Field Application of Modified In Situ Soil Flushing in Combination with Air Sparging at a Military Site Polluted by Diesel and Gasoline in Korea
Abstract
:1. Introduction
2. Materials and Methods
2.1. Explanation of the Target Area
| Item | Reclamation Soil | Weathering Soil |
|---|---|---|
| pH | 6.0 | 5.9 |
| Porosity (%) | 39.0 | 29.7 |
| Density (g/cm3) | 1.62 | 1.73 |
| Distribution of soil (%) | Granule 3.6% | Granule 1.3% |
| Sand 94.1% | Sand 89.7% | |
| Silt/Clay 2.3% | Silt/Clay 8.9% |
2.2. Pilot Scale Test


2.3. Full Scale Test

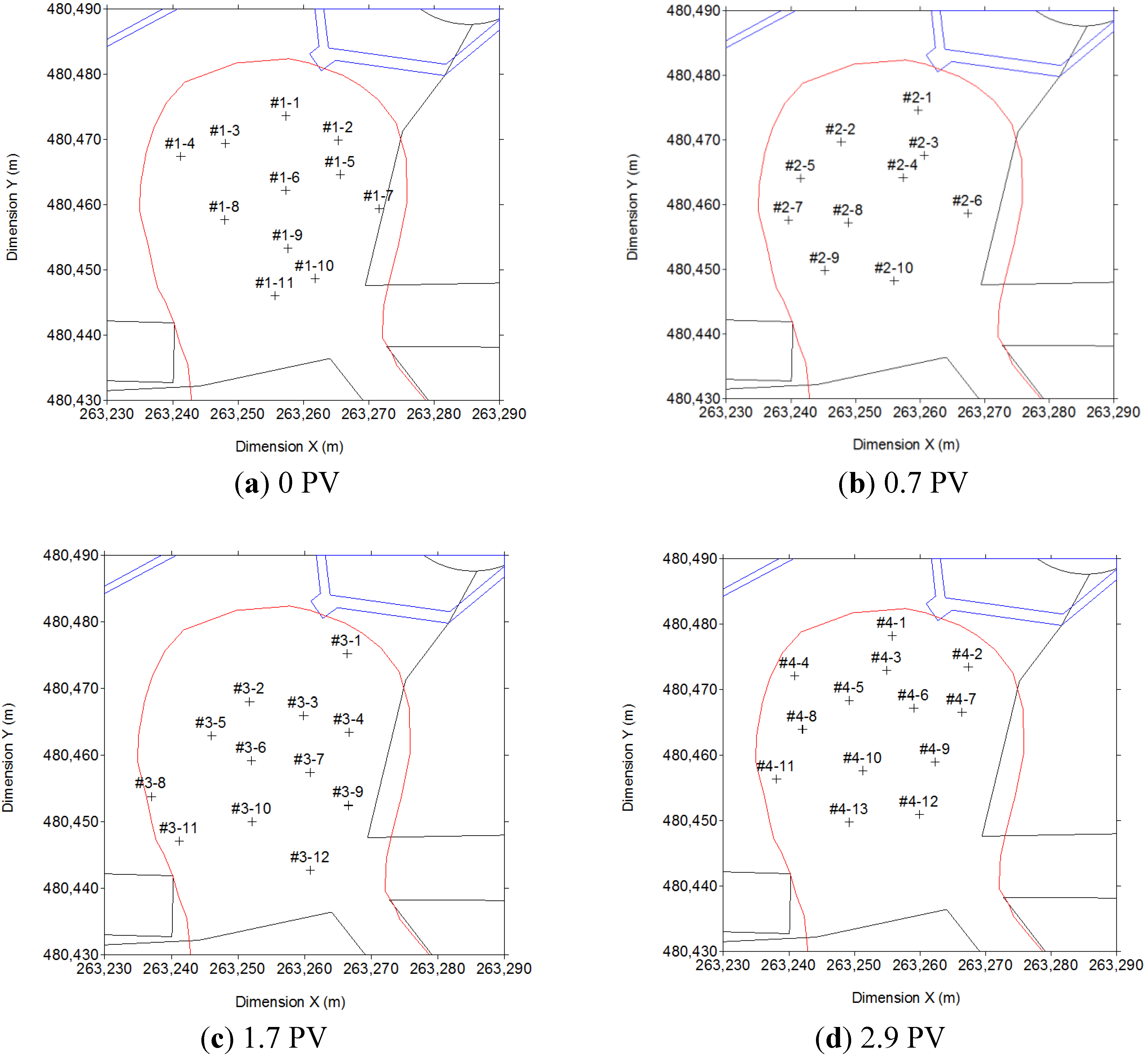
2.4. Experimental Methods
3. Results and Discussion
3.1. Pilot Scale Application
3.1.1. Efficiency Assessment with the Non-Pressure Dose

3.1.2. Pilot Scale Assessment of Air Sparging


| Pollutant | With Surfactant | Without Surfactant |
|---|---|---|
| TPH (g) | 187 | 131 |
| BTEX (g) | 458 | 440 |
3.2. Full Scale Application

| Depth (m) | Pore Volume 0 | Pore Volume 0.7 | Pore Volume 1.7 | Pore Volume 2.9 | ||||
|---|---|---|---|---|---|---|---|---|
| TPH (mg/kg) | *SD | TPH (mg/kg) | SD | TPH (mg/kg) | SD | TPH (mg/kg) | SD | |
| 6–7 m | 654 | 90 | 684 | 68 | 441 | 44 | 309 | 90 |
| 7–8 m | 1170 | 98 | 892 | 91 | 662 | 83 | 498 | 97 |
| 8−9 m | 1575 | 93 | 1203 | 58 | 829 | 78 | 601 | 93 |
| Depth (m) | Pore Volume 0 | Pore Volume 0.7 | Pore Volume 1.7 | Pore Volume 2.9 | ||||
|---|---|---|---|---|---|---|---|---|
| BTEX (mg/kg) | *SD | BTEX (mg/kg) | SD | BTEX (mg/kg) | SD | BTEX (mg/kg) | SD | |
| 6–7 m | 73 | 6 | 92 | 8 | 15 | 6 | 14 | 8 |
| 7–8 m | 119 | 10 | 100 | 6 | 35 | 9 | 30 | 7 |
| 8–9 m | 166 | 7 | 97 | 8 | 30 | 9 | 21 | 6 |
| Depth | Before Operation | After Operation | ||
|---|---|---|---|---|
| Average TPH Mass(kg) | Average BTEX Mass(kg) | Average TPH Mass(kg) | Average BTEX Mass(kg) | |
| 6–7 m | 1589 | 178 | 751 | 34 |
| 7–8 m | 3036 | 309 | 1292 | 78 |
| 8–9 m | 4087 | 430 | 1560 | 54 |
| Total | 8712 | 917 | 3603 (*58.6%) | 165 (*82.0%) |
| Depth | TPH (>500 mg/kg) | BTEX (>80 mg/kg) | ||
|---|---|---|---|---|
| Pore Volume 0 | Pore Volume 2.9 | Pore Volume 0 | Pore Volume 2.9 | |
| 6–7 m | 739 | 241 | 160 | 1 |
