1. Introduction
The dangers of excessive ultraviolet (UV) radiation to the human body has prompted the use of cosmetic products, such as sunscreen creams, lotions, etc., containing UV-absorbing chemicals (UV filters) that block UV light mainly through physical function. The most commonly used UV filters are composed of benzophenone-2 (BP-2), benzophenone-3 (BP-3), benzophenone-4 (BP-4), and 4-methylbenzylidene camphor (4-MBC), which block UV light mainly through physical function. In the production and consumption processes, these substances are released into the environment in two ways: wastewater discharge in the production process or by showering and swimming. In a study conducted in Spain, at least one kind of UV filter was detected in 51 water samples from 17 pools and hot springs [
1]. In the southeastern part of Brazil, UV filters have been detected in samples from six drinking water treatment plants at different time points from June to December, where the BP-3 concentration ranged from 18.00 to 115.00 ng/L [
2]. In addition, studies have shown that indoor dust also contained UV filters, where the concentrations of BP-1, BP-2, and BP-3 ranged from 80.00 to 600.00 ng/g [
3]. Since the hydrophobicity and degradation rate of UV filters are poor, these compounds are likely to accumulate in the environment and produce harmful effects. Reportedly, the half maximal effective concentration at 48.h (48 h-EC
50) of 4-MBC in the small planktonic crustacean
Daphnia magna was 0.56 mg/L and concentrations as low as 50.00 μg/L produced negative effects on the reproduction of
D. magna [
4]. Studies have shown that UV filters impact biological reproduction. For example, BP-2 was shown to interfere with expression of the precursor egg yolk protein vitellogenin in male fathead minnows (
Pimephales promelas) [
5].
However, few studies have investigated the ecotoxicological effects of these substances, thus they have not been listed in any environmental criteria and discharge standards. Furthermore, it is reported that most processes in sewage treatment plants cannot remove UV filter chemicals effectively [
6], therefore, the discharge of these chemicals is still uncontrolled. Although they are low concentration chemicals, the great mount discharging and unknown fates in the aquatic ecosystem should alarm us to pay attention to them. In our previous study [
7], the acute toxic effects of Benzophenone (BP) on three species of organisms were tested and also the mixing toxic with
N,N-diethyl-3-methylbenzamide (DEET). The acute toxicity of BP to the 3 tested organisms were classified as high or medium-level toxicity [
7]. The results sparked our interests to study the toxicity of all BPs series chemicals. BPs series chemicals include 14 BP-types which are used in UV protection products [
8]. Furthermore, Benzophenone (BP-1), BP-3, BP-4, 2,4,4’-Trihydroxy benzophenone (BP-8) have been frequently detected within multiple environmental matrices [
9,
10,
11]. In the present study, the results about the toxic effects of BP-3 and BP-4 to aquatic ecosystems and the consequent environmental risk assessment are discussed.
BP-3, a broad-spectrum UV absorber, can effectively absorb UV light at 290–440 nm, but almost does not absorb visible light. BP-3 can be absorbed through the mouth and skin of humans because of its lipophilicity, light stability, and bioaccumulation [
9]. As one of the most widely used UV filters, BP-3 has been detected in surface water samples collected in Hong Kong and New York, with a maximum concentration of 5429.00 ng/L in Hong Kong [
10]. A recent study of endocrine disrupting effects found that BP-3 can disrupt the agonistic behavior of male Siamese fighting fish (
Betta splendens) [
12].
BP-4, a broad-spectrum UV filter with good light and thermal stability, is the most frequently detected UV filter in waste water, groundwater, and surface water in Spain. The median concentration in wastewater is 2.10 μg/L with higher concentrations in surface water and groundwater, and a maximum concentration in drinking water of 62.00 ng/L [
13]. At present, there are very few studies about the toxicity of BP-4. There was only one study on the toxicity of BP-4, which found that the 48 h-EC
50 value of
D. magna was 50 mg/L.
The aim of this study was to investigate the ecological toxicity of BP-3 and BP-4 to aquatic organisms. The experiment employed three different freshwater species: green algae (Chlorella vulgaris), D. magna, and zebrafish (Brachydanio rerio). The acute ecological toxicity of the two chemicals, both singularly and in combination, were evaluated.
