Force-Induced Visualization of Nucleic Acid Functions with Single-Nucleotide Resolution
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Principle and Demonstration of FIV
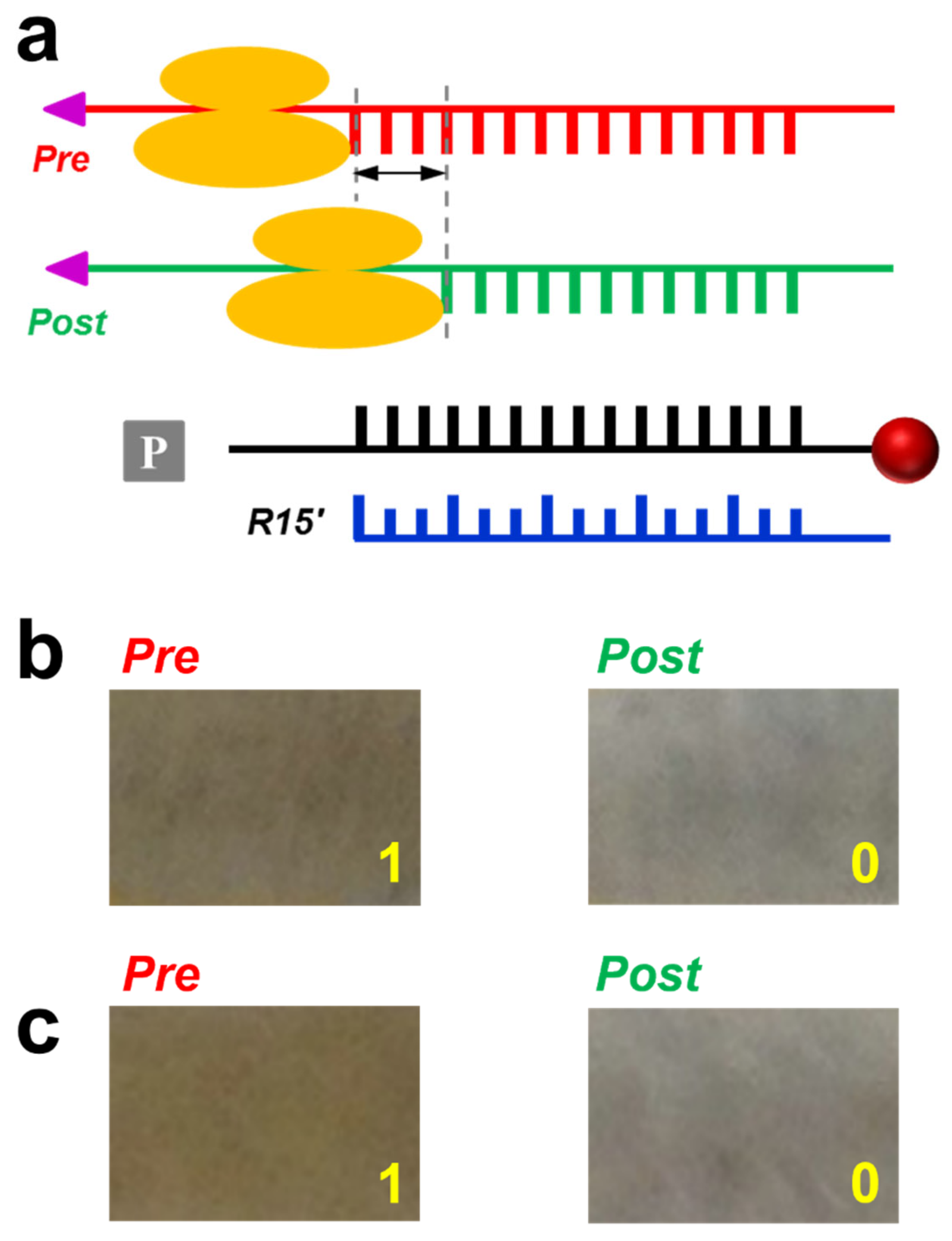
3.2. Application in Probing Ribosome Translocation
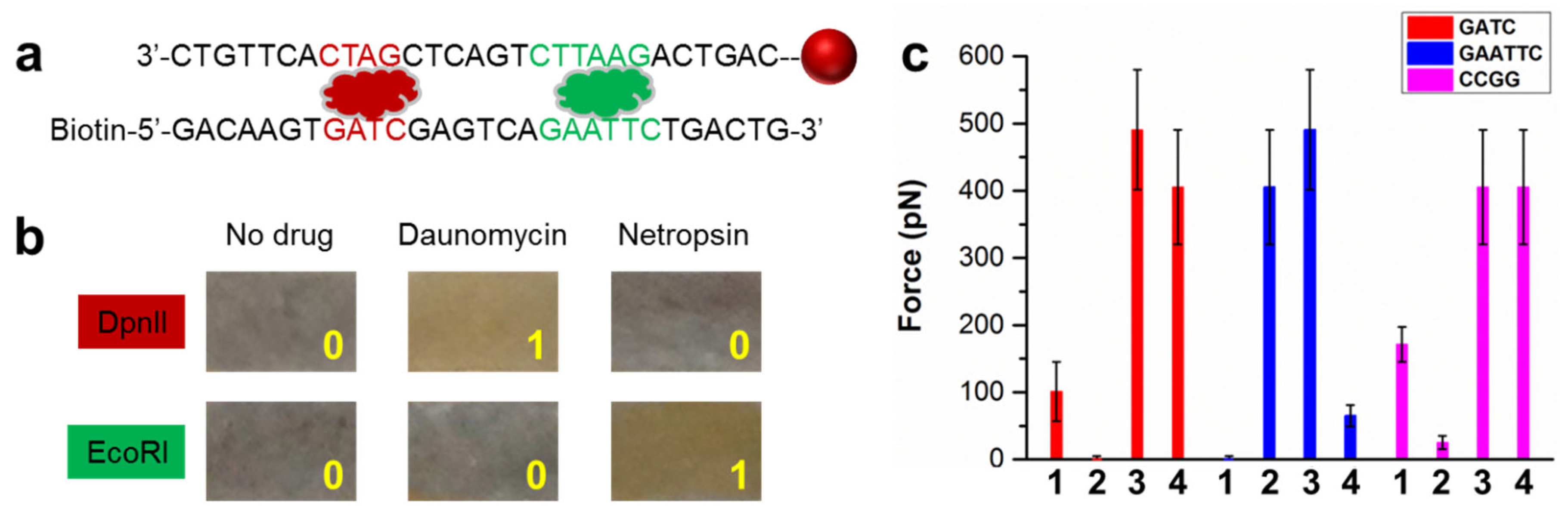
3.3. Application in Sequence-Specific Drug Screening
3.4. A device for Recording and Quantification
3.5. Multiplexed Detection Using Acoustic Radiation Force
4. Discussion
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bloomfield, V.A.; Crothers, D.M.; Tinoco, I., Jr. Nucleic Acids: Structure, Properties, and Functions. In University Science Books: Sausalito; AIP Publishing: Woodbury, NY, USA, 2000; ISBN 0-935702-49-0. [Google Scholar]
- Zhu, Y.; Zhang, M.; Jie, Z.; Tao, S. Nucleic acid testing of SARS-CoV-2: A review of current methods, challenges, and prospects. Front. Microbiol. 2002, 13, 1074289. [Google Scholar] [CrossRef] [PubMed]
- Owczarzy, R.; Moreira, B.G.; You, Y.; Behlke, M.A.; Walder, J.A. Predicting stability of DNA duplexes in solutions containing magnesium and monovalent cations. Biochemistry 2008, 47, 5336–5353. [Google Scholar] [CrossRef]
- He, M.; Xu, X.; Wang, H.; Wu, Q.; Zhang, L.; Zhou, D.; Tong, Y.; Zu, X.; Liu, H. Nanozyme-based colorimetric SARS-CoV-2 nucleic acid detection by naked eye. Small 2023, 19, 2208167. [Google Scholar] [CrossRef]
- Schiffer, J.T.; Aubert, M.; Weber, N.D.; Mintzer, E.; Stone, D.; Jerome, K.R. Targeted DNA mutagenesis for the cure of chronic viral infections. J. Virol. 2012, 86, 8920–8936. [Google Scholar] [CrossRef]
- Palchaudhuri, R.; Hergenrother, P.J. DNA as a target for anticancer compounds: Methods to determine the mode of binding and the mechanism of action. Curr. Opin. Biotechnol. 2007, 18, 497–503. [Google Scholar] [CrossRef]
- Moazed, D.; Noller, H.F. Intermediate states in the movement of transfer RNA in the ribosome. Nature 1989, 342, 142–148. [Google Scholar] [CrossRef]
- Marotta, N.E.; Beavers, K.R.; Bottomley, L.A. Limitations of surface enhanced Raman scattering in sensing DNA hybridization demonstrated by label-free DNA oligos as molecular rulers of distance-dependent enhancement. Anal. Chem. 2013, 85, 1440–1446. [Google Scholar] [CrossRef]
