Association between Asthma Control and Exposure to Greenness and Other Outdoor and Indoor Environmental Factors: A Longitudinal Study on a Cohort of Asthmatic Children
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.1.1. Inclusion and Exclusion Criteria
2.1.2. Clinical Assessments
2.1.3. Questionnaire
2.1.4. Skin Prick Tests
2.1.5. Childhood Asthma Control Test (C-ACT) and Asthma Control Test (ACT)
2.1.6. Spirometry Parameters
2.2. Environmental Exposures
2.2.1. CORINE Land Cover Classes
2.2.2. Nitrogen Dioxide (NO2) Concentrations
2.2.3. Normalized Difference Vegetation Index (NDVI)
2.3. Sample Size & Statistical Analysis
3. Results
3.1. Characteristics of the Study Sample
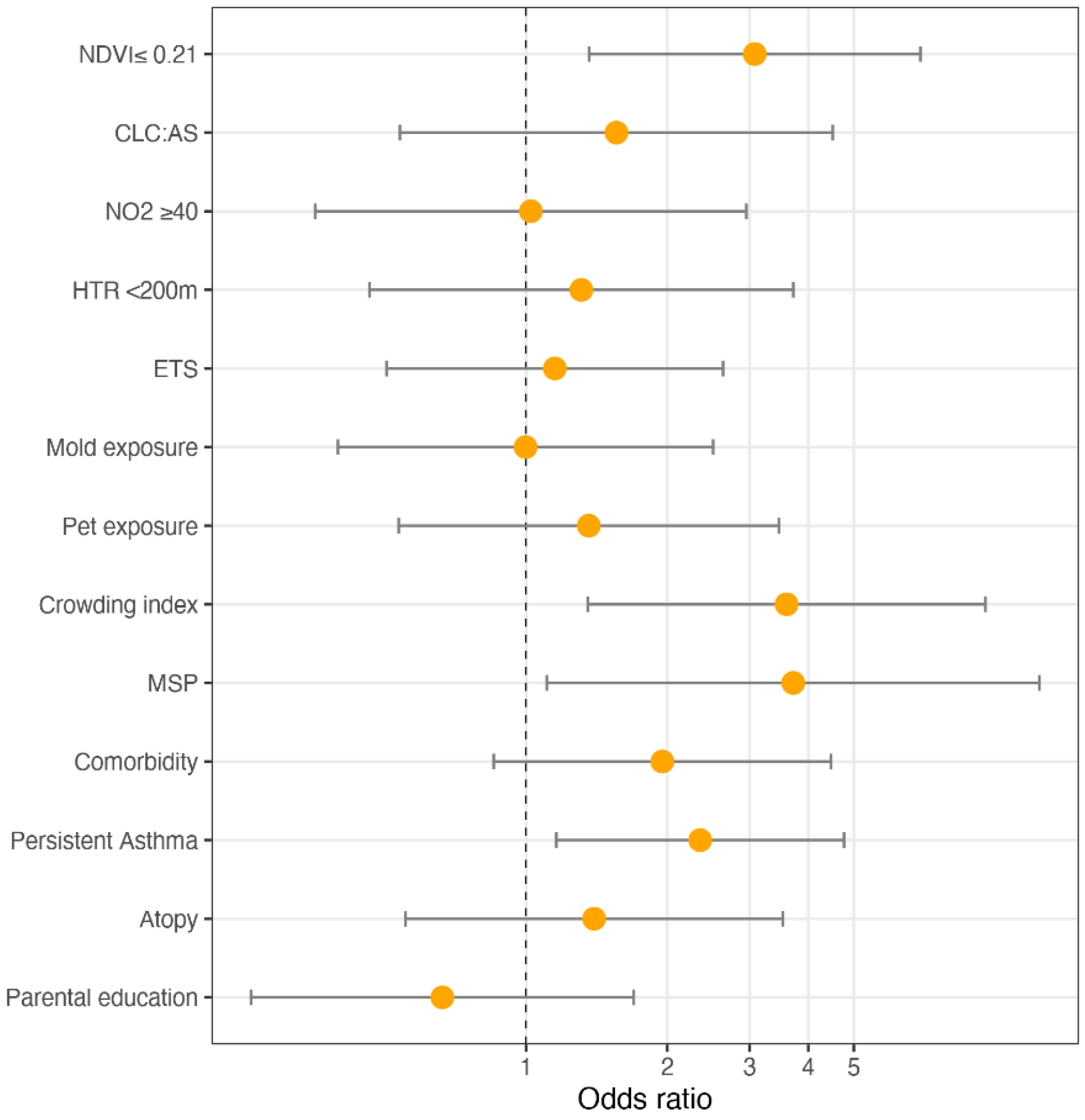
3.2. Multivariable Analyses
3.3. Sensitivity Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dharmage, S.C.; Perret, J.; Custovic, A. Epidemiology of asthma in children and adults. Front. Pediatr. 2019, 7, 246. [Google Scholar] [CrossRef] [PubMed]
- Bateman, E.; Hurd, S.; Barnes, P.; Bousquet, J.; Drazen, J.; FitzGerald, M.; Gibson, P.; Ohta, K.; O’Byrne, P.; Pedersen, S.; et al. Global strategy for asthma management and prevention: GINA executive summary. Eur. Respir. J. 2008, 31, 143–178. [Google Scholar] [CrossRef] [PubMed]
- Montalbano, L.; Cilluffo, G.; Gentile, M.; Ferrante, G.; Malizia, V.; Cibella, F.; Viegi, G.; Passalacqua, G.; La Grutta, S. Development of a nomogram to estimate the quality of life in asthmatic children using the Childhood Asthma Control Test. Pediatr. Allergy Immunol. 2016, 27, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Etzel, R.A. How environmental exposures influence the development and exacerbation of asthma. Pediatrics 2003, 112, 233–239. [Google Scholar] [CrossRef]
- Ferrante, G.; Asta, F.; Cilluffo, G.; De Sario, M.; Michelozzi, P.; La Grutta, S. The effect of residential urban greenness on allergic respiratory diseases in youth: A narrative review. World Allergy Organ. J. 2020, 13, 100096. [Google Scholar] [CrossRef] [Green Version]
- Andrusaityte, S.; Grazuleviciene, R.; Kudzyte, J.; Bernotiene, A.; Dedele, A.; Nieuwenhuijsen, M.J. Associations between neighbourhood greenness and asthma in preschool children in Kaunas, Lithuania: A case–control study. BMJ Open 2016, 6, e010341. [Google Scholar] [CrossRef] [Green Version]
