New Immunotherapy Strategies in Breast Cancer
Abstract
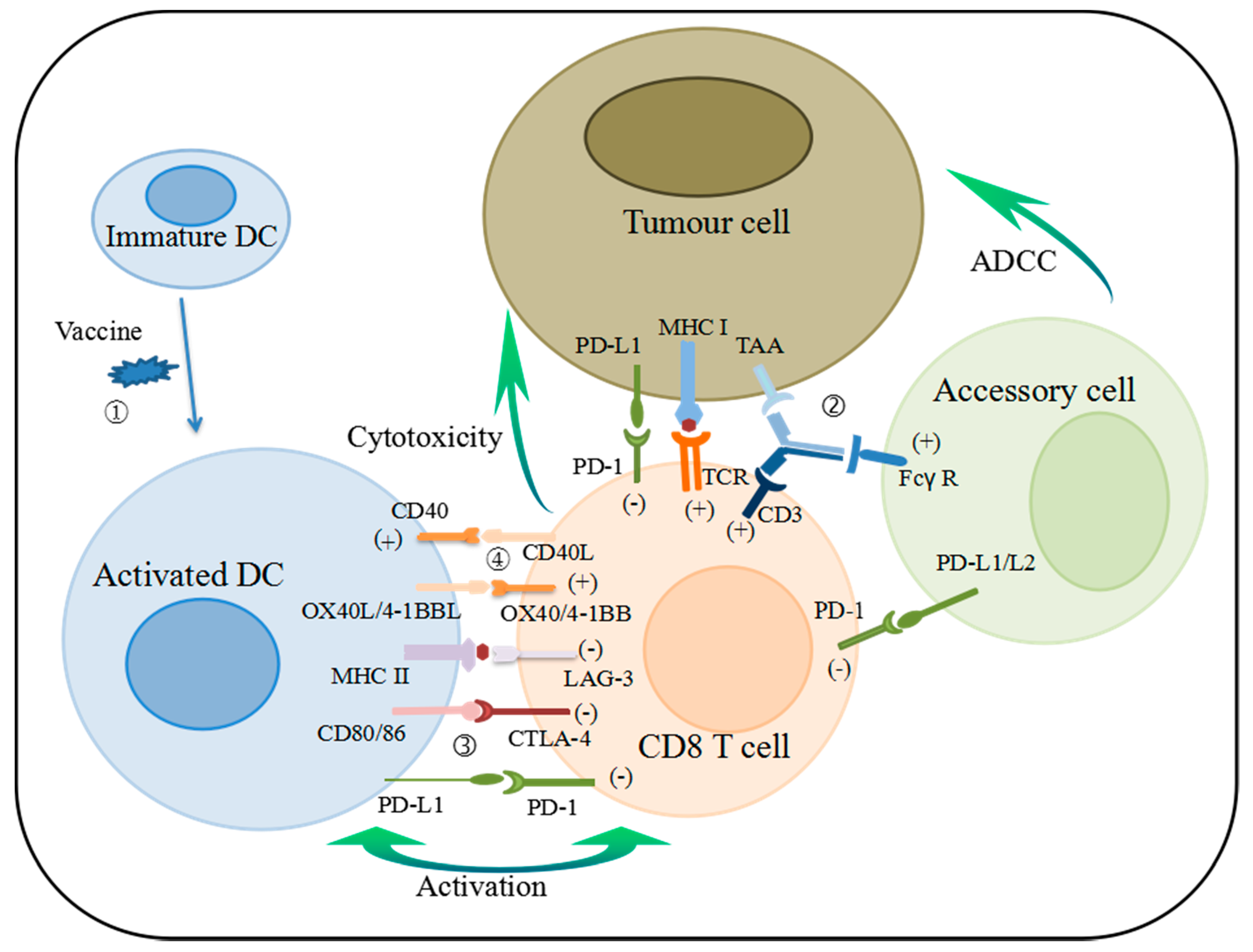
:1. Introduction
2. Cancer Vaccines
2.1. Antigen-Specific Vaccines
2.1.1. HER2-Derived Vaccines
2.1.2. MUC1-Derived Vaccines
2.2. Cell-Based Vaccines
3. Bispecific Antibodies
4. Immune Checkpoint Therapy
4.1. CTLA-4 Inhibitors
4.2. PD-1/PD-L1 Inhibitors
4.3. LAG-3 Target Therapy
5. Stimulatory Molecule Agonist Antibodies
5.1. OX40 Agonist Antibodies
5.2. 4-1BB Agonist Antibodies
5.3. CD40 Agonist Antibodies
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Chlebowski, R.T.; Manson, J.E.; Anderson, G.L.; Cauley, J.A.; Aragaki, A.K.; Stefanick, M.L.; Lane, D.S.; Johnson, K.C.; Wactawski-Wende, J.; Chen, C.; et al. Estrogen plus progestin and breast cancer incidence and mortality in the women’s health initiative observational study. J. Natl. Cancer Inst. 2013, 105, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Coley, H.M. Mechanisms and strategies to overcome chemotherapy resistance in metastatic breast cancer. Cancer Treat. Rev. 2008, 34, 378–390. [Google Scholar] [CrossRef] [PubMed]
- Foukakis, T.; Fornander, T.; Lekberg, T.; Hellborg, H.; Adolfsson, J.; Bergh, J. Age-specific trends of survival in metastatic breast cancer: 26 Years longitudinal data from a population-based cancer registry in Stockholm, Sweden. Breast Cancer Res. Treat. 2011, 130, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Emens, L.A. Breast cancer immunobiology driving immunotherapy: Vaccines and immune checkpoint blockade. Expert Rev. Anticancer Ther. 2012, 12, 1597–1611. [Google Scholar] [CrossRef] [PubMed]
- Kiewe, P.; Hasmuller, S.; Kahlert, S.; Heinrigs, M.; Rack, B.; Marme, A.; Korfel, A.; Jager, M.; Lindhofer, H.; Sommer, H.; et al. Phase I trial of the trifunctional anti-HER2 × anti-CD3 antibody ertumaxomab in metastatic breast cancer. Clin. Cancer Res. 2006, 12, 3085–3091. [Google Scholar] [CrossRef] [PubMed]
- Iwamoto, T.; Bianchini, G.; Booser, D.; Qi, Y.; Coutant, C.; Shiang, C.Y.; Santarpia, L.; Matsuoka, J.; Hortobagyi, G.N.; Symmans, W.F.; et al. Gene pathways associated with prognosis and chemotherapy sensitivity in molecular subtypes of breast cancer. J. Natl. Cancer Inst. 2011, 103, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Gingras, I.; Azim, H.A., Jr.; Ignatiadis, M.; Sotiriou, C. Immunology and breast cancer: Toward a new way of understanding breast cancer and developing novel therapeutic strategies. Clin. Adv. Hematol. Oncol. 2015, 13, 372–382. [Google Scholar] [PubMed]
- Coventry, B.J.; Weightman, M.J.; Bradley, J.; Skinner, J.M. Immune profiling in human breast cancer using high-sensitivity detection and analysis techniques. JRSM Open 2015, 6, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Ruffell, B.; Au, A.; Rugo, H.S.; Esserman, L.J.; Hwang, E.S.; Coussens, L.M. Leukocyte composition of human breast cancer. Proc. Natl. Acad. Sci. USA 2012, 109, 2796–2801. [Google Scholar] [CrossRef] [PubMed]
- Pardoll, D.M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 2012, 12, 252–264. [Google Scholar] [CrossRef] [PubMed]
- Yamane, H.; Paul, W.E. Cytokines of the γc family control CD4+ T-cell differentiation and function. Nat. Immunol. 2012, 13, 1037–1044. [Google Scholar] [CrossRef]
- Espinosa, V.; Rivera, A. Cytokines and the regulation of fungus-specific CD4 T-cell differentiation. Cytokine 2012, 58, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Raju, K.; Rabinovich, B.A.; Radvanyi, L.G.; Spaner, D.; Miller, R.G. A central role for IL-2 in fate determination of mature T-cells—I: Role in determining the Th1/Th2 profile in primary T-cell cultures. Int. Immunol. 2001, 13, 1453–1459. [Google Scholar] [CrossRef] [PubMed]
