Low p-SYN1 (Ser-553) Expression Leads to Abnormal Neurotransmitter Release of GABA Induced by Up-Regulated Cdk5 after Microwave Exposure: Insights on Protection and Treatment of Microwave-Induced Cognitive Dysfunction
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethics and Methods Statement
2.2. Animals and Groups
2.3. Synaptosome Isolation
2.4. Cell Culture
2.5. Microwave Exposure and Dosimetry
2.6. Timm Staining of the Hippocampus
2.7. Protein Extraction and Western Blotting
2.8. Western Blotting
2.9. Immunohistochemistry
2.10. Neurotransmitter Release Assays
2.11. Amino Acid Neurotransmitter Measurements
2.12. Short Hairpin RNA (shRNA) Transfection of SYN1
2.13. Inhibition of Mitogen-Activated Protein Kinase Kinase (MAPKK/MEK) and Cdk5 with U0126 and Roscovitine
2.14. Statistical Analysis
3. Results
3.1. Abnormality in Mossy Fiber Sprouting of the Hippocampus after Microwave Exposure
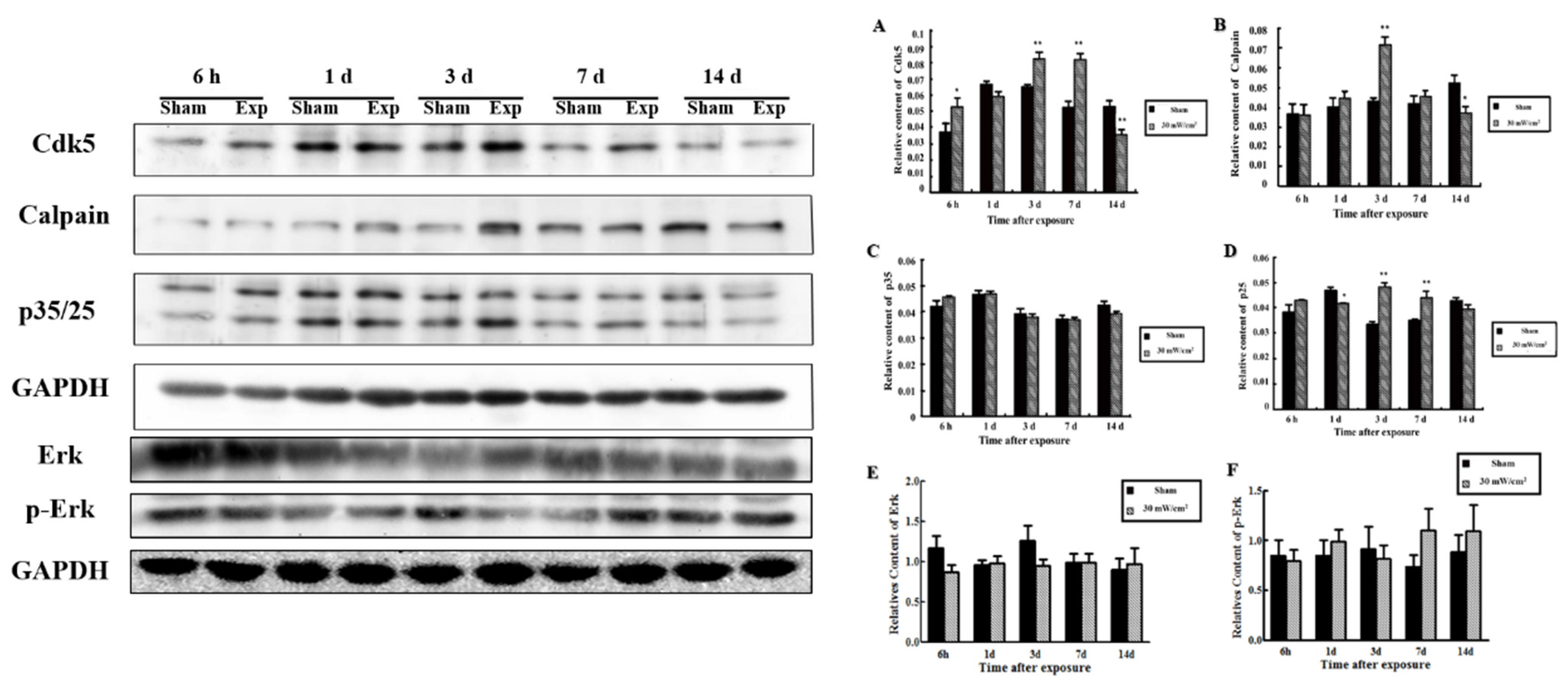
3.2. Expression of SYN1 and Related Proteins in the Rat Hippocampus
3.3. Alteration of GABA, SYN1 and Its Phosphorylated Proteins in PC12 Cells after Microwave Exposure
3.4. Silencing of P-SYN1 Results in Abnormal Neurotransmitter Release
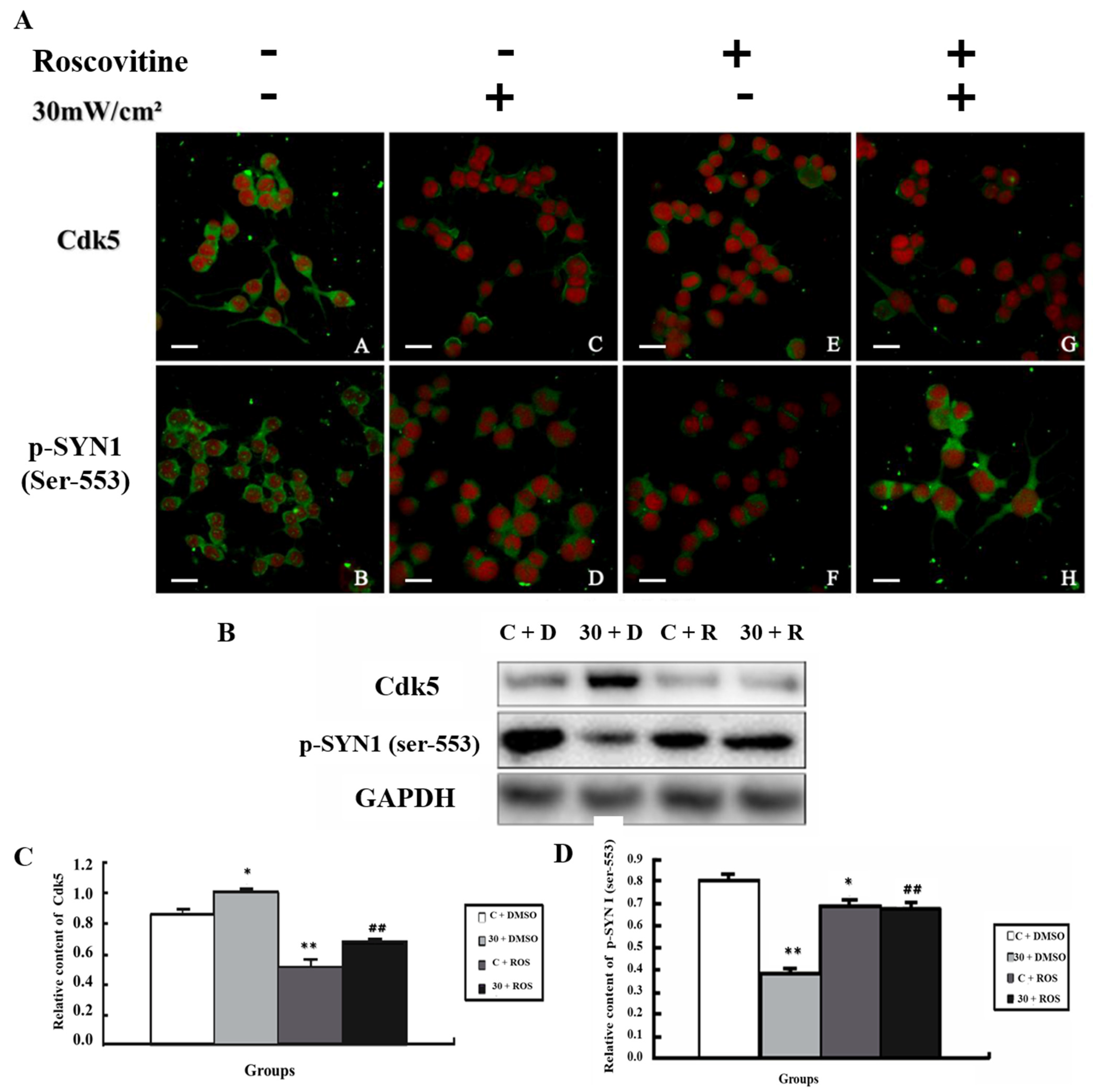
3.5. Alteration of p-SYN1 and Its Related Kinases after Inhibition of MEK and Cdk5
