Journal Description
Colloids and Interfaces
Colloids and Interfaces
is an international, peer-reviewed, open access journal on colloids and interfaces chemistry published bimonthly online by MDPI.
- Open Access— free for readers, no limits on space and color.
- High Visibility: indexed within Scopus, ESCI (Web of Science), CAPlus / SciFinder, Inspec, and other databases.
- Journal Rank: CiteScore - Q2 (Chemistry (miscellaneous))
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 27.6 days after submission; acceptance to publication is undertaken in 4.6 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
2.4 (2022);
5-Year Impact Factor:
2.9 (2022)
Latest Articles
A Review of Investigations and Applications of Biocides in Nanomaterials and Nanotechnologies
Colloids Interfaces 2024, 8(3), 31; https://doi.org/10.3390/colloids8030031 - 16 May 2024
Abstract
In recent years, the development of nanomaterials with biocidal properties has received considerable attention due to their potential applications in various industries, including food, medicine, and cultural heritage preservation. The growing demand for coatings with antibacterial properties has sparked interest from industrial sectors
[...] Read more.
In recent years, the development of nanomaterials with biocidal properties has received considerable attention due to their potential applications in various industries, including food, medicine, and cultural heritage preservation. The growing demand for coatings with antibacterial properties has sparked interest from industrial sectors in exploring the incorporation of biocides into these materials. Coatings are prone to microbial growth, which can cause damage such as cracking, discoloration, and staining. To combat these problems, the integration of biocides into coatings is a crucial strategy. Biocide-embedded nanomaterials offer numerous advantages, including high efficiency in small quantities, ease of application, good chemical stability, low toxicity, and non-bioaccumulation. Encapsulated nanobiocides are particularly attractive to the agro-industry, because they can be less toxic than traditional biocides while still effectively controlling microbial contamination. To fully exploit the benefits of nanobiocides, future research should focus on optimizing their synthesis, formulation, and delivery methods. The purpose of this review is to summarize the current status of biocide nanomaterials, discuss potential future research directions, and highlight research methods, the development of new forms of nanomaterials, and studies of their physico-chemical properties. Biocide nanocapsules of DCOIT (4,5-Dichloro-2-octyl-2H-isothiazol-3-one) are chosen as an example to illustrate the research pathways.
Full article
(This article belongs to the Special Issue Exclusive Papers of the Editorial Board Members of Colloids and Interfaces 2024)
►
Show Figures
Open AccessArticle
Effect of Asphaltenes and Asphaltene Dispersants on Wax Precipitation and Treatment
by
Oualid M’barki, John Clements and Quoc P. Nguyen
Colloids Interfaces 2024, 8(3), 30; https://doi.org/10.3390/colloids8030030 - 14 May 2024
Abstract
A detailed understanding of the interactions between wax and asphaltenes with other components of crude oils and the effect of treatments with paraffin inhibitors (PIs) and asphaltene dispersants (ADs), with a focus on identifying specific structure-activity relationships, is necessary to develop effective flow
[...] Read more.
A detailed understanding of the interactions between wax and asphaltenes with other components of crude oils and the effect of treatments with paraffin inhibitors (PIs) and asphaltene dispersants (ADs), with a focus on identifying specific structure-activity relationships, is necessary to develop effective flow assurance strategies. The morphological and rheological consequences of treating wax and asphaltenes in oils of differing composition with a series of ADs having structural features in common with an alpha olefin-maleic anhydride (AO-MA) comb-like copolymer PI were assessed alone and in combination with said PI. Of the four ADs studied, two were identified as being effective dispersants of asphaltenes in heptane-induced instability tests and in a West Texas (WT) crude. The degree to which a low concentration of asphaltenes stabilizes wax in the absence of treatment additives is lessened in oils having greater aromatic fractions. This is because these stabilizing interactions are replaced by more energetically favorable aromatic–asphaltene interactions, increasing oil viscosity. Treatment with AD alone also reduces the extent of wax–asphaltene interactions, increasing oil viscosity. In concert with the PI, treatment with the AD having greater structural similarity with the PI appears to improve wax solubility in both the presence and absence of asphaltenes. However, the viscosity of the treated oils is greater than that of the oil treated with PI alone, while treatment with AD having lesser structural similarity with the PI does not adversely affect oil viscosity. These data suggest that rather than treating both wax and asphaltenes, AD may poison the function of the PI. These data illuminate the pitfalls of designing flow assurance additives to interact with both wax and asphaltenes and developing treatment plans.
Full article
(This article belongs to the Special Issue Crude Oil Recovery)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Vesicle Morphogenesis in Amphiphilic Triblock Copolymer Solutions
by
Senyuan Liu, Mohammad Sadegh Samie and Radhakrishna Sureshkumar
Colloids Interfaces 2024, 8(3), 29; https://doi.org/10.3390/colloids8030029 - 6 May 2024
Abstract
►▼
Show Figures
Coarse-grained molecular dynamics simulations are employed to investigate the spatiotemporal evolution of vesicles (polymersomes) through the self-assembly of randomly distributed amphiphilic BAB triblock copolymers with hydrophilic A and hydrophobic B blocks in an aqueous solution. The vesiculation pathway consists of several intermediate structures,
[...] Read more.