- Forbes, E.; van Panhuys, N.; Min, B.; le Gros, G. Differential requirements for IL-4/STAT6 signalling in CD4 T-cell fate determination and TH2-immune effector responses. Immunol. Cell Biol. 2010, 88, 240–243. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.Y.; Choi, B.K.; Lee, D.G.; Kim, Y.H.; Kim, C.H.; Lee, S.J.; Kwon, B.S. 4-1BB signaling activates the T-cell factor 1 effector/beta-catenin pathway with delayed kinetics via ERK signaling and delayed PI3K/AKT activation to promote the proliferation of CD8+ T-cells. PLoS ONE 2013, 8, e69677. [Google Scholar]
- Schreiber, R.D.; Old, L.J.; Smyth, M.J. Cancer immunoediting: Integrating immunity’s roles in cancer suppression and promotion. Science 2011, 331, 1565–1570. [Google Scholar] [CrossRef] [PubMed]
- Drake, C.G.; Lipson, E.J.; Brahmer, J.R. Breathing new life into immunotherapy: Review of melanoma, lung and kidney cancer. Nat. Rev. Clin. Oncol. 2014, 11, 24–37. [Google Scholar] [CrossRef] [PubMed]
- Coley, W.B. The treatment of inoperable sarcoma by bacterial toxins (the mixed toxins of the streptococcus erysipelas and the bacillus prodigiosus). Proc. R. Soc. Med. 1910, 3, 1–48. [Google Scholar] [PubMed]
- Disis, M.L.; Pupa, S.M.; Gralow, J.R.; Dittadi, R.; Menard, S.; Cheever, M.A. High-titer HER-2/NEU protein-specific antibody can be detected in patients with early-stage breast cancer. J. Clin. Oncol. 1997, 15, 3363–3367. [Google Scholar] [PubMed]
- Miles, D.; Papazisis, K. Rationale for the clinical development of STN-KLH (theratope) and anti-MUC-1 vaccines in breast cancer. Clin. Breast Cancer 2003, 3, S134–S138. [Google Scholar] [CrossRef] [PubMed]
- Mittendorf, E.A.; Clifton, G.T.; Holmes, J.P.; Schneble, E.; van Echo, D.; Ponniah, S.; Peoples, G.E. Final report of the phase I/II clinical trial of the E75 (nelipepimut-S) vaccine with booster inoculations to prevent disease recurrence in high-risk breast cancer patients. Ann. Oncol. 2014, 25, 1735–1742. [Google Scholar] [CrossRef] [PubMed]
- Sears, A.K.; Perez, S.A.; Clifton, G.T.; Benavides, L.C.; Gates, J.D.; Clive, K.S.; Holmes, J.P.; Shumway, N.M.; Van Echo, D.C.; Carmichael, M.G.; et al. AE37: A novel T-cell-eliciting vaccine for breast cancer. Expert Opin. Biol. Ther. 2011, 11, 1543–1550. [Google Scholar] [CrossRef] [PubMed]
- Disis, M.L.; Wallace, D.R.; Gooley, T.A.; Dang, Y.; Slota, M.; Lu, H.; Coveler, A.L.; Childs, J.S.; Higgins, D.M.; Fintak, P.A.; et al. Concurrent trastuzumab and HER2/NEU-specific vaccination in patients with metastatic breast cancer. J. Clin. Oncol. 2009, 27, 4685–4692. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, E.; Blackwell, K.; Hobeika, A.C.; Clay, T.M.; Broadwater, G.; Ren, X.R.; Chen, W.; Castro, H.; Lehmann, F.; Spector, N.; et al. Phase 1 clinical trial of HER2-specific immunotherapy with concomitant Her2 kinase inhibition [corrected]. J. Transl. Med. 2012, 10, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Kimura, T.; Finn, O.J. MUC1 immunotherapy is here to stay. Expert Opin. Biol. Ther. 2013, 13, 35–49. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, N.K.; Murray, J.L.; Zhou, D.; Mittendorf, E.A.; Sample, D.; Tautchin, M.; Miles, D. Survival advantage in patients with metastatic breast cancer receiving endocrine therapy plus sialyl TN-KLH vaccine: Post Hoc analysis of a large randomized trial. J. Cancer 2013, 4, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Mohebtash, M.; Tsang, K.Y.; Madan, R.A.; Huen, N.Y.; Poole, D.J.; Jochems, C.; Jones, J.; Ferrara, T.; Heery, C.R.; Arlen, P.M.; et al. A pilot study of MUC-1/CEA/tricom poxviral-based vaccine in patients with metastatic breast and ovarian cancer. Clin. Cancer Res. 2011, 17, 7164–7173. [Google Scholar] [CrossRef] [PubMed]
- Degregorio, M.; Degregorio, M.; Wurz, G.T.; Wurz, G.T.; Gutierrez, A.; Gutierrez, A.; Wolf, M. L-BLP25 vaccine plus letrozole for breast cancer: Is translation possible? Oncoimmunology 2012, 1, 1422–1424. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Degliangeli, F.; Palitzsch, B.; Gerlitzki, B.; Kunz, H.; Schmitt, E.; Fiammengo, R.; Westerlind, U. Glycopeptide-functionalized gold nanoparticles for antibody induction against the tumor associated mucin-1 glycoprotein. Bioorgan. Med. Chem. 2016, 24, 1132–1135. [Google Scholar] [CrossRef] [PubMed]