- Sönnichsen, C.; Reinhard, B.M.; Liphardt, J.; Alivisatos, A.P. A molecular ruler based on plasmon coupling of single gold and silver nanoparticles. Nat. Biotechnol. 2005, 22, 741–745. [Google Scholar] [CrossRef]
- Lewis, F.D.; Zhang, L.; Liu, X.; Zuo, X.; Tiede, D.M.; Long, H.; Schatz, G.C. DNA as helical ruler: Exciton-coupled circular dichroism in DNA conjugates. J. Am. Chem. Soc. 2005, 127, 14445–14453. [Google Scholar] [CrossRef]
- Yang, A.H.J.; Hsieh, K.; Patterson, A.S.; Ferguson, B.S.; Eisenstein, M.; Plaxco, K.W.; Soh, H.T. Accurate zygote-specific SNP discrimination using microfluidic electrochemical DNA melt curves. Angew. Chem. Int. Ed. 2014, 53, 3163–3167. [Google Scholar] [CrossRef]
- Chen, S.X.; Seelig, G. An engineered kinetic amplification mechanism for single nucleotide variant discrimination by DNA hybridization probes. J. Am. Chem. Soc. 2016, 138, 5076–5086. [Google Scholar] [CrossRef]
- Steinhauer, C.; Jungmann, R.; Sobey, T.L.; Simmel, F.C.; Tinnefeld, P. DNA origami as a nanoscopic ruler for super-resolution microscopy. Angew. Chem. Int. Ed. 2009, 48, 8870–8873. [Google Scholar] [CrossRef]
- Liu, G.L.; Yin, Y.; Kunchakarra, S.; Mukherjee, B.; Gerion, D.; Jett, S.D.; Bear, D.G.; Gray, J.W.; Alivisatos, A.P.; Lee, L.P.; et al. A nanoplasmonic molecular ruler for measuring nuclease activity and DNA footprinting. Nat. Nanotechnol. 2006, 1, 47–52. [Google Scholar] [CrossRef]
- Howorka, S.; Cheley, S.; Bayley, H. Sequence-specific detection of individual DNA strands using engineered nanopores. Nat. Biotechnol. 2001, 19, 636–639. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Yan, Y.; Zhang, Y.; Zhang, X.; Yin, Y.; Ding, S. DNA transducer-triggered signal switch for visual colorimetric bioanalysis. Sci. Rep. 2015, 5, 11190. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Chen, S.; Ho, J.K.L.; Chieng, C.-C.; Chen, T.-H. Visual detection of nucleic acids based on Mie scattering and the magnetophoretic effect. Analyst 2015, 140, 7876–7885. [Google Scholar] [CrossRef] [PubMed]
