Synthesis and Characterization of Anti-HER2 Antibody Conjugated CdSe/CdZnS Quantum Dots for Fluorescence Imaging of Breast Cancer Cells
Abstract
:1. Introduction
2. Results and Discussion
2.1. Surface Modification of QDs with Glutathione (GSH)
2.2. Characterization of GSH-QDs
2.2.1. Fluorescence quantum yields
2.2.2. Hydrodynamic size and dispersibility of QDs
2.2.3. Apparent molecular weights of GSH-QDs
2.2.4. Stability of GSH-QDs in aqueous solution
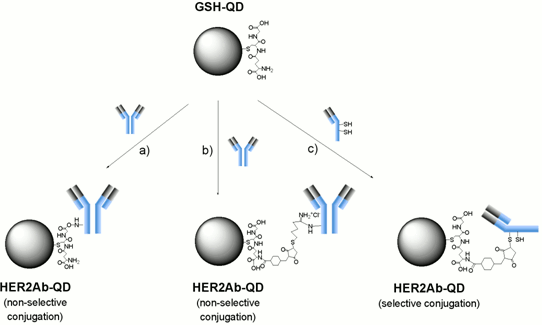
2.3. Conjugation of Anti-HER2 Antibody to GSH-QDs
2.4. Confocal Fluorescence Imagimg of KPL-4 Breast Cancer Cells
3. Experimental Section
3.1. Chemicals
3.2. QD Synthesis
3.2.1. Preparation of Se (TBP) and Cd-Zn-S stock solution
3.2.2. Synthesis of CdSe/CdZnS with a 540 nm emission peak
3.2.3. Synthesis of CdSe/CdZnS with a 585 nm emission peak
3.2.4. Synthesis of CdSe/CdZnS with a 650 nm emission peak
3.3. Surface Modification of CdSe/CdZnS QDs with Glutathione (GSH)
3.4. Synthesis of Sulfo-SMCC Conjugated QDs (SMCC-QDs)
3.5. Conjugation of Anti-HER2 Antibody to QDs (HER2Ab-QDs)
3.5.1. EDC/sulfo-NHS coupling
3.5.2. Iminothiolane/sulfo-SMCC coupling
3.5.3. Sulfo-SMCC coupling
3.6. Synthesis of Anti-GFP Polyclonal Antibody Conjugated QDs (anti-GFP QDs)
3.7. Characterization of QDs
3.8. Cell Lines
3.9. Confocal Fluorescence Microscopy
4. Conclusions
Acknowledgments
References and Notes
- Slamon, D.J.; Clark, G.M.; Wong, S.G.; Levin, W.J.; Ullrich, A.; McGuire, W.L. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 1987, 235, 177–182. [Google Scholar]
- Slamon, D.J.; Godolphin, W.; Jones, L.A.; Holt, J.A.; Wong, S.G.; Keith, D.E.; Levin, W.J.; Stuart, S.G.; Udove, J.; Ullrich, A.; Press, M.F. Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science 1989, 244, 707–712. [Google Scholar]
- Cobleigh, M.A.; Vogel, C.L.; Tripathy, D.; Rober, N.J.; Scholl, S.; Fehrenbacher, L.; Wolter, J.M.; Paton, V.; Shak, S.; Lieberman, G.; Slamon, D.J. Multinational study of the efficacy and safety of humanized anti-HER2 monoclonal antibody in women who have HER2-overexpressing metastatic breast cancer that has progressed after chemotherapy for metastatic disease. J. Clin. Oncol. 1999, 17, 2639–2648. [Google Scholar]
- Haudis, C.A. Trastuzumab-mechanism of action and use in clinical practice. N. Engl. J. Med. 2007, 357, 39–51. [Google Scholar]
- Liberato, N.L.; Marchetti, M.; Barosi, G. Cost effectiveness of adjuvant trastuzumab in human epidermal growth factor receptor-2 positive breast cancer. J. Clin. Oncol. 2007, 25, 625–633. [Google Scholar]
- Jimenez, R.E.; Willis, T.; Tabasczka, P.; Visscher, D.W. Determination of Her2-/Neu status in breast carcinoma: comparative analysis of immunohistochemistry and fluorescent in situ hybridization. Mod. Pathol. 2000, 13, 37–45. [Google Scholar]
- Taylor, C.R.; Levenson, R.M. Quantification of immunohistochemistry-issues concerning methods, utility and semiquantitative assessment II. Histopathology 2006, 49, 411–24. [Google Scholar]
- Payne, S.J.L.; Bowen, R.L.; Jones, J.L.; Wells, C.A. Predictive markers in breast cancer-the present. Histopathology 2008, 52, 82–90. [Google Scholar]
