Association between PM2.5 Exposure and All-Cause, Non-Accidental, Accidental, Different Respiratory Diseases, Sex and Age Mortality in Shenzhen, China
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Mortality Data
2.1.2. Air Pollution Data
2.1.3. Meteorological Data
2.2. Methods
2.2.1. Basic Description
2.2.2. Autocorrelation Analysis
2.2.3. Analysis of Time Series
2.2.4. Generalized Additive Models, GAM
2.2.5. Analysis of Concentration-Response Relationship
3. Results
3.1. Basic Information of Mortality Residents
3.2. Information of Air Pollutants and Meteorological Factors
3.3. Autocorrelation Analysis of PM2.5 Concentration
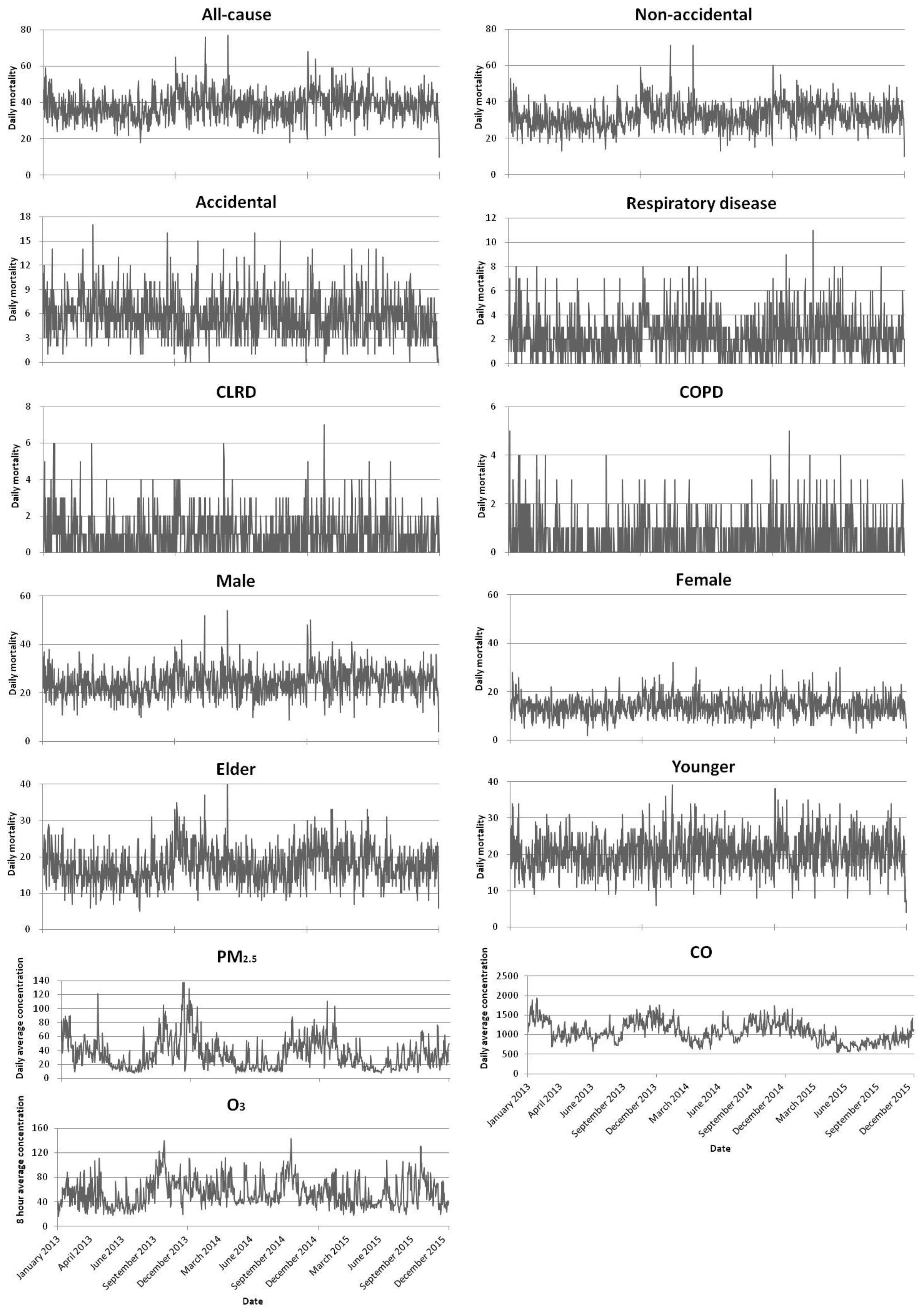
3.4. Time Series Chart on Residents’ Mortality versus Air Pollutants Concentration
3.5. Analysis of Generalized Additive Model, GAM
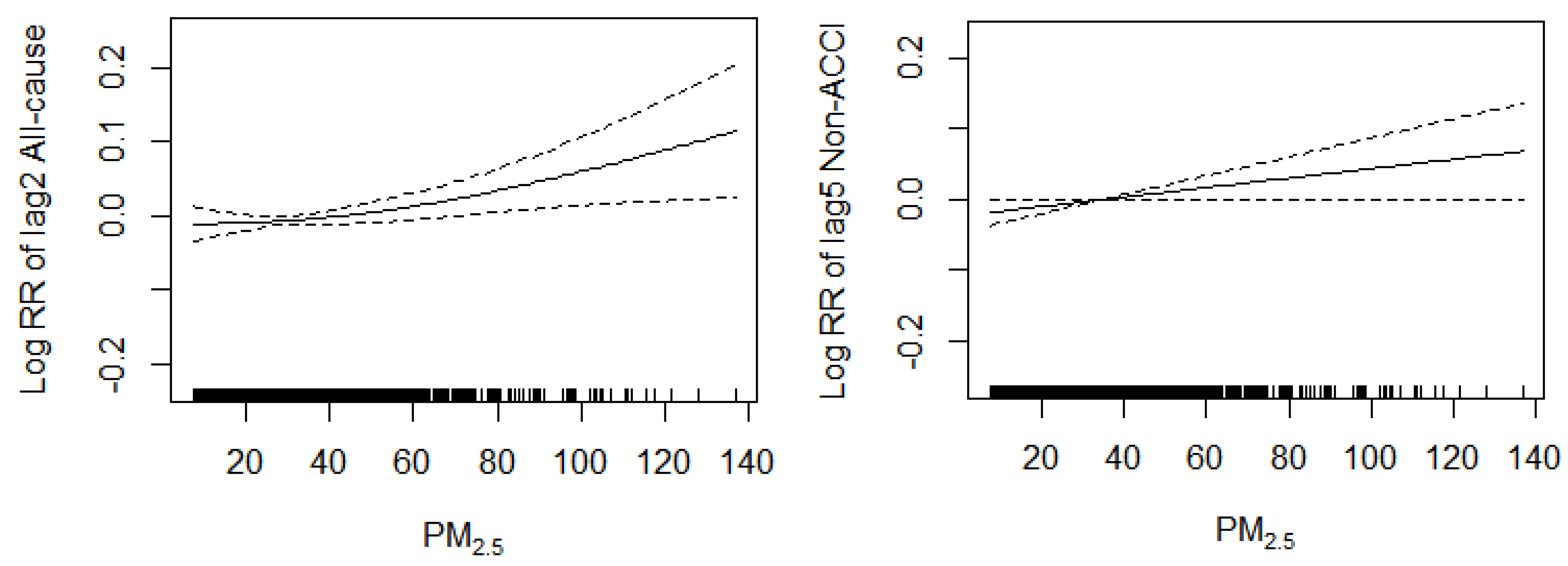
3.5.1. Effects of PM2.5 Concentration on All-Cause Mortality
3.5.2. Effects of PM2.5 Concentration on Non-Accidental Mortality
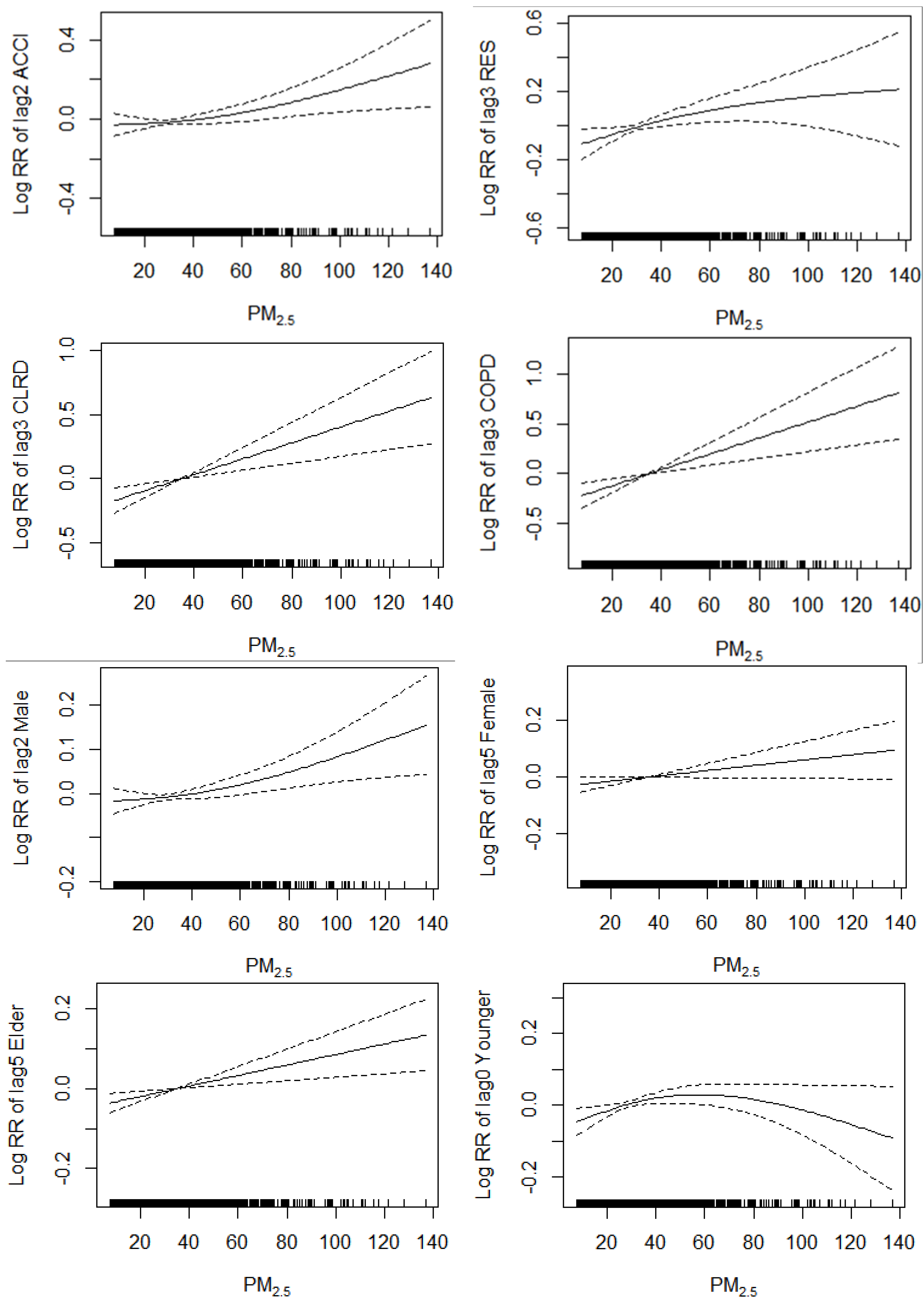
3.5.3. Effects of PM2.5 Concentration on Accidental Mortality
3.5.4. Effects of PM2.5 Concentration on Total Respiratory Disease Mortality
3.5.5. Effects of PM2.5 Concentration on CLRD Mortality
3.5.6. Effects of PM2.5 Concentration on COPD Mortality
3.5.7. Effects of PM2.5 Concentration on Male Mortality
3.5.8. Effects of PM2.5 Concentration on Female Mortality
3.5.9. Effects of PM2.5 Concentration on Elderly Mortality
3.5.10. Effects of PM2.5 Concentration on Younger Mortality
3.6. Concentration-Response Relationship
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dai, L.Z.; Zanobetti, A.; Koutrakis, P.; Schwartz, J.D. Associations of fine particulate matter species with mortality in the United States: A multicity time-series analysis. Environ. Health Perspect. 2014, 122, 837–842. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.H.; Zanobetti, A.; Kloog, I.; Coull, B.A.; Koutrakis, P.; Melly, S.J.; Schwartz, J.D. Low-Concentration PM2.5 and Mortality: Estimating Acute and Chronic Effects in a Population-Based Study. Environ. Health Perspect. 2016, 124, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Cohen, A.J.; Brauer, M.; Burnett, R.; Anderson, H.R.; Frostad, J.; Estep, K.; Balakrishnan, K.; Brunekreef, B.; Dandona, L.; Dandona, R.; et al. Estimates and 25-year trends of the global burden of disease attributable to ambient air pollution: An analysis of data from the Global Burden of Diseases Study 2015. Lancet 2017, 389, 1907–1918. [Google Scholar] [CrossRef]
- Yang, G.H.; Wang, Y.; Zeng, Y.X.; Gao, G.F.; Liang, X.F.; Zhou, M.G.; Wan, X.; Yu, S.C.; Jiang, Y.H.; Naghavi, M.; et al. Rapid health transition in China; 1990–2010: Findings from the Global Burden of Disease Study 2010. Lancet 2013, 381, 1987–2015. [Google Scholar] [CrossRef]
- Ito, K.; Mathes, R.; Ross, Z.; Nádas, A.; Thurston, G.; Matte, T. Fine particulate matter constituents associated with cardiovascular hospitalizations and mortality in New York City. Environ. Health Perspect. 2011, 119, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Ostro, B.; Feng, W.Y.; Broadwin, R.; Green, S.; Lipsett, M. The effects of components of fine particulate air pollution on mortality in california: Results from CALFINE. Environ. Health Perspect. 2007, 115, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Ito, K.; Lall, R.; Lippmann, M.; Thurston, G. Time-series analysis of mortality effects of fine particulate matter components in Detroit and Seattle. Environ. Health Perspect. 2011, 119, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Wang, J.N.; Ma, G.X.; Zhang, Y.S. China tackles the health effects of air pollution. Lancet 2013, 382, 1959–1960. [Google Scholar] [CrossRef]
