 146, 815–823. [Google Scholar] [CrossRef]
- Shirogane, Y.; Takemoto, R.; Suzuki, T.; Kameda, T.; Nakashima, K.; Hashiguchi, T.; Yanagi, Y. CADM1 and CADM2 Trigger Neuropathogenic Measles Virus-Mediated Membrane Fusion by Acting in cis. J. Virol. 2021, 95, e00528-21. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, S.; Shirogane, Y.; Sato, Y.; Hashiguchi, T.; Yanagi, Y. New Insights into Measles Virus Brain Infections. Trends Microbiol. 2019, 27, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Ning, X.; Ayata, M.; Kimura, M.; Komase, K.; Furukawa, K.; Seto, T.; Ito, N.; Shingai, M.; Matsunaga, I.; Yamano, T.; et al. Alterations and diversity in the cytoplasmic tail of the fusion protein of subacute sclerosing panencephalitis virus strains isolated in Osaka, Japan. Virus Res. 2002, 86, 123–131. [Google Scholar] [CrossRef]
- Schmid, A.; Spielhofer, P.; Cattaneo, R.; Baczko, K.; Ter Meulen, V.; Billeter, M.A. Subacute sclerosing panencephalitis is typically characterized by alterations in the fusion protein cytoplasmic domain of the persisting measles virus. Virology 1992, 188, 910–915. [Google Scholar] [CrossRef]
- Brunel, J.; Kweder, H.H.; Ainouze, M.M.; Brunel, J.J. Measles Virus: Identification in the M Protein Primary Sequence of a Potential Molecular Marker for Subacute Sclerosing Panencephalitis. Adv. Virol. 2015, 2015, 769837. [Google Scholar] [CrossRef]
- Watanabe, S.; Shirogane, Y.; Suzuki, S.O.; Ikegame, S.; Koga, R.; Yanagi, Y. Mutant fusion proteins with enhanced fusion activity promote measles virus spread in human neuronal cells and brains of suckling hamsters. J. Virol. 2013, 87, 2648–2659. [Google Scholar] [CrossRef]
- Watanabe, S.; Ohno, S.; Shirogane, Y.; Suzuki, S.O.; Koga, R.; Yanagi, Y. Measles virus mutants possessing the fusion protein with enhanced fusion activity spread effectively in neuronal cells, but not in other cells, without causing strong cytopathology. J. Virol. 2015, 89, 2710–2717. [Google Scholar] [CrossRef]
- Mathieu, C.; Bovier, F.T.; Ferren, M.; Lieberman, N.A.P.; Predella, C.; Lalande, A.; Peddu, V.; Lin, M.J.; Addetia, A.; Patel, A.; et al. Molecular Features of the Measles Virus Viral Fusion Complex That Favor Infection and Spread in the Brain. MBio 2021, 12, e00799-21. [Google Scholar] [CrossRef]
- Shirogane, Y.; Watanabe, S.; Yanagi, Y. Cooperation between different RNA virus genomes produces a new phenotype. Nat. Commun. 2012, 3, 1235. [Google Scholar] [CrossRef]
- Tahara, M.; Takeda, M.; Yanagi, Y. Altered interaction of the matrix protein with the cytoplasmic tail of hemagglutinin modulates measles virus growth by affecting virus assembly and cell-cell fusion. J. Virol. 2007, 81, 6827–6836. [Google Scholar] [CrossRef]
- Angius, F.; Smuts, H.; Rybkina, K.; Stelitano, D.; Eley, B.; Wilmshurst, J.; Ferren, M.; Lalande, A.; Mathieu, C.; Moscona, A.; et al. Analysis of a Subacute Sclerosing Panencephalitis Genotype B3 Virus from the 2009–2010 South African Measles Epidemic Shows That Hyperfusogenic F Proteins Contribute to Measles Virus Infection in the Brain. J. Virol. 2019, 93, e01700-18. [Google Scholar] [CrossRef]
- Cathomen, T.; Naim, H.Y.; Cattaneo, R. Measles viruses with altered envelope protein cytoplasmic tails gain cell fusion competence. J. Virol. 1998, 72, 1224–1234. [Google Scholar] [CrossRef]
- Gascon, G.G.; Frosch, M.P. Case records of the Massachusetts General Hospital. Weekly clinicopathological exercises. Case 15-1998. A 34-year-old woman with confusion and visual loss during pregnancy. N. Engl. J. Med. 1998, 338, 1448–1456. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Watanabe, S.; Fukuda, Y.; Hashiguchi, T.; Yanagi, Y.; Ohno, S. Cell-to-Cell Measles Virus Spread between Human Neurons Is Dependent on Hemagglutinin and Hyperfusogenic Fusion Protein. J. Virol. 2018, 92, e02166-17. [Google Scholar] [CrossRef] [PubMed]