Coarse-grained molecular dynamics simulations are employed to investigate the spatiotemporal evolution of vesicles (polymersomes) through the self-assembly of randomly distributed amphiphilic BAB triblock copolymers with hydrophilic A and hydrophobic B blocks in an aqueous solution. The vesiculation pathway consists of several intermediate structures, such as an interconnected network of copolymer aggregates, a cage of cylindrical micelles, and a lamellar cage. The cage-to-vesicle transition occurs at a constant aggregation number and practically eliminates the hydrophobic interfacial area between the B block and solvent. Molecular reorganization underlying the sequence of morphology transitions from a cage-like aggregate to a vesicle is nearly isentropic. The end-to-end distances of isolated copolymer chains in solution and those within a vesicular assembly follow lognormal probability distributions. This can be attributed to the preponderance of folded chain configurations in which the two hydrophobic end groups of a given chain stay close to each other. However, the probability distribution of end-to-end distances is broader for chains within the vesicle as compared with that of a single chain. This is due to the swelling of the folded configurations within the hydrophobic bilayer. Increasing the hydrophobicity of the B block reduces the vesiculation time without qualitatively altering the self-assembly pathway.
Full article

Graphical abstract
Open AccessArticle
Water-Based Bi2S3 Nano-Inks Obtained with Surfactant-Assisted Liquid Phase Exfoliation and Their Direct Processing into Thin Films
by
Micaela Pozzati, Felix Boll, Matteo Crisci, Sara Domenici, Francesco Scotognella, Bernd Smarsly, Teresa Gatti and Mengjiao Wang
Colloids Interfaces 2024, 8(3), 28; https://doi.org/10.3390/colloids8030028 - 30 Apr 2024
Abstract
►▼
Show Figures
Bi2S3 has gained considerable attention as a semiconductor for its versatile functional properties, finding application across various fields, and liquid phase exfoliation (LPE) serves as a straightforward method to produce it in nano-form. Till now, the commonly used solvent for
[...] Read more.
Bi2S3 has gained considerable attention as a semiconductor for its versatile functional properties, finding application across various fields, and liquid phase exfoliation (LPE) serves as a straightforward method to produce it in nano-form. Till now, the commonly used solvent for LPE has been N-Methyl-2-pyrrolidone, which is expensive, toxic and has a high boiling point. These limitations drive the search for more sustainable alternatives, with water being a promising option. Nonetheless, surfactants are necessary for LPE in water due to the hydrophobic nature of Bi2S3, and organic molecules with amphoteric characteristics are identified as suitable surfactants. However, systematic studies on the use of ionic surfactants in the LPE of Bi2S3 have remained scarce until now. In this work, we used sodium dodecyl sulfate (SDS), sodium dodecylbenzene sulfonate (SDBS) and sodium hexadecyl sulfonate (SHS) as representative species and we present a comprehensive investigation into their effects on the LPE of Bi2S3. Through characterizations of the resulting products, we find that all surfactants effectively exfoliate Bi2S3 into few-layer species. Notably, SDBS demonstrates superior stabilization of the 2D layers compared to the other surfactants, while SHS becomes the most promising surfactant for obtaining products with high yield. Moreover, the resulting nano-inks are used for fabricating films using spray-coating, reaching a fine tuning of band gap by controlling the number of cycles, and paving the way for the utilization of 2D Bi2S3 in optoelectronic devices.
Full article

Figure 1
Open AccessArticle
Rheological Behaviors and Fractional Viscoelastic Modeling of Glucopone (APG)/Water/Hydrocarbons Solutions
by
Mohamed A. Siddig
Colloids Interfaces 2024, 8(3), 27; https://doi.org/10.3390/colloids8030027 - 24 Apr 2024
Abstract
The aims of this work are to study the rheological behaviors of a microemulsion of Glucopone–water–hydrocarbon systems and to use a fractional model to describe several experimental results of these systems. Four different types of hydrocarbons were considered. The frequency dependent storage, G
[...] Read more.
The aims of this work are to study the rheological behaviors of a microemulsion of Glucopone–water–hydrocarbon systems and to use a fractional model to describe several experimental results of these systems. Four different types of hydrocarbons were considered. The frequency dependent storage, G′, and loss modulus, G″, were investigated below the critical strain. The critical strain was found to decrease as the alkane chain lengths increased, while the opposite behavior was observed for zero shear viscosity, η0. Most of the microemulsions exhibited stable elastic fluid behavior (G′ > G″) below 10 rad s−1 angular frequency. For all systems, elastic modulus values were found to be greater than loss modulus in the frequency range studied, indicating more elastic behaviors. Shear-thinning behaviors were observed, and the complex viscosity decreased with an increase in hydrocarbon chain lengths. The effects of hydrocarbon types on the rheological behaviors were more profound in the dodecane systems which showed maximum values of G′ and η0. The Friedrich–Braun model was introduced and was used to describe several experimental results on Alkyl polyglocoside solutions. Fractional rheology successfully described the viscoelastic phenomena in Glucopone surfactant solutions and the comparisons between the experimental results and the theoretical predictions were found to be satisfactory.
Full article
(This article belongs to the Special Issue Rheology of Complex Fluids and Interfaces)
►▼
Show Figures
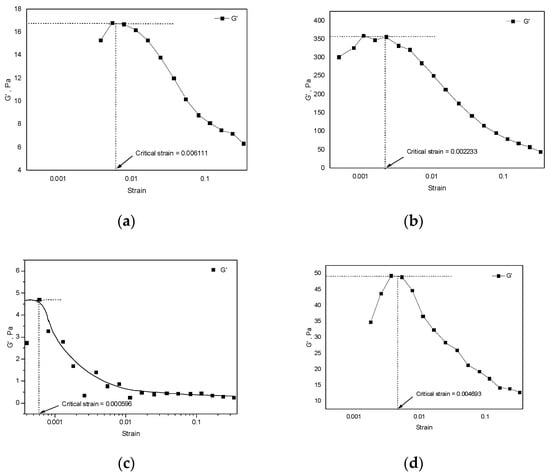
Figure 1
Open AccessArticle
Caffeine Adsorption on a Thermally Modified Bentonite: Adsorbent Characterization, Experimental Design, Equilibrium and Kinetics
by
Javier A. Quintero-Jaramillo, Javier Ignacio Carrero and Nancy R. Sanabria-González
Colloids Interfaces 2024, 8(2), 26; https://doi.org/10.3390/colloids8020026 - 15 Apr 2024
Abstract
►▼
Show Figures
Caffeine is a chemical compound found in various products such as coffee, tea, and energy drinks; therefore, it is common in wastewater and surface water. The present study investigated caffeine adsorption on a thermally modified bentonite-type clay. The effects of the heat treatment
[...] Read more.