- Tabulation On The 2010 Population Census Of The Peoples Republic Of China. Available online: http://www.stats.gov.cn/tjsj/pcsj/rkpc/6rp/indexch.htm (accessed on 24 September 2018).
- Donkelaar, A.V.; Martin, R.V.; Brauer, M.; Kahn, R.; Levy, R.; Verduzco, C.; Villeneuve, P.J. Global estimates of ambient fine particulate matter concentrations from satellite-based aerosol optical depth: Development and application. Environ. Health Perspect. 2010, 118, 847–855. [Google Scholar] [CrossRef]
- Chen, R.J.; Yin, P.; Meng, X.; Liu, C.; Wang, L.J.; Xu, X.H.; Ross, J.A.; Tse, L.A.; Zhao, Z.H.; Kan, H.D.; et al. Fine Particulate Air Pollution and Daily Mortality. A Nationwide Analysis in 272 Chinese Cities. Am. J. Respir. Crit. Care Med. 2017, 196, 73–81. [Google Scholar] [CrossRef]
- China National Environmental Monitoring Centre. Available online: http://www.cnemc.cn (accessed on 12 September 2018).
- The Report of the 19th Session of National Congress of the Communist Party of China, China. 24 October 2017. Available online: http://cpc.people.com.cn/n1/2017/1028/c64094-29613660.html (accessed on 21 July 2018).
- Cao, J.J.; Xu, H.M.; Xu, Q.; Chen, B.H.; Kan, H.D. Fine particulate matter constituents and cardiopulmonary mortality in a heavily polluted Chinese city. Environ. Health Perspect. 2012, 120, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.M.; Jia, Y.P.; Pan, X.C.; Liu, L.Q.; Wichmann, H.E. The association between fine particulate air pollution and hospital emergency room visits for cardiovascular diseases in Beijing; China. Sci. Total Environ. 2009, 407, 4826–4830. [Google Scholar] [CrossRef] [PubMed]
- Kan, H.D.; London, S.J.; Chen, G.H.; Zhang, Y.H.; Song, G.X.; Jiang, L.L.; Zhao, N.Q.; Chen, B.H. Differentiating the effects of fine and coarse particles on daily mortality in Shanghai, China. Environ. Int. 2007, 3, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.Y.; Chen, R.J.; Pan, G.W.; Xu, X.H.; Song, W.M.; Chen, B.H.; Kan, H.D. Fine particulate air pollution and daily mortality in Shenyang, China. Sci. Total Environ. 2011, 409, 2473–2477. [Google Scholar] [CrossRef] [PubMed]
- Venners, S.A.; Wang, B.Y.; Xu, Z.G.; Schlatter, Y.; Wang, L.H.; Xu, XP. Particulate matter; sulfur dioxide; and daily mortality in Chongqing; China. Environ. Health Perspect. 2003, 111, 562–567. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.S.; Ma, G.X.; Yu, F.; Cao, D. Health damage assessment due to PM2.5 exposure during haze pollution events in Beijing-Tianjin-Hebei region in January 2013. Natl. Med. J. China 2013, 34, 2707–2710. [Google Scholar] [CrossRef]
- Huang, J.; Pan, X.C.; Guo, X.B.; Li, G.X. Health impact of China’s Air Pollution Prevention and Control Action Plan_ an analysis of national air quality monitoring and mortality data. Lancet Planet. Health 2018, 2, 313–323. [Google Scholar] [CrossRef]
- Shenzhen Staistical Yearbook. Available online: http://www.sztj.gov.cn (accessed on 6 October 2018).
- China Staistical Yearbook. Available online: http://www.stats.gov.cn/tjsj/tjcbw/201512/t20151210_1287840.html (accessed on 6 October 2018).
- Ministry of Environmental Protection of the People’s Republic of China. Ambient Air Quality Standards. GB3095-2012. Available online: http://kjs.mep.gov.cn/hjbhbz/bzwb/dqhjbh/dqhjzlbz/201203/t20120302_224165.htm (accessed on 6 February 2018).