Based on the results of acute toxicity testing of BP-3 and BP-4, the toxic grades of these substances were determined according to the corresponding acute toxicity standards. The degree of combined toxicity was analyzed based on mixing tests. The predicted no-effect concentration (PNEC) was reduced to evaluate potential ecological risks. The results will provide a scientific basis for formulating environmental criteria and further protection of aquatic ecosystems.
All experiments were conducted in accordance with Chinese National Standards: GB/T 21805-2008 (algae) [
14], GB/T 16125-2012 (
D. magna Straus) [
15], and GB/T 27861-2011 (fish) [
16], as well as the Analytical Methods for Water and Wastewater [
17].
2. Materials and Methods
2.1. Preparation of Chemicals
BP-3 and BP-4 at 99% purity were purchased from Aladdin Industrial Corporation (Shanghai, China). Analytical purity grade dimethyl sulfoxide (Sinopharm Chemical Reagent Co., Ltd., Shanghai, China) was used as a cosolvent.
2.2. Experimental Biota for Toxicity Testing
C. vulgaris (FACHB-8) was obtained from the Freshwater Algae Culture Collection of the Institute of Hydrobiology, Chinese Academy of Sciences (Wuhan, China). The algae were cultured for three generations in the laboratory before the toxicity experiments. Algal cells in the logarithmic growth phase were used for toxicity experiments, which were conducted in an illumination incubator to maintain the same condition: 2000–3000 lx, 25 ± 2 °C, pH 7.1, and a 12-h light: dark cycle.
D. magna were obtained from the Institute of Hydrobiology, Chinese Academy of Sciences, cultured in the laboratory, and then tested for sensitivity to potassium dichromate before the toxicity experiments. D. magna at 6–24 h old were tested. The experiments were conducted using an illumination incubator to maintain the same conditions: 25 ± 2 °C, pH 7.0–8.0, and a 12-h light: dark cycle.
B. rerio (AB strains; average body length, 2.60 ± 0.20 cm; average body weight, 0.33 ± 0.06 g) were obtained from Nanjing YSY Biotech Company, Ltd. (Nanjing, China). Potassium dichromate was used to test the sensitivity of B. rerio before the toxicity experiments. The experiments were carried out at 25 ± 2 °C, pH 7.0–8.0, and a 12-h light: dark cycle.
2.3. Experimental Design
2.3.1. Acute Toxicity Experiments (96 h) with Chlorella vulgaris
Stock solutions of BP-3 and BP-4 were prepared using sterilized BG11 liquid medium. According to the results of a pre-experiment, a series of concentrations of the experimental solution (100 mL) were prepared in 250-mL glass flasks. The initial concentration of the algae in solution was 106 cell/mL and the test lasted for 96 h. A blank control group and solvent control group at the highest and different concentrations were included for analysis. Three parallel samples of each group were tested. The correlation between the algal density and absorbance at 680 nm were plotted before the experiment. During the test period, the absorbance was measured every 24 h and the growth rate of algae, according to an algal density-absorbance curve, was calculated.
2.3.2. Acute Toxicity Experiments (48 h) with Daphnia magna
Test stock solutions of BP-3 and BP-4 were prepared using aerated tap water. According to the results of the pre-experiment, a series of concentrations of the experimental solution (40 mL) were prepared in 100-mL glass beakers. Ten D. magna were added to each glass beaker. The test was continued for 48 h. A blank control and solvent control group at the highest concentration and different concentrations were included for analysis. Three parallel samples of each group were tested. The numbers of dead D. magna at every 24 h were recorded.
2.3.3. Acute Toxicity Experiments (96 h) with Brachydanio rerio
Stock solutions of BP-3 and BP-4 were prepared using aerated tap water. According to the results of the pre-experiment, a series of concentrations of the experimental solution (4000 mL) were prepared in 5000-mL glass beakers. Ten B. rerio were added to each glass beaker. The test was continued for 96 h. A blank control group and solvent control group at the highest and different concentrations were included for analysis. Three parallel samples of each group were tested. The numbers of dead B. rerio were recorded every 24 h.
2.3.4. The Mixing Toxicity Experiment
Testing of the toxicity of a mixture of BP-3 and BP-4 was conducted with the same method as for individual compounds. The mixing ratio and concentration of BP-3 and BP-4 for the different tested organisms were determined by referring to the obtained EC50 or 50% lethal concentration (LC50) of the independent acute toxicity results.