- Makhortova, N.R.; Askovich, P.; Patterson, C.E.; Gechman, L.A.; Gerard, N.P.; Rall, G.F. Neurokinin-1 enables measles virus trans-synaptic spread in neurons. Virology 2007, 362, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, T.; Matsuishi, T.; Iwamoto, R.; Handa, K.; Yoshioka, H.; Kato, H.; Ueda, S.; Hara, H.; Tabira, T.; Mekada, E. Elevated levels of anti-CD9 antibodies in the cerebrospinal fluid of patients with subacute sclerosing panencephalitis. J. Infect. Dis. 2002, 185, 1346–1350. [Google Scholar] [CrossRef] [PubMed]
- Takemoto, R.; Suzuki, T.; Hashiguchi, T.; Yanagi, Y.; Shirogane, Y. Short-Stalk Isoforms of CADM1 and CADM2 Trigger Neuropathogenic Measles Virus-Mediated Membrane Fusion by Interacting with the Viral Hemagglutinin. J. Virol. 2022, 96, e00528-21. [Google Scholar] [CrossRef] [PubMed]
- Generous, A.R.; Harrison, O.J.; Troyanovsky, R.B.; Mateo, M.; Navaratnarajah, C.K.; Donohue, R.C.; Pfaller, C.K.; Alekhina, O.; Sergeeva, A.P.; Indra, I.; et al. Trans-endocytosis elicited by nectins transfers cytoplasmic cargo, including infectious material, between cells. J. Cell Sci. 2019, 132, jcs235507. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, J.; Issacson, R.S.; Koppel, B.S.; Koppel, B. Subacute sclerosing panencephalitis: An update. Dev. Med. Child Neurol. 2010, 52, 901–907. [Google Scholar] [CrossRef] [PubMed]
- Torisu, H.; Kusuhara, K.; Kira, R.; Bassuny, W.M.; Sakai, Y.; Sanefuji, M.; Takemoto, M.; Hara, T. Functional MxA promoter polymorphism associated with subacute sclerosing panencephalitis. Neurology 2004, 62, 457–460. [Google Scholar] [CrossRef] [PubMed]
- Nakao, F.; Inoue, T.; Kira, R.; Nakao, F. Contribution of the interleukin 4 gene to susceptibility to subacute sclerosing panencephalitis. Arch. Neurol. 2002, 59, 822–827. [Google Scholar] [CrossRef]
- Gadoth, N.; Kott, E.; Levin, S.; Hahn, T. The interferon system in subacute sclerosing panencephalitis and its response to isoprinosine. Brain Dev. 1989, 11, 308–312. [Google Scholar] [CrossRef]
- Anlar, B.; Yalaz, K.; Öktem, F.; Köse, G. Long-term follow-up of patients with subacute sclerosing panencephalitis treated with intraventricular α-interferon. Neurology 1997, 48, 526–528. [Google Scholar] [CrossRef]
- Oldstone, M.B.A.; Dales, S.; Tishon, A.; Lewicki, H.; Martin, L. A role for dual viral hits in causation of subacute sclerosing panencephalitis. J. Exp. Med. 2005, 202, 1185–1190. [Google Scholar] [CrossRef] [PubMed]
- Jabbour, J.T.; Garcia, J.H.; Lemmi, H.; Ragland, J.; Duenas, D.A.; Sever, J.L. Subacute sclerosing panencephalitis. A multidisciplinary study of eight cases. JAMA 1969, 207, 2248–2254. [Google Scholar] [CrossRef]
- Mekki, M.; Eley, B.; Hardie, D.; Wilmshurst, J.M. Subacute sclerosing panencephalitis: Clinical phenotype, epidemiology, and preventive interventions. Dev. Med. Child Neurol. 2019, 61, 1139–1144. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, M.M.; Weitman, S.D.; Winick, N.J.; Bellini, W.J.; Timmons, C.F.; Siegel, J.D. Subacute measles encephalitis in the young immunocompromised host: Report of two cases diagnosed by polymerase chain reaction and treated with ribavirin and review of the literature. Clin. Infect. Dis. 1993, 16, 654–660. [Google Scholar] [CrossRef]
- Dyken, P.R.; Swift, A.; Durant, R.H. Long-term follow-up of patients with subacute sclerosing panencephalitis treated with inosiplex. Ann. Neurol. 1982, 11, 359–364. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.E.; Huttenlocher, P.R.; Dyken, P.R.; Jabbour, J.T.; Maxwell, K.W. Inosiplex therapy in subacute sclerosing panencephalitis. A multicentre, non-randomised study in 98 patients. Lancet 1982, 1, 1034–1037. [Google Scholar] [CrossRef]
