1. Introduction
Fossil fuels such as oil, coal, and gas represent the main energy source worldwide. However, these turn out to be limited resources and contribute to environmental pollution and global warming.This scenario demands policies that stimulate the use of renewable and sustainable energy sources in order to mitigate these effects [
1,
2]. In this context, biodiesel has been glimpsed as a viable replace respect to engine conventional fuels. Among the several biodiesel alternatives, the use of Fatty Methyl Ester (FAME) in combustion engines can be found [
3]. Biodiesel has advantages over conventional diesel fuel, such as it can be obtained from oils and animal fats; also, it is biodegradable, non-toxic, and environmentally sustainable, avoiding incorporate new CO
2 to the carbon cycle [
4,
5].
Currently, the biggest obstacle to the substitution of conventional diesel by the biodiesel is its high costs, where the feedstock represents 75% of its manufacturing cost [
6]. Therefore, it is necessary to use lower-cost feedstocks in its manufacture. Among the alternatives for producing biodiesel at a reasonable cost, are the cooking vegetable oil, agricultural waste, animal fats, and unrefined vegetable oils [
7,
8]. For example, the cost of biodiesel made from used cooking oil and animal fats waste is estimated between
$0.09–
$0.20 USD per pound [
9]. The cost of biodiesel from crude African palm oil has been estimated between
$0.49 and
$0.53 USD per liter [
10]. Unfortunately, those feedstocks have a high content of free fatty acids (FFAs) which present soap formation during the process of conventional direct base-catalyzed transesterification, making their use not viable [
11]. As an alternative, the use of a multistep process allows employing feedstocks with high FFAs concentrations by first carrying out the acid-catalyzed pre-esterification of the FFAs prior to the base-catalyzed triglycerides transesterification [
12].
The usual esterification procedure implies the use of p-toluene sulphonic acid, sulfuric, hydrochloric, and phosphoric acids as a catalyst. However, the use of these catalysts has several disadvantages: (a) during the esterification reaction water is generated, favoring the reversibility of the reaction, which lowers the yield of methyl esters; (b) these acids are highly corrosive, which reduces the lifetime of the equipment used in the process; (c) toxic wastes that are harmful to the environment are generated; and, (d) costs of purification of biodiesel are high increasing the biodiesel cost [
13]. Another method that can be used for esterification of FFAs is the supercritical process. Its main advantages consist of reduce mass-transfer limitations and improve the phase solubility. Furthermore, the reaction rate is augmented, completing it in shorter periods and simpler separations and purification of FAME [
14]. Nevertheless, to present satisfactory FAME yield the supercritical method needs high molarity of alcohol to oil and high pressure and temperature conditions for the reaction. This implies high processing costs and causes the degradation of the fatty acids esters formed [
15].
A very attractive alternative to carry out the esterification of FFAs is the use of heterogeneous catalyst systems, which include cationic-exchanged resins. Examples of these resins are Amberlyst-15, metal oxides, heteropolyacids supported on silica and carbon, aluminophosphates, and silicoalumino-phosphate molecular sieves and clays [
16]. The clays have presented a substantial interest in several organic syntheses as catalysts [
17]. Among these, the montmorillonite possesses both Lewis and Brönsted acidities sites. Due to these properties, there is particular interest in using montmorillonite as catalysts in the biodiesel manufacture, as they have several advantages: (a) availability and low cost; (b) environmental compatible; (c) easy recovered by filtration and reusable; (d) high selectivity and thermal stability; and, (e) allow the development of continuous processes to increase the production volume of biodiesel, which reduces manufacturing costs. In particular, the montmorillonite KSF (commercial name for clays activated with acid treatment) presents high concentrations of Lewis and Brönsted acid sites, thus oil esterification can be employed with high FFAs concentrations [
18].
The esterification reaction of FFAs is represented by the following reversible chemical equation
This reaction is affected by temperature changes, reaction time, and the change of catalyst and alcohol concentrations. These variations can enhance or reduce the FFAs to FAME conversion. For this reason, it is important to evaluate the effect of the process variables to determine the optimal reaction conditions of FFAs to FAME conversion and to maximize the yield and reduce costs in the manufacture of biodiesel. It is also necessary to develop the experimental design Response Surface Methodology (RSM) and a quadratic model to calculate the best fit. The RSM is a group of mathematical and statistical tools of important use in the modeling, analysis and optimization problems. The response of interest is influenced by various factors in RMS [
11]. In this methodology, a suitable experiment can significantly reduce the number of tests, and provide the information needful for process optimization. The RSM uses polynomial models to characterize the response of the surface [
19].