- Technical Regulation for Ambient Air Quality Assessment (On Trial). HJ663-243. Available online: http://www.doc88.com/p-9903770969556.html (accessed on 12 September 2018).
- Chen, R.J.; Kan, H.D.; Chen, B.H.; Huang, W.; Bai, Z.P.; Song, G.X.; Pan, G.W. Association of particulate air pollution with daily mortality: The China Air Pollution and Health Effects Study. Am. J. Epidemiol. 2012, 175, 1173–1181. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.H.; Zhang, Y.H.; Ma, W.J.; Xu, Y.J.; Xu, X.J.; Cai, Q.M.; Pan, B.; Zeng, S.Q. Association between temperature and daily mortality in Guangzhou; 2006–2009: A time series study. Zhonghua Liu Xing Bing Xue Za Zhi 2011, 32, 9–12. [Google Scholar] [CrossRef]
- Hastie, T.; Tibshirani, R. Generalized Additive Models; Chapman and Hall: London, UK, 1990. [Google Scholar]
- Hoek, G.; Schwartz, J.D.; Groot, B.; Eilers, P. Effects of ambient particulate matter and ozone on daily mortality in Rotterdam, The Netherlands. J. Arch. Environ. Health 1997, 52, 455–463. [Google Scholar] [CrossRef]
- Schwartz, J. Air pollution and daily mortality: A review and meta-analysis. Environ. Health Perspect. 1994, 64, 36–52. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, J.; Laden, F.; Zanobetti, A. The Concentration-Response Relation between PM2.5 and Daily Deaths. Environ. Health Perspect. 2002, 110, 1025–1029. [Google Scholar] [CrossRef] [PubMed]
- Katsouyanni, K.; Touloumi, G.; Samoli, E.; Gryparis, A.; Tertre, A.L.; Monopolis, Y.; Rossi, G.; Zmirou, D.; Ballester, F.; Boumghar, A.; et al. Confounding and effect modification in the short-term effects of ambient particles on total mortality: Results from 29 European Cities within the APHEA2 Project. Epidemiology 2001, 12, 521–531. [Google Scholar] [CrossRef] [PubMed]
- Michelozzi, P.; Forastiere, F.; Fusco, D.; Perucci, C.A.; Ostro, B.; Ancona, C.; Pallotti, G. Air pollution and daily mortality in Rome, Italy. Occup. Environ. Med. 1998, 5, 605–610. [Google Scholar] [CrossRef]
- Kloog, I.; Ridgway, B.; Koutrakis, P.; Coull, B.A.; Schwartz, J.D. Long- and short-term exposure to PM2.5 and mortality: Using novel exposure models. Epidemiology 2013, 24, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Ver Hoef, J.M.; Boveng, P.L. Quasi-Poisson Vs. Negative Binomial Regression: How Should We Model Overdispersed Count Data? Ecology 2007, 88, 2766–2772. [Google Scholar] [CrossRef] [PubMed]
- Qiu, H.; Yu, I.T.S.; Tian, L.W.; Wang, X.R.; Tse, L.A.; Tam, W.; Wong, T.W. Effects of Coarse Particulate Matter on Emergency Hospital Admissions for Respiratory Diseases: A Time-Series Analysis in Hong Kong. Environ. Health Perspect. 2012, 120, 572–576. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, T.W.; Tam, W.; Tak, S.Y.I.; Wun, Y.T.; Wong, A.H.; Wong, C.M. Association between air pollution and general practitioner visits for respiratory diseases in Hong Kong. Thorax 2006, 61, 585–591. [Google Scholar] [CrossRef] [Green Version]
- Wood, S.N. Generalized Additive Models: An Introduction with R; CRC Press: Boca Raton, FL, USA, 2006; Available online: https://www.crcpress.com/Generalized-Additive-Models-An-Introduction-with-R/Wood/9781584884743 (accessed on 20 August 2018).
- Cifuentes, L.A.; Vega, J.; Köpfer, K.; Lave, L.B. Effect of the Fine Fraction of Particulate Matter versus the Coarse Mass and Other Pollutants on Daily Mortality in Santiago; Chile. J. Air Waste Manag. Assoc. 2000, 50, 1287–1298. [Google Scholar] [CrossRef]
- Krall, J.R.; Anderson, G.B.; Dominici, F.; Bell, M.L.; Peng, R.D. Short-term exposure to particulate matter constituents and mortality in a national study of U.S. urban communities. Environ. Health Perspect. 2013, 121, 1148–1153. [Google Scholar] [CrossRef]
- Bell, M.L.; Samet, J.M.; Dominici, F. Time-series studies of particulate matter. Annu. Rev. Public Health 2004, 25, 247–280. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.F.; Yu, S.Y.; Pei, Y.X.; Peng, C.Q.; Liao, Y.X.; Liu, N.; Ji, J.J.; Cheng, J.Q. Association between Airborne Fine Particulate Matter and Residents’ Cardiovascular Diseases; Ischemic Heart Disease and Cerebral Vascular Disease Mortality in Areas with Lighter Air Pollution in China. Int. J. Environ. Res. Public Health 2018, 15, E1918. [Google Scholar] [CrossRef] [PubMed]
- Roberts, S. A new model for investigating the mortality effects of multiple air pollutants in air pollution mortality time-series studies. J. Toxicol. Environ. Health A 2006, 69, 417–435. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.H.; Huang, W.; London, S.J.; Song, G.X.; Chen, G.H.; Jiang, L.L.; Zhao, N.Q.; Chen, B.H.; Kan, H.D. Ozone and daily mortality in Shanghai, China. Environ. Health Perspect. 2006, 114, 1227–1232. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, R.W.; Analitis, A.; Samoli, E.; Fuller, G.W.; Green, D.C.; Mudway, I.S.; Anderson, H.R.; Kelly, F.J. Short-term exposure to traffic-related air pollution and daily mortality in London, UK. J. Expo. Sci. Environ. Epidemiol. 2016, 26, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Rush, B.; Wiskar, K.; Fruhstorfer, C.; Celi, L.A.; Walley, K.R. The Impact of Chronic Ozone and Particulate Air Pollution on Mortality in Patients With Sepsis Across the United States. J. Intensive Care Med. 2018. [Google Scholar] [CrossRef]
- Janssen, N.A.H.; Fischer, P.; Marra, M.; Ameling, C.; Cassee, F.R. Short-term effects of PM2.5; PM10 and PM2.5–10 on daily mortality in The Netherlands. Sci. Total Environ. 2013, 463–464, 20–26. [Google Scholar] [CrossRef]
- Lepeule, J.; Laden, F.; Dockery, D.; Schwartz, J. Chronic exposure to fine particles and mortality: An extended follow-up of the Harvard Six Cities study from 1974 to 2009. Environ. Health Perspect. 2012, 120, 965–970. [Google Scholar] [CrossRef]