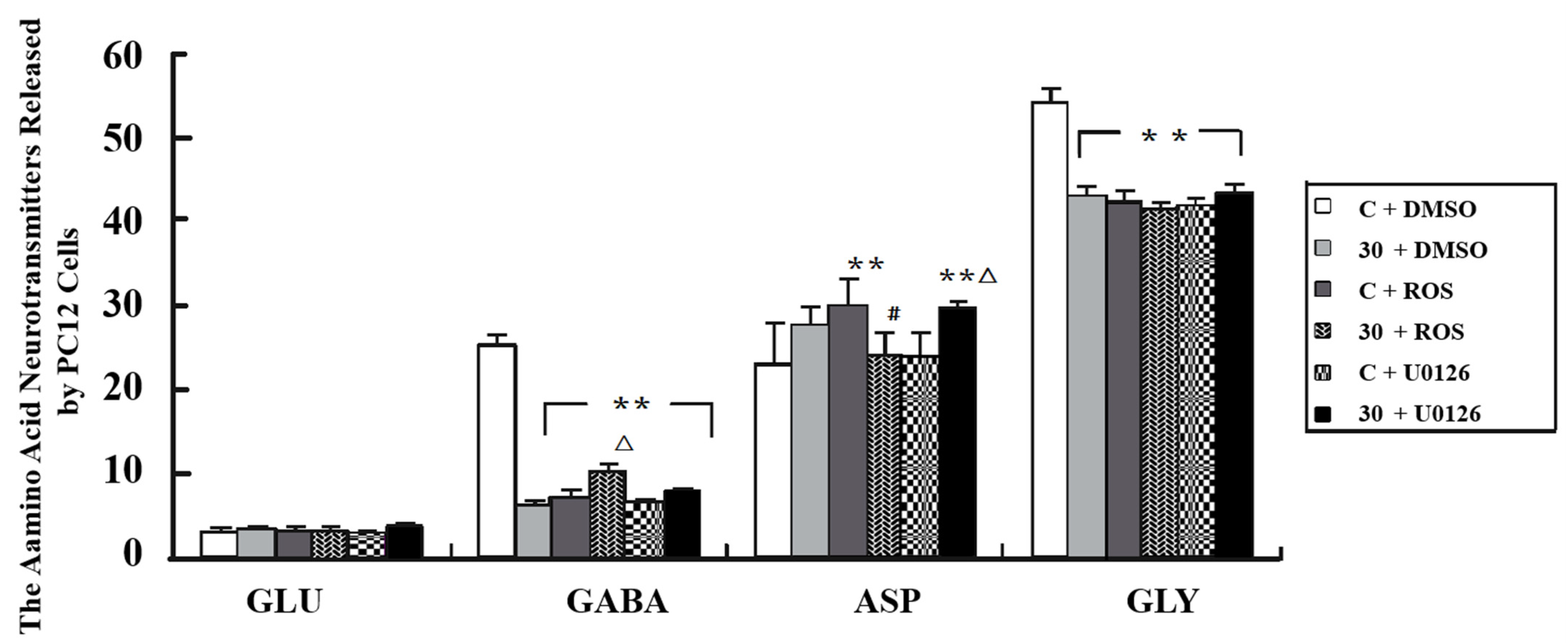
3.6. Alteration of Neurotransmitter Levels after Inhibition of MEK and Cdk5
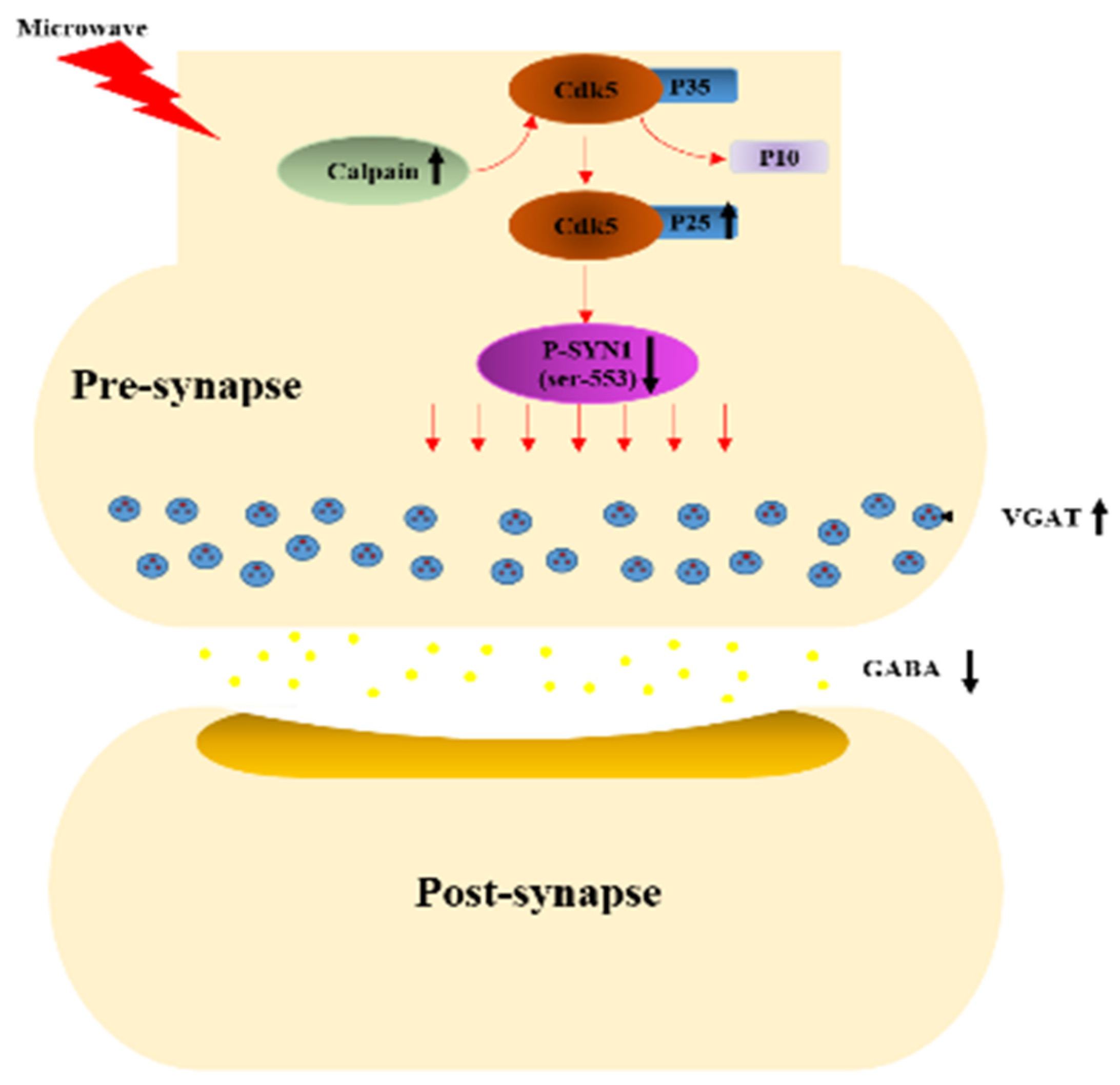
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cusack, P.T. Havana Syndrome Possible Cause. Biomed. J. Sci. Tech. Res. 2021, 36, 28508–28510. [Google Scholar] [CrossRef]
- Bartholomew, R.E.; Baloh, R.W. Challenging the diagnosis of ‘Havana Syndrome’ as a novel clinical entity. J. R. Soc. Med. 2019, 113, 014107681987755. [Google Scholar] [CrossRef]
- Zhao, L.; Yang, Y.F.; Gao, Y.B.; Wang, S.M.; Wang, L.F.; Zuo, H.Y.; Dong, J.; Xu, X.P.; Su, Z.T.; Zhou, H.M. Upregulation of HIF-1α via activation of ERK and PI3K pathway mediated protective response to microwave-induced mitochondrial injury in neuron-like cells. Mol. Neurobiol. 2014, 50, 1024–1034. [Google Scholar] [CrossRef]
- Pall, M.L. Wi-Fi is an important threat to human health. Environ. Res. 2018, 164, 405. [Google Scholar] [CrossRef] [PubMed]
- Altunkaynak, B.Z.; Altun, G.; Yahyazadeh, A.; Kaplan, A.A.; Deniz, O.G.; Türkmen, A.P.; Önger, M.E.; Kaplan, S. Different methods for evaluating the effects of microwave radiation exposure on the nervous system. J. Chem. Neuroanat. 