Caffeine is a chemical compound found in various products such as coffee, tea, and energy drinks; therefore, it is common in wastewater and surface water. The present study investigated caffeine adsorption on a thermally modified bentonite-type clay. The effects of the heat treatment of the adsorbent over the temperature range of 60–500 °C, as well as the initial pH of the solution, stirring speed, and contact time, on the removal of caffeine were analyzed. The adsorbent was characterized by XRF, XRD, FT–IR, thermal analysis (TGA–DSC), and N2 physisorption at 77 K. The response surface methodology (RSM) based on a central composite design (CCD) was used to evaluate and optimize the adsorption of caffeine in aqueous solution. The maximum adsorption capacity of caffeine obtained with the Langmuir model was 80.3 ± 2.1 mg/g (0.41 ± 0.01 mmol/g) at 25 °C under equilibrium conditions (initial pH = 8.0, stirring speed = 400 rpm, contact time = 120 min). A kinetic study showed that the pseudo-second-order and Elovich models adequately describe the adsorption process. Bentonite thermally modified at 400 °C can be considered a low-cost adsorbent with potential application for removing caffeine in aqueous media.
Full article
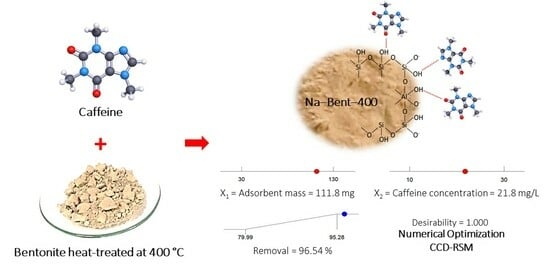
Graphical abstract
Open AccessArticle
Adsorption Layer Properties and Foam Behavior of Aqueous Solutions of Whey Protein Isolate (WPI) Modified by Vacuum Cold Plasma (VCP)
by
Elham Ommat Mohammadi, Samira Yeganehzad, Mohammad Ali Hesarinejad, Mohsen Dabestani, Regine von Klitzing, Reinhard Miller and Emanuel Schneck
Colloids Interfaces 2024, 8(2), 25; https://doi.org/10.3390/colloids8020025 - 9 Apr 2024
Abstract
For years, cold plasma processing has been used as a non-thermal technology in industries such as food. As interfacial properties of protein play a remarkable role in many processes, this study investigates the effect of cold plasma on the foaming and interfacial behavior
[...] Read more.
For years, cold plasma processing has been used as a non-thermal technology in industries such as food. As interfacial properties of protein play a remarkable role in many processes, this study investigates the effect of cold plasma on the foaming and interfacial behavior of WPI. The objective of this study is to evaluate the effect of different gases (air, 1:1 argon–air mixture, and sulfur hexafluoride (SF6)) used in low-pressure cold plasma (VCP) treatments of whey protein isolate (WPI) on the surface and foaming behavior of aqueous WPI solutions. Dynamic surface dilational elasticity, surface tension isotherms, surface layer thickness, and the foamability and foam stability were investigated in this study. VCP treatment did not significantly affect the adsorption layer thickness. However, an increase in induction time, surface pressure equilibrium value, and aggregated size is observed after SF6VCP treatment, which can be attributed to the reaction of WPI with the reactive SF6 species of the cold plasma. The surface dilational elastic modulus increased after VCP treatment, which can be related to the increased mechanical strength of the protein layer via sulfonation and aggregate formation. VCP treatment of WPI increases the foam stability, while the average diameter of foam bubbles and liquid drainage in the foam depends on the gas used for the cold plasma.
Full article
(This article belongs to the Special Issue Proteins at the Interface)
►▼
Show Figures
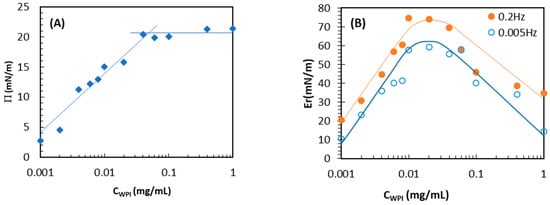
Figure 1
Open AccessArticle
In Vitro Gastrointestinal Release of Chlorogenic Acid and Curcumin Co-Encapsulated in Double Emulsions with the Outer Interface Stabilized by Cellulose Nanocrystals
by
Javier Paredes-Toledo, Javier Herrera, Paulo Díaz-Calderón, Paz Robert and Begoña Giménez
Colloids Interfaces 2024, 8(2), 24; https://doi.org/10.3390/colloids8020024 - 9 Apr 2024
Abstract
A Pickering double emulsion (DE) with an outer (O:W2) interface stabilized by cellulose nanocrystals (DE-CNC) was designed as a co-delivery systems for chlorogenic acid (CA) and curcumin, then compared with a control DE emulsion with an O:W2 interface stabilized with
[...] Read more.