- Thurston, G.D.; Ahn, J.; Cromar, K.R.; Shao, Y.Z.; Reynolds, H.R.; Jerrett, M.; Lim, C.C.; Shanley, R.; Park, Y.; Hayes, R.B. Ambient Particulate Matter Air Pollution Exposure and Mortality in the NIH-AARP Diet and Health Cohort. Environ. Health Perspect. 2016, 124, 484–490. [Google Scholar] [CrossRef] [Green Version]
- Atkinson, R.W.; Kang, S.; Anderson, H.R.; Mills, I.C.; Walton, H.A. Epidemiological time series studies of PM2.5 and daily mortality and hospital admissions: A systematic review and meta-analysis. Thorax 2014, 69, 660–665. [Google Scholar] [CrossRef]
- Chen, H.; Burnett, R.T.; Copes, R.; Kwong, J.C.; Villeneuve, P.J.; Goldberg, M.S.; Brook, R.D.; Donkelaar, A.V.; Jerrett, M.; Martin, R.V.; et al. Ambient Fine Particulate Matter and Mortality among Survivors of Myocardial Infarction: Population-Based Cohort Study. Environ. Health Perspect. 2016, 124, 124. [Google Scholar] [CrossRef]
- Crouse, D.L.; Peters, P.A.; Donkelaar, A.V.; Goldberg, M.S.; Villeneuve, P.J.; Brion, O.; Khan, S.; Atari, D.O.; Jerrett, M.; Pope, C.A.; et al. Risk of Non accidental and Cardiovascular Mortality in Relation to Long-term Exposure to Low Concentrations of Fine Particulate Matter: A Canadian National-Level Cohort Study. Environ. Health Perspect. 2012, 120, 708–714. [Google Scholar] [CrossRef]
- Pinault, L.L.; Weichenthal, S.; Crouse, D.L.; Brauer, M.; Erickson, A.; Donkelaar, A.V.; Martin, R.V.; Hystad, P.; Chen, H.; Finès, P.; et al. Associations between fine particulate matter and mortality in the 2001 Canadian Census Health and Environment Cohort. Environ. Res. 2017, 159, 406–415. [Google Scholar] [CrossRef] [PubMed]
- Pinault, L.; Tjepkema, M.; Crouse, D.L.; Weichenthal, S.; Donkelaar, A.V.; Martin, R.V.; Brauer, M.; Chen, H.; Burnett, R.T. Risk estimates of mortality attributed to low concentrations of ambient fine particulate matter in the Canadian community health survey cohort. Environ. Health 2016, 15, 18. [Google Scholar] [CrossRef] [PubMed]
- Vodonos, A.; Awad, Y.A.; Schwartz, J. The concentration-response between long-term PM2.5 exposure and mortality; A meta-regression approach. Environ. Res. 2018, 166, 677–689. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Koutrakis, P.; Coull, B.; Kloog, I.; Schwartz, J. Acute effect of fine particulate matter on mortality in three Southeastern states from 2007–2011. J. Expo. Sci. Environ. Epidemiol. 2016, 26, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Baxter, L.K.; Crooks, J.L.; Sacks, J.D. Influence of exposure differences on city-to-city heterogeneity in PM2.5-mortality associations in US cities. Environ. Health 2017, 16, 1. [Google Scholar] [CrossRef] [PubMed]
- Weichenthal, S.; Villeneuve, P.J.; Burnett, R.T.; Donkelaar, A.V.; Martin, R.V.; Jones, R.R.; DellaValle, C.T.; Sandler, D.P.; Ward, M.H.; Hoppin, J.A. Long-term exposure to fine particulate matter: Association with nonaccidental and cardiovascular mortality in the agricultural health study cohort. Environ. Health Perspect. 2014, 122, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Hao, Y.G.; Balluz, L.; Strosnider, H.; Wen, X.J.; Li, C.Y.; Qualters, J.R. Ozone, Fine Particulate Matter, and Chronic Lower Respiratory Disease Mortality in the United States. Am. J. Respir. Crit. Care Med. 2015, 92, 337–341. [Google Scholar] [CrossRef]
- Li, M.H.; Fan, L.C.; Mao, B.; Yang, J.W.; Choi, A.M.K.; Cao, W.J.; Xu, J.F. Short-term Exposure to Ambient Fine Particulate Matter Increases Hospitalizations and Mortality in COPD: A Systematic Review and Meta-analysis. Chest 2016, 149, 447–458. [Google Scholar] [CrossRef]
- Sun, Q.; Liu, C.; Chen, R.J.; Wang, C.; Li, J.D.; Sun, J.; Kan, H.D.; Cao, J.Y.; Bai, H.J. Association of fine particulate matter on acute exacerbation of chronic obstructive pulmonary disease in Yancheng, China. Sci. Total Environ. 2018, 650 (Pt 2), 1665–1670. [Google Scholar] [CrossRef]
- Villeneuve, P.J.; Weichenthal, S.A.; Crouse, D.; Miller, A.B.; To, T.; Martin, R.V.; Donkelaar, A.V.; Wall, C.; Burnett, R.T. Long-term Exposure to Fine Particulate Matter Air Pollution and Mortality Among Canadian Women. Epidemiology 2015, 4, 536–545. [Google Scholar] [CrossRef]
- Alessandrini, E.R.; Stafoggia, M.; Faustini, A.; Berti, G.; Canova, C.; Togni, A.D.; Biagio, K.D.; Gherardi, B.; Giannini, S.; Lauriola, P.; et al. Association Between Short-Term Exposure to PM2.5 and PM10 and Mortality in Susceptible Subgroups: A Multisite Case-Crossover Analysis of Individual Effect Modifiers. Am. J. Epidemiol. 2016, 184, 744–754. [Google Scholar] [CrossRef] [PubMed]
- Qiu, H.; Pun, V.C.; Tian, L.W. Short-term effects of fine and coarse particles on deaths in Hong Kong elderly population: An analysis of mortality displacement. Environ. Pollut. 2018, 241, 148–154. [Google Scholar] [CrossRef]
- Wong, C.M.; Lai, H.K.; Tsang, H.; Thach, T.Q.; Thomas, G.N.; Lam, K.B.H.; Chan, K.P.; Yang, L.; Lau, A.K.H.; Ayres, J.G.; et al. Satellite-Based Estimates of Long-Term Exposure to Fine Particles and Association with Mortality in Elderly Hong Kong Residents. Environ. Health Perspect. 2015, 123, 1167–1172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baxter, L.K.; Dionisio, K.; Pradeep, P.; Rappazzo, K.; Neas, L. Human exposure factors as potential determinants of the heterogeneity in city-specific associations between PM2.5 and mortality. J. Expo. Sci. Environ. Epidemiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Fang, B.; Wang, C.F.; Xia, T.; Bottai, M.; Fang, F.; Cao, Y. Relationship between fine particulate matter, weather condition and daily non-accidental mortality in Shanghai, China: A Bayesian approach. PLoS ONE 2017, 12, e0187933. [Google Scholar] [CrossRef]