2016, 75, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Tan, S.; Dong, J.; Zhang, J.; Yao, B.; Xu, X.; Hao, Y.; Yu, C.; Zhou, H.; Zhao, L.; et al. iTRAQ quantitatively proteomic analysis of the hippocampus in a rat model of accumulative microwave-induced cognitive impairment. Environ. Sci. Pollut. Res. 2019, 26, 17248–17260. [Google Scholar] [CrossRef] [PubMed]
- D’Andrea, J.A.; Adair, E.R.; De Lorge, J.O. Behavioral and cognitive effects of microwave exposure. Bioelectromagn. 2003, 24, S39–S62. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh, P.S.; Megha, K.; Nasare, N.; Banerjee, B.D.; Ahmed, R.S.; Abegaonkar, M.P.; Tripathi, A.K.; Mediratta, P.K. Effect of Low Level Subchronic Microwave Radiation on Rat Brain. Biomed. Environ. Sci. 2016, 29, 858–867. [Google Scholar] [PubMed]
- Vorobyov, V.; Petković, B.; Pesic, V.; Prolić, Z. Repeated exposure to low-level extremely low frequency-modulated microwaves affects cortex-hypothalamus interplay in freely moving rats: EEG study. Int. J. Radiat. Biol. 2010, 86, 376–383. [Google Scholar] [CrossRef]
- Stein, Y.; Udasin, I.G. Electromagnetic hypersensitivity (EHS, microwave syndrome) – Review of mechanisms. Environ. Res. 2020, 186, 109445. [Google Scholar] [CrossRef] [PubMed]
- Greengard, P.; Valtorta, F.; Czernik, A.J.; Benfenati, F. Synaptic vesicle phosphoproteins and regulation of synaptic function. Science 1993, 259, 780–785. [Google Scholar] [CrossRef] [Green Version]