- Palucka, K.; Banchereau, J. Cancer immunotherapy via dendritic cells. Nat. Rev. Cancer 2012, 12, 265–277. [Google Scholar] [CrossRef] [PubMed]
- Park, J.W.; Melisko, M.E.; Esserman, L.J.; Jones, L.A.; Wollan, J.B.; Sims, R. Treatment with autologous antigen-presenting cells activated with the HER-2 based antigen lapuleucel-T: Results of a phase I study in immunologic and clinical activity in HER-2 overexpressing breast cancer. J. Clin. Oncol. 2007, 25, 3680–3687. [Google Scholar] [CrossRef] [PubMed]
- Met, O.; Balslev, E.; Flyger, H.; Svane, I.M. High immunogenic potential of p53 mRNA-transfected dendritic cells in patients with primary breast cancer. Breast Cancer Res. Treat. 2011, 125, 395–406. [Google Scholar] [CrossRef] [PubMed]
- Svane, I.M.; Pedersen, A.E.; Johansen, J.S.; Johnsen, H.E.; Nielsen, D.; Kamby, C.; Ottesen, S.; Balslev, E.; Gaarsdal, E.; Nikolajsen, K.; et al. Vaccination with p53 peptide-pulsed dendritic cells is associated with disease stabilization in patients with p53 expressing advanced breast cancer; monitoring of serum YKL-40 and IL-6 as response biomarkers. Cancer Immunol. Immunother. 2007, 56, 1485–1499. [Google Scholar] [CrossRef] [PubMed]
- Hollander, N. Bispecific antibodies for cancer therapy. Immunotherapy 2009. [Google Scholar] [CrossRef] [PubMed]
- Heiss, M.M.; Strohlein, M.A.; Jager, M.; Kimmig, R.; Burges, A.; Schoberth, A.; Jauch, K.W.; Schildberg, F.W.; Lindhofer, H. Immunotherapy of malignant ascites with trifunctional antibodies. Int. J. Cancer 2005, 117, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Buie, L.W.; Pecoraro, J.J.; Horvat, T.Z.; Daley, R.J. Blinatumomab: A first-in-class bispecific T-cell engager for precursor B-cell acute lymphoblastic leukemia. Ann. Pharmacother. 2015, 49, 1057–1067. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Axup, J.Y.; Ma, J.S.; Wang, R.E.; Choi, S.; Tardif, V.; Lim, R.K.; Pugh, H.M.; Lawson, B.R.; Welzel, G.; et al. Multiformat T-cell-engaging bispecific antibodies targeting human breast cancers. Angew. Chem. Int. Ed. 2015, 54, 7022–7027. [Google Scholar] [CrossRef] [PubMed]
- Jager, M.; Schoberth, A.; Ruf, P.; Hess, J.; Lindhofer, H. The trifunctional antibody ertumaxomab destroys tumor cells that express low levels of human epidermal growth factor receptor 2. Cancer Res. 2009, 69, 4270–4276. [Google Scholar] [CrossRef] [PubMed]
- Lum, L.G.; Thakur, A.; Al-Kadhimi, Z.; Colvin, G.A.; Cummings, F.J.; Legare, R.D.; Dizon, D.S.; Kouttab, N.; Maizel, A.; Colaiace, W.; et al. Targeted T-cell therapy in stage iv breast cancer: A phase I clinical trial. Clin. Cancer Res. 2015, 21, 2305–2314. [Google Scholar] [CrossRef] [PubMed]
- Hacohen, N.; Fritsch, E.F.; Carter, T.A.; Lander, E.S.; Wu, C.J. Getting personal with neoantigen-based therapeutic cancer vaccines. Cancer Immunol. Res. 2013, 1, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Topalian, S.L.; Drake, C.G.; Pardoll, D.M. Immune checkpoint blockade: A common denominator approach to cancer therapy. Cancer Cell 2015, 27, 450–461. [Google Scholar] [CrossRef] [PubMed]
- Ghasemzadeh, A.; Bivalacqua, T.J.; Hahn, N.M.; Drake, C.G. New strategies in bladder cancer: A second coming for immunotherapy. Clin. Cancer Res. 2015, 22, 793–801. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, H.; Nose, M.; Hiai, H.; Minato, N.; Honjo, T. Development of lupus-like autoimmune diseases by disruption of the PD-1 gene encoding an itim motif-carrying immunoreceptor. Immunity 1999, 11, 141–151. [Google Scholar] [CrossRef]
- Gibson, J. Anti-PD-L1 for metastatic triple-negative breast cancer. Lancet Oncol. 2015, 16, e264. [Google Scholar] [CrossRef]
