Challenges of Application of Green Ammonia as Fuel in Onshore Transportation
Abstract
:1. Introduction
- As a carbon-free emission fuel:
- As a good economic solution:
- –
- Availability: NH is one of the most produced and distributed chemicals in the world [14];
- –
- Technological feasibility: infrastructure already exists for the production, transport, and distribution [15];
- –
- Low cost of ammonia as fuel: it is comparable to the cost of diesel fuel on an energy basis [16].
- –
- Volume expander to current fuels: as a mixture, ammonia can be blended with, for example, gasoline [13];
- –
- Low storage costs: ammonia can be stored at room temperature (300 K) with relatively low pressure (more-less 10 bar) [16];
- –
- Cheap long-distance transportation: ammonia’s energy cost is far lower than for transporting electricity over long distances (>2000 km) [13];
2. Current Status of Ammonia
3. Safety Aspects, Also Related to the Safe Powering of Vehicles
- Physical threats
- Flammable gas;
- Gas under pressure, which may explode if heated.
- Health hazards
- Toxic if inhaled;
- Causes severe skin burns and eye damage;
- Corrosive to the respiratory tract.
- Environmental hazards
- Very toxic to aquatic life, with long-lasting effects.
3.1. Human Toxicity
3.2. Environmental Toxicity
- LD50 is the amount of material administered at one time that causes the death of 50% (half) of a group of test animals. LD50 is one way to measure the short-term poisoning potential (acute toxicity) of a material.
- LC values usually refer to the concentration of a chemical in air, but in environmental tests, they can also mean the concentration of a chemical in water. According to the Organization for Economic Cooperation and Development (OECD) guidelines for the testing of chemicals, a traditional experiment involves groups of animals exposed to a concentration (or series of concentrations) for a specified period of time (usually 4 h). The animals are clinically observed for up to 14 days. The concentration of a chemical in the air that kills 50% of test animals during the observation period is the LC50 value. Other exposure times (as opposed to the traditional 4 h) may apply depending on specific regulations.
3.3. Flammability
3.4. Explosiveness
3.5. Corrosivity
- The yield strength of the material;
- Residual stresses, e.g., after welding processes;
- Oxygen content;