- Fornasiero, E.F.; Raimondi, A.; Guarnieri, F.C.; Orlando, M.; Fesce, R.; Benfenati, F.; Valtorta, F. Synapsins contribute to the dynamic spatial organization of synaptic vesicles in an activity-dependent manner. J. Neurosci. 2012, 32, 12214–12227. [Google Scholar] [CrossRef] [Green Version]
- Bogen, I.; Jensen, V.; Hvalby, O.; Walaas, S. Synapsin-dependent development of glutamatergic synaptic vesicles and presynaptic plasticity in postnatal mouse brain. Neuroscience 2009, 158, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Augustine, G. Synapsins and the Synaptic Vesicle Reserve Pool: Floats or Anchors? Cells 2021, 10, 658. [Google Scholar] [CrossRef] [PubMed]
- Longhena, F.; Faustini, G.; Brembati, V.; Pizzi, M.; Benfenati, F.; Bellucci, A. An updated reappraisal of synapsins: Structure, function and role in neurological and psychiatric disorders. Neurosci. Biobehav. Rev. 2021, 130, 33–60. [Google Scholar] [CrossRef]
- Cesca, F.; Baldelli, P.; Valtorta, F.; Benfenati, F. The synapsins: Key actors of synapse function and plasticity. Prog. Neurobiol. 2011, 91, 313–348, Erratum in Prog. Neurobiol. 2011, 93, 549. [Google Scholar] [CrossRef] [PubMed]