| Category | Total Deaths | Percent among Total Deaths (%) | Mean (SD) | Range | Median (Q1, Q3) |
|---|---|---|---|---|---|
| Health effects | |||||
| All-cause mortality | |||||
| 2013 | 13,126 | 100 | 36 (7) | 18–59 | 35 (31, 40) |
| 2014 | 14,116 | 100 | 39 (8) | 18–77 | 39 (33, 43) |
| 2015 | 14,573 | 100 | 40 (7) | 10–68 | 40 (35, 44) |
| 2013–2015 | 41,815 | 100 | 38 (8) | 10–77 | 38 ( 33, 43) |
| Non-accidental mortality (A00–R99) | |||||
| 2013 | 10,846 | 83 | 30 (6) | 13–53 | 29 (25, 34) |
| 2014 | 12,071 | 86 | 33 (7) | 13–71 | 33 (28, 37) |
| 2015 | 12,550 | 86 | 34 (7) | 10–60 | 35 (30, 39) |
| 2013–2015 | 35,467 | 85 | 32 (7) | 10–71 | 32 (27, 37) |
| Accidental mortality (S, T, V, W, X, Y) | |||||
| 2013 | 2278 | 17 | 6 (3) | 1–17 | 6 (4, 8) |
| 2014 | 2045 | 14 | 6 (3) | 0–16 | 5 (4, 7) |
| 2015 | 2023 | 14 | 6 (3) | 0–14 | 5 (4, 7) |
| 2013–2015 | 6348 | 15 | 6 (3) | 0–17 | 6 (4, 7) |
| Respiratory disease mortality (J00–J99) | |||||
| 2013 | 745 | 6 | 2 (2) | 0–8 | 2 (1, 3) |
| 2014 | 883 | 6 | 2 (2) | 0–8 | 2 (1, 3) |
| 2015 | 906 | 6 | 2 (2) | 0–11 | 2 (1, 3) |
| 2013–2015 | 2534 | 6 | 2 (2) | 0–11 | 2 (1, 3) |
| CLRD mortality (J40–J47) | |||||
| 2013 | 319 | 2 | 1 (1) | 0–6 | 1 (0, 1) |
| 2014 | 384 | 3 | 1 (1) | 0–6 | 1 (0, 2) |
| 2015 | 346 | 2 | 1 (1) | 0–7 | 1 (0, 1) |
| 2013–2015 | 1049 | 3 | 1 (1) | 0–7 | 1 (0, 2) |
| COPD mortality (J44) | |||||
| 2013 | 192 | 1 | 1 (1) | 0–5 | 0 (0, 1) |
| 2014 | 217 | 2 | 1 (1) | 0–4 | 0 (0, 1) |
| 2015 | 237 | 2 | 1 (1) | 0–5 | 0 (0, 1) |
| 2013–2015 | 646 | 2 | 1 (1) | 0–5 | 0 (0, 1) |
| Sex | |||||
| Male | |||||
| 2013 | 8258 | 63 | 23 (5) | 10–38 | 22 (19, 26) |
| 2014 | 8881 | 63 | 24 (6) | 9–54 | 24 (20, 27) |
| 2015 | 9428 | 65 | 26 (6) | 4–50 | 26 (22, 29) |
| 2013–2015 | 26,567 | 64 | 24 (6) | 4–54 | 24 (20, 28) |
| Female | |||||
| 2013 | 4868 | 37 | 13 (4) | 2–28 | 13 (11, 16) |
| 2014 | 5232 | 37 | 14 (4) | 5–32 | 14 (11, 17) |
| 2015 | 5143 | 35 | 14 (4) | 3–30 | 14 (11, 17) |
| 2013–2015 | 15,243 | 36 | 14 (4) | 2–32 | 14 (11, 17) |
| Sex-Unknown | |||||
| 2013 | 0 | 0 | 0 (0) | 0–0 | 0 (0, 0) |
| 2014 | 3 | 0 | 0 (0) | 0–1 | 0 (0, 0) |
| 2015 | 2 | 0 | 0 (0) | 0–1 | 0 (0, 0) |
| 2013–2015 | 5 | 0 | 0 (0) | 0–1 | 0 (0, 0) |
| Age (years) | |||||
| Elderly (≥65) | |||||
| 2013 | 5927 | 45 | 16 (4) | 5–31 | 15 (13, 19) |
| 2014 | 6633 | 47 | 18 (5) | 8–43 | 18 (15, 21) |
| 2015 | 6835 | 47 | 19 (5) | 6–33 | 19 (16, 22) |
| 2013–2015 | 19,395 | 46 | 18 (5) | 5–43 | 17 (14, 21) |
| Younger (<65) | |||||
| 2013 | 7199 | 55 | 20 (5) | 9–34 | 20 (16, 23) |
| 2014 | 7483 | 53 | 21 (5) | 6–39 | 20 (17, 24) |
| 2015 | 7738 | 53 | 21 (6) | 4–38 | 21 (17, 24) |
| 2013–2015 | 22,420 | 54 | 20 (5) | 4–39 | 20 (17, 24) |
| Indicator | Mean (SD) | Range | Median (Q1, Q3) | Days of Exceeding Grade 1 Criterion * | Days of Exceeding Grade 2 Criterion ** |
|---|---|---|---|---|---|
| Air pollutants | |||||
| PM2.5 (µg/m3) | |||||
| 2013 | 40 (26) | 7–137 | 35 (20, 53) | 183 | 39 |
| 2014 | 35 (20) | 7–107 | 31 (17, 48) | 159 | 13 |
| 2015 | 30 (17) | 7–111 | 27 (16, 41) | 116 | 6 |
| 2013–2015 | 35 (22) | 7–137 | 30 (17, 47) | 458 | 58 |
| PM10 (µg/m3) | |||||
| 2013 | 62 (36) | 11–182 | 52 (33, 83) | 195 | 8 |
| 2014 | 56 (28) | 12–169 | 49 (32, 73) | 178 | 2 |
| 2015 | 49 (24) | 13–174 | 44 (30, 63) | 140 | 1 |
| 2013–2015 | 56 (30) | 11–182 | 48 (31, 72) | 513 | 11 |
| SO2 (µg/m3) | |||||
| 2013 | 12 (6) | 4–55 | 10 (8, 15) | 1 | 0 |
| 2014 | 10 (4) | 4–31 | 9 (7, 11) | 0 | 0 |
| 2015 | 9 (3) | 4–19 | 9 (8, 11) | 0 | 0 |
| 2013–2015 | 10 (5) | 4–55 | 9 (7, 12) | 1 | 0 |
| NO2 (µg/m3) | |||||
| 2013 | 49 (21) | 17–134 | 44 (34, 59) | 34 | 34 |
| 2014 | 42 (16) | 15–130 | 39 (31, 51) | 9 | 9 |
| 2015 | 40 (14) | 16–128 | 37 (30, 47) | 4 | 4 |
| 2013–2015 | 44 (18) | 15–134 | 40 (32, 52) | 47 | 47 |
| CO (µg/m3) | |||||
| 2013 | 1163 (257) | 575–1930 | 1134 (963, 1350) | 0 | 0 |
| 2014 | 1126 (233) | 619–1759 | 1130 (945, 1277) | 0 | 0 |
| 2015 | 897 (202) | 543–1671 | 857 (757, 1029) | 0 | 0 |
| 2013–2015 | 1062 (260) | 543–1930 | 1034 (857, 1239) | 0 | 0 |
| O3 (µg/m3) | |||||
| 2013 | 53 (24) | 16–140 | 50 (33, 70) | 17 | 0 |
| 2014 | 60 (20) | 26–143 | 55 (44, 73) | 17 | 0 |
| 2015 | 52 (22) | 18–131 | 47 (35, 68) | 8 | 0 |
| 2013–2015 | 55 (22) | 16–143 | 51 (38, 70) | 42 | 0 |
| Meteorological factors | |||||
| Daily average temperature (°C) | |||||
| 2013 | 23 (5) | 10–31 | 24 (19, 28) | ||
| 2014 | 23 (6) | 6–31 | 25 (19, 29) | ||
| 2015 | 24 (5) | 12–33 | 26 (19, 28) | ||
| 2013–2015 | 23 (5) | 6–33 | 25 (19, 28) | ||
| Daily average RH (%) | |||||
| 2013 | 75 (16) | 24–100 | 78 (67, 87) | ||
| 2014 | 73 (13) | 19–96 | 76 (67, 82) | ||
| 2015 | 72 (11) | 28–93 | 73 (67, 79) | ||
| 2013–2015 | 73 (13) | 19–100 | 75 (67, 82) | ||
| Daily average atmosphere pressure (Kpa) | |||||
| 2013 | 1005 (6) | 987–1019 | 1005 (1001, 1011) | ||
| 2014 | 1006 (6) | 992–1021 | 1006 (1000, 1011) | ||
| 2015 | 1006 (6) | 991–1019 | 1006 (1001, 1011) | ||
| 2013–2015 | 1006 (6) | 987–1020 | 1006 (1001, 1011) | ||
| Daily average wind speed (m/s) | |||||
| 2013 | 2 (1) | 0–6 | 2 (2, 3) | ||
| 2014 | 2 (1) | 1–5 | 2 (2, 3) | ||
| 2015 | 2 (1) | 1–5 | 2 (2, 2) | ||
| 2013–2015 | 2 (1) | 0–6 | 2 (2, 3) | ||
| Daily rainfall (0.1 mm) | |||||
| 2013 | 6 (15) | 0–101 | 0 (0, 3) | ||
| 2014 | 5 (17) | 0–188 | 0 (0, 1) | ||
| 2015 | 4 (15) | 0–150 | 0 (0, 1) | ||
| 2013–2015 | 4 (16) | 0–188 | 0 (0, 1) | ||
| Sunshine (0.1 h) | |||||