- Water content
3.6. Gas under Pressure
3.7. Chilled Refrigerant
3.8. Summary
- Design rules;
- Specifications of the valve in relation to the required safety level;
- Specifications of the valve in relation to the performance of the installation;
- Requirements for all installation components to have certificates confirming compliance with the relevant regulations;
- Installation and maintenance requirements;
- Supervision requirements; and
- Requirements for periodic inspection.
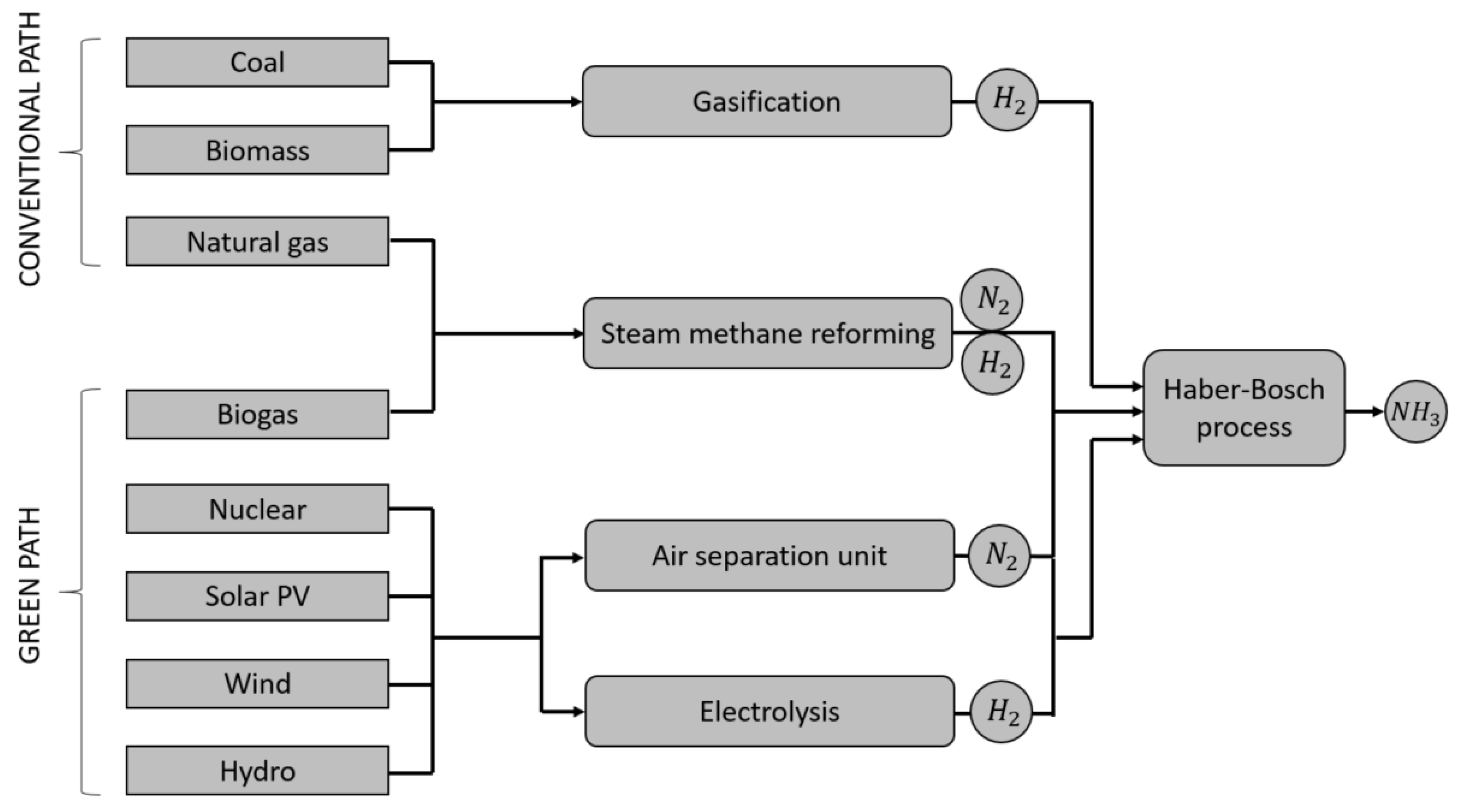
4. Production of Green Ammonia
| Reference [44] | Production Path Wind | LCOA 455–637 EUR | Remarks |
|---|---|---|---|
| [45] | wind +/ solar | 842 and 759 EUR | for off-grid and on-grid solutions analyzed for multiple locations |
| [46] | offshore wind | 1114 EUR | |
| [51] | hydro | 1248 EUR | |
| [47] | wind, solar | 430 EUR | 534 locations in 70 countries studied future improvement to 282 EUR possible island operation |
| [48] | solar, wind | 664 EUR | |
| [49] | solar, wind, hydro | 431–528 EUR | |
| [50] | solar | 653 EUR | future price 410 EUR |
Summary
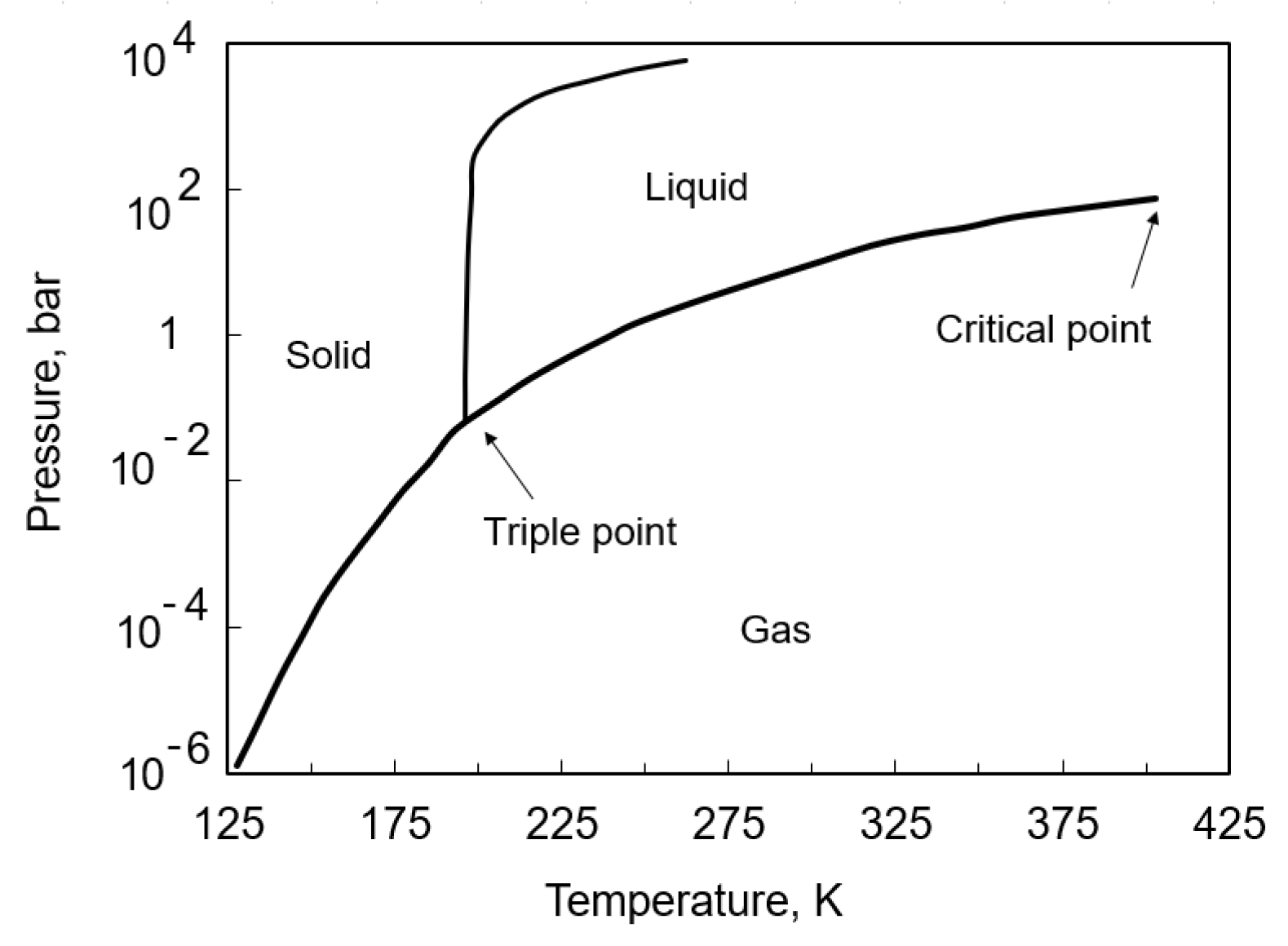
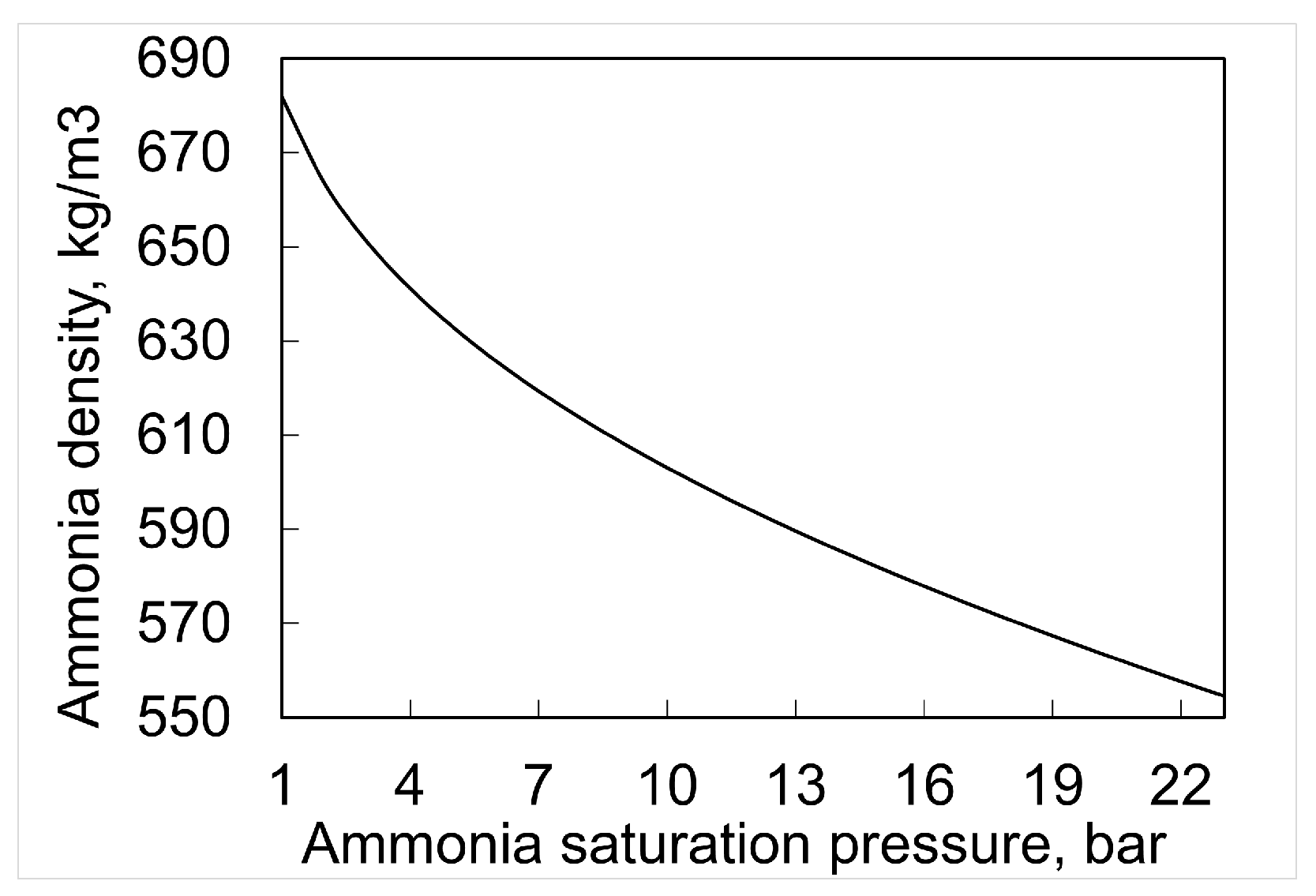