- Yamagata, Y.; Nairn, A.C. Contrasting features of ERK1/2 activity and synapsin I phosphorylation at the ERK1/2-dependent site in the rat brain in status epilepticus induced by kainic acid in vivo. Brain Res. 2015, 1625, 314–323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verstegen, A.M.J.; Tagliatti, E.; Lignani, G.; Marte, A.; Stolero, T.; Atias, M.; Corradi, A.; Valtorta, F.; Gitler, D.; Onofri, F.; et al. Phosphorylation of Synapsin I by Cyclin-Dependent Kinase-5 Sets the Ratio between the Resting and Recycling Pools of Synaptic Vesicles at Hippocampal Synapses. J. Neurosci. 2014, 34, 7266–7280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamagata, Y.; Jovanovic, J.N.; Czernik, A.J.; Greengard, P.; Obata, K. Bidirectional changes in synapsin I phosphorylation at MAP kinase-dependent sites by acute neuronal excitation in vivo. J. Neurochem. 2002, 80, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Costa, R.; Murphy, G.G.; Elgersma, Y.; Zhu, Y.; Gutmann, D.; Parada, L.F.; Mody, I.; Silva, A.J. Neurofibromin Regulation of ERK Signaling Modulates GABA Release and Learning. Cell 2008, 135, 549–560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiao, S.; Peng, R.; Yan, H.; Gao, Y.; Wang, C.; Wang, S.; Zou, Y.; Xu, X.; Zhao, L.; Dong, J.; et al. Reduction of Phosphorylated Synapsin I (Ser-553) Leads to Spatial Memory Impairment by Attenuating GABA Release after Microwave Exposure in Wistar Rats. PLoS ONE 2014, 9, e95503. [Google Scholar] [CrossRef]