| 2013 | 5 (4) | 0–13 | 6 (1, 9) | ||
| 2014 | 6 (4) | 0–12 | 6 (2, 9) | ||
| 2015 | 5 (4) | 0–12 | 6 (2, 9) | ||
| 2013–2015 | 5 (4) | 0–13 | 6 (2, 9) | ||
| Items | Single Pollutant Model | Two-Pollutant Model | Two-Pollutant Model | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PM2.5 | PM2.5 + CO | PM2.5 + O3 | ||||||||||
| β | StdErr | p | ER % (95% CI) | β | StdErr | p | ER % (95% CI) | β | StdErr | p | ER % (95% CI) | |
| All-cause mortality | ||||||||||||
| Lag0 | 0.0005 | 0.0003 | 0.16 | 0.48 (−0.19–1.17) | 0.0008 | 0.0004 | 0.05 * | 0.78 (0.00–1.56) | 0.0004 | 0.0004 | 0.31 | 0.37 (−0.35–1.10) |
| Lag1 | 0.0006 | 0.0003 | 0.07 @ | 0.61 (−0.04–1.27) | 0.0009 | 0.0004 | 0.01 # | 0.95 (0.23–1.68) | 0.0006 | 0.0004 | 0.09 @ | 0.61 (−0.11–1.33) |
| Lag2 | 0.0007 | 0.0003 | 0.02 * | 0.74 (0.11–1.38) | 0.0010 | 0.0004 | 0.00 # | 1.00 (0.30–1.70) | 0.0008 | 0.0004 | 0.04 * | 0.76 (0.05–1.48) |
| Lag3 | 0.0004 | 0.0003 | 0.26 | 0.36 (−0.26–0.98) | 0.0006 | 0.0003 | 0.10 @ | 0.57 (−0.10–1.26) | 0.0006 | 0.0004 | 0.11 | 0.57 (−0.14–1.27) |
| Lag4 | 0.0004 | 0.0003 | 0.16 | 0.44 (−0.17–1.05) | 0.0008 | 0.0003 | 0.02 * | 0.79 (0.12–1.47) | 0.0005 | 0.0004 | 0.20 | 0.46 (−0.24–1.16) |
| Lag5 | 0.0007 | 0.0003 | 0.02 * | 0.71 (0.10–1.32) | 0.0008 | 0.0003 | 0.02 * | 0.83 (0.16–1.50) | 0.0011 | 0.0004 | 0.00 # | 1.07 (0.37–1.77) |
| Lag01 | 0.0007 | 0.0004 | 0.06 @ | 0.70 (−0.04–1.45) | 0.0010 | 0.0004 | 0.01 * | 1.05 (0.23–1.88) | 0.0006 | 0.0004 | 0.14 | 0.62 (−0.20–1.45) |
| Lag02 | 0.0009 | 0.0004 | 0.02 * | 0.93 (0.14–1.73) | 0.0013 | 0.0004 | 0.00 # | 1.26 (0.40–2.12) | 0.0009 | 0.0005 | 0.05 @ | 0.89 (−0.02–1.81) |
| Lag03 | 0.0009 | 0.0004 | 0.03 * | 0.92 (0.09–1.75) | 0.0012 | 0.0004 | 0.01 # | 1.25 (0.36–2.14) | 0.0010 | 0.0005 | 0.04 * | 1.04 (0.06–2.02) |
| Lag04 | 0.0010 | 0.0004 | 0.03 * | 0.97 (0.11–1.83) | 0.0013 | 0.0005 | 0.01 # | 1.30 (0.39–2.22) | 0.0011 | 0.0005 | 0.03 * | 1.12 (0.09–2.16) |
| Non-accidental mortality | ||||||||||||
| Lag0 | 0.0004 | 0.0004 | 0.31 | 0.38 (−0.36–1.11) | 0.0007 | 0.0004 | 0.08 @ | 0.75 (−0.09–1.60) | 0.0003 | 0.0004 | 0.49 | 0.27 (−0.50–1.06) |
| Lag1 | 0.0005 | 0.0004 | 0.15 | 0.51 (−0.19–1.22) | 0.0009 | 0.0004 | 0.03 * | 0.86 (0.08–1.65) | 0.0005 | 0.0004 | 0.17 | 0.54 (−0.24–1.32) |
| Lag2 | 0.0006 | 0.0003 | 0.09 @ | 0.60 (−0.09–1.29) | 0.0008 | 0.0004 | 0.03 * | 0.83 (0.08–1.59) | 0.0007 | 0.0004 | 0.07 @ | 0.72 (−0.05–1.50) |
| Lag3 | 0.0003 | 0.0003 | 0.32 | 0.34 (−0.33–1.01) | 0.0006 | 0.0004 | 0.10 | 0.61 (−0.12–1.35) | 0.0006 | 0.0004 | 0.12 | 0.60 (−0.16–1.36) |
| Lag4 | 0.0004 | 0.0003 | 0.18 | 0.45 (−0.21–1.11) | 0.0009 | 0.0004 | 0.02 * | 0.89 (0.16–1.62) | 0.0005 | 0.0004 | 0.16 | 0.54 (−0.21–1.30) |
| Lag5 | 0.0007 | 0.0003 | 0.05 * | 0.67 (0.01–1.33) | 0.0009 | 0.0004 | 0.02 * | 0.88 (0.16–1.61) | 0.0011 | 0.0004 | 0.00 # | 1.10 (0.35–1.86) |
| Lag01 | 0.0006 | 0.0004 | 0.16 | 0.57 (−0.23–1.38) | 0.0010 | 0.0005 | 0.03 * | 0.96 (0.07–1.86) | 0.0005 | 0.0005 | 0.26 | 0.51 (−0.37–1.41) |
| Lag02 | 0.0007 | 0.0004 | 0.09 @ | 0.74 (−0.11–1.60) | 0.0011 | 0.0005 | 0.02 * | 1.10 (0.18–2.04) | 0.0008 | 0.0005 | 0.11 | 0.79 (−0.19–1.78) |
| Lag03 | 0.0008 | 0.0005 | 0.10 @ | 0.76 (−0.14–1.66) | 0.0011 | 0.0005 | 0.02 * | 1.13 (0.17–2.10) | 0.0010 | 0.0005 | 0.07 @ | 0.98 (−0.07–2.04) |
| Lag04 | 0.0008 | 0.0005 | 0.08 @ | 0.83 (−0.09–1.76) | 0.0012 | 0.0005 | 0.01 * | 1.24 (0.25–2.23) | 0.0011 | 0.0006 | 0.05 * | 1.11 (0.01–2.23) |
| Accidental mortality | ||||||||||||
| Lag0 | 0.0012 | 0.0009 | 0.17 | 1.19 (−0.50–2.91) | 0.0010 | 0.0010 | 0.29 | 1.04 (−0.89–3.01) | 0.0012 | 0.0009 | 0.20 | 1.20 (−0.64–3.07) |
| Lag1 | 0.0013 | 0.0008 | 0.10 | 1.36 (−0.28–3.02) | 0.0016 | 0.0009 | 0.08 @ | 1.63 (−0.18–3.47) | 0.0013 | 0.0009 | 0.15 | 1.36 (−0.47–3.22) |
| Lag2 | 0.0018 | 0.0008 | 0.03 * | 1.81 (0.22–3.42) | 0.0021 | 0.0009 | 0.02 * | 2.16 (0.41–3.94) | 0.0013 | 0.0009 | 0.15 | 1.33 (−0.48–3.17) |
| Lag3 | 0.0007 | 0.0008 | 0.36 | 0.73 (−0.82–2.31) | 0.0006 | 0.0009 | 0.49 | 0.61 (−1.09–2.34) | 0.0008 | 0.0009 | 0.40 | 0.77 (−1.02–2.60) |
| Lag4 | 0.0007 | 0.0008 | 0.39 | 0.67 (−0.86–2.22) | 0.0005 | 0.0009 | 0.56 | 0.50 (−1.18–2.20) | 0.0004 | 0.0009 | 0.69 | 0.36 (−1.41–2.17) |
| Lag5 | 0.0012 | 0.0008 | 0.14 | 1.16 (−0.36–2.71) | 0.0008 | 0.0009 | 0.38 | 0.76 (−0.92–2.46) | 0.0012 | 0.0009 | 0.18 | 1.22 (−0.56–3.03) |
| Lag01 | 0.0016 | 0.0009 | 0.09 @ | 1.61 (−0.24–3.49) | 0.0017 | 0.0010 | 0.11 | 1.69 (−0.35–3.78) | 0.0016 | 0.0011 | 0.12 | 1.64 (−0.44–3.77) |
| Lag02 | 0.0022 | 0.0010 | 0.02 * | 2.26 (0.30–4.25) | 0.0024 | 0.0011 | 0.03 * | 2.40 (0.27–4.56) | 0.0020 | 0.0011 | 0.08 @ | 2.01 (−0.26–4.33) |
| Lag03 | 0.0022 | 0.0010 | 0.03 * | 2.19 (0.16–4.27) | 0.0022 | 0.0011 | 0.04 * | 2.24 (0.05–4.47) | 0.0020 | 0.0012 | 0.10 | 2.03 (−0.39–4.50) |
| Lag04 | 0.0021 | 0.0010 | 0.05 * | 2.08 (0.02–4.17) | 0.0019 | 0.0011 | 0.08 @ | 1.95 (−0.25–4.19) | 0.0018 | 0.0013 | 0.15 | 1.85 (−0.64–4.40) |
| Total respiratory mortality | ||||||||||||
| Lag0 | −0.0014 | 0.0014 | 0.30 | −1.43 (−4.06–1.27) | −0.0006 | 0.0016 | 0.73 | −0.55 (−3.58–2.57) | −0.0019 | 0.0015 | 0.21 | −1.83 (−4.64–1.06) |
| Lag1 | 0.0004 | 0.0013 | 0.74 | 0.44 (−2.12–3.07) | 0.0009 | 0.0015 | 0.56 | 0.85 (−1.98–3.77) | −0.0012 | 0.0015 | 0.43 | −1.15 (−3.95–1.74) |
| Lag2 | 0.0020 | 0.0013 | 0.12 | 2.02 (−0.49–4.58) | 0.0011 | 0.0014 | 0.41 | 1.14 (−1.56–3.91) | 0.0018 | 0.0014 | 0.20 | 1.85 (−0.96–4.75) |
| Lag3 | 0.0030 | 0.0012 | 0.01 * | 3.04 (0.60–5.55) | 0.0026 | 0.0013 | 0.05 @ | 2.62 (−0.04–5.34) | 0.0041 | 0.0014 | 0.00 # | 4.17 (1.40–7.02) |
| Lag4 | 0.0021 | 0.0012 | 0.08 @ | 2.17 (−0.25–4.65) | 0.0020 | 0.0013 | 0.13 | 2.05 (−0.59–4.77) | 0.0015 | 0.0014 | 0.28 | 1.53 (−1.21–4.36) |