- Dadvand, P.; Villanueva, C.M.; Font-Ribera, L.; Martinez, D.; Basagaña, X.; Belmonte, J.; Vrijheid, M.; Gražulevičienė, R.; Kogevinas, M.; Nieuwenhuijsen, M.J. Risks and benefits of green spaces for children: A cross-sectional study of associations with sedentary behavior, obesity, asthma, and allergy. Environ. Health Perspect. 2014, 122, 1329. [Google Scholar] [CrossRef]
- Wu, M.; Xie, J.; Wang, Y.; Tian, Y. Greenness and eosinophilic asthma: Findings from the UK Biobank. Eur. Respir. J. 2021, 58, 2101597. [Google Scholar] [CrossRef]
- Hanski, I.; von Hertzen, L.; Fyhrquist, N.; Koskinen, K.; Torppa, K.; Laatikainen, T.; Karisola, P.; Auvinen, P.; Paulin, L.; Mäkelä, M.J.; et al. Environmental biodiversity, human microbiota, and allergy are interrelated. Proc. Natl. Acad. Sci. USA 2012, 109, 8334–8339. [Google Scholar] [CrossRef] [Green Version]
- Lovasi, G.S.; Quinn, J.W.; Neckerman, K.M.; Perzanowski, M.S.; Rundle, A. Children living in areas with more street trees have lower prevalence of asthma. J. Epidemiol. Community Health 2008, 62, 647–649. [Google Scholar] [CrossRef] [Green Version]
- Feng, X.; Astell-Burt, T. Is neighborhood green space protective against associations between child asthma, neighborhood traffic volume and perceived lack of area safety? Multilevel analysis of 4447 Australian children. Int. J. Environ. Res. Public. Health 2017, 14, 543. [Google Scholar] [CrossRef] [Green Version]
- Lovasi, G.S.; O’Neil-Dunne, J.P.; Lu, J.W.; Sheehan, D.; Perzanowski, M.S.; MacFaden, S.W.; King, K.L.; Matte, T.; Miller, R.L.; Hoepner, L.A.; et al. Urban tree canopy and asthma, wheeze, rhinitis, and allergic sensitization to tree pollen in a New York City birth cohort. Environ. Health Perspect. 2013, 121, 494. [Google Scholar] [CrossRef] [PubMed]
- Aerts, R.; Dujardin, S.; Nemery, B.; Van Nieuwenhuyse, A.; Van Orshoven, J.; Aerts, J.-M.; Somers, B.; Hendrickx, M.; Bruffaerts, N.; Bauwelinck, M.; et al. Residential green space and medication sales for childhood asthma: A longitudinal ecological study in Belgium. Environ. Res. 2020, 189, 109914. [Google Scholar] [CrossRef] [PubMed]