- Hodi, F.S.; O’Day, S.J.; McDermott, D.F.; Weber, R.W.; Sosman, J.A.; Haanen, J.B.; Gonzalez, R.; Robert, C.; Schadendorf, D.; Hassel, J.C.; et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 2010, 363, 711–723. [Google Scholar] [CrossRef] [PubMed]
- Powles, T.; Eder, J.P.; Fine, G.D.; Braiteh, F.S.; Loriot, Y.; Cruz, C.; Bellmunt, J.; Burris, H.A.; Petrylak, D.P.; Teng, S.L.; et al. MPDL3280A (anti-PD-L1) treatment leads to clinical activity in metastatic bladder cancer. Nature 2014, 515, 558–562. [Google Scholar] [CrossRef] [PubMed]
- Sgambato, A.; Casaluce, F.; Sacco, P.C.; Palazzolo, G.; Maione, P.; Rossi, A.; Ciardiello, F.; Gridelli, C. Anti PD-1 and PDL-1 immunotherapy in the treatment of advanced non- small cell lung cancer (NSCLC): A review on toxicity profile and its management. Curr. Drug Saf. 2016, 11, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Leach, D.R.; Krummel, M.F.; Allison, J.P. Enhancement of antitumor immunity by CTLA-4 blockade. Science 1996, 271, 1734–1736. [Google Scholar] [CrossRef] [PubMed]
- Linsley, P.S.; Greene, J.L.; Brady, W.; Bajorath, J.; Ledbetter, J.A.; Peach, R. Human B7-1 (CD80) and B7-2 (CD86) bind with similar avidities but distinct kinetics to CD28 and CTLA-4 receptors. Immunity 1994, 1, 793–801. [Google Scholar] [CrossRef]
- Maker, A.V.; Attia, P.; Rosenberg, S.A. Analysis of the cellular mechanism of antitumor responses and autoimmunity in patients treated with CTLA-4 blockade. J. Immunol. 2005, 175, 7746–7754. [Google Scholar] [CrossRef] [PubMed]
- Simpson, T.R.; Li, F.; Montalvo-Ortiz, W.; Sepulveda, M.A.; Bergerhoff, K.; Arce, F.; Roddie, C.; Henry, J.Y.; Yagita, H.; Wolchok, J.D.; et al. Fc-dependent depletion of tumor-infiltrating regulatory T-cells co-defines the efficacy of anti-CTLA-4 therapy against melanoma. J. Exp. Med. 2013, 210, 1695–1710. [Google Scholar] [CrossRef] [PubMed]
- Vonderheide, R.H.; LoRusso, P.M.; Khalil, M.; Gartner, E.M.; Khaira, D.; Soulieres, D.; Dorazio, P.; Trosko, J.A.; Ruter, J.; Mariani, G.L.; et al. Tremelimumab in combination with exemestane in patients with advanced breast cancer and treatment-associated modulation of inducible costimulator expression on patient T-cells. Clin. Cancer Res. 2010, 16, 3485–3494. [Google Scholar] [CrossRef] [PubMed]
- Wimberly, H.; Brown, J.R.; Schalper, K.; Haack, H.; Silver, M.R.; Nixon, C.; Bossuyt, V.; Pusztai, L.; Lannin, D.R.; Rimm, D.L. PD-L1 expression correlates with tumor-infiltrating lymphocytes and response to neoadjuvant chemotherapy in breast cancer. Cancer Immunol. Res. 2015, 3, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Soliman, H.; Khalil, F.; Antonia, S. PD-L1 expression is increased in a subset of basal type breast cancer cells. PLoS ONE 2014, 9, e88557. [Google Scholar] [CrossRef] [PubMed]
- Sabatier, R.; Finetti, P.; Mamessier, E.; Adelaide, J.; Chaffanet, M.; Ali, H.R.; Viens, P.; Caldas, C.; Birnbaum, D.; Bertucci, F. Prognostic and predictive value of PDL1 expression in breast cancer. Oncotarget 2015, 6, 5449–5464. [Google Scholar] [CrossRef] [PubMed]
- Herbst, R.S.; Soria, J.C.; Kowanetz, M.; Fine, G.D.; Hamid, O.; Gordon, M.S.; Sosman, J.A.; McDermott, D.F.; Powderly, J.D.; Gettinger, S.N.; et al. Predictive correlates of response to the anti-PD-l1 antibody MPDL3280A in cancer patients. Nature 2014, 515, 563–567. [Google Scholar] [CrossRef] [PubMed]
- Josefowicz, S.Z.; Lu, L.F.; Rudensky, A.Y. Regulatory T-cells: Mechanisms of differentiation and function. Annu. Rev. Immunol. 2012, 30, 531–564. [Google Scholar] [CrossRef] [PubMed]
- Topalian, S.L.; Sznol, M.; McDermott, D.F.; Kluger, H.M.; Carvajal, R.D.; Sharfman, W.H.; Brahmer, J.R.; Lawrence, D.P.; Atkins, M.B.; Powderly, J.D.; et al. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J. Clin. Oncol. 2014, 32, 1020–1030. [Google Scholar] [CrossRef] [PubMed]