- Ginestier, C.; Charafe-Jauffret, E.; Penault-Llorca, F.; Geneix, J.; Adelaide, J.; Chaffanet, M.; Mozziconacci, M.J.; Hassoun, J.; Viens, P.; Birnbaum, D.; Jacquemier, J. Comparative multi-methodological measurement of ERBB2 status in breast cancer. J. Pathol. 2004, 202, 286–298. [Google Scholar]
- Yaziji, H.; Goldstein, L.C.; Barry, T.S.; Werling, R.; Hwang, H.; Ellis, G.K.; Gralow, J.R.; Livingston, R.B.; Gown, A.M. HER-2 testing in breast cancer using parallel tissue-based methods. J. Am. Med. Assn. 2004, 29, 1972–1977. [Google Scholar]
- Wu, X.; Liu, H.; Liu, J.; Haley, K.N.; Treadway, J.A.; Larson, J.P.; Ge, N.; Peale, F.; Bruchez, M.P. Immunofluorescent labeling of cancer marker Her2 and other cellular targets with semiconductor quantum dots. Nat. Biotechnol. 2003, 21, 41–46. [Google Scholar]
- Li-Schishido, S.; Watanabe, T.M.; Tada, H.; Higuchi, H.; Ohuchi, N. Reduction in nonfluorescence state of quantum dots on an immunofluorescence staining. Biochem. Biophys. Res. Commun. 2006, 351, 7–13. [Google Scholar]
- Tada, H.; Higuchi, H.; Watanabe, T.M.; Ohuchi, N. In vivo real-time tracking of single quantum dots conjugated with monoclonal anti-HER2 antibody in tumors of mice. Cancer Res. 2007, 67, 1138–1144. [Google Scholar]
- Watanabe, T.M.; Higuchi, H. Stepwise movements in visible transport of HER2 by motor proteins in living cells. Biophys. J. 2007, 92, 4109–4120. [Google Scholar]
- Yu, W.W.; Chang, E.; Falkner, J.C.; Zhang, J.; Al-Somali, A.M.; Sayes, C.M.; Johns, J.; Drezek, R.; Colvin, V.L. Forming biocompatible and nonaggregated nanocrystals in water using amphiphilic polymers. J. Am. Chem. Soc. 2007, 129, 2871–79. [Google Scholar]
- Xing, Y.; Chaudry, Q.; Shen, C.; Kong, K.Y.; Zhau, H.E.; Chung, L.W.; Petros, J.A.; O'Regan, R.M.; Yezhelyev, M.V.; Simons, J.W.; Wang, M.D.; Nie, S. Bioconjugated quantum dots for multiplexed and quantitative immunohistochemistry. Nat. Protoc. 2007, 2, 1152–1165. [Google Scholar]
- Takeda, M.; Tada, H.; Higuchi, H.; Kobayashi, Y.; Kobayashi, M.; Sakurai, Y.; Ishida, T.; Ohuchi, N. In vivo single molecular imaging and sentinel node navigation by nanotechnology for molecular targeting drug-delivery systems and tailor-made medicine. Breast Cancer 2008, 15, 145–152. [Google Scholar]
- Xiao, Y.; Gao, X.; Gannot, G.; Emmert-Buck, M.R.; Srivastava, S.; Wagner, P.D.; Amos, M.D.; Barker, P.E. Quantitation of HER2 and telomerase biomarkers in solid tumors with IgY antibodies and nanocrystal detection. Int. J. Cancer 2008, 122, 2178–2186. [Google Scholar]
- Chen, C.; Peng, J.; Xia, H.S.; Yang, G.F.; Wu, Q.S.; Chen, L.D.; Zeng, L.B.; Zhang, Z.L.; Pang, D.W.; Li, Y. Quantum dots-based immunofluorescence technology for the quantitative determination of HER2 expression in breast cancer. Biomaterials 2009, 30, 2912–2918. [Google Scholar]
- Yang, Y.; Mao, H.; Wang, Y.A.; Cao, Z.; Peng, X.; Wang, X.; Duan, H.; Ni, C.; Yuan, Q.; Adams, G.; Smith, M.Q.; Wood, W.C.; Gao, X.; Nie, S. Single chain epidermal growth factor antibody conjugated nanoparticles for in vivo tumor targeting and imaing. Small 2009, 5, 235–243. [Google Scholar]