- Jenison, R.; Yang, S.; Haeberli, A.; Polisky, B. Interference-based detection of nucleic acid targets on optically coated silicon. Nat. Biotechnol. 2001, 19, 62–65. [Google Scholar] [CrossRef]
- Xia, F.; Zuo, X.; Yang, R.; Xiao, Y.; Kang, D.; Vallée-Bélisle, A.; Gong, X.; Yuen, J.D.; Hsu, B.B.Y.; Heeger, A.J.; et al. Colorimetric detection of DNA, small molecules, proteins, and ions using unmodified gold nanoparticles and conjugated polyelectrolytes. Proc. Natl. Acad. Sci. USA 2010, 107, 10837–10841. [Google Scholar] [CrossRef]
- Lee, A.C.; Ye, J.-S.; Tan, S.N.; Poenar, D.; Sheu, F.-S.; Heng, C.K.; Lim, T.M. Carbon nanotube-based labels for highly sensitive colorimetric and aggregation-based visual detection of nucleic acids. Nanotechnology 2007, 18, 455102. [Google Scholar] [CrossRef]
- Yokota, H.; Sunwoo, J.; Sarikaya, M.; van den Engh, G.; Aebersold, R. Spin-stretching of DNA and protein molecules for detection by fluorescence and atomic force microscopy. Anal. Chem. 1999, 71, 4418–4422. [Google Scholar] [CrossRef]
- Nawaz, A.A.; Soteriou, D.; Xu, C.K.; Goswami, R.; Herbig, M.; Guck, J.; Girardo, S. Image-based cell sorting using focused travelling surface acoustic waves. Lap Chip 2023, 23, 372–387. [Google Scholar] [CrossRef]
- Florin, E.-L.; Moy, V.T.; Gaub, H.E. Adhesion forces between individual ligand-receptor pairs. Science 1994, 264, 415–417. [Google Scholar] [CrossRef]
- Steffens, C.; Leite, F.L.; Bueno, C.C.; Manzoli, A.; Herrmann, P.S.D.P. Atomic force microscopy as a tool applied to nano/biosensors. Sensors 2012, 12, 8278–8300. [Google Scholar] [CrossRef]
- Yangyuoru, P.M.; Dhakal, S.; Yu, Z.; Koirala, D.; Mwongela, S.M.; Mao, H. Single-molecule measurements of the binding between small molecules and DNA aptamers. Anal. Chem. 2012, 84, 5298–5303. [Google Scholar] [CrossRef] [PubMed]
- Bustamante, C.; Bryant, Z.; Smith, S.B. Ten years of tension: Single-molecule DNA mechanics. Nature 2003, 421, 423–427. [Google Scholar] [CrossRef] [PubMed]
- Halvorsen, K.; Wong, W.P. Massively parallel single-molecule manipulation using centrifugal force. Biophys. J. 2010, 98, L53–L55. [Google Scholar] [CrossRef]