A Pickering double emulsion (DE) with an outer (O:W2) interface stabilized by cellulose nanocrystals (DE-CNC) was designed as a co-delivery systems for chlorogenic acid (CA) and curcumin, then compared with a control DE emulsion with an O:W2 interface stabilized with sodium caseinate (DE-NaCas). DE-CNC was more resistant to creaming during storage (6.79%, day 42) and showed higher encapsulation efficiency (EE) of CA (>90%). Conversely, both DEs exhibited similarly high EE for curcumin (>97%). The ζ-potential values were highly negative in both DEs, but tended to be lower in DE-CNC due to the highly negative charge of the CNCs. DE-CNC allowed for a steady release of CA during the oral, gastric, and intestinal phases of digestion, while a total release of CA was already observed in the gastric phase in case of DE-NaCas. The bioaccessibility of CA was similar in both DEs (~57–58%). Curcumin was mainly released in the intestinal phase with both DEs, reaching slightly lower bioaccessibility values with DE-CNC. The use of CNCs as a stabilizer for the outer interface of DEs is a promising strategy to increase the stability and EE of these systems, providing oral co-delivery vehicles capable of releasing significantly bioactive compounds during the intestinal phase of digestion.
Full article
(This article belongs to the Special Issue Recent Advances on Emulsions and Applications: Volume II)
►▼
Show Figures
Figure 1
Open AccessArticle
Oil/Brine Screening for Improved Fluid/Fluid Interactions during Low-Salinity Water Flooding
by
Jose Villero-Mandon, Peyman Pourafshary and Masoud Riazi
Colloids Interfaces 2024, 8(2), 23; https://doi.org/10.3390/colloids8020023 - 1 Apr 2024
Abstract
Low-salinity water flooding/smart water flooding (LSWF/SWF) are used for enhanced oil recovery (EOR) because of the improved extraction efficiency. These methods are more environmentally friendly and in many scenarios more economical for oil recovery. They are proven to increase recovery factors (RFs) by
[...] Read more.
Low-salinity water flooding/smart water flooding (LSWF/SWF) are used for enhanced oil recovery (EOR) because of the improved extraction efficiency. These methods are more environmentally friendly and in many scenarios more economical for oil recovery. They are proven to increase recovery factors (RFs) by between 6 and 20%, making LSWF/SWF technologies that should be further evaluated to replace conventional water flooding or other EOR methods. Fluid/fluid interaction improvements include interfacial tension (IFT) reduction, viscoelastic behavior (elastic properties modification), and microemulsion generation, which could complement the main mechanisms, such as wettability alteration. In this research, we evaluate the importance of fluid/fluid mechanisms during LSWF/SWF operations. Our study showed that a substantial decrease in IFT occurs when the oil asphaltene content is in the range of 0% to 3 wt.%. An IFT reduction was observed at low salinity (0–10,000 ppm) and a specific oil composition condition. Optimal IFT occurs at higher divalent ion concentrations when oil has low asphaltene content. For the oil with high asphaltene content, the sulfates concentration controls the IFT alteration. At high asphaltene concentrations, the formation of micro-dispersion is not effective to recover oil, and only a 5% recovery factor improvement was observed. The presence of asphaltene at the oil/low-salinity brine interface increases the energy required to disrupt it, inducing significant changes in the elastic moduli. In cases of low asphaltene content, the storage modulus demonstrates optimal performance at higher divalent concentrations. Conversely, at high asphaltene concentrations, the dominant factors to control the interface are paraffin content and temperature.
Full article
(This article belongs to the Special Issue Crude Oil Recovery)
►▼
Show Figures
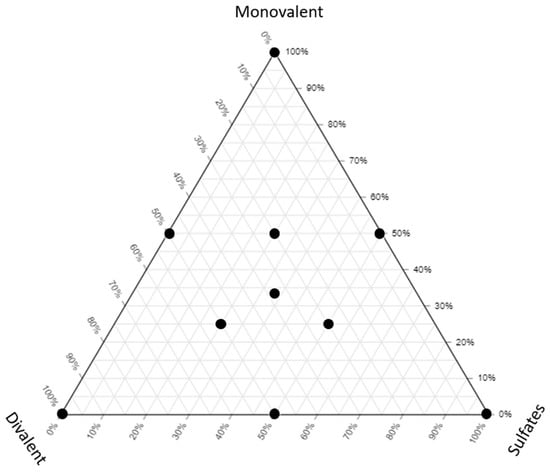
Figure 1
Open AccessArticle
Shrimp Oil-Enriched Mayonnaise Prepared Using Fish Myofibrillar Protein as a Substitute for Egg Yolk: Physical, Rheological, and Sensory Properties
by
Bharathipriya Rajasekaran, Avtar Singh, Bin Zhang, Hui Hong, Thummanoon Prodpran and Soottawat Benjakul
Colloids Interfaces 2024, 8(2), 22; https://doi.org/10.3390/colloids8020022 - 18 Mar 2024
Abstract
The effect of SO (shrimp oil) at various levels (5, 10, and 15%) on the stability of mayonnaise was investigated. Droplet size (d32 and d43), polydispersity index, and microstructure results showed an upsurge in droplet sizes with augmenting level of
[...] Read more.