Stock solutions of BP-3 and BP-4 were prepared for
C. vulgaris (14 and 400 mg/L, respectively),
D. magna (10 and 300 mg/L, respectively), and
B. rerio (1000 and 20 mg/L, respectively). The toxic effects of the mixtures were determined by calculating the toxicity units (TU) [
5] with the following formula:
where C
i is the concentration of i in the tested solution at the EC
50 of the mixture.
The total TU value was the sum of the TUi values of BP-3 and BP-4. If the total TUi was greater than 1, the toxicity was considered to be antagonistic, if TUi was equal to 1, the toxicity was considered to be a simple additive effect, and if TUi was less than 1, the toxicity was considered to be synergistic.
2.4. Classification Criteria for Acute Toxicity Test
The classification criteria for acute toxicity of BP-3 and BP-4 to the 3 species of organisms was referred to the 1) acute toxicity test classification criteria for algae (
Table 1 and
Table 2) acute toxicity test classification criteria for Daphnia (
Table 2 and
Table 3) acute toxicity test classification criteria for fish. All of the classification criteria were recorded in Analytical Methods for Water and Wastewater [
17].
2.5. Statistical Analysis
OriginPro 9 software (OriginLab, Northampton, MA, USA) was used to draw graphs depicting the experimental results. IBM SPSS version 20 software (IBM Corp., Armonk, NY, USA) was applied to calculate the 96-h EC50 value of C. vulgaris, the 48-h LC50 value of D. magna, and the 96-h LC50 value of B. rerio.
2.6. Ecological Risk Assessment
AIST-MeRAM (National institute of Advanced Industrial Science and Technology-Multi-Purpose Ecological Risk Assessment and Management Tool, Japan [
18]) was used to assess the ecological risks of BP-3 and BP-4 to aquatic ecosystems. PNEC values are calculated by the present toxicity data and those pre-registered in the database of AIST-MeRAM. The assessment factor (AF) and species sensitivity distribution (SSD) methods were employed to determine the PNEC. OECD recommended UF value 100 was used in the AF method. HC5 (concentration at which 5% of the species are potentially affected) was selected as an endpoint in the SSD method.
Experiments using dimethyl sulfoxide as a cosolvent and the solvent control were set at the highest concentrations in the experimental group. The results showed that there was no significant difference between the growth rate of the blank and solvent control groups, indicating that dimethyl sulfoxide has no significant effect on C. vulgaris, D. magna, and B. rerio, and did not interfere with the experimental results.
4. Discussion
4.1. Assessment of the PNEC Values by AF and SSD
In this study, the PNEC values of BP-3 and BP-4 were deduced by the AF and SSD methods. The PNEC values derived from the SSD method were 1–2 orders of magnitude higher than that derived by the AF method. According to the instruction to the SSD method in the software, the number and quality of toxic data have a great influence on the final results. A good accurate SSD assessment is usually defined as the results of chronic data (REACH Technical Guidance) of at least eight species. If the result was derived from SSD analysis just based on the acute toxicity data or a small amount of chronic toxicity data, the accuracy of the data is not well, and the results should be just taken as a reference value. Since studies about BPs are rare, available toxicity data remain incomplete. There were 11 and four sets of acute and chronic toxicity data, respectively, for BP-3 and BP-4 from algae, daphnia, and fish in the database of AIST-MeRAM software Therefore, the deduced PNEC values derived by the SSD method should be considered unreliable. In this case, PNEC values derived by the AF method were selected for ecological risk assessment in the present study. Because the PNEC values deduced by the AF method were lower, the environmental criteria based on this value may be overestimated. However, since presently available toxicity data are insufficient, and the SSD method cannot obtain reliable results, in order to protect the ecological environment as much as possible, the AF method was employed in the present study.