The RSM has been widely reported as a suitable tool for the modeling and optimization of biodiesel yield, for example: Jahirul et al. [
11] used RSM based in a Box-Behnken design to determine a method to optimize the biodiesel production process from beauty-leaf oil. The authors considered catalyst concentration, methanol to oil molar ratio and reaction temperature as the main factors in the conversion process. FFAs and FAME concentration prediction and reaction conditions optimization were achieved through linear and full quadratic regression models. Ali et al. [
20] investigated on fuel properties and engine performance of blended palm biodiesel-diesel using diethyl ether as an additive. The independent variables considered were the engine revolution per minute and the diethyl ether blend. A design of experiment based on RSM was employed to optimize the fuel properties viscosity, density, acid value, heating value, pour point, and cloud point. Corral Bobadilla et al. [
21] used RSM to improve the production of biodiesel by finding the best combination of input variables used in the transesterification reactions. Various biodiesel optimization scenarios were proposed based on the objective to improve the biodiesel production and higher heating value, and decrease the viscosity, density, and turbidity. Omar et al. [
22] used the RSM to study the relationship of different variables in the waste palm cooking oil heterogeneous transesterification to biodiesel by Sr/ZrO
2 catalyst.
According to Ramos et al. [
23], soybean, canola, sunflower, and African palm (
Elaeis guinnensis Jacq) oils are the most widely used for the production of biodiesel in the world. The African palm is an oil-producing species, its expansion around the world is due to its easy adaptation to tropical environments. The high oil content that has its fruit allows for yields of 5.5 ton ha