The effect of SO (shrimp oil) at various levels (5, 10, and 15%) on the stability of mayonnaise was investigated. Droplet size (d32 and d43), polydispersity index, and microstructure results showed an upsurge in droplet sizes with augmenting level of SO in mayonnaise (5 to 15%) (p < 0.05). SO imparted a bright orange color to the mayonnaise as evidenced by increased a* and b* values with lower L* values (p < 0.05). Moreover, the impact of a fish myofibrillar protein (FMP) substitution for egg yolk (0, 25, 50, 75%) in mayonnaise containing SO (5% and 10%) was also studied. Increasing the level of FMP substitution in SO-added mayonnaise showed a dilution effect and reduced a* and b* values (p < 0.05). In addition, excessive FMP substitution up to 75% drastically increased centrifugal and thermal creaming indices, indicating lowered stability (p < 0.05). Nevertheless, with the augmenting FMP substitutions, the viscosity, texture, and rheological properties in mayonnaise became lower (p < 0.05). However, there were no differences in overall acceptability scores between 5% SO-added mayonnaise with 25% FMP substitution (SO5:FMP25) and 5% SO-added mayonnaise without FMP substitution (SO5:FMP0) (p > 0.05). A confocal laser scanning microscopic (CLSM) study revealed a smaller droplet and less aggregation in the SO5:FMP0 sample, compared to SO5:FMP25. The incorporation of SO and FMP substitution yielded the resulting mayonnaise, which met the requirements of a healthy food since SO is rich in PUFA and the replacement of egg yolk by FMP can contribute several health benefits. The incorporation of SO as well as FMP as substitution for egg yolk therefore has potential in the development of functional foods.
Full article
(This article belongs to the Special Issue Food Colloids II)
►▼
Show Figures

Figure 1
Open AccessArticle
Theoretical and Experimental Determinations of the Hydrophilic–Lipophilic Balance (HLB) of Representative Oils and Lecithins
by
Gary Smejkal, Vera Gross and Alexander Lazarev
Colloids Interfaces 2024, 8(2), 21; https://doi.org/10.3390/colloids8020021 - 16 Mar 2024
Abstract
The hydrophilic–lipophilic balance (HLB) is a valuable parameter used to determine the relative hydrophobicity of a compound based on its chemical structure. This semi-empirical parameter has been instrumental in formulating oil-in-water and water-in-oil emulsions using well-characterized ingredients with known HLB values. However, recent
[...] Read more.
The hydrophilic–lipophilic balance (HLB) is a valuable parameter used to determine the relative hydrophobicity of a compound based on its chemical structure. This semi-empirical parameter has been instrumental in formulating oil-in-water and water-in-oil emulsions using well-characterized ingredients with known HLB values. However, recent trends toward the use of minimally processed “virgin” oils of therapeutic or nutritional value may render the reported “required HLB” values inaccurate. Minimally processed oils can contain numerous compounds at varying or unknown concentrations, rendering the HLB value incalculable. Factors such as regional source, growing season, and processing method contribute to the variability in oil composition. Furthermore, the solubilization of lipophilic bioactives in oils can significantly alter the HLB of the oil phase in a concentration-dependent manner. This complicates the formulation of emulsions, as the HLB values of both the oil phase and emulsifiers must be closely matched to achieve stable formulations. This study presents a simple and efficient experimental method to determine an HLB value of a complex ingredient without resorting to lengthy Design-of-Experiment (DoE) matrices and trial-and-error approaches. The optimal HLB of a formulation can be determined from a series of experiments in which two well-characterized surfactants of known HLB values are mixed at varying proportions with an unknown oil phase, where the HLB of the oil is considered to match the HLB value of the surfactants combined at the proportion at which nanoemulsions with the smallest, most stable oil droplets are formed. Similarly, when the HLB values of the oil phase and other contributing components are precisely known, the unknown HLB of a complex natural surfactant can be calculated. These calculations assist in formulating emulsions efficiently and effectively by ensuring optimal compatibility among all the components.
Full article
(This article belongs to the Special Issue Recent Advances on Emulsions and Applications: Volume II)
►▼
Show Figures

Figure 1
Open AccessArticle
Start-Up Rotation of a Porous Colloidal Sphere in a Cavity
by
Chan W. Yu and Huan J. Keh
Colloids Interfaces 2024, 8(2), 20; https://doi.org/10.3390/colloids8020020 - 11 Mar 2024
Abstract
►▼
Show Figures
The starting rotation of a porous sphere induced by the sudden application of a continuous torque about its diameter at the center of a spherical cavity filled with an incompressible Newtonian fluid at low Reynolds numbers is analyzed. The unsteady Stokes and Brinkman
[...] Read more.
The starting rotation of a porous sphere induced by the sudden application of a continuous torque about its diameter at the center of a spherical cavity filled with an incompressible Newtonian fluid at low Reynolds numbers is analyzed. The unsteady Stokes and Brinkman equations governing the fluid velocities outside and inside the porous particle, respectively, are solved via the Laplace transform, and an explicit formula of its dynamic angular velocity as a function of the pertinent parameters is obtained. The behavior of the start-up rotation of an isolated porous particle and the cavity wall effect on the particle rotation are interesting. The angular velocity of the particle grows incessantly over time from an initial zero to its final value, while the angular acceleration declines with time continuously. In general, the transient angular velocity is an increasing function of the porosity of the particle. A porous sphere with higher fluid permeability rotates at higher angular velocity and acceleration relative to the reference particle at any elapsed time but lags behind the reference particle in the percentage growth of angular velocity towards the respective terminal values. The transient angular velocity decreases with increasing particle-to-cavity radius ratio, but it is not a sensitive function of the radius ratio when the resistance to fluid flow inside the porous particle or the radius ratio itself is small.