- Josefsson, A.; Nedrow, J.R.; Park, S.; Banerjee, S.R.; Rittenbach, A.; Jammes, F.; Tsui, B.; Sgouros, G. Imaging, biodistribution, and dosimetry of radionuclide-labeled PD-L1 antibody in an immunocompetent mouse model of breast cancer. Cancer Res. 2016, 76, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Loi, S.; Dushyanthen, S.; Beavis, P.A.; Salgado, R.; Denkert, C.; Savas, P.; Combs, S.; Rimm, D.L.; Giltnane, J.M.; Estrada, M.V.; et al. RAS/MAPK activation is associated with reduced tumor-infiltrating lymphocytes in triple-negative breast cancer: Therapeutic cooperation between mek and PD-1/PD-L1 immune checkpoint inhibitors. Clin. Cancer Res. 2015, 22, 1499–1509. [Google Scholar] [CrossRef] [PubMed]
- Black, M.; Barsoum, I.B.; Truesdell, P.; Cotechini, T.; Macdonald-Goodfellow, S.K.; Petroff, M.; Siemens, D.R.; Koti, M.; Craig, A.W.; Graham, C.H. Activation of the PD-1/PD-L1 immune checkpoint confers tumor cell chemoresistance associated with increased metastasis. Oncotarget 2016, 7, 10557–10567. [Google Scholar] [PubMed]
- Nanda, R.; Chow, L.Q.; Claire Dees, E.; Berger, R.; Gupta, S.; Geva, R.; Pusztai, L.; Dolled-Filhart, M.; Emancipator, K.; Gonzalez, E.J.; et al. Abstract S1-09: A phase IB study of pembrolizumab (MK-3475) in patients with advanced triple-negative breast cancer. Cancer Res. 2015, 75. [Google Scholar] [CrossRef]
- Rugo, H.S.; Delord, J.P.; Im, S.A.; Ott, P.A.; Pihapaul, S.A.; Bedard, P.L.; Sachdev, J.; Le Tourneau, C.; van Brummelen, E.; Varga, A.; et al. Abstract s5-07: Preliminary efficacy and safety of pembrolizumab (MK-3475) in patients with PD-L1-positive, estrogen receptor-positive (Er+)/HER2-negative advanced breast cancer enrolled in keynote-028. Cancer Res. 2016, 76. [Google Scholar] [CrossRef]
- Adams, S.; Diamond, J.; Hamilton, E.; Pohlmann, P.; Tolaney, S.; Molinero, L.; Zou, W.; Liu, B.; Waterkamp, D.; Funke, R.; et al. Abstract P2-11-06: Safety and clinical activity of atezolizumab (anti-PDL1) in combination with nab-paclitaxel in patients with metastatic triple-negative breast cancer. Cancer Res. 2016, 76. [Google Scholar] [CrossRef]
- Dirix, L.Y.; Takacs, I.; Nikolinakos, P.; Jerusalem, G.; Arkenau, H.T.; Hamilton, E.P.; von Heydebreck, A.; Grote, H.J.; Chin, K.; Lippman, M.E. Abstract s1-04: Avelumab (Msb0010718c), an anti-PD-L1 antibody, in patients with locally advanced or metastatic breast cancer: A phase IB javelin solid tumor trial. Cancer Res. 2016, 76. [Google Scholar] [CrossRef]
- Huang, C.T.; Workman, C.J.; Flies, D.; Pan, X.; Marson, A.L.; Zhou, G.; Hipkiss, E.L.; Ravi, S.; Kowalski, J.; Levitsky, H.I.; et al. Role of Lag-3 in regulatory T-cells. Immunity 2004, 21, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Brignone, C.; Gutierrez, M.; Mefti, F.; Brain, E.; Jarcau, R.; Cvitkovic, F.; Bousetta, N.; Medioni, J.; Gligorov, J.; Grygar, C.; et al. First-line chemoimmunotherapy in metastatic breast carcinoma: Combination of paclitaxel and IMP321 (Lag-3IG) enhances immune responses and antitumor activity. J. Transl. Med. 2010, 8, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Xie, F.; Wang, Q.; Chen, Y.; Gu, Y.; Mao, H.; Zeng, W.; Zhang, X. Costimulatory molecule ox40/ox40l expression in ductal carcinoma in situ and invasive ductal carcinoma of breast: An immunohistochemistry-based pilot study. Pathol. Res. Pract. 2010, 206, 735–739. [Google Scholar] [CrossRef] [PubMed]
- Aspeslagh, S.; Postel-Vinay, S.; Rusakiewicz, S.; Soria, J.C.; Zitvogel, L.; Marabelle, A. Rationale for anti-OX40 cancer immunotherapy. Eur. J. Cancer 2016, 52, 50–66. [Google Scholar] [CrossRef] [PubMed]
- Kjaergaard, J.; Tanaka, J.; Kim, J.A.; Rothchild, K.; Weinberg, A.; Shu, S. Therapeutic efficacy of OX-40 receptor antibody depends on tumor immunogenicity and anatomic site of tumor growth. Cancer Res. 2000, 60, 5514–5521. [Google Scholar] [PubMed]
- Weinberg, A.D.; Rivera, M.M.; Prell, R.; Morris, A.; Ramstad, T.; Vetto, J.T.; Urba, W.J.; Alvord, G.; Alvord, C.; Shields, J. Engagement of the OX-40 receptor in vivo enhances antitumor immunity. J. Immunol. 2000, 164, 2160–2169. [Google Scholar] [CrossRef] [PubMed]