- Kamsma, D.; Creyghton, R.; Sitters, G.; Wuite, G.J.L.; Peterman, E.J.G. Tuning the music: Acoustic force spectroscopy (AFS) 2.0. Methods 2016, 105, 26–33. [Google Scholar] [CrossRef] [PubMed]
- De Silva, L.; Yao, L.; Wang, Y.; Xu, S.-J. Well-defined and sequence-specific noncovalent binding forces of DNA. J. Phys. Chem. B 2013, 117, 7554–7558. [Google Scholar] [CrossRef]
- Jia, H.; Tsai, T.-W.; Xu, S.-J. Probing drug-DNA interactions using super-resolution force spectroscopy. Appl. Phys. Lett. 2018, 113, 193702. [Google Scholar] [CrossRef]
- Yao, L.; Li, Y.; Tsai, T.-W.; Xu, S.-J.; Wang, Y. Noninvasive measurement of the mechanical force generated by motor protein EF-G during ribosome translocation. Angew. Chem. Int. Ed. 2013, 52, 14041–14044. [Google Scholar] [CrossRef]
- Hu, Q.; Yang, H.; Wang, Y.; Xu, S.-J. Controlled formation and precise resolution of multiple noncovalent bonds. Chem. Commun. 2016, 52, 3705–3708. [Google Scholar] [CrossRef] [PubMed]
- Visser, E.W.A.; van IJzendoorn, L.J.; Prins, M.W.J. Particle motion analysis reveals nanoscale bond characteristics and enhanced dynamic range for biosensing. ACS Nano 2016, 10, 3093–3101. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Bae, J.H.; Zhang, D.Y. Native characterization of nucleic acid motif thermodynamics via non-covalent catalysis. Nat. Commun. 2016, 7, 10319. [Google Scholar] [CrossRef] [PubMed]
- Sitters, G.; Kamsma, D.; Thalhammer, G.; Ritsch-Marte, M.; Peterman, E.J.G.; Wuite, G.J.L. Acoustic force spectroscopy. Nat. Methods 2015, 12, 47–50. [Google Scholar] [CrossRef] [PubMed]
- Luo, G.-Z.; Wang, F.; Weng, X.; Chen, K.; Hao, Z.; Yu, M.; Deng, X.; Liu, J.; He, C. Charterization of eukaryotic DNA N6-methyladenine by a highly sensitive restrictioon enzyme-assisted sequencing. Nat. Comm. 2016, 7, 11301. [Google Scholar] [CrossRef]
- Lacks, S.A.; Springhorn, S.S. Cloning in Streptococcus pneumoniae of the gene for DpnII DNA methylase. J. Bacteriol. 1984, 157, 934–936. [Google Scholar] [CrossRef]
- Chaires, J. Calorimetry and thermodynamics in drug design. Annu. Rev. Biophys. 2008, 37, 135–151. [Google Scholar] [CrossRef]