- Tischer, C.; Gascon, M.; Fernández-Somoano, A.; Tardón, A.; Materola, A.L.; Ibarluzea, J.; Ferrero, A.; Estarlich, M.; Cirach, M.; Vrijheid, M.; et al. Urban green and grey space in relation to respiratory health in children. Eur. Respir. J. 2017, 49, 1502112. [Google Scholar] [CrossRef] [Green Version]
- Cavaleiro Rufo, J.; Paciência, I.; Hoffimann, E.; Moreira, A.; Barros, H.; Ribeiro, A.I. The neighbourhood natural environment is associated with asthma in children: A birth cohort study. Allergy 2021, 76, 348–358. [Google Scholar] [CrossRef]
- Chen, E.; Miller, G.E.; Shalowitz, M.U.; Story, R.E.; Levine, C.S.; Hayen, R.; Sbihi, H.; Brauer, M. Difficult family relationships, residential greenspace, and childhood asthma. Pediatrics 2017, 139, e20163056. [Google Scholar] [CrossRef] [Green Version]
- Cowan, K.N.; Qin, X.; Ruiz Serrano, K.; Sircar, K.; Pennington, A.F. Uncontrolled asthma and household environmental exposures in Puerto Rico. J. Asthma 2020, 1–11. [Google Scholar] [CrossRef]
- Matsui, E.C.; Abramson, S.L.; Sandel, M.T.; Dinakar, C.; Irani, A.M.; Kim, J.S.; Mahr, T.A.; Pistiner, M.; Wang, J.; Lowry, J.A.; et al. Indoor environmental control practices and asthma management. Pediatrics 2016, 138, e20162589. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guarnieri, M.; Balmes, J.R. Outdoor air pollution and asthma. Lancet 2014, 383, 1581–1592. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.; Xu, X.; Thompson, L.A.; Gross, H.E.; Shenkman, E.A.; DeWalt, D.A.; Huang, I.-C. Longitudinal effect of ambient air pollution and pollen exposure on asthma control: The Patient-Reported Outcomes Measurement Information System (PROMIS) pediatric asthma study. Acad. Pediatr. 2019, 19, 615–623. [Google Scholar] [CrossRef]
- Cilluffo, G.; Ferrante, G.; Fasola, S.; Montalbano, L.; Malizia, V.; Piscini, A.; Romaniello, V.; Silvestri, M.; Stramondo, S.; Stafoggia, M.; et al. Associations of greenness, greyness and air pollution exposure with children’s health: A cross-sectional study in Southern Italy. Environ. Health 2018, 17, 86. [Google Scholar] [CrossRef] [Green Version]
- Squillacioti, G.; Bellisario, V.; Levra, S.; Piccioni, P.; Bono, R. Greenness availability and respiratory health in a population of urbanised children in North-Western Italy. Int. J. Environ. Res. Public. Health 2019, 17, 108. [Google Scholar] [CrossRef] [Green Version]