- Gough, M.J.; Ruby, C.E.; Redmond, W.L.; Dhungel, B.; Brown, A.; Weinberg, A.D. OX40 agonist therapy enhances CD8 infiltration and decreases immune suppression in the tumor. Cancer Res. 2008, 68, 5206–5215. [Google Scholar] [CrossRef] [PubMed]
- Piconese, S.; Valzasina, B.; Colombo, M.P. OX40 triggering blocks suppression by regulatory T-cells and facilitates tumor rejection. J. Exp. Med. 2008, 205, 825–839. [Google Scholar] [CrossRef] [PubMed]
- Curti, B.D.; Kovacsovics-Bankowski, M.; Morris, N.; Walker, E.; Chisholm, L.; Floyd, K.; Walker, J.; Gonzalez, I.; Meeuwsen, T.; Fox, B.A.; et al. OX40 is a potent immune-stimulating target in late-stage cancer patients. Cancer Res. 2013, 73, 7189–7198. [Google Scholar] [CrossRef] [PubMed]
- Watts, T.H. TNF/TNFR family members in costimulation of T-cell responses. Annu. Rev. Immunol. 2005, 23, 23–68. [Google Scholar] [CrossRef] [PubMed]
- Shuford, W.W.; Klussman, K.; Tritchler, D.D.; Loo, D.T.; Chalupny, J.; Siadak, A.W.; Brown, T.J.; Emswiler, J.; Raecho, H.; Larsen, C.P.; et al. 4-1BB costimulatory signals preferentially induce CD8+ T-cell proliferation and lead to the amplification in vivo of cytotoxic T-cell responses. J. Exp. Med. 1997, 186, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Melero, I.; Johnston, J.V.; Shufford, W.W.; Mittler, R.S.; Chen, L. Nk1.1 cells express 4-1BB (CDW137) costimulatory molecule and are required for tumor immunity elicited by anti-4-1BB monoclonal antibodies. Cell. Immunol. 1998, 190, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Ascierto, P.A.; Simeone, E.; Sznol, M.; Fu, Y.X.; Melero, I. Clinical experiences with anti-CD137 and anti-PD1 therapeutic antibodies. Semin. Oncol. 2010, 37, 508–516. [Google Scholar] [CrossRef] [PubMed]
- Kohrt, H.E.; Houot, R.; Weiskopf, K.; Goldstein, M.J.; Scheeren, F.; Czerwinski, D.; Colevas, A.D.; Weng, W.K.; Clarke, M.F.; Carlson, R.W.; et al. Stimulation of natural killer cells with a CD137-specific antibody enhances trastuzumab efficacy in xenotransplant models of breast cancer. J. Clin. Investig. 2012, 122, 1066–1075. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moran, A.E.; Kovacsovics-Bankowski, M.; Weinberg, A.D. The Tnfrs OX40, 4-1BB, and CD40 as targets for cancer immunotherapy. Curr. Opin. Immunol. 2013, 25, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Vonderheide, R.H.; Flaherty, K.T.; Khalil, M.; Stumacher, M.S.; Bajor, D.L.; Hutnick, N.A.; Sullivan, P.; Mahany, J.J.; Gallagher, M.; Kramer, A.; et al. Clinical activity and immune modulation in cancer patients treated with CP-870,893, a novel CD40 agonist monoclonal antibody. J. Clin. Oncol. 2007, 25, 876–883. [Google Scholar] [CrossRef] [PubMed]
- Kawaguchi, K.; Suzuki, E.; Yamaguchi, A.; Yamamoto, M.; Morita, S.; Toi, M. Altered expression of major immune regulatory molecules in peripheral blood immune cells associated with breast cancer. Breast Cancer 2016, 24, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Hirano, A.; Longo, D.L.; Taub, D.D.; Ferris, D.K.; Young, L.S.; Eliopoulos, A.G.; Agathanggelou, A.; Cullen, N.; Macartney, J.; Fanslow, W.C.; et al. Inhibition of human breast carcinoma growth by a soluble recombinant human CD40 ligand. Blood 1999, 93, 2999–3007. [Google Scholar] [PubMed]
- Slobodova, Z.; Ehrmann, J.; Krejci, V.; Zapletalova, J.; Melichar, B. Analysis of CD40 expression in breast cancer and its relation to clinicopathological characteristics. Neoplasma 2011, 58, 189–197. [Google Scholar] [CrossRef] [PubMed]
- De Iuliis, F.; Salerno, G.; Taglieri, L.; Scarpa, S. Are pharmacogenomic biomarkers an effective tool to predict taxane toxicity and outcome in breast cancer patients? Literature review. Cancer Chemother. Pharmacol. 2015, 76, 679–690. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.C.; Rha, S.Y.; Jeung, H.; Choi, Y.; Park, K.; Sohn, J.; Kim, C.; Kim, G.E.; Yoo, J.H.; Kim, S. Gemcitabine pathway genotype analysis to predict toxicity in phase II gemcitabine monotherapy in heavily pre-treated metastatic breast cancer. J. Clin. Oncol. 2004, 22, 2066. [Google Scholar]