- Zdobnova, T.A.; Drofeev, S.G.; Tananaev, P.T.; Vasiliev, R.B.; Balandin, T.G.; Edelweiss, E.F.; Stremovskiy, O.A.; Balalaeva, I.V.; Turchin, I.V.; Lebedenko, E.N.; Zlomanov, V.P.; Deyev, S.M. Fluorescent immunolabeling of cancer cells by quantum dots and antibody scFv fragment. J. Biomed. Opt. 2009, 14, 021004–021001. [Google Scholar]
- Xiao, Y.; Gao, X.; Maragh, S.; Telford, W.G.; Tona, A. Cell lines candidate reference materials for quality control of ERBB2 amplification and expression assays in breast cancer. Clin. Chem. 2009, 55, 1307–1315. [Google Scholar]
- Hermanson, G.T. Bioconjugate Techniques; Academic Press: New York, NY, USA, 1996; pp. 137–226. [Google Scholar]
- Liu, W.; Choi, H.S.; Zimmer, J.P.; Tanaka, E.; Frangioni, J.V.; Bawendi, M. Compact cystein-coated CdSe(ZnCdS) quantum dots for in vivo applications. J. Am. Chem. Soc. 2007, 129, 14530–14531. [Google Scholar]
- Hines, M.A.; Guyot-Sionnest, P. Synthesis and characterization of strongly luminescent ZnS-capped CdSe nanocrystals. J. Phys. Chem. 1996, 100, 468–471. [Google Scholar]
- Murray, C.B.; Norris, D.J.; Bawendi, M.G. Synthesis and characterization of nearly monodisperse CdE (E = S, Se, Te) semiconductor nanocrystallites. J. Am. Chem. Soc. 1993, 115, 8706–8715. [Google Scholar]
- Peng, X.; Schlamp, M.C.; Kadavanish, A.V.; Alivisatos, A.P. Epitaxial growth of highly luminescent CdSe/CdS core/shell nanocrystals with photostability and electronic accessibility. J. Am. Chem. Soc. 1997, 119, 7019–7029. [Google Scholar]
- Dabbousi, B.O; Rodriguez-Viejo, J.; Mikulec, V.; Hine, J.R.; Mattoussi, H.; Ober, R.; Jensen, K.F.; Bawendi, M.G. (CdSe)ZnS coe-shell quantum dots: synthesis and characterization of a size series of highly luminescent nanocrystallites. J. Phys. Chem. B 1997, 101, 9463–9475. [Google Scholar]
- Qu, L.; Peng, X. Control of photoluminescence properties of CdSe nanocrystals in growth. J. Am. Chem. Soc. 2002, 124, 2049–2055. [Google Scholar]
- Gill, R.; Bahshi, L.; Freeman, R.; Willner, I. Optical detection of glucose and acetylcholine esterase inhibitors by H2O2-sensitive CdSe/ZnS quantum dots. Angew. Chem. Int. Ed. 2008, 47, 1676–1679. [Google Scholar]
- Jin, T.; Fujii, F.; Komai, Y.; Seki, J.; Seiyama, A.; Yoshioka, Y. Preparation and characterization of highly fluorescent, glutathione-coated near infrared quantum dots for in vivo fluorescence imaging. Int. J. Mol. Sci. 2008, 9, 2044–2061. [Google Scholar]
- Jin, T.; Yoshioka, Y.; Fujii, F.; Komai, Y.; Seki, J.; Seiyama, A. Gd3+-functionalized near-infrared quantum dots for in vivo dual modal (fluorescence/magnetic resonance) imaging. Chem. Commun. 2008, 5764–5766. [Google Scholar]
- Thelwall, P.E.; Yemin, A.Y.; Gillian, T.L.; Simpson, N.E.; Kasibhatla, M.S.; Rabbani, Z.N.; Macdonald, J.M.; Blackband, S.J.; Gamcsik, M.P. Nonivasive in vivo detection of glutathione metabolism in tumors. Cancer. Res. 2005, 65, 10149–10153. [Google Scholar]
- Satoh, T.; Yoshioka, Y. Contribution of reduced and oxidized glutathione to signals detected by magnetic resonance spectroscopy as indicators of local brain redox state. Neurosci. Res. 2006, 55, 34–39. [Google Scholar]