Full article

Figure 1
Open AccessArticle
Dynamic Interfacial Properties and Foamability of DoTAB/SiO2 Mixtures
by
Fariza Amankeldi, Maratbek Gabdullin, Miras Issakhov, Zhanar Ospanova, Altynay Sharipova, Saule Aidarova and Reinhard Miller
Colloids Interfaces 2024, 8(2), 19; https://doi.org/10.3390/colloids8020019 - 11 Mar 2024
Abstract
The interaction between nanoparticles and cationic surfactants is an exciting and emerging field in interfacial science. This area of research holds significant promise, linking fundamental principles to practical applications in a variety of industries, including chemical processes, biomedical applications and the petroleum industry.
[...] Read more.
The interaction between nanoparticles and cationic surfactants is an exciting and emerging field in interfacial science. This area of research holds significant promise, linking fundamental principles to practical applications in a variety of industries, including chemical processes, biomedical applications and the petroleum industry. This study explores the interaction between dodecyltrimethylammonium bromide (DoTAB) and silica (SiO2) nanoparticles, investigating their influence on dynamic interfacial properties and foam characteristics. Through equilibrium and dynamic surface tension measurements, along with examining the dilational visco-elasticity behavior, this research reveals the complex surface behavior of DoTAB/SiO2 mixtures compared to individual surfactant solutions. The foamability and stability experiments indicate that the addition of SiO2 significantly improves the foam stability. Notably, stable foams are achieved at low SiO2 concentrations, suggesting a cost-effective approach to enhancing the foam stability. This study identifies the optimal stability conditions for 12 mM DoTAB solutions, emphasizing the crucial role of the critical aggregation concentration region. These findings offer valuable insights for designing surfactant-nanoparticle formulations to enhance foam performance in various industrial applications.
Full article
(This article belongs to the Special Issue Exclusive Papers of the Editorial Board Members of Colloids and Interfaces 2024)
►▼
Show Figures
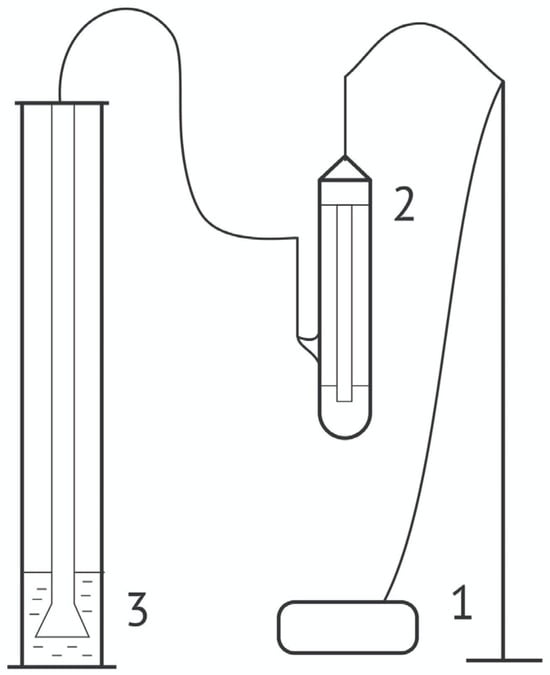
Figure 1
Open AccessArticle
The Synthesis and Reactivity of Mesoporous and Surface-Rough Vinyl-Containing ORMOSIL Nanoparticles
by
Nathan I. Walton, Eric M. Brozek, Courtney C. Gwinn and Ilya Zharov
Colloids Interfaces 2024, 8(2), 18; https://doi.org/10.3390/colloids8020018 - 7 Mar 2024
Abstract
►▼
Show Figures
Silica nanoparticles synthesized solely from organosilanes naturally possess a greater number of organic functionalities than silica nanoparticles surface-modified with organosilanes. We report the synthesis of organically modified silica (ORMOSIL) nanoparticles with a mesoporous and surface-rough morphology and with a high surface area, made
[...] Read more.
Silica nanoparticles synthesized solely from organosilanes naturally possess a greater number of organic functionalities than silica nanoparticles surface-modified with organosilanes. We report the synthesis of organically modified silica (ORMOSIL) nanoparticles with a mesoporous and surface-rough morphology and with a high surface area, made solely from vinyltrimethoxy silane. We chemically modified these vinyl silica nanoparticles using bromination and hydroboration, and demonstrated the high accessibility and reactivity of the vinyl groups with an ~85% conversion of the functional groups for the bromination of both particle types, a ~60% conversion of the functional groups for the hydroboration of surface-rough particles and a 90% conversion of the functional groups for the hydroboration of mesoporous particles. We determined that the mesoporous vinyl silica nanoparticles, while having a surface area that lies between the non-porous and surface-rough vinyl silica nanoparticles, provide the greatest accessibility to the vinyl groups for boronation and allow for the incorporating of up to 3.1 × 106 B atoms per particle, making the resulting materials attractive for boron neutron capture therapy.
Full article

Figure 1
Open AccessArticle
Influence of Surface Roughness on Interfacial Properties of Particle Networks
by
Elton L. Correia, Nick Brown, Dimitrios V. Papavassiliou and Sepideh Razavi
Colloids Interfaces 2024, 8(2), 17; https://doi.org/10.3390/colloids8020017 - 4 Mar 2024
Abstract
►▼
Show Figures
The behavior of colloidal particles near fluid interfaces has attracted significant scientific interest, as particles minimize the contact area between the two fluid phases, stabilizing interfacial systems. This study explores the influence of surface roughness on the properties of particle monolayers at the
[...] Read more.