- Maio, S.; Baldacci, S.; Tagliaferro, S.; Angino, A.; Parmes, E.; Pärkkä, J.; Pesce, G.; Maesano, C.; Annesi-Maesano, I.; Viegi, G. Urban grey spaces are associated with increased allergy in the general population. Environ. Res. 2021, 112428. [Google Scholar] [CrossRef]
- Renzoni, E.; Sestini, P.; Corbo, G.; Biggeri, A.; Viegi, G.; Forastiere, F.; on behalf of the SIDRIA Collaborative Group. Asthma and respiratory symptoms in 6-7 yr old Italian children: Gender, latitude, urbanization and socioeconomic factors. Eur. Respir. J. 1997, 10, 1780–1786. [Google Scholar] [CrossRef]
- Liu, A.H.; Zeiger, R.; Sorkness, C.; Mahr, T.; Ostrom, N.; Burgess, S.; Rosenzweig, J.C.; Manjunath, R. Development and cross-sectional validation of the Childhood Asthma Control Test. J. Allergy Clin. Immunol. 2007, 119, 817–825. [Google Scholar] [CrossRef]
- ATS/ERS European Respiratory Society. ATS/ERS recommendations for standardized procedures for the online and offline measurement of exhaled lower respiratory nitric oxide and nasal nitric oxide, 2005. Am. J. Respir. Crit Care Med. 2005, 171, 912–930. [Google Scholar] [CrossRef]
- Quanjer, P.H.; Hall, G.L.; Stanojevic, S.; Cole, T.J.; Stocks, J. Age-and height-based prediction bias in spirometry reference equations. Eur. Respir. J. 2012, 40, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Beelen, R.; Hoek, G.; Vienneau, D.; Eeftens, M.; Dimakopoulou, K.; Pedeli, X.; Tsai, M.-Y.; Künzli, N.; Schikowski, T.; Marcon, A.; et al. Development of NO2 and NOx land use regression models for estimating air pollution exposure in 36 study areas in Europe–the ESCAPE project. Atmos. Environ. 2013, 72, 10–23. [Google Scholar] [CrossRef]
- Vienneau, D.; De Hoogh, K.; Beelen, R.; Fischer, P.; Hoek, G.; Briggs, D. Comparison of land-use regression models between Great Britain and the Netherlands. Atmos. Environ. 2010, 44, 688–696. [Google Scholar] [CrossRef]
- Weier, J.; Herring, D. Measuring Vegetation (NDVI&EVI). 2011. Available online: https://earthobservatory.nasa.gov/Features/MeasuringVegetation/ (accessed on 27 July 2017).
- Lüdecke, D. Sjstats: Statistical Functions for Regression Models (Version 0.18.1). 2021. Available online: https://CRAN.R-project.org/package=sjstats (accessed on 1 September 2021).