4.2. Acute Toxicity and Ecological Risk Assessment of BP-3
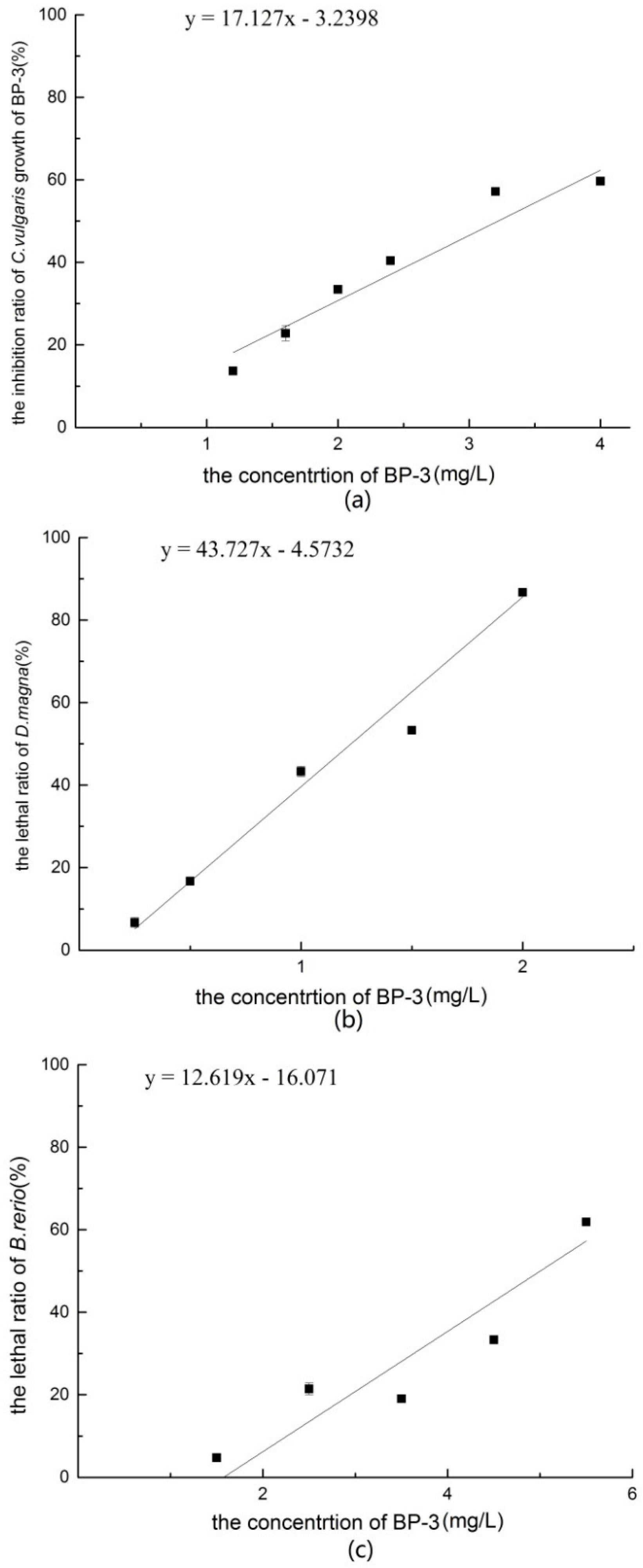
According to the individual acute toxicity results of BP-3, the 96 h-EC50 value of C. vulgaris was 2.98 mg/L, the 48 h-LC50 value of D. magna was 1.09 mg/L, and the 96 h-LC50 value of B. rerio was 3.89 mg/L. The sensitivity of D. magna was greater than that of C. vulgaris and B. rerio.
A query of the AIST-MeRAM database (
Table 4) showed that the 72 h-EC
50 values of BP-3 for algae were 0.67 and 0.96 mg/L, respectively (Ministry of the Environment, Japan). The sensitivity of algae to BP-3 was greater than in this study. The EC
50 values of BP-3 to
D. magna, as determined with the 48-h acute toxicity test, were 1.90 mg/L (Ministry of the Environment, Japan) and 1.67 mg/L [
19], which were the same order of magnitude as the 48 h-LC
50 value in this study. In addition, the 96 h-LC
50 value of
Oryzias latipes in the database was 3.80 mg/L, which was very close to the result of
B. rerio in the present experiment. Based on the above data, the sensitivity of different trophic level organisms to BP-3 was Daphnia > algae > fish.