| 7–8 m | 902 | 437 | 510 | 24 |
| 8–9 m | 889 | 512 | 420 | 52 |
| Total | 2530 | 1190 | 1090 | 77 |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mercer, J.W.; Cohen, R.M. A review of immiscible fluids in the subsurface: Properties, models, characterization and remediation. J. Contami. Hydrol. 1990, 6, 107–163. [Google Scholar] [CrossRef]
- Tsakiroglou, C.D.; Aggelopoulos, C.A.; Tzovolou, D.N.; Theodoropoulou, M.A.; Avraam, D.G. Dynamics of surfactant-enhanced oil mobilization and dissolution in porous media: Experiments and numerical modeling. Int. J. Multiphase Flow. 2013, 55, 11–23. [Google Scholar] [CrossRef]
- Long, A.; Zhang, H.; Lei, Y. Surfactant flushing remediation of toluene contaminated soil: Optimization with response surface methodology and surfactant recovery by selective oxidation with sulfate radicals. Sep. Purif. Technol. 2013, 118, 612–619. [Google Scholar] [CrossRef]
- Lundegard, P.D.; LaBrecque, D. Air sparging in a sandy aquifer (Florence, Oregon, USA): Actual and apparent radius of influence. J. Contam. Hydrol. 1995, 19, 1–27. [Google Scholar]
- Marley, M.C.; Hazebroudck, D.J.; Walch, M.T. The application of in situ air sparging as an innovative soil and ground water technology. Ground Water Monit. Rev. 1992, 12, 137–145. [Google Scholar]
- Rabiduar, A.J.; Blayden, J.M.; Ganguly, C. Field performance of air-sparging system for removing TCE from ground water. Environ. Sci. Technol. 1999, 33, 157–162. [Google Scholar] [CrossRef]
- Reddy, K.R.; Kosgi, S.; Zhou, J. A Review of in-situ air sparging for the remediation of VOC-contaminated saturated soils and ground water. Haz. Waste Haz. Mat. 1995, 12, 97–118. [Google Scholar] [CrossRef]
- Unger, A.J.A.; Sudicky, E.; Forsyth, P.A. Mechanisms controlling vacuum extraction coupled with air sparging for remediation of heterogeneous formation contaminated by dense nonaquesous phase liquids. Water Resour. Res. 1995, 31, 1913–1925. [Google Scholar] [CrossRef]
- Braida, W.J.; Ong, S.K. Air sparging: Air-water mass transfer coefficients. Water Resour. Res. 1998, 34, 3245–3253. [Google Scholar]