- Stekhoven, D.J.; Bühlmann, P. MissForest—non-parametric missing value imputation for mixed-type data. Bioinformatics 2012, 28, 112–118. [Google Scholar] [CrossRef] [Green Version]
- Lu, N.; Han, Y.; Chen, T.; Gunzler, D.D.; Xia, Y.; Julia, Y.; Lin, J.Y.; Tu, X.M. Power analysis for cross-sectional and longitudinal study designs. Shanghai Arch. Psychiatry 2013, 25, 259. [Google Scholar]
- Fuertes, E.; Markevych, I.; von Berg, A.; Bauer, C.-P.; Berdel, D.; Koletzko, S.; Sugiri, D.; Heinrich, J. Greenness and allergies: Evidence of differential associations in two areas in Germany. J Epidemiol. Community Health 2014, 68, 787–790. [Google Scholar] [CrossRef]
- Remmers, T.; Thijs, C.; Ettema, D.; De Vries, S.; Slingerland, M.; Kremers, S. Critical hours and important environments: Relationships between afterschool physical activity and the physical environment using GPS, GIS and accelerometers in 10–12-year-old children. Int. J. Environ. Res. Public. Health 2019, 16, 3116. [Google Scholar] [CrossRef] [Green Version]
- Ward, J.S.; Duncan, J.S.; Jarden, A.; Stewart, T. The impact of children’s exposure to greenspace on physical activity, cognitive development, emotional wellbeing, and ability to appraise risk. Health Place 2016, 40, 44–50. [Google Scholar] [CrossRef]
- Matsunaga, N.Y.; Oliveira, M.S.; Morcillo, A.M.; Ribeiro, J.D.; Ribeiro, M.A.; Toro, A.A. Physical activity and asthma control level in children and adolescents. Respirology 2017, 22, 1643–1648. [Google Scholar] [CrossRef]
- Macaubas, C.; De Klerk, N.H.; Holt, B.J.; Wee, C.; Kendall, G.; on behalf of the Raine Study group; Firth, M.; Sly, P.D.; Holt, P.G. Association between antenatal cytokine production and the development of atopy and asthma at age 6 years. Lancet 2003, 362, 1192–1197. [Google Scholar] [CrossRef]
- Hollams, E.M.; De Klerk, N.H.; Holt, P.G.; Sly, P.D. Persistent effects of maternal smoking during pregnancy on lung function and asthma in adolescents. Am. J. Respir. Crit. Care Med. 2014, 189, 401–407. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.-F.; Gilliland, F.D.; Berhane, K.; McConnell, R.; James Gauderman, W.; Rappaport, E.B.; Peters, J.M. Effects of in utero and environmental tobacco smoke exposure on lung function in boys and girls with and without asthma. Am. J. Respir. Crit. Care Med. 2000, 162, 2097–2104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zacharasiewicz, A. Maternal smoking in pregnancy and its influence on childhood asthma. ERJ Open Res. 2016, 2. [Google Scholar] [CrossRef] [Green Version]
- Cohen, R.T.; Raby, B.A.; Van Steen, K.; Fuhlbrigge, A.L.; Celedón, J.C.; Rosner, B.A.; Strunk, R.C.; Zeiger, R.S.; Weiss, S.T.; The CAMP Research Group. In utero smoke exposure and impaired response to inhaled corticosteroids in children with asthma. J. Allergy Clin. Immunol. 2010, 126, 491–497. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burr, J.A.; Mutchler, J.E.; Gerst, K. Patterns of residential crowding among Hispanics in later life: Immigration, assimilation, and housing market factors. J. Gerontol. B Psychol. Sci. Soc. Sci. 2010, 65, 772–782. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gray, A. Definitions of Crowding and the Effects of Crowding on Health; A Literature Review Prepared for the Ministry of Social Policy; Ministry of Social Development (MSD): Wellington, New Zealand, 2001; pp. 1–40.