In this study, the PNEC value derived from the AF method was 1.8 × 10
−3 mg/L. In Nanjing, BP-3 was detected in surface water at a concentration of 3.63–164 ng/L [
10]. Moreover, several UV filters, including BP-3, 2-ethyl-hexyl-4-trimethoxycinnamate, and homosalate, have been detected in lakes and rivers at concentrations of 25.00, 74.00, and 92.00 ng/L, respectively. In addition, the concentration of BP-3 in the Glatt River in Switzerland was 0.1 μg/POCIS (polar organic chemical integrative samplers) from July to August 2006, and the concentration of BP-3 was up to 0.18 μg/POCIS from May to August 2017 [
9]. These data indicate that the concentration of BP-3 in the aquatic environment is much lower than the deduced PNEC value. So, BP-3 may have little effect on the ecological environment at present.
However, Heffernan collected urine samples from males and females at various ages and detected BP-3 at concentrations ranging from 16.50 to 312.00 ng/L [
12]. In addition, the BP-3 concentrations were greater in samples collected from adults and females, as compared to children and males. These findings may be due to the fact that adult females are more likely to use sunscreen products. Another study reported that BP-3 has an effect on endocrine function and neurodevelopment in fish [
21]. Meanwhile, the concentration of BP-3 in children (aged 0–4 years) is reportedly 17.00–55.30 ng/L, which may be injurious to human health. Although very low concentrations of BP-3 have been detected in natural ecosystems, the potential for harmful effects on human health and potential endocrine effects to fish are alarming. Hence, the biotransformation process from environmental media to humans suggest potential harm of BP-3 to human health.
4.3. Acute Toxicity and Ecological Risk Assessments of BP-4
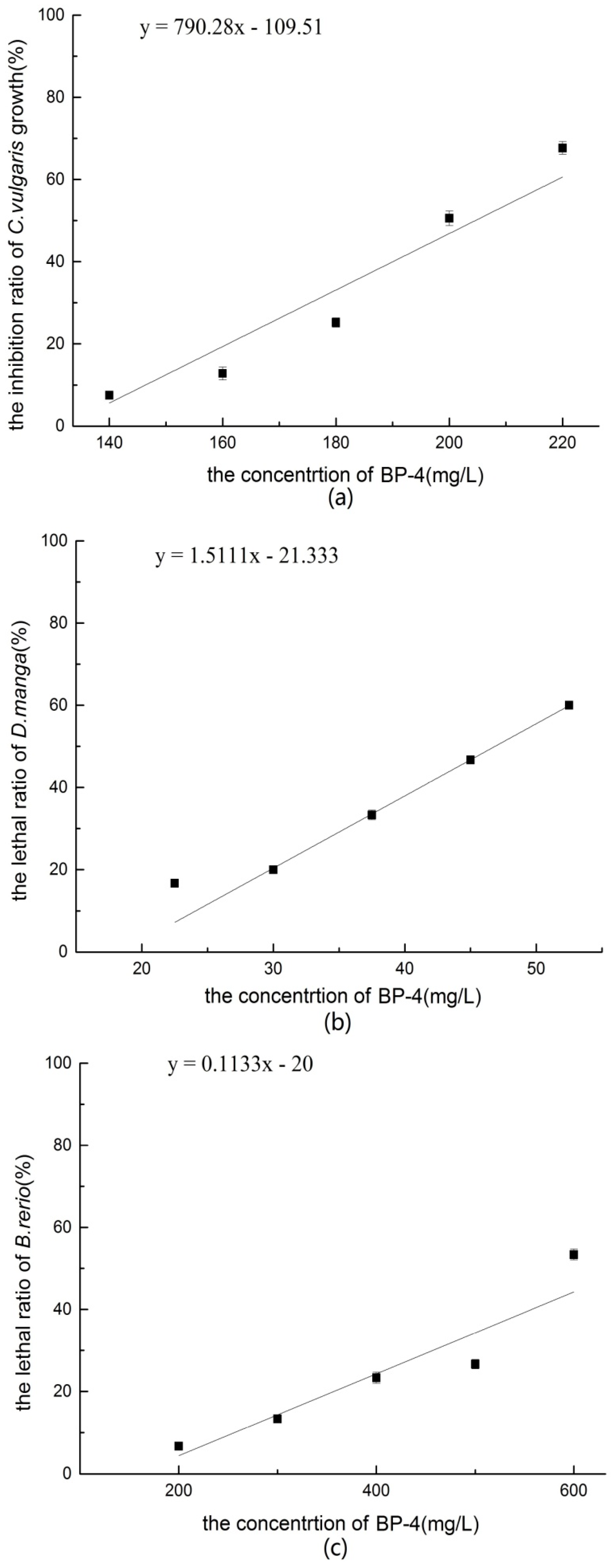
According to the individual acute toxicity results of BP-4, the 96 h-EC50 value of C. vulgaris was 201.00 mg/L, the 48 h-LC50 value of D. magna was 47.47 mg/L, and the 96 h-LC50 value of B. rerio was 633.00 mg/L. Based on the acute toxicity classification criteria, the toxicity of BP-4 to C. vulgaris and B. rerio were low-level, and that to D. magna was medium-level. The sensitivity of D. magna to BP-4 was greater than that of C. vulgaris and B. rerio.