−1, generating the decrease of production costs. This makes the crude African palm oil (CAPO) a viable option for its use as raw material in the manufacture of low-cost biodiesel [
24]. The main producing CAPO countries in the world are Malaysia, Indonesia, Thailand, Nigeria, and Colombia, among others. In Mexico, the African palm cultivation takes place in the states of Chiapas, Campeche, Tabasco, and Veracruz with 54,434.00 ha planted in 2010 [
24]. There is a significant possibility to project 2.5 million ha of African palm plantation in these areas. Based on these conditions, the African palm may be considered as a potential alternative to increasing the manufacture of biodiesel in Mexico [
25].
The objective of this study is to evaluate the efficiency of the montmorillonite KSF for the esterification of the FFAs present in the CAPO. Furthermore, the determination the optimal reaction conditions that yield the optimization of FFAs to FAME conversion at a laboratory level, through the application of a central composite rotatable design (CCRD) and RSM techniques.
4. Conclusions
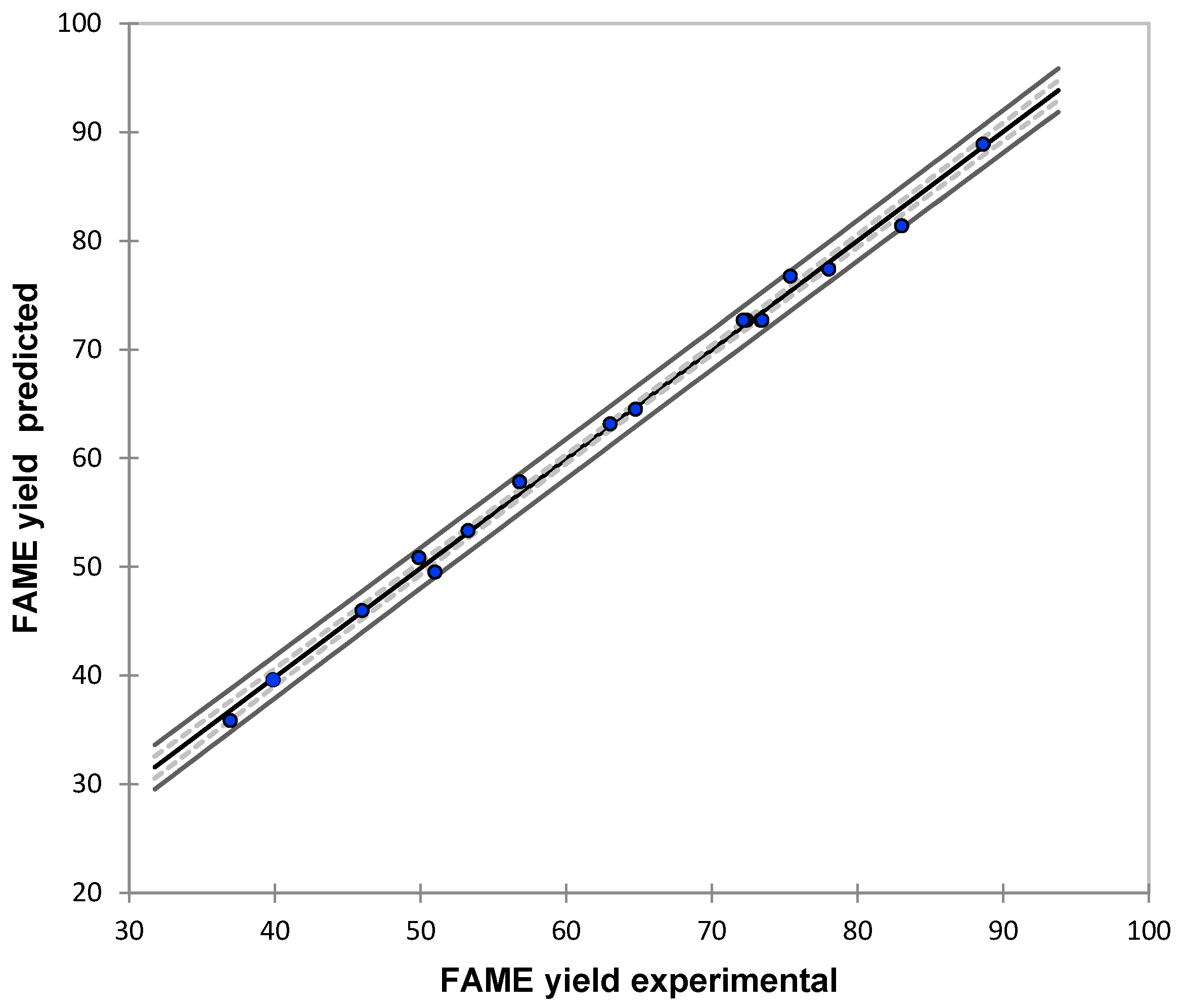
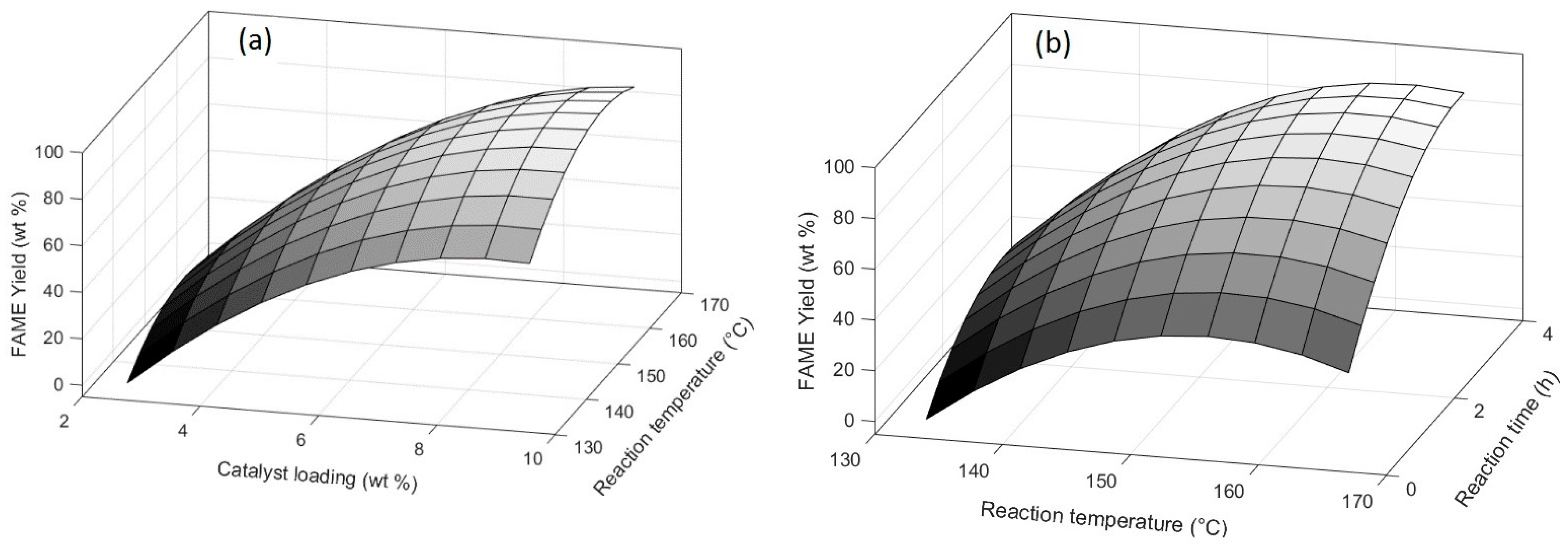
This study is focused in optimize the Fatty Methyl Ester yield of the effect of montmorillonite KSF in the esterification of FFAs of CAPO. The operation parameters catalyst loading, reaction time, and reaction temperature were optimized using a response surface methodology that was based on a central composite rotatable design to achieve the maximum Fatty Methyl Ester yield. According to the statistical results, the second-order model generated is a feasible tool for estimation of Fatty Methyl Ester yield with minimums error levels (MAPE = 2.13, RMSE = 1.43, and R2 = 97.23%). Additionally, ANOVA results demonstrate that the three operation parameters present relative importance for the model output.