The behavior of colloidal particles near fluid interfaces has attracted significant scientific interest, as particles minimize the contact area between the two fluid phases, stabilizing interfacial systems. This study explores the influence of surface roughness on the properties of particle monolayers at the air–water interface, focusing on colloidal silica particles and fumed silica particles of similar hydrodynamic diameter. This research involves comparing low-surface-area (LSA) and medium-surface-area (MSA) fumed silica particles with spherical colloidal silica particles (250 nm in diameter). Utilizing a Langmuir trough, the interfacial particle networks are compressed and expanded. Analysis of surface pressure isotherms reveals that fumed silica particle monolayers form networks at a lower particle surface coverage compared to spherical particles. The spherical particle monolayer exhibits a higher apparent surface elasticity, indicating greater resistance to the applied compression compared to fumed silica networks. Additionally, monolayers formed by fumed silica particles display hysteresis even after successive compressions and expansions due to irreversible particle interlocking and the formation of multilayered aggregates. These findings provide insights into the impact of surface roughness on the behavior of particle monolayers at fluid interfaces, offering valuable information for designing and optimizing mechanisms involved in emulsion and foam stabilization.
Full article
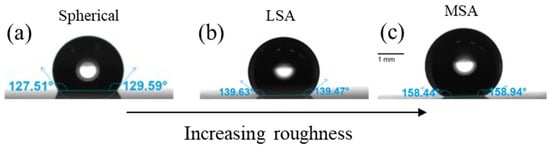
Figure 1
Open AccessFeature PaperArticle
Ionic Strength Effect in the Equilibrium and Rheological Behavior of an Amphiphilic Triblock Copolymer at the Air/Solution Interface
by
Carlo Carbone, Eduardo Guzmán, Julia Maldonado-Valderrama, Ramón G. Rubio and Francisco Ortega
Colloids Interfaces 2024, 8(2), 16; https://doi.org/10.3390/colloids8020016 - 1 Mar 2024
Abstract
This study investigates the effect of an inert salt (NaCl) on the equilibrium interfacial tension and dilatational modulus of Pluronic F-68 copolymer, a triblock copolymer consisting of two terminal blocks of poly(ethylene oxide) and a less hydrophilic central block of poly(propylene oxide). Interfacial
[...] Read more.
This study investigates the effect of an inert salt (NaCl) on the equilibrium interfacial tension and dilatational modulus of Pluronic F-68 copolymer, a triblock copolymer consisting of two terminal blocks of poly(ethylene oxide) and a less hydrophilic central block of poly(propylene oxide). Interfacial tension measurements were carried out using a surface force balance and a drop shape tensiometer, while rheological measurements were carried out in two different frequency ranges. This involved the use of the oscillatory barrier/droplet method and electrocapillary wave measurements, complemented by an appropriate theoretical framework. This work aimed to elucidate the influence of NaCl on the interfacial behavior of Gibbs monolayers of Pluronic F-68. In addition, this study highlights some of the technical and theoretical limitations associated with obtaining reliable dilatational rheological data at high frequencies (<1 kHz) using electrocapillary wave measurements. The results provide valuable insights into the interplay between salt presence and interfacial properties of Pluronic F-68 and highlight the challenges of obtaining accurate dilatational rheological data under specific measurement conditions.
Full article
(This article belongs to the Special Issue B&D 2023)
►▼
Show Figures

Figure 1
Open AccessCorrection
Correction: Veronico et al. Enhancing Oil-Uptake Efficiency with an Alkyl Polyglycoside–Dodecanol Formulation. Colloids Interfaces 2024, 8, 6
by
Lorenzo Veronico, Giuseppe Colafemmina and Luigi Gentile
Colloids Interfaces 2024, 8(2), 15; https://doi.org/10.3390/colloids8020015 - 29 Feb 2024
Abstract
In the original publication [...]
Full article
(This article belongs to the Special Issue Progress in Surfactants with Low Environmental Impact)
Open AccessArticle
A New Method for Calculating the Hamaker Constant Based on the Hansen Solubility Parameters for Non-Polar Liquids
by
Hiroyuki Ohshima and Shin-ichi Takeda
Colloids Interfaces 2024, 8(2), 14; https://doi.org/10.3390/colloids8020014 - 22 Feb 2024
Abstract
►▼
Show Figures
A simple relationship between the Hamaker constant and the Hansen solubility parameters for non-polar liquids is derived by combining a Hamaker constant/surface tension relationship derived by Israelachvili and a Hansen solubility parameters/surface tension relationship derived by Abbott. With this relationship, one can easily
[...] Read more.
A simple relationship between the Hamaker constant and the Hansen solubility parameters for non-polar liquids is derived by combining a Hamaker constant/surface tension relationship derived by Israelachvili and a Hansen solubility parameters/surface tension relationship derived by Abbott. With this relationship, one can easily estimate the Hamaker constant of non-polar liquids on the basis of the database of the Hansen solubility parameters. This is an entirely new method for calculating the Hamaker constant without recourse to data on the frequency-dependent dielectric permittivity of those substances (which are required for the rigorous Lifshitz theory) and laborious numerical calculations.
Full article

Figure 1
Open AccessArticle
Factors Influencing the Rheology of Methane Foam for Gas Mobility Control in High-Temperature, Proppant-Fractured Reservoirs
by
Aashish T. Parekh, Amit Katiyar and Quoc P. Nguyen
Colloids Interfaces 2024, 8(1), 13; https://doi.org/10.3390/colloids8010013 - 4 Feb 2024
Abstract
Gas-enhanced oil recovery (EOR) through huff-n-puff (HnP) is an important method of recovering oil from fracture-stimulated reservoirs. HnP productivity is hampered by fracture channeling, leading to early gas breakthroughs and gas losses. To mitigate these issues, foam-generating surfactants have been developed as a
[...] Read more.