- Reddy, K.R.; Adams, J.A. System effect on benzene removal from saturated soils and groundwater using air sparging. J. Environ. Eng. 1998, 124, 288–299. [Google Scholar] [CrossRef]
- Adams, J.A.; Reddy, K.R. Removal of dissolved and free-phase benzene pools from ground water using in situ air sparging. J. Environ. Eng. 2000, 126, 697–707. [Google Scholar] [CrossRef]
- Kim, H.K.; Soh, H.E.; Annable, M.D.; Kim, D.J. Surfactant-enhanced air sparging in saturated sand. Environ. Sci. Technol. 2004, 38, 1170–1175. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Cho, K.M.; Moon, J.W.; Annable, M.D. Changes in air saturation and air-water interfacial area during surfactant-enhances air sparging in saturated sand. J. Conatam. Hydrol. 2006, 88, 23–35. [Google Scholar]
- Lee, H.; Lee, Y.J. A field scale study of serial treatment using slurping and in situ soil flushing to remediate an oil contaminated site. Asian J. Chem. 2010, 22, 1535–1549. [Google Scholar]
- Ministry of Environment in Korea. Korean Standard Method for Soil. Available online: https://sgis.nier.go.kr (accessed on 10 March 2009).
- Choi, S.I.; So, J.H.; Cho, C.H. A Study on the basic characteristics of in-situ soil flushing using surfactant. (In Korean)J. KoSSGE. 2002, 7, 87–91. [Google Scholar]
- Couto, H.J.B.; Massarani, G; Biscaia, E.C., Jr. Remediation of sandy soils using surfactant solutions and foams. J. Hazard. Mater. 2009, 164, 1325–1334. [Google Scholar]
- Seo, H.G.; Lee, M.H.; Chung, S.Y. Study of surfactant enhanced remediation methods for organic pollutant distributed over the heterogeneous medium. (In Korean)J. KoSSGE. 2001, 6, 51–59. [Google Scholar]
- Lee, M.H.; Kang, H.M.; Do, W.H. Application of nonionic surfactant-enhanced in situ flushing to a diesel contaminated site. Water Res. 2005, 39, 139–146. [Google Scholar]
- Song, S.Y.; Kwon, H.J.; Yang, S.Y.; Kim, H.K. Removal of volatile organic contaminant from specific depth in aquifer using selective surfactant-enhanced air sparging. (In Korean)Econ. Environ. Geol. 2010, 43, 565–571. [Google Scholar]
- U.S. Environmental Protection Agency. A technology assessment of soil vapor extraction and air sparging; EPA/600/R-92/173; Office of research and development: Washington, DC, USA, 1992.