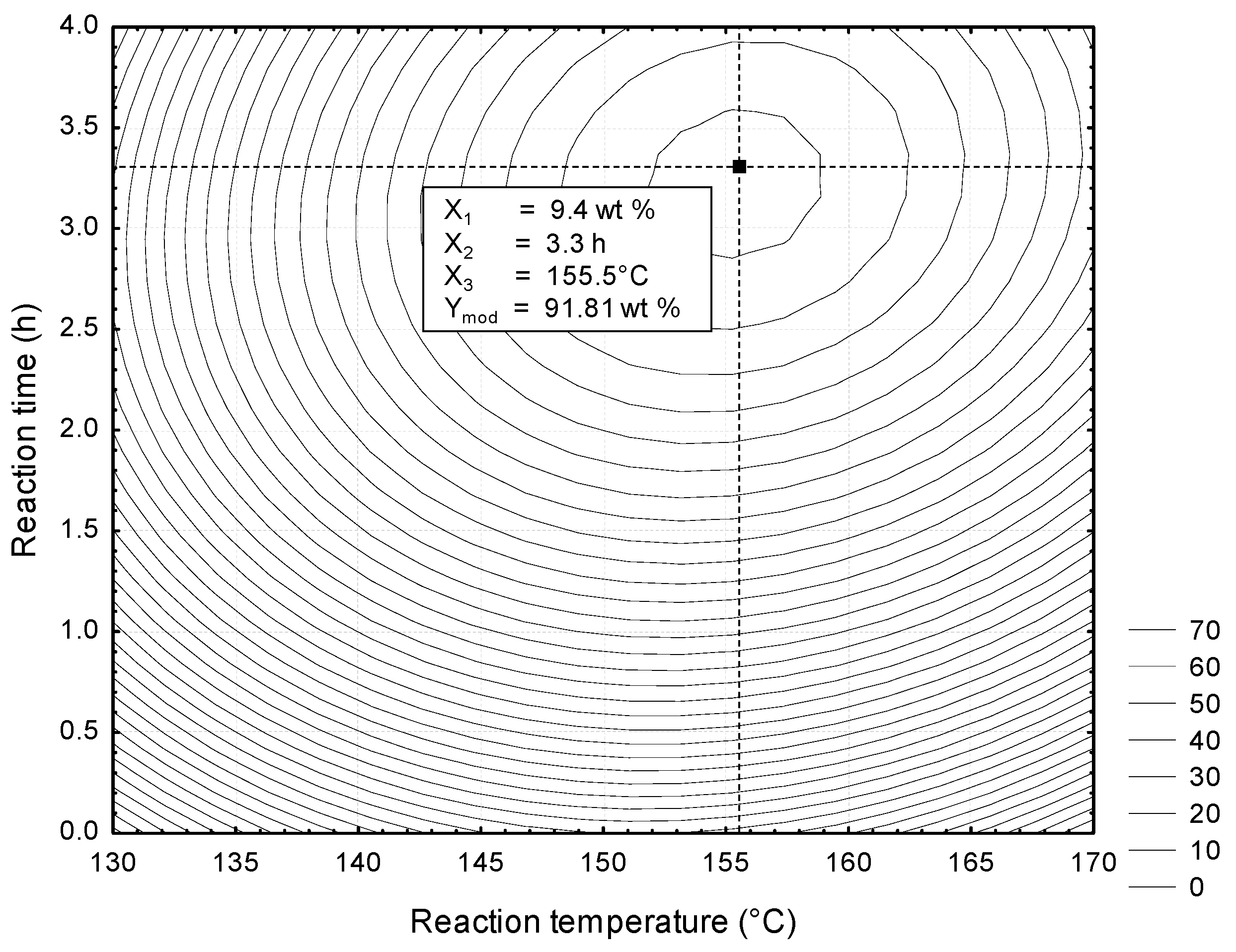
According to the experimental results, the montmorillonite KSF could be used as an effective heterogeneous catalyst for the conversion of FFAs of CAPO to biodiesel. The optimum reaction conditions were a catalyst loading 9.4 wt %, reaction temperature 155.5 °C and reaction time, 3.3 h. Under these conditions, was possible to obtain 91.81 wt % Fatty Methyl Ester yield. After the esterification steps, the FFAs to less than 1.1 wt %, this allows for the use of alkaline catalysis. This would allow for developing a two-step process for the synthesis of biodiesel, with high efficiency and low energy cost.