- Dunkley, P.R.; Jarvie, P.E.; Heath, J.W.; Kidd, G.; Rostas, J.A. A rapid method for isolation of synaptosomes on Percoll gradients. Brain Res. 1986, 372, 115–129. [Google Scholar] [CrossRef]
- Levi, A.; Biocca, S.; Cattaneo, A.; Calissano, P. The mode of action of nerve growth factor in PC12 cells. Mol. Neurobiol. 1988, 2, 201–226. [Google Scholar] [CrossRef]
- Wang, H.; Peng, R.; Zhou, H.; Wang, S.; Gao, Y.; Wang, L.; Yong, Z.; Zuo, H.; Zhao, L.; Dong, J.; et al. Impairment of long-term potentiation induction is essential for the disruption of spatial memory after microwave exposure. Int. J. Radiat. Biol. 2013, 89, 1100–1107. [Google Scholar] [CrossRef] [PubMed]
- Schuderer, J.; Kuster, N. Effect of the meniscus at the solid/liquid interface on the SAR distribution in Petri dishes and flasks. Bioelectromagnetics 2003, 24, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Icnirp, G. Guidelines for limiting exposure to time-varying electric, magnetic, and electromagnetic fields (up to 300 Ghz). Health Phys. 1998, 74, 257–258. [Google Scholar]
- IEEE Std C95.1; IEEE Standard for Safety Levels with Respect to Human Exposure to Radiofrequency Electromagnetic Fields, 3 kHz to 300 GHz. IEEE: Piscataway, NJ, USA, 2015.