5. Storage (Refrigeration and Pressure) and Regasification of Ammonia
Summary
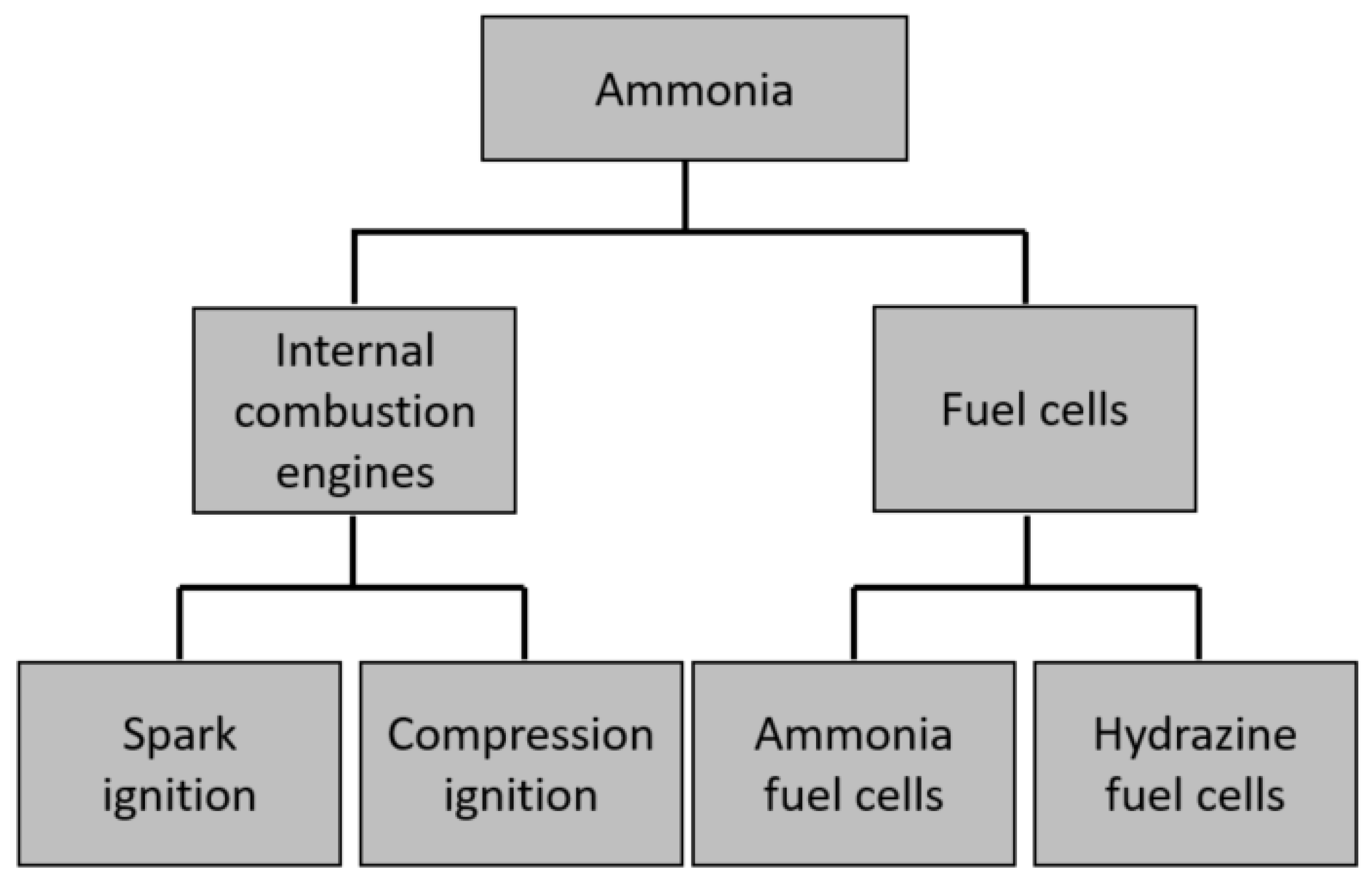
6. Ammonia Fueling Solutions
6.1. Hydrogen-Based Fuels in Transport Industry
6.2. Internal Combustion Engines (ICE)—Issues Related with Ammonia as Fuel Source
6.3. Spark Ignition Engines
6.4. Compression Ignition Engines
6.5. Summary
6.6. Fuel Cells—Current State of Development
6.7. Selected Methods of Supplying Fuel Cells with Ammonia
6.8. Ammonia-Fed Solid Oxide Fuel Cells
| Technology | Volumetric Power Density, kW/m | Gravimetric Power Density, kW/t | Efficiency, % |
|---|---|---|---|
| SOFC stack | 200–400 | <100 | - |
| SOFC system | 2–100 | <70 | 35–65 |
| PEMFC stack | 50–200 | 100–600 | - |
| PEMFC system | 4–500 | 10–500 | 40–60 |
| Diesel engine (500 kW) | 235 | 330 | - |
6.9. Summary
7. Ammonia Fuel in the Context of Clean Air Policies
Summary
8. Summary: Indication of the Strengths and Weaknesses of Green Ammonia as a Fuel and Further Research and Optimization Directions Required
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AFC | Alkaline Fuel Cell |
| AIT | Autoignition |
| CAPEX | Capital Expenditures |
| CEM | Clean Energy Ministerial |
| CI | Compression Ignition |