| Lag5 | 0.0023 | 0.0012 | 0.06 @ | 2.36 (−0.05–4.83) | 0.0018 | 0.0013 | 0.18 | 1.79 (−0.83–4.48) | 0.0019 | 0.0014 | 0.18 | 1.88 (−0.86–4.69) |
| Lag01 | −0.0006 | 0.0015 | 0.69 | −0.59 (−3.47–2.38) | 0.0001 | 0.0017 | 0.94 | 0.13 (−3.08–3.43) | −0.0021 | 0.0017 | 0.22 | −2.07 (−5.26–1.22) |
| Lag02 | 0.0006 | 0.0016 | 0.72 | 0.58 (−2.50–3.76) | 0.0007 | 0.0017 | 0.68 | 0.70 (−2.62–4.14) | −0.0009 | 0.0019 | 0.64 | −0.85 (−4.39–2.81) |
| Lag03 | 0.0018 | 0.0016 | 0.27 | 1.82 (−1.41–5.16) | 0.0017 | 0.0017 | 0.33 | 1.73 (−1.69–5.28) | 0.0012 | 0.0020 | 0.54 | 1.22 (−2.59–5.17) |
| Lag04 | 0.0024 | 0.0017 | 0.16 | 2.42 (−0.93–5.88) | 0.0021 | 0.0018 | 0.24 | 2.12 (−1.40–5.77) | 0.0015 | 0.0021 | 0.46 | 1.53 (−2.48–5.71) |
| Chronic lower respiratory disease mortality | ||||||||||||
| Lag0 | 0.0015 | 0.0021 | 0.48 | 1.48 (−2.54–5.66) | 0.0006 | 0.0024 | 0.80 | 0.60 (−3.93–5.35) | 0.0015 | 0.0022 | 0.50 | 1.47 (−2.75–5.87) |
| Lag1 | 0.0012 | 0.0020 | 0.54 | 1.22 (−2.65–5.24) | 0.0005 | 0.0022 | 0.81 | 0.53 (−3.68–4.92) | 0.0010 | 0.0022 | 0.66 | 0.96 (−3.21–5.32) |
| Lag2 | 0.0043 | 0.0019 | 0.02 * | 4.43 (0.71–8.29) | 0.0022 | 0.0020 | 0.28 | 2.20 (−1.77–6.34) | 0.0056 | 0.0020 | 0.01 # | 5.81 (1.68–10.11) |
| Lag3 | 0.0062 | 0.0018 | 0.00 # | 6.38 (2.78–10.11) | 0.0043 | 0.0019 | 0.03 * | 4.40 (0.50–8.44) | 0.0093 | 0.0019 | 0.00 # | 9.71 (5.60–13.97) |
| Lag4 | 0.0034 | 0.0018 | 0.06 @ | 3.41 (−0.14–7.08) | 0.0021 | 0.0020 | 0.27 | 2.17 (−1.66–6.16) | 0.0039 | 0.0020 | 0.05 @ | 3.93 (−0.06–8.08) |
| Lag5 | 0.0028 | 0.0018 | 0.12 | 2.80 (−0.74–6.46) | 0.0015 | 0.0020 | 0.43 | 1.56 (−2.26–5.53) | 0.0025 | 0.0020 | 0.22 | 2.51 (−1.47–6.65) |
| Lag01 | 0.0016 | 0.0023 | 0.47 | 1.66 (−2.74–6.26) | 0.0008 | 0.0025 | 0.75 | 0.81 (−3.98–5.83) | 0.0015 | 0.0024 | 0.54 | 1.52 (−3.23–6.51) |
| Lag02 | 0.0035 | 0.0024 | 0.13 | 3.60 (−1.08–8.50) | 0.0020 | 0.0025 | 0.42 | 2.07 (−2.89–7.29) | 0.0043 | 0.0026 | 0.10 | 4.36 (−0.83–9.83) |
| Lag03 | 0.0057 | 0.0024 | 0.02 * | 5.84 (0.94–10.98) | 0.0038 | 0.0026 | 0.14 | 3.87 (−1.27–9.28) | 0.0079 | 0.0027 | 0.00 # | 8.26 (2.66–14.16) |
| Lag04 | 0.0059 | 0.0025 | 0.02 * | 6.05 (1.01–11.35) | 0.0036 | 0.0026 | 0.18 | 3.64 (−1.59–9.15) | 0.0083 | 0.0028 | 0.00 # | 8.64 (2.75–14.86) |
| Chronic obstructive pulmonary disease mortality | ||||||||||||
| Lag0 | −0.0011 | 0.0027 | 0.68 | −1.09 (−6.10–4.19) | −0.0012 | 0.0030 | 0.69 | −1.18 (−6.83–4.81) | −0.0006 | 0.0028 | 0.83 | −0.61 (−5.91–4.99) |
| Lag1 | 0.0000 | 0.0026 | 0.99 | −0.03 (−4.92–5.12) | 0.0006 | 0.0028 | 0.84 | 0.56 (−4.84–6.26) | −0.0001 | 0.0028 | 0.98 | −0.05 (−5.37–5.56) |
| Lag2 | 0.0052 | 0.0024 | 0.03 * | 5.37 (0.55–10.41) | 0.0035 | 0.0026 | 0.18 | 3.57 (−1.57–8.98) | 0.0070 | 0.0026 | 0.01 # | 7.29 (1.89–12.98) |
| Lag3 | 0.0079 | 0.0023 | 0.00 # | 8.24 (3.53–13.17) | 0.0063 | 0.0025 | 0.01 * | 6.52 (1.43–11.86) | 0.0122 | 0.0025 | 0.00 # | 13.01 (7.53–18.77) |
| Lag4 | 0.0044 | 0.0023 | 0.06 @ | 4.46 (−0.18–9.32) | 0.0029 | 0.0025 | 0.26 | 2.89 (−2.09–8.14) | 0.0053 | 0.0026 | 0.04 * | 5.40 (0.12–10.95) |
| Lag5 | 0.0052 | 0.0023 | 0.03 * | 5.29 (0.61–10.20) | 0.0030 | 0.0025 | 0.24 | 3.01 (−1.98–8.25) | 0.0055 | 0.0026 | 0.03 * | 5.70 (0.39–11.29) |
| Lag01 | −0.0007 | 0.0029 | 0.82 | −0.67 (−6.14–5.13) | −0.0003 | 0.0032 | 0.92 | −0.33 (−6.35–6.09) | −0.0003 | 0.0032 | 0.92 | −0.33 (−6.31–6.02) |
| Lag02 | 0.0024 | 0.0030 | 0.43 | 2.40 (−3.51–8.69) | 0.0019 | 0.0033 | 0.56 | 1.92 (−4.40–8.65) | 0.0037 | 0.0034 | 0.27 | 3.81 (−2.85–10.94) |
| Lag03 | 0.0057 | 0.0031 | 0.07 @ | 5.83 (−0.47–12.53) | 0.0047 | 0.0033 | 0.16 | 4.82 (−1.81–11.90) | 0.0093 | 0.0036 | 0.01 # | 9.72 (2.34–17.62) |
| Lag04 | 0.0062 | 0.0032 | 0.06 @ | 6.36 (−0.18–13.32) | 0.0043 | 0.0034 | 0.20 | 4.42 (−2.30–11.61) | 0.0106 | 0.0038 | 0.01 # | 11.15 (3.18–19.74) |
| Male | ||||||||||||
| Lag0 | 0.0007 | 0.0004 | 0.10 | 0.70 (−0.14–1.56) | 0.0010 | 0.0005 | 0.05 * | 0.98 (0.01–1.97) | 0.0005 | 0.0005 | 0.24 | 0.55 (−0.36–1.46) |
| Lag1 | 0.0006 | 0.0004 | 0.14 | 0.62 (−0.20–1.44) | 0.0009 | 0.0005 | 0.06 @ | 0.87 (−0.03–1.78) | 0.0005 | 0.0005 | 0.26 | 0.51 (−0.39–1.42) |
| Lag2 | 0.0010 | 0.0004 | 0.01 # | 1.04 (0.25–1.84) | 0.0011 | 0.0004 | 0.01 * | 1.09 (0.23–1.97) | 0.0011 | 0.0005 | 0.02 * | 1.09 (0.19–1.99) |
| Lag3 | 0.0005 | 0.0004 | 0.25 | 0.45 (−0.32–1.23) | 0.0004 | 0.0004 | 0.39 | 0.37 (−0.48–1.22) | 0.0008 | 0.0004 | 0.07 @ | 0.81 (−0.07–1.70) |
| Lag4 | 0.0004 | 0.0004 | 0.35 | 0.36 (−0.40–1.13) | 0.0005 | 0.0004 | 0.28 | 0.46 (−0.38–1.30) | 0.0004 | 0.0004 | 0.33 | 0.43 (−0.44–1.31) |
| Lag5 | 0.0007 | 0.0004 | 0.09 @ | 0.65 (−0.10–1.42) | 0.0004 | 0.0004 | 0.33 | 0.41 (−0.42–1.25) | 0.0010 | 0.0004 | 0.02 * | 1.03 (0.17–1.91) |
| Lag01 | 0.0009 | 0.0005 | 0.07 @ | 0.86 (−0.07–1.79) | 0.0011 | 0.0005 | 0.03 * | 1.13 (0.10–2.17) | 0.0007 | 0.0005 | 0.20 | 0.67 (−0.36–1.71) |
| Lag02 | 0.0012 | 0.0005 | 0.02 * | 1.22 (0.23–2.22) | 0.0014 | 0.0005 | 0.01 # | 1.40 (0.34–2.48) | 0.0011 | 0.0006 | 0.06 @ | 1.11 (−0.02–2.25) |
| Lag03 | 0.0012 | 0.0005 | 0.02 * | 1.21 (0.18–2.25) | 0.0013 | 0.0006 | 0.02 * | 1.31 (0.21–2.43) | 0.0013 | 0.0006 | 0.03 * | 1.36 (0.14–2.58) |
| Lag04 | 0.0012 | 0.0005 | 0.02 * | 1.21 (0.15–2.27) | 0.0013 | 0.0006 | 0.03 * | 1.26 (0.14–2.40) | 0.0014 | 0.0006 | 0.02 * | 1.45 (0.19– 2.72) |
| Female | ||||||||||||
| Lag0 | 0.0002 | 0.0006 | 0.77 | 0.16 (−0.92–1.25) | 0.0005 | 0.0006 | 0.45 | 0.47 (−0.76–1.73) | 0.0002 | 0.0006 | 0.77 | 0.17 (−0.98–1.34) |
| Lag1 | 0.0006 | 0.0005 | 0.25 | 0.61 (−0.43–1.66) | 0.0011 | 0.0006 | 0.07 @ | 1.09 (−0.07–2.26) | 0.0009 | 0.0006 | 0.14 | 0.86 (−0.29–2.03) |