- Singhal, R.K.; Anderson, M.E.; Meister, A. Glutathione, a first defense against cadmium toxicity. FASEB J. 1987, 1, 220–223. [Google Scholar]
- Howarth, M.; Liu, W.; Puthenveetil, S.; Zheng, Y.; Marshall, L.F.; Schmidt, M.M.; Wittrup, K.D.; Bawendi, M.G. Monovalent, reduced-size quantum dots for imaging receptors on living cells. Nat. Methods 2008, 5, 397–399. [Google Scholar]
- Liu, W.; Howarth, M.; Greytak, A.B.; Zheng, Y.; Nocera, D.G.; Ting, A.Y.; Bawendi, M.G. Compact biocompatible quantum dots functionalized for cellular imaging. J. Am. Chem. Soc. 2008, 130, 1274–1284. [Google Scholar]
- Denardo, S.J.; Yao, Z.; Lam, K.S.; Song, A.; Burke, P.A.; Mirick, G.R.; Lamborn, K.R.; O'Donnel, R.T.; DeNardo, G.L. Effect of molecular size of pegylated peptide on the pharmacokinetics and tumor targeting in lymphoma-nearing mice. Clin. Can. Res. 2003, 9, 3854s–3864s. [Google Scholar]
- Choi, H.S.; Liu, W.; Misra, P.; Tanaka, E.; Zimmer, J.P.; Ipe, B.I.; Bawendi, M.G.; Frangioni, J.V. Renal clearence of quantum dots. Nat. Biotechnol. 2007, 25, 1165–1170. [Google Scholar]
- Jin, T.; Fujii, F.; Sakata, H.; Tamura, M.; Kinjo, M. Calixarene-coated water-soluble CdSe-ZnS semiconductor quantum dots that are highly fluorescent and stable in aqueous solution. Chem. Commun. 2005, 22, 2829–2831. [Google Scholar]
- Jin, T.; Fujii, F.; Sakata, H.; Tamura, M.; Kinjo, M. Amphiphilic p-sulfonatocalix[4]arene-coated CdSe/ZnS quantum dots for the optical detection of the neurotransmitter acetylcholine. Chem. Commun. 2005, 34, 4300–4302. [Google Scholar]
- Jin, T.; Fujii, F.; Yamada, E.; Nodasaka, Y.; Kinjo, M. Control of the optical properties of quantum dots by surface coating with calix[n]arene carboxylic acids. J. Am. Chem. Soc. 2006, 128, 9288–9289. [Google Scholar]
- Heuff, R.F.; Swift, J.L.; Cramb, D.T. Fluorescence correlation spectroscopy using quantum dots: advances, challenges and opportunities. Phys. Chem. Chem. Phys. 2007, 9, 1870–1880. [Google Scholar]
- Jin, T.; Fujii, F.; Yamada, E.; Nodasaka, Y.; Kinjo, M. Preparation and characterization of thiacalix[4]arene coated water-soluble CdSe/ZnS quantum dots as a fluorescent probe for Cu2+ ions. Comb. Chem. High Throughput Scr. 2007, 10, 473–479. [Google Scholar]
- Kobayashi, H.; Hama, Y.; Koyama, Y.; Barrett, T.; Regino, C.A.S.; Urano, Y.; Choyke, P. Simultaneous multicolor imaging of five different lymphatic basins using quantum dots. Nano Lett. 2007, 7, 1711–1716. [Google Scholar]
- Ormö, M.; Cubitt, A.B.; Kallio, K.; Gross, L.A.; Tsien, R.Y.; Remingston, S.J. Crystal structure of the aequorea Victoria green fluorescent protein. Science 1986, 273, 747–753. [Google Scholar]
- Amit, A.G.; Mariuzza, R.A.; Phillips, S.E.V.; Poljak, R.J. Three-dimensional structure of an antigen-antibody complex at 2.5 Å resulution. Science 1996, 273, 1392–1395. [Google Scholar]
- Ouerghi, O.; Touhami, A.; Othmane, A.; Ouada, H.B.; Martelet, C.; Fretigny, C.; Jaffrezic-renault, N. Investigation specific antigen/antibody binding with the atomic force microscope. Biomol. Eng. 2002, 19, 183–188. [Google Scholar]