Gas-enhanced oil recovery (EOR) through huff-n-puff (HnP) is an important method of recovering oil from fracture-stimulated reservoirs. HnP productivity is hampered by fracture channeling, leading to early gas breakthroughs and gas losses. To mitigate these issues, foam-generating surfactants have been developed as a method of reducing injected gas phase mobility and increasing oil recovery. This work investigates foam generation and propagation by a proprietary surfactant blend in high-temperature, high-pressure, high-permeability, and high-shear conditions that simulate the environment of a proppant-packed fracture. Bulk foam tests confirmed the aqueous stability and foaming viability of the surfactant at the proposed conditions. Through several series of floods co-injecting methane gas and the surfactant solution through a proppant pack at residual oil saturation, the effects of several injection parameters on apparent foam viscosity were investigated. The foam exhibited an exceptionally high transition foam quality (>95%) and strong shear-thinning behavior. The foam viscosity also linearly decreased with increasing pressure. Another flood series conducted in an oil-free proppant pack showed that swelling of residual oil had no effect on the apparent foam viscosity and was not the reason for the inversely linear pressure dependency. An additional flood series with nitrogen as the injection gas was completed to see if the hydrophobic attraction between the methane and surfactant tail was responsible for the observed pressure trend, but the trend persisted even with nitrogen. In a previous study, the dependence of foam viscosity on pressure was found to be much weaker with a different foaming surfactant under similar conditions. Thus, a better understanding of this important phenomenon requires additional tests with a focus on the effect of pressure on interfacial surfactant adsorption.
Full article
(This article belongs to the Special Issue Crude Oil Recovery)
►▼
Show Figures

Figure 1
Open AccessFeature PaperArticle
Energetic and Entropic Motifs in Vesicle Morphogenesis in Amphiphilic Diblock Copolymer Solutions
by
Senyuan Liu and Radhakrishna Sureshkumar
Colloids Interfaces 2024, 8(1), 12; https://doi.org/10.3390/colloids8010012 - 4 Feb 2024
Cited by 1
Abstract
►▼
Show Figures
Coarse-grained molecular dynamic simulations are employed to investigate the spatiotemporal evolution of vesicles (polymersomes) via self-assembly of randomly distributed amphiphilic diblock copolymers PB-PEO (Poly(Butadiene)-b-Poly(Ethylene Oxide)) in water. The vesiculation pathway consists of several intermediate structures, such as spherical/rodlike aggregates, wormlike micelles, lamellae, and
[...] Read more.
Coarse-grained molecular dynamic simulations are employed to investigate the spatiotemporal evolution of vesicles (polymersomes) via self-assembly of randomly distributed amphiphilic diblock copolymers PB-PEO (Poly(Butadiene)-b-Poly(Ethylene Oxide)) in water. The vesiculation pathway consists of several intermediate structures, such as spherical/rodlike aggregates, wormlike micelles, lamellae, and cavities. The lamella-to-vesicle transition occurs at a constant aggregation number and is accompanied by a reduction in the solvent-accessible surface area. Simulation predictions are in qualitative agreement with the mechanism of vesicle formation in which the unfavorable hydrophobic interactions between water molecules and polymer segments, along the edge of the lamella, are eliminated at the expense of gaining curvature energy. However, rod–lamella–vesicle transition is accompanied by an increase in copolymer packing density. Hence, the change in the surface area accompanying vesiculation predicted by the simulations is significantly lower than theoretical estimates. Changes in information entropy, quantified by the expectation of the logarithm of the probability distribution function of the segmental stretch parameter s, defined as the difference between the maximum and instantaneous segmental extension, are statistically insignificant along the vesiculation pathway. For rods, lamellae, and polymersomes, s follows a log normal distribution. This is explained based on the configurational dynamics of a single diblock chain in water.
Full article
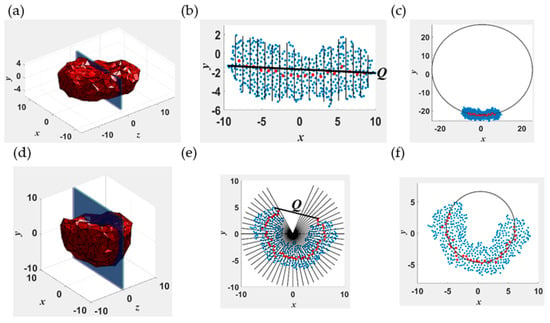
Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Coatings, Materials, Membranes, Nanomaterials, Solar, Colloids and Interfaces, Molecules
Advances in Functional Thin Films
Topic Editors: Ricardo Lopez Anton, Jose Maria De Teresa, Sion Federico Olive MéndezDeadline: 30 September 2024

Conferences
Special Issues
Special Issue in
Colloids and Interfaces
Crude Oil Recovery
Guest Editor: Plamen TchoukovDeadline: 30 June 2024
Special Issue in
Colloids and Interfaces
Recent Advances on Emulsions and Applications: Volume II
Guest Editors: César Burgos-Díaz, Carla Arancibia, Karla A. Garrido-MirandaDeadline: 31 July 2024
Special Issue in
Colloids and Interfaces
Rheology of Complex Fluids and Interfaces
Guest Editors: Cecile Lemaitre, Philippe MarchalDeadline: 31 October 2024
Special Issue in
Colloids and Interfaces
Food Colloids—Volume Three
Guest Editors: Eleni P. Kalogianni, Julia Maldonado-Valderrama, Reinhard MillerDeadline: 20 December 2024