- Tury, S.; Becette, V.; Assayag, F.; Vacher, S.; Benoist, C.; Kamal, M.; Marangoni, E.; Bièche, I.; Lerebours, F.; Callens, C. Combination of COX-2 expression and PIK3CA mutation as prognostic and predictive markers for celecoxib treatment in breast cancer. Oncotarget 2016. [Google Scholar] [CrossRef] [PubMed]
- Jamieson, D.; Sunter, N.; Muro, S.; Pouche, L.; Cresti, N.; Lee, J.; Sludden, J.; Griffin, M.J.; Allan, J.M.; Verrill, M.W.; et al. Pharmacogenetic association of MBL2 and CD95 polymorphisms with grade 3 infection following adjuvant therapy for breast cancer with doxorubicin and cyclophosphamide. Eur. J. Cancer 2016, 71, 15–24. [Google Scholar] [CrossRef] [PubMed]
| Trial ID | Phase | Breast Cancer Subtype | Primary Endpoint | Study |
|---|---|---|---|---|
| Vaccines | ||||
| NCT02427581 | I | TNBC | Safety | Poly ICLC |
| NCT01730118 | I | HER2+ BC | Cardiac toxicity and anti-HER2 antibody concentration | Autologous Ad HER2 dendritic cell vaccine |
| NCT02018458 | I/II | phase1: LA TNBC; phase 2: ER+/HER2− BC | Safety | DC vaccination + Preoperative chemotherapy |
| NCT01570036 | II | HER2+ BC | DFS | E75 + Trastuzumab |
| NCT02061332 | I/II | BC | Blood pressure, temperature, pulse | HER2 Pulsed Dendritic Cell Vaccine |
| NCT01376505 | I | BC | Immune response and clinical benefit | HER2 vaccine |
| NCT02140996 | I | BC | Safety and tolerability | Ad-sig-hMUC-1/ecdCD40L vector vaccine |
| BsAbs | ||||
| NCT01730612 | I/II | HER2−CEA+ BC | Tumor targeting and signal/noise ratio | TF2 + 68 Ga-IMP-288 |
| CTLA-4 | ||||
| NCT02536794 | II | HER2− BC | Response rate | MEDI4736 + Tremelimumab |
| NCT02381314 | I | TNBC | Adverse event | MGA271 + Ipilimumab |
| PD-1 | ||||
| NCT02661100 | I/II | Advanced TNBC | DLT | Pembrolizumab + CDX-1401 + Poly ICLC |
| NCT02453620 | I | HER2− BC | Adverse event | Entinostat + Ipilimumab + Nivolumab |
| NCT02129556 | I/II | HER2+BC (Trastuzumab-resistant) | DLT | Pembrolizumab |
| NCT02309177 | I | BC | DLT, Safety, Grade 3 or 4 TEAE | Nab-Paclitaxel + Nivolumab + Gemcitabine + Carboplatin |
| NCT02404441 | I/II | TNBC | DLT and ORR | PDR001 |
| NCT02555657 | III | TNBC | PFS and OS | Pembrolizumab + Capecitabine + Eribulin + Gemcitaine + Vinorelbine |
| PD-L1 | ||||
| NCT02643303 | I/II | BC | Phase 1 Safety and tolerability Phase 2 ORR, PFS, OS | Durvalumab + Tremelimumab + Poly ICLC |
| NCT02628132 | I/II | TNBC | Adverse event | Paclitaxel + Durvalumab |
| NCT02685059 | II | TNBC | pCR | Durvalumab + Placebo + nab-Paclitaxel + Epirubicin + Cyclophosphamide |
| NCT02725489 | II/III | TNBC | Tolerability and adverse event | Vigil™ + Durvalumab |
| NCT02425891 | III | Metastatic BC/TNBC | PFS and OS | Atezolizumab + Nab-Paclitaxel + Placebo |
| NCT02478099 | II | TNBC | ORR | Atezolizumab |
| NCT02649686 | I | HER2+ Metastatic BC | Confirm phase II dose | Durvalumab + Trastuzumab |
| NCT02708680 | I/II | Advanced TNBC | DLT, MTD and PFS | Entinostat + Atezolizumab |
| LAG-3 | ||||
| NCT02614833 | II | Stage IV BC | PFS | IMP321 + Placebo + Paclitaxel |
| OX40 | ||||
| NCT01862900 | I/II | Metastatic BC | DLT and safety profile | MEDI6469 |
| 4-1BB | ||||
| NCT02554812 | II | TNBC | DLT and ORR | PF-05082566 + Avelumab |
| Phase | Breast Cancer Subtype | n | Study | Immune-Related Response | Clinical Benefit | Reference |
|---|---|---|---|---|---|---|
| Vaccines | ||||||
| I/II | HER2+ BC | 195 | E75 + GM-CSF | All patients developed a DTH response to E75 after vaccination, and that DTH reactions were dose dependent | Toxicities were mild; improved 5-year DFS | [22] |
| II | Early stage BC | 206 | AE37 + GM-CSF | Increase in DTH response to AE37, decrease in CD4+CD25high CD127low regulatory T-cells | A reduction in recurrence | [23] |