| Lag2 | 0.0003 | 0.0005 | 0.55 | 0.31 (−0.71–1.34) | 0.0009 | 0.0006 | 0.13 | 0.87 (−0.25–2.00) | 0.0004 | 0.0006 | 0.53 | 0.37 (−0.78–1.54) |
| Lag3 | 0.0003 | 0.0005 | 0.59 | 0.28 (−0.72–1.28) | 0.0009 | 0.0006 | 0.09 @ | 0.95 (−0.15–2.06) | 0.0003 | 0.0006 | 0.57 | 0.33 (−0.81–1.48) |
| Lag4 | 0.0007 | 0.0005 | 0.18 | 0.68 (−0.31–1.67) | 0.0014 | 0.0006 | 0.01 * | 1.42 (0.32–2.53) | 0.0007 | 0.0006 | 0.22 | 0.70 (−0.43–1.85) |
| Lag5 | 0.0009 | 0.0005 | 0.06 @ | 0.94 (−0.05–1.93) | 0.0016 | 0.0006 | 0.00 # | 1.62 (0.52–2.73) | 0.0014 | 0.0006 | 0.02 * | 1.39 (0.26–2.53) |
| Lag01 | 0.0005 | 0.0006 | 0.43 | 0.47 (−0.70–1.66) | 0.0009 | 0.0007 | 0.17 | 0.92 (−0.39–2.24) | 0.0007 | 0.0007 | 0.32 | 0.67 (−0.64–1.99) |
| Lag02 | 0.0005 | 0.0006 | 0.43 | 0.51 (−0.74–1.78) | 0.0011 | 0.0007 | 0.13 | 1.06 (−0.31–2.44) | 0.0007 | 0.0007 | 0.31 | 0.75 (−0.70–2.22) |
| Lag03 | 0.0005 | 0.0007 | 0.42 | 0.54 (−0.77–1.86) | 0.0012 | 0.0007 | 0.10 @ | 1.20 (−0.21–2.63) | 0.0008 | 0.0008 | 0.30 | 0.82 (−0.73–2.40) |
| Lag04 | 0.0007 | 0.0007 | 0.30 | 0.72 (−0.63–2.09) | 0.0015 | 0.0007 | 0.05 * | 1.47 (0.01–2.96) | 0.0010 | 0.0008 | 0.22 | 1.02 (−0.62–2.68) |
| Elder | ||||||||||||
| Lag0 | 0.0006 | 0.0005 | 0.22 | 0.61 (−0.35–1.57) | 0.0009 | 0.0006 | 0.09 @ | 0.95 (−0.15–2.06) | 0.0005 | 0.0005 | 0.31 | 0.54 (−0.49–1.57) |
| Lag1 | 0.0009 | 0.0005 | 0.07 @ | 0.85 (−0.07–1.78) | 0.0011 | 0.0005 | 0.03 * | 1.14 (0.11–2.17) | 0.0009 | 0.0005 | 0.08 @ | 0.91 (−0.11–1.93) |
| Lag2 | 0.0011 | 0.0005 | 0.02 * | 1.10 (0.20–2.00) | 0.0012 | 0.0005 | 0.02 * | 1.22 (0.23–2.21) | 0.0010 | 0.0005 | 0.06 @ | 0.98 (−0.04–2.01) |
| Lag3 | 0.0009 | 0.0004 | 0.04 * | 0.91 (0.03–1.79) | 0.0011 | 0.0005 | 0.03 * | 1.09 (0.12–2.06) | 0.0008 | 0.0005 | 0.12 | 0.78 (−0.21–1.79) |
| Lag4 | 0.0010 | 0.0004 | 0.03 * | 0.98 (0.12–1.86) | 0.0013 | 0.0005 | 0.01 # | 1.30 (0.34–2.26) | 0.0010 | 0.0005 | 0.06 @ | 0.97 (−0.02–1.96) |
| Lag5 | 0.0013 | 0.0004 | 0.00 # | 1.32 (0.46–2.19) | 0.0013 | 0.0005 | 0.01 # | 1.32 (0.37–2.28) | 0.0017 | 0.0005 | 0.00 # | 1.74 (0.75–2.74) |
| Lag01 | 0.0009 | 0.0005 | 0.08 @ | 0.92 (−0.12–1.98) | 0.0013 | 0.0006 | 0.03 * | 1.26 (0.10–2.43) | 0.0009 | 0.0006 | 0.12 | 0.91 (−0.25–2.08) |
| Lag02 | 0.0013 | 0.0006 | 0.03 * | 1.27 (0.15–2.40) | 0.0015 | 0.0006 | 0.01 * | 1.54 (0.33–2.77) | 0.0012 | 0.0007 | 0.06 @ | 1.22 (−0.06–2.52) |
| Lag03 | 0.0014 | 0.0006 | 0.02 * | 1.44 (0.27–2.62) | 0.0017 | 0.0006 | 0.01 # | 1.70 (0.45–2.96) | 0.0014 | 0.0007 | 0.05 @ | 1.37 (−0.01–2.76) |
| Lag04 | 0.0016 | 0.0006 | 0.01 # | 1.57 (0.38–2.78) | 0.0018 | 0.0006 | 0.00 # | 1.82 (0.55–3.11) | 0.0015 | 0.0007 | 0.04 * | 1.53 (0.10–2.99) |
| Younger | ||||||||||||
| Lag0 | 0.0004 | 0.0005 | 0.39 | 0.41 (−0.51–1.34) | 0.0007 | 0.0005 | 0.22 | 0.67 (−0.39–1.74) | 0.0003 | 0.0005 | 0.54 | 0.31 (−0.68–1.31) |
| Lag1 | 0.0004 | 0.0005 | 0.40 | 0.39 (−0.50–1.28) | 0.0008 | 0.0005 | 0.12 | 0.78 (−0.21–1.78) | 0.0004 | 0.0005 | 0.45 | 0.38 (−0.60–1.38) |
| Lag2 | 0.0004 | 0.0004 | 0.35 | 0.42 (−0.45–1.29) | 0.0008 | 0.0005 | 0.11 | 0.79 (−0.17–1.75) | 0.0006 | 0.0005 | 0.22 | 0.61 (−0.37–1.60) |
| Lag3 | −0.0001 | 0.0004 | 0.80 | −0.11 (−0.96–0.74) | 0.0001 | 0.0005 | 0.77 | 0.14 (−0.79–1.07) | 0.0005 | 0.0005 | 0.36 | 0.45 (−0.51–1.43) |
| Lag4 | 0.0000 | 0.0004 | 0.98 | −0.01 (−0.85–0.83) | 0.0004 | 0.0005 | 0.45 | 0.35 (−0.57–1.28) | 0.0001 | 0.0005 | 0.86 | 0.09 (−0.87–1.06) |
| Lag5 | 0.0002 | 0.0004 | 0.65 | 0.19 (−0.64–1.03) | 0.0004 | 0.0005 | 0.36 | 0.43 (−0.49–1.35) | 0.0005 | 0.0005 | 0.28 | 0.53 (−0.43–1.49) |
| Lag01 | 0.0005 | 0.0005 | 0.33 | 0.51 (−0.51–1.53) | 0.0009 | 0.0006 | 0.13 | 0.86 (−0.26–2.00) | 0.0004 | 0.0006 | 0.45 | 0.43 (−0.69–1.57) |
| Lag02 | 0.0006 | 0.0005 | 0.26 | 0.62 (−0.46–1.71) | 0.0010 | 0.0006 | 0.10 @ | 0.99 (−0.18–2.18) | 0.0007 | 0.0006 | 0.29 | 0.68 (−0.57–1.94) |
| Lag03 | 0.0004 | 0.0006 | 0.44 | 0.45 (−0.68–1.58) | 0.0008 | 0.0006 | 0.18 | 0.83 (−0.38–2.05) | 0.0008 | 0.0007 | 0.22 | 0.85 (−0.49–2.20) |
| Lag04 | 0.0004 | 0.0006 | 0.53 | 0.38 (−0.79–1.56) | 0.0008 | 0.0006 | 0.23 | 0.77 (−0.48–2.02) | 0.0008 | 0.0007 | 0.26 | 0.82 (−0.59–2.25) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cai, J.; Peng, C.; Yu, S.; Pei, Y.; Liu, N.; Wu, Y.; Fu, Y.; Cheng, J. Association between PM2.5 Exposure and All-Cause, Non-Accidental, Accidental, Different Respiratory Diseases, Sex and Age Mortality in Shenzhen, China. Int. J. Environ. Res. Public Health 2019, 16, 401. https://doi.org/10.3390/ijerph16030401
Cai J, Peng C, Yu S, Pei Y, Liu N, Wu Y, Fu Y, Cheng J. Association between PM2.5 Exposure and All-Cause, Non-Accidental, Accidental, Different Respiratory Diseases, Sex and Age Mortality in Shenzhen, China. International Journal of Environmental Research and Public Health. 2019; 16(3):401. https://doi.org/10.3390/ijerph16030401
Chicago/Turabian StyleCai, Junfang, Chaoqiong Peng, Shuyuan Yu, Yingxin Pei, Ning Liu, Yongsheng Wu, Yingbin Fu, and Jinquan Cheng. 2019. "Association between PM2.5 Exposure and All-Cause, Non-Accidental, Accidental, Different Respiratory Diseases, Sex and Age Mortality in Shenzhen, China" International Journal of Environmental Research and Public Health 16, no. 3: 401. https://doi.org/10.3390/ijerph16030401
APA StyleCai, J., Peng, C., Yu, S., Pei, Y., Liu, N., Wu, Y., Fu, Y., & Cheng, J. (2019). Association between PM2.5 Exposure and All-Cause, Non-Accidental, Accidental, Different Respiratory Diseases, Sex and Age Mortality in Shenzhen, China. International Journal of Environmental Research and Public Health, 16(3), 401. https://doi.org/10.3390/ijerph16030401