- Pathak, S.; Choi, S.K.; Arnheim, N.; Thompson, M.E. Hydroxylated quantum dots as luminescent probes for in situ hybridization. J. Am. Chem. Soc. 2001, 123, 4103–4104. [Google Scholar]
- Pereira, M.; Lai, E.P.C. Capillary electrophoresis for the characterization of quantum dots after non-selective or selective bioconjugation with antibodies for immunoassay. J. Nanotechnol. 2008, 6, 10. [Google Scholar]
- Austin, C.D.; De Maziere, A.M.; Pisacane, P.L.; van Dijk, S.M.; Eigenbrot, C.; Sliwkowski, M.X.; Klumperman, J.; Scheller, R.H. Endocytosis and sorting of ErbB2 and the site of action of cancer therapeutics trastuzumab and geldanamycin. Mol. Biol. Cell. 2004, 15, 5268–5282. [Google Scholar]
- Jia, L.-T.; Zhang, L.-H.; Yu, C.-J.; Zhao, J.; Xu, Y.-M.; Gui, J.-H.; Jin, M.; Ji, Z.-L.; Wen, W.-H.; Wang, C.-J.; Yang, A.-G. Specific tumoricidal activity of a secreted proaptotic protein consisting of HER2 antibody and constutively active caspase-3. Cancer Res. 2003, 63, 3275–3262. [Google Scholar]
- Rigler, R.; Elson, E.S. Fluorescence correlation spectroscopy; Springer-Verlag: Berlin, Germany, 2001. [Google Scholar]
- Kurebayashi, J.; Otsuki, T.; Tang, C.K. Isolation and characterization of a new human breat cancer cell line, KPL-4, expressing the Erb B family receptors and interleukin-6. Br. J. Cancer 1998, 79, 707–717. [Google Scholar]
- Ouchi, F.; Sekiguchi, K.; Tanaka, Y. Antitumor activity of combinations of anti-HER-2 antibody trastuzumab and oral fluoropyrimidines capecitabine/5′-dFUrd in human breast cancer models. Cancer Chemother. Pharmacol. 2002, 49, 211–216. [Google Scholar]











| GSH-QD (540 nm) | GSH-QD (585 nm) | GSH-QD (650 nm) | |
|---|---|---|---|
| Quantum yield | 0.33 a | 0.23 b | 0.39 b |
| Diameter (nm) | 4.5 ± 0.6 c | 5.9 ± 0.5 c | Not determined |
| 4.4 ± 0.2d | 5.1± 0.3 d | 6.9 ± 0.5 d | |
| Apparent MW (kDa) | 75 | 150 | 300 |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tiwari, D.K.; Tanaka, S.-I.; Inouye, Y.; Yoshizawa, K.; Watanabe, T.M.; Jin, T. Synthesis and Characterization of Anti-HER2 Antibody Conjugated CdSe/CdZnS Quantum Dots for Fluorescence Imaging of Breast Cancer Cells. Sensors 2009, 9, 9332-9354. https://doi.org/10.3390/s91109332
Tiwari DK, Tanaka S-I, Inouye Y, Yoshizawa K, Watanabe TM, Jin T. Synthesis and Characterization of Anti-HER2 Antibody Conjugated CdSe/CdZnS Quantum Dots for Fluorescence Imaging of Breast Cancer Cells. Sensors. 2009; 9(11):9332-9354. https://doi.org/10.3390/s91109332
Chicago/Turabian StyleTiwari, Dhermendra K., Shin-Ichi Tanaka, Yasushi Inouye, Keiko Yoshizawa, Tomonobu M. Watanabe, and Takashi Jin. 2009. "Synthesis and Characterization of Anti-HER2 Antibody Conjugated CdSe/CdZnS Quantum Dots for Fluorescence Imaging of Breast Cancer Cells" Sensors 9, no. 11: 9332-9354. https://doi.org/10.3390/s91109332
APA StyleTiwari, D. K., Tanaka, S.-I., Inouye, Y., Yoshizawa, K., Watanabe, T. M., & Jin, T. (2009). Synthesis and Characterization of Anti-HER2 Antibody Conjugated CdSe/CdZnS Quantum Dots for Fluorescence Imaging of Breast Cancer Cells. Sensors, 9(11), 9332-9354. https://doi.org/10.3390/s91109332