- Tan, S.; Wang, H.; Xu, X.; Zhao, L.; Zhang, J.; Dong, J.; Yao, B.; Wang, H.; Hao, Y.; Zhou, H.; et al. Acute effects of 2.856 GHz and 1.5 GHz microwaves on spatial memory abilities and CREB-related pathways. Sci. Rep. 2021, 11, 1–10. [Google Scholar] [CrossRef]
- Tan, S.; Wang, H.; Xu, X.; Zhao, L.; Zhang, J.; Dong, J.; Yao, B.; Wang, H.; Zhou, H.; Gao, Y.; et al. Study on dose-dependent, frequency-dependent, and accumulative effects of 1.5 GHz and 2.856 GHz microwave on cognitive functions in Wistar rats. Sci. Rep. 2017, 7, 10781. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, H.J.; Peng, R.Y.; Wang, C.Z.; Qiao, S.M.; Yong, Z.; Gao, Y.B.; Xu, X.P.; Wang, S.X.; Dong, J.; Zuo, H.Y.; et al. Alterations of cognitive function and 5-HT system in rats after long term microwave exposure. Physiol. Behav. 2015, 140, 236–246. [Google Scholar] [CrossRef]
- Meyer-Franke, A.; Wilkinson, G.A.; Kruttgen, A.; Hu, M.; Munro, E.; Hanson, M.G.; Reichardt, L.F.; Barres, B.A. Depolarization and cAMP elevation rapidly recruit TrkB to the plasma membrane of CNS neurons. Neuron 1998, 21, 681–693. [Google Scholar] [CrossRef] [Green Version]
- Minami, A.; Saito, M.; Mamada, S.; Ieno, D.; Hikita, T.; Takahashi, T.; Otsubo, T.; Ikeda, K.; Suzuki, T. Role of Sialidase in Long-Term Potentiation at Mossy Fiber-CA3 Synapses and Hippocampus-Dependent Spatial Memory. PLoS ONE 2016, 11, e0165257. [Google Scholar] [CrossRef] [Green Version]
- Heimrich, B.; Claus, H.; Schwegler, H.; Haas, H. Hippocampal mossy fiber distribution and long-term potentiation in two inbred mouse strains. Brain Res. 1989, 490, 404–406. [Google Scholar] [CrossRef]
- Derrick, B.E.; York, A.D.; Martinez, J.L., Jr. Increased granule cell neurogenesis in the adult dentate gyrus following mossy fiber stimulation sufficient to induce long-term potentiation. Brain Res. 2000, 857, 300–307. [Google Scholar] [CrossRef]
- Mitsuno, K.; Sasa, M.; Ishihara, K.; Ishikawa, M.; Kikuchi, H. LTP of mossy fiber-stimulated potentials in CA3 during learning in rats. Physiol. Behav. 1994, 55, 633–638. [Google Scholar] [CrossRef]
- Shu, Y.; Xiang, M.; Zhang, P.; Qi, G.; He, F.; Zhang, Q.; Zhang, Z.; Lv, Z.; Peng, X.; Cai, H. Wnt-5a Promotes Neural Development and Differentiation by Regulating CDK5 via Ca2+/Calpain Pathway. Cell. Physiol. Biochem. 2018, 51, 2604–2615. [Google Scholar] [CrossRef] [PubMed]
- Antoniou, X.; Gassmann, M.; Ogunshola, O. Cdk5 interacts with Hif-1α in neurons: A new hypoxic signalling mechanism? Brain Res. 2011, 1381, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Zhao, J.; Guo, F.-L.; Gao, X.; Xie, X.; Liu, S.; Yang, X.; Yang, X.; Zhang, L.; Ye, Y.; et al. Metformin Ameliorates Synaptic Defects in a Mouse Model of AD by Inhibiting Cdk5 Activity. Front. Cell. Neurosci. 2020, 14, 170. [Google Scholar] [CrossRef] [PubMed]