- Illi, S.; Depner, M.; Genuneit, J.; Horak, E.; Loss, G.; Strunz-Lehner, C.; Büchele, G.; Boznanski, A.; Danielewicz, H.; Cullinan, P.; et al. Protection from childhood asthma and allergy in Alpine farm environments—the GABRIEL Advanced Studies. J. Allergy Clin. Immunol. 2012, 129, 1470–1477. [Google Scholar] [CrossRef] [PubMed]
- Kopel, L.S.; Phipatanakul, W.; Gaffin, J.M. Social disadvantage and asthma control in children. Paediatr. Respir. Rev. 2014, 15, 256–263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, Y.; Zhong, T.; Ge, D.; Li, Q.; Wu, J. Multi-factor analysis of single-center asthma control in Xiamen, China. Front. Pediatr. 2019, 7, 498. [Google Scholar] [CrossRef] [Green Version]
- Ranzi, A.; Porta, D.; Badaloni, C.; Cesaroni, G.; Lauriola, P.; Davoli, M.; Forastiere, F. Exposure to air pollution and respiratory symptoms during the first 7 years of life in an Italian birth cohort. Occup. Environ. Med. 2014, 71, 430–436. [Google Scholar] [CrossRef]




| All | C | UA | p-Value | |
|---|---|---|---|---|
| n | 179 | 129 | 50 | |
| Age, years, mean (SD) | 8.70 (2.64) | 8.52 (2.52) | 9.16 (2.90) | 0.161 |
| Gender: Female, n (%) | 72 (40.22) | 47 (36.43) | 25 (50.00) | 0.136 |
| BMI, kg/m2, mean (SD) | 19.36 (4.10) | 19.15 (4.31) | 19.90 (3.51) | 0.099 |
| Persistent asthma, n (%) | 99 (55.31) | 62 (48.06) | 37 (74.00) | 0.003 |
| Median ICS dose (fluticasone propionate) μg·day−1 | 240.32 (149.02) | 208.27 (132.81) | 297.43 (159.58) | 0.020 |
| Asthma onset, years, mean (SD) | 5.05 (2.59) | 4.96 (2.45) | 5.28 (2.32) | 0.522 |
| Severe exacerbations during the last year, mean (SD) | 0.72 (1.63) | 0.64 (1.31) | 0.94 (2.48) | 0.532 |
| Emergency visits (at least one during last year), n (%) | 26 (14.5%) | 19 (14.7%) | 7 (14.0%) | 1.000 |
| Atopy, n (%) | 131 (76.16) | 89 (72.95) | 42 (84.00) | 0.178 |
| Indoor sensitization, n (%) | 155 (73.81) | 81 (69.23) | 39 (81.25) | 0.167 |
| Outdoor sensitization, n (%) | 97 (45.97) | 27 (42.19) | 6 (46.15) | 1.000 |
| Parental education (<8 years), n (%) | 50 (28.57) | 36 (28.57) | 14 (28.57) | 1.000 |
| Physical activity (>3 times per week), n (%) | 88 (38.60) | 58 (45.31) | 15 (30.00) | 0.065 |
| Comorbidity, n (%) | 117 (65.73) | 82 (64.06) | 35 (70.00) | 0.566 |
| Environmental exposures | ||||
| Outdoor | ||||
| NDVI ≤ 0.21 | 39 (26.53) | 28 (26.17) | 11 (27.50) | 1.000 |
| CLC, Artificial Surface | 148 (82.68) | 106 (82.17) | 42 (84.00) | 0.944 |
| NO2 LUR ≥ 40, µg/m3, n (%) | 150 (83.80) | 106 (82.17) | 44 (88.00) | 0.469 |
| HTRs < 200 m, n (%) | 145 (81.92) | 104 (81.89) | 41 (82.00) | 1.000 |
| Indoor | ||||
| Current ETS | 62 (35.03) | 43 (33.86) | 19 (38.00) | 0.730 |
| Current mold exposure | 38 (21.59) | 22 (17.46) | 16 (32.00) | 0.056 |
| Current pet exposure | 38 (21.47) | 28 (22.05) | 10 (20.00) | 1.000 |
| Crowding index | 1.10 (0.47) | 1.05 (0.44) | 1.25 (0.50) | 0.020 |
| MSP | 16 (9.04) | 7 (5.51) | 9 (18.00) | 0.020 |