| CNG | Compressed Natural Gas |
| CR | Compression Ratio |
| DMFC | Direct Methanol Fuel Cell |
| EU | European Union |
| EVI | Electric Vehicles Initiative |
| FC | Fuel cell |
| GHG | Green House Gases |
| HRR | Heat Release Rate |
| ICE | Internal Combustion Engine |
| IEA | International Energy Agency |
| IMO | International Marine Organization |
| IRENA | International Renewable Energy Agency |
| LC | Lethal Concentration |
| LCOA | Levelized Cost of Ammonia |
| LD | Lethal Dose |
| LNG | Liquefied Natural Gas |
| LPG | Liquid Petroleum Gas |
| MCFC | Molten Carbonite Fuel Cell |
| MIE | Minimum Ignition Energy |
| NIOSH | National Institute for Occupational Safety and Health |
| OECD | Organization for Economic Cooperation and Development |
| PAFC | Phosphoric Acid Fuel Cell |
| PEMFC | Proton Exchange Membrane Fuel Cell |
| PM | Particulate Matter |
| RON | research Octane Number |
| SI | Spark Ignition |
| SOFC | Solid Oxide Fuel Cell |
| TRL | Technology Readiness Level |
| US | United States |
References
- Phase-Out of Fossil Fuel Vehicles. 2023. Available online: https://en.wikipedia.org/wiki/Phase-out_of_fossil_fuel_vehicles (accessed on 10 May 2023).