- Haddad, F.S.; Risk, W.S. Isoprinosine treatment in 18 patients with subacute sclerosing panencephalitis: A controlled study. Ann. Neurol. 1980, 7, 185–188. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, K.; Hashimoto, K.; Suyama, K.; Sato, M.; Abe, Y.; Watanabe, M.; Kanno, S.; Maeda, H.; Kawasaki, Y.; Hosoya, M. Maintaining Concentration of Ribavirin in Cerebrospinal Fluid by a New Dosage Method; 3 Cases of Subacute Sclerosing Panencephalitis Treated Using a Subcutaneous Continuous Infusion Pump. Pediatr. Infect. Dis. J. 2019, 38, 496–499. [Google Scholar] [CrossRef] [PubMed]
- Hosoya, M.; Mori, S.; Tomoda, A.; Mori, K.; Sawaishi, Y.; Kimura, H.; Shigeta, S.; Suzuki, H. Pharmacokinetics and effects of ribavirin following intraventricular administration for treatment of subacute sclerosing panencephalitis. Antimicrob. Agents Chemother. 2004, 48, 4631–4635. [Google Scholar] [CrossRef]
- Hosoya, M.; Shigeta, S.; Mori, S.; Tomoda, A.; Shiraishi, S.; Miike, T.; Suzuki, H. High-dose intravenous ribavirin therapy for subacute sclerosing panencephalitis. Antimicrob. Agents Chemother. 2001, 45, 943–945. [Google Scholar] [CrossRef]
- Gascon, G.G. International Consortium on Subacute Sclerosing Panencephalitis; Panencephalitis, International Consortium 439 on Subacute Sclerosing. Randomized treatment study of inosiplex versus combined inosiplex and intraventricular interferon-440 alpha in subacute sclerosing panencephalitis (SSPE): International multicenter study. J. Child Neurol. 2003, 18, 819–827. [Google Scholar] [CrossRef] [PubMed]
- Tomoda, A.; Shiraishi, S.; Hosoya, M.; Hamada, A.; Miike, T. Combined treatment with interferon-alpha and ribavirin for subacute sclerosing panencephalitis. Pediatr. Neurol. 2001, 24, 54–59. [Google Scholar] [CrossRef]
- Faruk Aydin, Ö.; Şenbil, N.; Kuyucu, N.; Gürer, Y.K.Y. Combined treatment with subcutaneous interferon-alpha, oral isoprinosine, and lamivudine for subacute sclerosing panencephalitis. J. Child Neurol. 2003, 18, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Gascon, G.; Yamani, S.; Crowell, J.; Stigsby, B.; Nester, M.; Kanaan, I.; Jallu, A. Combined oral isoprinosine-intraventricular alpha-interferon therapy for subacute sclerosing panencephalitis. Brain Dev. 1993, 15, 346–355. [Google Scholar] [CrossRef]
- Sliva, J.; Pantzartzi, C.N.; Votava, M. Inosine Pranobex: A Key Player in the Game Against a Wide Range of Viral Infections and Non-Infectious Diseases. Adv. Ther. 2019, 36, 1878–1905. [Google Scholar] [CrossRef] [PubMed]
- Tomoda, A.; Nomura, K.; Shiraishi, S.; Hamada, A.; Ohmura, T.; Hosoya, M.; Miike, T.; Sawaishi, Y.; Kimura, H.; Takashima, H.; et al. Trial of intraventricular ribavirin therapy for 431 subacute sclerosing panencephalitis in Japan. Brain Dev. 2003, 25, 514–517. [Google Scholar] [CrossRef]
- Hashimoto, K.; Hosoya, M.; Hosoya, M. Advances in Antiviral Therapy for Subacute Sclerosing Panencephalitis. Molecules 2021, 26, 427. [Google Scholar] [CrossRef] [PubMed]
- Ohya, T.; Yamashita, Y.; Shibuya, I.; Hara, M.; Nagamitsu, S.; Kaida, H.; Kurata, S.; Ishibashi, M.; Matsuishi, T. A serial 18FDG-436 PET study of a patient with SSPE who had good prognosis by combination therapy with interferon alpha and ribavirin. Eur. J. Paediatr. Neurol. 2014, 18, 536–539. [Google Scholar] [CrossRef]
- Takahashi, T.; Hosoya, M.; Kimura, K.; Ohno, K.; Mori, S.; Takahashi, K.; Shigeta, S. The cooperative effect of interferon-α and ribavirin on subacute sclerosing panencephalitis (SSPE) virus infections, in vitro and in vivo. Antivir. Res. 1998, 37, 29–35. [Google Scholar] [CrossRef]