| I/II | Stage IV HER2+ MBC | 22 | HER2 vaccine + Trastuzumab | Increase the HER2-specific immune responses | Well tolerated | [24] |
| I | HER2+ BC (trastuzumab-refractory) | 12 | HER2 vaccine + Lapatinib | Anti-HER2-specific antibodies and HER2-specific T-cells were induced in 100% and 8% of patients respectively | Well tolerated; no objective clinical responses | [25] |
| III | MBC | 1208 | Theratope + Endocrine | Antibody response to theratope | Longer TTP and OS than control group | [27] |
| I | MBC | 12 | PANVAC | Limited tumor burden, better CD4 response or higher number of CEA specific T-cells appeared to benefit from this vaccine | 33% SD and 8% CR | [28] |
| I | HER2+ MBC | 18 | Lapuleucel-T | Significant HER2-specific T-cell proliferation | Without grade 3 or 4 adverse events; 5.5% PR, 16.6% experienced SD lasting >1 years | [32] |
| II | MBC | 26 | P53 DC vaccine | The efficacy was associated with tumor p53 expression, p53 specific T-cells and serum YKL-40 and IL-6 levels | 8/19 evaluable patients attained SD | [34] |
| BsAbs | ||||||
| I | HER2+ MBC | 15 | Ertumaxomab | A strong T helper cell type 1-associated immune response | Most drug-related adverse events were mild; The ORR was 33% | [6,39] |
| I | MBC | 23 | Anti-CD3/anti-HER2 BsAb armed ATC along with low-dose IL-2 and GM-CSF | Induce both PBMC specific anti-SK-BR-3 and innate immune responses | No dose-limiting toxicities was observed; 59.1% evaluable patients had SD or better, and the median OS was 36.2 months | [41] |
| CTLA-4 | ||||||
| I | MBC | 26 | Tremelimumab + Exemestane | Treatment was associated with increased peripheral CD4+ and CD8+ T-cells expressing ICOS and a marked increase in the ratio of ICOS+ T-cells to FoxP3+ regulatory T-cells. | Tolerable, and 42% patients experienced SD lasting ≥12 weeks. | [54] |
| PD-1/PD-L1 | ||||||
| I | PD-L1+ mTNBC | 32 | Pembrolizumab | NR | 15.6% experienced at least one drug-related serious adverse event; 16.1% PR, 9.7% SD | [64] |
| I | PD-L1+ TNBC | 21 | Atezolizumab | Treatment was associated with increased plasma cytokine concentrations and proliferating CD8 cells | 24% ORs, 29% patients had PFS of 24 weeks or longer; several adverse reactions | [46] |
| I | mTNBC | 11 | Atezolizumab + Nab-paclitaxel | NR | Tolerable, 4 PRs and 1 SD | [66] |
| I | Locally MBC | 168 | Avelumab | NR | Among all patients with PD-L1 expressing, 33.3% (4 of 12) had PRs. | [67] |
| LAG-3 | ||||||
| I/II | MBC | 30 | IMP321 + paclitaxel | Increase the number of activated APC, percentage of NK and long-lived cytotoxic effector-memory CD8 T-cells | ORR was 50%, and clinical benefit was noted in 90% in 6 months with no clinically significant IMP321-related adverse events | [69] |
| OX40 | ||||||
| I | Advanced cancer (refractory to conventional therapy) | 30 | 9B12 | Immunologic effects were increased including proliferation of circulation CD4 and CD8 T-cells, responses to recall and naive reporter antigens, and endogenous tumor-specific immune responses | Induced the regression of at least one metastatic lesion in 40% patients | [76] |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, L.-Y.; Tang, J.; Zhang, C.-M.; Zeng, W.-J.; Yan, H.; Li, M.-P.; Chen, X.-P. New Immunotherapy Strategies in Breast Cancer. Int. J. Environ. Res. Public Health 2017, 14, 68. https://doi.org/10.3390/ijerph14010068
Yu L-Y, Tang J, Zhang C-M, Zeng W-J, Yan H, Li M-P, Chen X-P. New Immunotherapy Strategies in Breast Cancer. International Journal of Environmental Research and Public Health. 2017; 14(1):68. https://doi.org/10.3390/ijerph14010068
Chicago/Turabian StyleYu, Lin-Yu, Jie Tang, Cong-Min Zhang, Wen-Jing Zeng, Han Yan, Mu-Peng Li, and Xiao-Ping Chen. 2017. "New Immunotherapy Strategies in Breast Cancer" International Journal of Environmental Research and Public Health 14, no. 1: 68. https://doi.org/10.3390/ijerph14010068