At present, other than the three data sets provided by this study, there are no toxicity data for BP-4 in the AIST-MeRAM database (
Table 5). There was one acute toxicity test of BP-4 to
D. magna by Fent et al. [
22], who reported a 48 h-LC
50 value of 50.00 mg/L, which is very close to the value determined in the present study. In addition, a 14-day chronic test with the rainbow trout (
Oncorhynchus mykiss) as the test species showed that the LOEC value was 4897.00 μg/L [
11].
Rodil et al. [
23] identified BP-4 as a potential contaminant in the water of Galicia in northwestern Spain with median concentrations of up to 2.10 μg/L in wastewater and maximum concentrations in surface water and tap water of 62.00 ng/L. In addition, the concentration of BP-4 in surface water in Tokyo, New York, and Bangkok were 71.00–136.00, 89.00–574.00, and 80.00–95.00 ng/L, respectively [
20].
The PNEC value, as determined with the AF method, was 0.47 mg/L, which was significantly higher than the concentration of BP-4 detected in aquatic environments. BP-4 can be considered as an ecologically safe chemical at present.
Although the current concentration of BP-4 in the aquatic environment is low, the levels of BP-4 in the influent and outflow of a German sewage treatment plant were 2120.00 and 572.00 ng/L, respectively [
24]. These concentrations did not change much and the concentration of BP-4 in sludge was 29.00 ng/g-dw. This result indicated that the sewage treatment was insufficient to effectively remove concentrated BP-4 from sludge. As a result, with the discharge of sewage, BP-4 may accumulate in the environment, thereby endangering the ecological environment and human health.
In addition, few studies have analyzed the toxicity of BP-4. We tested the acute toxicity of BP-4 on three typical organisms in the aquatic ecosystem and calculated the corresponding PNEC values. Nonetheless, future toxicity studies of different species of organisms are necessary to analyze the risks of BP-4 to the environment.
4.4. Toxicity Assessment of a Mixture of BP-3 and BP-4
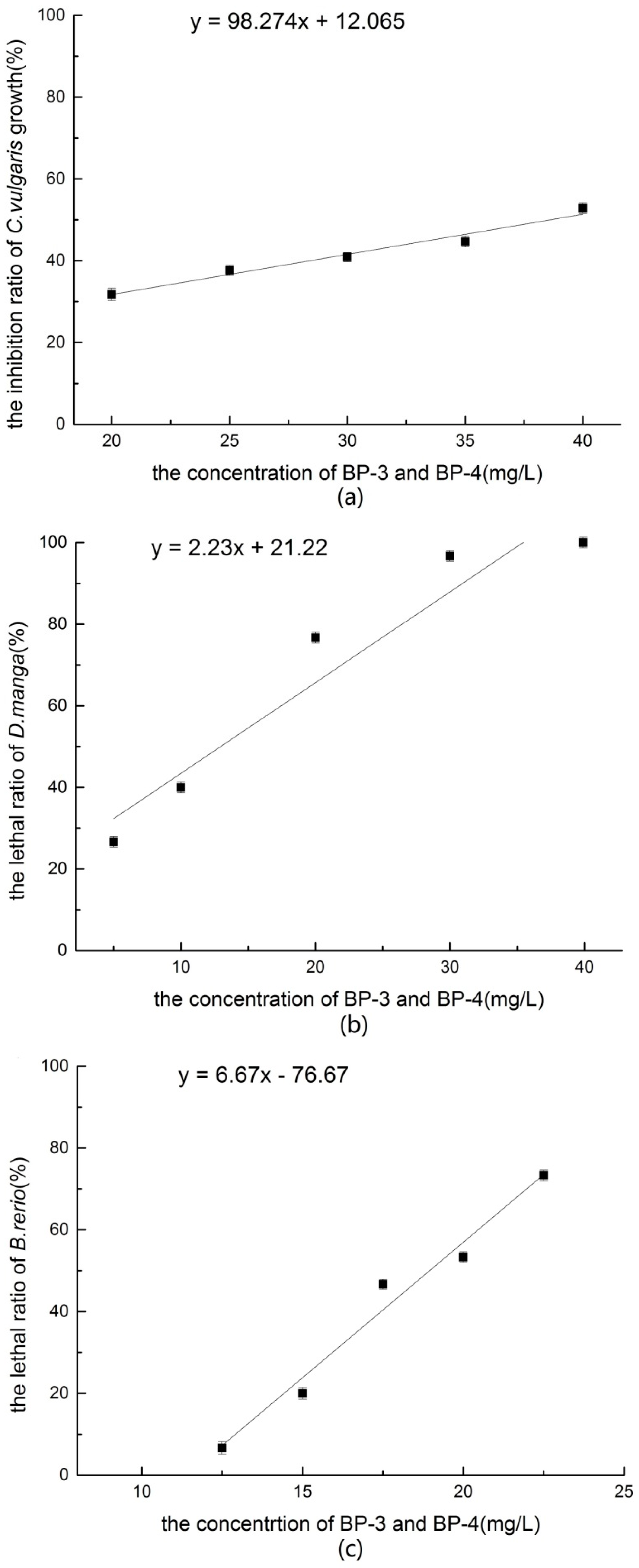
According to the results of the acute toxicity experiments, the effects of the mixture of BP-3 and BP-4 to
C. vulgaris,
D. magna, and
B. rerio were all antagonistic. Therefore, the mixture of the two chemicals is likely to exhibit antagonism in the aquatic environment. It is worth noting that BP-3 and BP-4 both contain hydroxyl and benzophenone groups. The number and position of these hydroxyl groups are the same, with the only difference being that BP-4 contains a sulfonic acid group. Usually, an antagonistic effect is considered to be a result of similar chemical properties that play a competitive role in the active parts of the cell surface and the metabolic system, thus affecting the interactions of matter [