- Magurano, F.; Baggieri, M.; Marchi, A.; Rezza, G.; Nicoletti, L.; Eleonora, B.; Concetta, F.; Stefano, F.; Maedeh, K.; Paola, B.; et al. SARS-CoV-2 infection: The environmental endurance of the virus can be influenced by the increase of temperature. Clin. Microbiol. Infect. 2021, 27, 289.e5–289.e7. [Google Scholar] [CrossRef]
- Magurano, F.; Baggieri, M.; Fortuna, C.; Bella, A.; Filia, A.; Rota, M.C.; Benedetti, E.; Bucci, P.; Marchi, A.; Nicoletti, L. Measles elimination in Italy: Data from laboratory activity, 2011–2013. J. Clin. Virol. 2015, 64, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Pınar, A.; Tuncer Kurne, A.; Lay, İ.; Acar, N.P.; Karahan, S.; Karabudak, R.; Akbıyık, F. Cerebrospinal fluid oligoclonal banding patterns and intrathecal immunoglobulin synthesis: Data comparison from a wide patient group. Neurol. Sci. Neurophysiol. 2018, 35, 21–28. [Google Scholar] [CrossRef]
- Yum, M.S.; Kwak, M.; Yeh, H.; Yum, M. A long-term subacute sclerosing panencephalitis survivor treated with intraventricular interferon-alpha for 13 years. Korean J. Pediatr. 2019, 62, 108–112. [Google Scholar] [CrossRef]
- Anlar, B.; Aydin, O.F.; Guven, A.; Sonmez, F.M.; Kose, G.; Herguner, O. Retrospective evaluation of interferon-beta treatment in subacute sclerosing panencephalitis. Clin. Ther. 2004, 26, 1890–1894. [Google Scholar] [CrossRef]
- D’Souza, R.; D’Souza, R. Vitamin A for treating measles in children. Cochrane Database Syst. Rev. 2001. [Google Scholar] [CrossRef]
- Gungor, S.; Olmez, A.; Firat, P.A.; Haliloǧlu, G.; Anlar, B. Serum retinol and beta-carotene levels in subacute sclerosing panencephalitis. J. Child Neurol. 2007, 22, 341–343. [Google Scholar] [CrossRef]
- Arciuolo, R.J.; Jablonski, R.R.; Zucker, J.R.; Rosen, J.B. Effectiveness of Measles Vaccination and Immune Globulin Post- Exposure Prophylaxis in an Outbreak Setting-New York City, 2013. Clin. Infect. Dis. 2017, 65, 1843–1847. [Google Scholar] [CrossRef]
- Barrabeig, I.; Rovira, A.; Rius, C.; Muñoz, P.; Soldevila, N.; Batalla, J.; Domínguez, A. Effectiveness of measles vaccination for control of exposed children. Pediatr. Infect. Dis. J. 2011, 30, 78–80. [Google Scholar] [CrossRef] [PubMed]
- Young, M.K. The indications and safety of polyvalent immunoglobulin for post-exposure prophylaxis of hepatitis A, rubella and measles. Hum. Vaccin. Immunother. 2019, 15, 2060–2065. [Google Scholar] [CrossRef] [PubMed]
- Young, M.K.; Nimmo, G.R.; Cripps, A.W.; Jones, M.A. Post-exposure passive immunisation for preventing measles. Cochrane Database Syst. Rev. 2014. [Google Scholar] [CrossRef] [PubMed]
- Temte, J.L.; McLean, H.Q.H.; Fiebelkorn, A.P.A.; Temte, J.L.J. Prevention of measles, rubella, congenital rubella syndrome, and mumps, 2013: Summary recommendations of the Advisory Committee on Immunization Practices (ACIP). Morb. Mortal. Wkly. Rep. Recomm. Rep. 2022, 62, 1–34. [Google Scholar]
- Ferren, M.; Horvat, B.; Mathieu, C. Measles encephalitis: Towards new therapeutics. Viruses 2019, 11, 1017. [Google Scholar] [CrossRef]
- Endo, A.; Izumi, H.; Miyashita, M.; Taniguchi, K.; Okubo, O.; Harada, K. Current efficacy of postexposure prophylaxis against measles with immunoglobulin. J. Pediatr. 2001, 138, 926–928. [Google Scholar] [CrossRef]
- Reiber, H.; Lange, P. Quantification of virus-specific antibodies in cerebrospinal fluid and serum: Sensitive and specific detection of antibody synthesis in brain. Clin. Chem. 1991, 37, 1153–1160. [Google Scholar] [CrossRef]
- Jacobi, C.; Lange, P.; Reiber, H.; Reiber, H. Quantitation of intrathecal antibodies in cerebrospinal fluid of subacute sclerosing panencephalitis, herpes simplex encephalitis and multiple sclerosis: Discrimination between microorganism-driven and polyspecific immune response. J. Neuroimmunol. 2007, 187, 139–146. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).