- Johnston, C.D.; Rayner, J.L.; Briegel, D. Effectiveness of in situ air sparging for removing NAPL gasoline from a sandy aquifer near Perth, Western Australia. J. Contam. Hydrol. 2002, 59, 87–111. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Kwon, H. Surface tension-water saturation relationship as the function of soil particle size and aquifer depth during groundwater air sparging. (In Korean)J. KoSSGE. 2009, 14, 65–72. [Google Scholar]
- Bass, D.H.; Hasting, N.A.; Brown, R.A. Performance of air sparing systems: A review of case studied. J. Haz. Mat. 2000, 72, 101–119. [Google Scholar] [CrossRef]
- Johnston, C.D.; Rayner, J.L.; Patterson, B.M.; Davis, G.B. Volatilization and biodegradation during air sparging of dissolved BTEX-contaminated ground water. J. Contam. Hydrol. 1998, 33, 377–404. [Google Scholar] [CrossRef]
- Rothmel, R.K.; Peters, R.W.; Martin, E.; Deflaun, M.F. Surfactant foam/bioaugmentation technology for in situ treatment of TCE-DNAPLs. Environ. Sci. Technol. 1998, 32, 1667–1675. [Google Scholar] [CrossRef]
- Zhang, C.U.; Werth, C.J.; Webb, A.G. Investigation of surfactant-enhanced mass removal and flux reduction in 3D correlated permeability fields using magnetic resonance imaging. J. Contam. Hydrol. 2008, 100, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Atteia, O.; Del Campo Estrada, E.; Bertin, H. Soil flushing: A review of the origin of efficiency variability. Rev. Environ. Sci. Biotechnol. 2013, 12, 379–389. [Google Scholar]
- Ministry of Environment in Korea. Soil Environmental Law. (In Korean). Available online: http://www.law.go.kr (accessed on 10 March 2008).
- Ransohoff, T.C.; Radke, C.J. Mechanisms of foam generation in gas-bead packs. SPE Reserv Eng. 1988, 3, 573–585. [Google Scholar]
- Tanzil, D.; Hirasaki, G.J.; Miller, C.A. Conditions for foam generation in homogeneous porous media. In Proceedings of SPA/DOE Improved Oil Recovery Symposium, Tulsa, OK, USA, 13–17 April 2002.
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lee, H.; Lee, Y.; Kim, J.; Kim, C. Field Application of Modified In Situ Soil Flushing in Combination with Air Sparging at a Military Site Polluted by Diesel and Gasoline in Korea. Int. J. Environ. Res. Public Health 2014, 11, 8806-8824. https://doi.org/10.3390/ijerph110908806
Lee H, Lee Y, Kim J, Kim C. Field Application of Modified In Situ Soil Flushing in Combination with Air Sparging at a Military Site Polluted by Diesel and Gasoline in Korea. International Journal of Environmental Research and Public Health. 2014; 11(9):8806-8824. https://doi.org/10.3390/ijerph110908806
Chicago/Turabian StyleLee, Hwan, Yoonjin Lee, Jaeyoung Kim, and Choltae Kim. 2014. "Field Application of Modified In Situ Soil Flushing in Combination with Air Sparging at a Military Site Polluted by Diesel and Gasoline in Korea" International Journal of Environmental Research and Public Health 11, no. 9: 8806-8824. https://doi.org/10.3390/ijerph110908806
APA StyleLee, H., Lee, Y., Kim, J., & Kim, C. (2014). Field Application of Modified In Situ Soil Flushing in Combination with Air Sparging at a Military Site Polluted by Diesel and Gasoline in Korea. International Journal of Environmental Research and Public Health, 11(9), 8806-8824. https://doi.org/10.3390/ijerph110908806