- More Than 25 Countries & US States Are Planning Gas Car Bans. 2021. Available online: https://insideevs.com/news/534890/countries-states-gas-car-bans/ (accessed on 11 May 2023).
- CO2 Emissions from Cars: Facts and Figures (Infographics). 2019. Available online: https://www.europarl.europa.eu/news/en/headlines/society/20190313STO31218/co2-emissions-from-cars-facts-and-figures-infographics (accessed on 11 May 2023).
- Unijny Zakaz Sprzedaży Nowych Samochodów Spalinowych od 2035 r.—co to Oznacza? 2022. Available online: https://www.europarl.europa.eu/news/pl/headlines/economy/20221019STO44572/unijny-zakaz-sprzedazy-nowych-samochodow-spalinowych-od-2035-r-co-to-oznacza (accessed on 11 May 2023).
- China Joins List of Nations Banning the Sale of Old-Style Fossil-Fuelled Vehicles. 2020. Available online: https://europeansting.com/2020/11/17/china-joins-list-of-nations-banning-the-sale-of-old-style-fossil-fuelled-vehicles/ (accessed on 11 May 2023).
- Powell, A. California Dreaming? Nope. 2022. Available online: https://environment.harvard.edu/news/california-dreaming-nope (accessed on 11 May 2023).
- DNV. Alternative Fuels for Containerships; Technical Report 4; DNV GL: Bærum, Norway, 2021. [Google Scholar]
- IEA. Global EV Outlook 2020: Technology Report; IEA: Paris, France, 2020; pp. 39–85. [Google Scholar]
- How to Increase the Use of Alternative Fuels for Cars. 2022. Available online: https://www.europarl.europa.eu/news/en/headlines/economy/20221013STO43019/how-to-increase-the-use-of-alternative-fuels-for-cars (accessed on 11 May 2023).
- European Biogas Association. The Potential of Biomethane for a Faster Decarbonisation of Transport Should Not Go to Waste Photo Credits: Deva Darshan; Technical Report; European Biogas Association: Brussels, Belgium, 2020. [Google Scholar]
- Chiong, M.C.; Chong, C.T.; Ng, J.H.; Mashruk, S.; Chong, W.W.F.; Samiran, N.A.; Mong, G.R.; Valera-Medina, A. Advancements of combustion technologies in the ammonia-fuelled engines. Energy Convers. Manag. 2021, 244, 114460. [Google Scholar] [CrossRef]
- Frigo, S.; Gentili, R. Analysis of the behaviour of a 4-stroke Si engine fuelled with ammonia and hydrogen. Int. J. Hydrogen Energy 2013, 38, 1607–1615. [Google Scholar] [CrossRef]
- Chehade, G.; Dincer, I. Progress in green ammonia production as potential carbon-free fuel. Fuel 2021, 299, 120845. [Google Scholar] [CrossRef]
- Tornatore, C.; Marchitto, L.; Sabia, P.; De Joannon, M. Ammonia as Green Fuel in Internal Combustion Engines: State-of-the-Art and Future Perspectives. Front. Mech. Eng. 2022, 8, 944201. [Google Scholar] [CrossRef]
- Elbaz, A.M.; Wang, S.; Guiberti, T.F.; Roberts, W.L. Review on the recent advances on ammonia combustion from the fundamentals to the applications. Fuel Commun. 2022, 10, 100053. [Google Scholar] [CrossRef]
- Ryu, K.; Zacharakis-Jutz, G.E.; Kong, S.C. Performance enhancement of ammonia-fueled engine by using dissociation catalyst for hydrogen generation. Int. J. Hydrogen Energy 2014, 39, 2390–2398. [Google Scholar] [CrossRef]