25]. So, the similar structures of BP-3 and BP-4 exhibited a competitive role with antagonistic effects to the aquatic organisms in the present study. There has not been much discussion about the toxic mechanism of BP-3 and BP-4. It has been reported that a mixture of 4-MBC and BP-3 induced a strong increase in mRNA levels at lower concentrations with a slight induction at higher concentrations [
26].
The toxicity of chemical mixtures is important to explain the behavior and comprehensive effects of individual chemicals in the environment. The different combinations of chemicals show different effects. In our previous studies, the mixture toxicity of BP and
N,N-diethyl-3-methylbenzamide (DEET), on
C. vulgaris,
D. magna, and
B. rerio all showed an additive effect [
7], which behaved different with the combination of BP-3 and BP-4. The results of this study revealed the effects of a mixture of BP-3 and BP-4 to three trophic level organisms in an aquatic ecosystem that should help us understand the behavior and effects of these chemicals in the environment.
Based on published data, environmental concentrations and toxicity of BPs have been largely overlooked in comparison to pharmaceutical compounds. Current published literature for environmental concentrations is fairly substantial for some personal care products (PCP) compounds (ex. Triclosan, DEET, fragrances) but relatively little is available for UV filters [
11]. To date, most conducted studies have investigated compounds individually but not as mixtures. The results of this study revealed the acute toxicity data of BP-3 and BP-4, and deduced the corresponding PNEC values and mixing effects. These results are significant for the assessment of the toxicity to the environmental and provide a scientific basis for critical environmental policies.
In future studies, more relevant toxicity tests, especially chronic toxicity tests, are expected to enrich the toxicity test database and assessment of chemicals used as UV filters. Continuously, all chemicals used in UV filters are expected to be evaluated. Furthermore, the potential for biomagnification and potential effects on higher trophic level organisms of these chemicals are needing to be examined, in order to protect the health of humans.