- Haq, I. Thermodynamics of drug-DNA interactions. Arch. Biochem. Biophys. 2002, 403, 1–15. [Google Scholar] [CrossRef]
- Doughty, B.; Rao, Y.; Kazer, S.W.; Kwok, S.J.J.; Turro, N.J.; Eisenthal, K.B. Binding of the anti-cancer drug daunomycin to DNA probed by second harmonic generation. J. Phys. Chem. B 2013, 117, 15285–15289. [Google Scholar] [CrossRef]
- McClelland, M.; Belson, M.; Raschke, E. Effect of site-specific modification on restriction endonucleases and DNA modification methyltransferases. Nucleic Acids Res. 1994, 22, 3640–3659. [Google Scholar] [CrossRef]
- Yamaguchi, N.; Fujii, T.; Aoi, S.; Kozuch, P.S.; Hortobagyi, G.N.; Blum, R.H. Comparison of cardiac events associated with liposomal doxorubicin, epirubicin and doxorubicin in breast cancer: A Bayesian network meta-analysis. Eur. J. Cancer 2015, 51, 2314–2320. [Google Scholar] [CrossRef] [PubMed]
- Khasraw, M.; Bell, R.; Dang, C. Epirubicin: Is it like doxorubicin in breast cancer? A clinical review. Breast 2012, 21, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Xu, S.-J. Sequence and chiral selectivity of DNA-drug interactions revealed by force spectroscopy. Angew. Chem. Int. Ed. 2014, 53, 14135–14138. [Google Scholar] [CrossRef] [PubMed]
- Glynne-Jones, P.; Hill, M. Acoustofluidics 23: Acoustic manipulation combined with other force fields. Lab Chip 2013, 13, 1003–1010. [Google Scholar] [CrossRef] [PubMed]
- Brierley, I.; Dos Ramos, F.J. Programmed ribosomal frameshifting in HIV-1 and the SARS-CoV. Virus Res. 2006, 119, 29–42. [Google Scholar] [CrossRef] [PubMed]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, Q.; Jia, H.; Wang, Y.; Xu, S. Force-Induced Visualization of Nucleic Acid Functions with Single-Nucleotide Resolution. Sensors 2023, 23, 7762. https://doi.org/10.3390/s23187762
Hu Q, Jia H, Wang Y, Xu S. Force-Induced Visualization of Nucleic Acid Functions with Single-Nucleotide Resolution. Sensors. 2023; 23(18):7762. https://doi.org/10.3390/s23187762
Chicago/Turabian StyleHu, Qiongzheng, Haina Jia, Yuhong Wang, and Shoujun Xu. 2023. "Force-Induced Visualization of Nucleic Acid Functions with Single-Nucleotide Resolution" Sensors 23, no. 18: 7762. https://doi.org/10.3390/s23187762