- Yüzbaşıoğlu, A.E.; Avşar, C.; Gezerman, A.O. The current situation in the use of ammonia as a sustainable energy source and its industrial potential. Curr. Res. Green Sustain. Chem. 2022, 5, 100307. [Google Scholar] [CrossRef]
- IRENA; AEA. Innovation Outlook: Renewable Ammonia; Technical Report; International Renewable Energy Agency: Abu Dhabi, United Arab Emirates, 2022. [Google Scholar]
- Pearson, A. Refrigeration with ammonia. Int. J. Refrig. 2008, 31, 545–551. [Google Scholar] [CrossRef]
- Khudhur, D.A.; Tuan Abdullah, T.A.; Norazahar, N. A Review of Safety Issues and Risk Assessment of Industrial Ammonia Refrigeration System. Acs Chem. Health Saf. 2022, 29, 394–404. [Google Scholar] [CrossRef]
- Machaj, K.; Kupecki, J.; Malecha, Z.; Morawski, A.; Skrzypkiewicz, M.; Stanclik, M.; Chorowski, M. Ammonia as a potential marine fuel: A review. Energy Strategy Rev. 2022, 44, 100926. [Google Scholar] [CrossRef]
- Linde. Ammonia Datasheet; Technical Report; Linge AG: Dublin, Ireland, 2023. [Google Scholar]
- Birken, G.A.; Fabri, P.J.; Carey, L.C. Acute ammonia intoxication complicating multiple trauma. J. Trauma 1981, 21, 820–822. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Occupational Safety and Health. NIOSH recommended standards for occupational exposure to ammonia and benzene. Int. J. Occup. Health Saf. 1974, 43, 32–33. [Google Scholar]
- The Fertilizer Institute. Health Effects of Ammonia; Technical Report; The Fertilizer Institute: Arlington, VA, USA, 2020. [Google Scholar]
- EC-European Commission. Commission Regulation (EU) No 582/2011 of 25 May 2011 Implementing and Amending Regulation (EC) No 595/2009 of the European Parliament and of the Council with Respect to Emissions from Heavy Duty Vehicles (Euro VI) and Amending Annexes I and III to Directive 2007/46/EC of the European Parliament and of the Council; Technical Report; EC-European Commission: Brussels, Belgium, 2011. [Google Scholar]
- EC-European Commission. Commission Regulation (EU) No 64/2012 of 23 January 2012 Amending Regulation (EU) No 582/2011 Implementing and Amending Regulation (EC) No 595/2009 of the European Parliament and of the Council with Respect to Emissions from Heavy Duty Vehicles (Euro VI) Text with EEA Relevance; Technical Report; EC-European Commission: Brussels, Belgium, 2012. [Google Scholar]
- Ziegler, B.; Trepp, C. Equation of state for ammonia-water mixtures. Int. J. Refrig. 1984, 7, 101–106. [Google Scholar] [CrossRef]
- Patterson, P.; Adrizal. Management strategies to reduce air emissions: Emphasis—Dust and ammonia. J. Appl. Poult. Res. 2005, 14, 638–650. [Google Scholar] [CrossRef]
- Wang, X.B.; Xiang, Y.; Li, X.Y. Environmental risk for application of ammonia-soda white mud in soils in China. J. Integr. Agric. 2020, 19, 601–611. [Google Scholar] [CrossRef]
- United States Environmetnal Protection Agency. 1999 Update Of Ambient Water Quality Criteria For Ammonia. EPA-822-R-99-014; Technical Report; USEPA: Washington, DC, USA, 1999.