| All | C | UA | p-Value | |
|---|---|---|---|---|
| n | 179 | 129 | 50 | |
| FEV1, L | 1.76 (0.61) | 1.74 (0.60) | 1.77 (0.63) | 0.787 |
| Z-score | −0.35 (1.26) | −0.23 (1.02) | −0.43 (1.79) | 0.044 |
| % pred | 95.74 (14.71) | 97.22 (12.27) | 94.63 (20.09) | 0.042 |
| FVC, L | 2.08 (0.76) | 2.03 (0.72) | 2.16 (0.78) | 0.276 |
| Z-score | −0.05 (1.19) | −0.03 (0.96) | 0.16 (1.72) | 0.921 |
| % pred | 99.54 (14.23) | 99.70 (11.69) | 102.01 (20.38) | 0.930 |
| FEV1/FVC | 0.85 (0.07) | 0.86 (0.06) | 0.83 (0.09) | 0.032 |
| Z-score | −0.51 (1.01) | −0.37 (0.91) | −0.91 (1.07) | 0.014 |
| % pred | 95.78 (7.99) | 96.97 (6.43) | 92.64 (9.36) | 0.015 |
| FEF25–75%, L/s | 1.95 (0.84) | 1.92 (0.73) | 1.90 (0.90) | 0.345 |
| Z-score | −0.67 (1.29) | −0.61 (0.90) | −0.88 (1.73) | 0.022 |
| % pred | 86.55 (35.21) | 86.84 (20.14) | 82.55 (40.42) | 0.023 |
| VIF | |
|---|---|
| NDVI ≤ 0.21 | 1.138 |
| CLC, AS | 1.318 |
| NO2 LUR ≥ 40 µg/m3 | 1.340 |
| HTR < 200 m | 1.214 |
| Current ETS | 1.338 |
| Current mold exposure | 1.283 |
| Current pet exposure | 1.313 |
| Crowding index | 1.975 |
| MSP | 1.222 |
| Comorbidity | 1.164 |
| Persistent asthma | 1.137 |
| Atopy | 1.144 |
| Parental education | 1.794 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cilluffo, G.; Ferrante, G.; Fasola, S.; Malizia, V.; Montalbano, L.; Ranzi, A.; Badaloni, C.; Viegi, G.; La Grutta, S. Association between Asthma Control and Exposure to Greenness and Other Outdoor and Indoor Environmental Factors: A Longitudinal Study on a Cohort of Asthmatic Children. Int. J. Environ. Res. Public Health 2022, 19, 512. https://doi.org/10.3390/ijerph19010512
Cilluffo G, Ferrante G, Fasola S, Malizia V, Montalbano L, Ranzi A, Badaloni C, Viegi G, La Grutta S. Association between Asthma Control and Exposure to Greenness and Other Outdoor and Indoor Environmental Factors: A Longitudinal Study on a Cohort of Asthmatic Children. International Journal of Environmental Research and Public Health. 2022; 19(1):512. https://doi.org/10.3390/ijerph19010512
Chicago/Turabian StyleCilluffo, Giovanna, Giuliana Ferrante, Salvatore Fasola, Velia Malizia, Laura Montalbano, Andrea Ranzi, Chiara Badaloni, Giovanni Viegi, and Stefania La Grutta. 2022. "Association between Asthma Control and Exposure to Greenness and Other Outdoor and Indoor Environmental Factors: A Longitudinal Study on a Cohort of Asthmatic Children" International Journal of Environmental Research and Public Health 19, no. 1: 512. https://doi.org/10.3390/ijerph19010512
APA StyleCilluffo, G., Ferrante, G., Fasola, S., Malizia, V., Montalbano, L., Ranzi, A., Badaloni, C., Viegi, G., & La Grutta, S. (2022). Association between Asthma Control and Exposure to Greenness and Other Outdoor and Indoor Environmental Factors: A Longitudinal Study on a Cohort of Asthmatic Children. International Journal of Environmental Research and Public Health, 19(1), 512. https://doi.org/10.3390/ijerph19010512