- Juliet, R.; Grace, I.; Hossein, A.; Mario, R.; Inez, V.; Steven, C.; Bland, R.J.; Slack, R.S.; During, M.J.; Park, D.S. Multiple cyclin-dependent kinases signals are critical mediators of ischemia/hypoxic neuronal death in vitro and in vivo. Proc. Natl. Acad. Sci. USA 2005, 102, 14080–14085. [Google Scholar]
- Zhou, M.; Huang, T.; Collins, N.; Zhang, J.; Shen, H.; Dai, X.; Xiao, N.; Wu, X.; Wei, Z.; York, J.; et al. APOE4 induces site-specific tau phosphorylation through calpain-CDK5 signaling pathway in EFAD-Tg mice. Curr. Alzheimer Res. 2016, 13, 1048–1055. [Google Scholar] [CrossRef]
- Allnutt, A.B.; Waters, A.K.; Kesari, S.; Yenugonda, V.M. Physiological and Pathological Roles of Cdk5: Potential Directions for Therapeutic Targeting in Neurodegenerative Disease. ACS Chem. Neurosci. 2020, 11, 1218–1230. [Google Scholar] [CrossRef]
- Lee, J.H.; Kim, K.T. Induction of cyclin-dependent kinase 5 and its activator p35 through the extracellular-signal-regulated kinase and protein kinase A pathways during retinoic-acid mediated neuronal differentiation in human neuroblastoma SK-N-BE(2)C cells . J. Neurochem. 2004, 91, 634–647. [Google Scholar]
- Ikiz, B.; Przedborski, S. A Sequel to the Tale of p25/Cdk5 in Neurodegeneration. Neuron 2008, 60, 731–732. [Google Scholar] [CrossRef] [PubMed] [Green Version]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhi, W.-J.; Qiao, S.-M.; Zou, Y.; Peng, R.-Y.; Yan, H.-T.; Ma, L.-Z.; Dong, J.; Zhao, L.; Yao, B.-W.; Zhao, X.-L.; et al. Low p-SYN1 (Ser-553) Expression Leads to Abnormal Neurotransmitter Release of GABA Induced by Up-Regulated Cdk5 after Microwave Exposure: Insights on Protection and Treatment of Microwave-Induced Cognitive Dysfunction. Curr. Issues Mol. Biol. 2022, 44, 206-221. https://doi.org/10.3390/cimb44010015
Zhi W-J, Qiao S-M, Zou Y, Peng R-Y, Yan H-T, Ma L-Z, Dong J, Zhao L, Yao B-W, Zhao X-L, et al. Low p-SYN1 (Ser-553) Expression Leads to Abnormal Neurotransmitter Release of GABA Induced by Up-Regulated Cdk5 after Microwave Exposure: Insights on Protection and Treatment of Microwave-Induced Cognitive Dysfunction. Current Issues in Molecular Biology. 2022; 44(1):206-221. https://doi.org/10.3390/cimb44010015
Chicago/Turabian StyleZhi, Wei-Jia, Si-Mo Qiao, Yong Zou, Rui-Yun Peng, Hai-Tao Yan, Li-Zhen Ma, Ji Dong, Li Zhao, Bin-Wei Yao, Xue-Long Zhao, and et al. 2022. "Low p-SYN1 (Ser-553) Expression Leads to Abnormal Neurotransmitter Release of GABA Induced by Up-Regulated Cdk5 after Microwave Exposure: Insights on Protection and Treatment of Microwave-Induced Cognitive Dysfunction" Current Issues in Molecular Biology 44, no. 1: 206-221. https://doi.org/10.3390/cimb44010015