- Wicks, B.; Joensen, R.; Tang, Q.; Randall, D. Swimming and ammonia toxicity in salmonids: The effect of sub lethal ammonia exposure on the swimming performance of coho salmon and the acute toxicity of ammonia in swimming and resting rainbow trout. Aquat. Toxicol. 2002, 59, 55–69. [Google Scholar] [CrossRef] [PubMed]
- Tepper, J.S.; Weiss, B.; Wood, R.W. Alterations in behavior produced by inhaled ozone or ammonia. Fundam. Appl. Toxicol. 1985, 5, 1110–1118. [Google Scholar] [CrossRef]
- Johnson, I.; Sorokin, N.; Atkinson, C.; Rule, K.; Hope, S.J. Proposed EQS for Water Framework Directive Annex VIII Substances: Ammonia (un-Ionised); Technical Report; Environment Agency: Bristol, UK, 2007.
- Levit, S.M. Review of the Literature on the Effects of Ammonia on Fish; Technical Report; Center for Science in Public Participation: Bozeman, MT, USA, 2010. [Google Scholar]
- Kobayashi, H.; Hayakawa, A.; Somarathne, K.; Okafor, E. Science and technology of ammonia combustion. Proc. Combust. Inst. 2019, 37, 109–133. [Google Scholar] [CrossRef]
- American Bureau of Shipping, Guide for Ammonia Fueled Vessels September 2021; Technical Report; American Bureau of Shipping: Houston, TX, USA, 2021.
- DNV. RU-SHIP Part 6 Additional Class Notations Chapter 2 Propulsion, Power Generation and Auxiliary Systems Section 14 Gas Fuelled Ship Installations—Gas Fuelled Ammonia; Technical Report; DNV: Bærum, Norway, 2021. [Google Scholar]
- International Maritime Organization. IMO Strategy on Reduction of GHG Emissions from Ships. Available online: https://unfccc.int/sites/default/files/resource/250_IMO%20submission_Talanoa%20Dialogue_April%202018.pdf (accessed on 10 May 2023).
- Group Technology & Research, D&G. Ammonia as a Marine Fuel; Technical Report; DNV-GL: Bærum, Norway, 2020. [Google Scholar]
- Rogala, Z.; Stanclik, M.; Łuszkiewicz, D.; Malecha, Z. Perspectives for the Use of Biogas and Biomethane in the Context of the Green Energy Transformation on the Example of an EU Country. Energies 2023, 16, 1911. [Google Scholar] [CrossRef]
- Bertilsson, O.B.; Kirchmann, H. Sustainable N fertilizer production based on a loop: Straw—Biogas—’Haber-Bosch’ process. Agric. Syst. 2021, 190, 103100. [Google Scholar] [CrossRef]
- Laval, A.; Hafnia, H.T.; Vestas, S.G. Ammonfuel—An Industrial View of Ammonia as a Marine Fuel. Hafnia 2020, 7, 32–59. [Google Scholar]
- Ofori-Bah, C.O.; Amanor-Boadu, V. Directing the wind: Techno-economic feasibility of green ammonia for farmers and community economic viability. Front. Environ. Sci. 2023, 10, 1070212. [Google Scholar] [CrossRef]
- Campion, N.; Nami, H.; Swisher, P.R.; Vang Hendriksen, P.; Münster, M. Techno-economic assessment of green ammonia production with different wind and solar potentials. Renew. Sustain. Energy Rev. 2023, 173, 113057. [Google Scholar] [CrossRef]
- Morgan, E.R. Techno-Economic Feasibility Study of Ammonia Plants Powered by Offshore Wind. Ph.D. Thesis, University of Massachusetts Amherst, Amherst, MA, USA, 2013. [Google Scholar]
- Nayak-Luke, R.M.; Bañares-Alcántara, R. Techno-economic viability of islanded green ammonia as a carbon-free energy vector and as a substitute for conventional production. Energy Environ. Sci. 2020, 13, 2957–2966. [Google Scholar] [CrossRef]
- Nayak-Luke, R.; Bañares-Alcántara, R.; Wilkinson, I. “Green” Ammonia: Impact of Renewable Energy Intermittency on Plant Sizing and Levelized Cost of Ammonia. Ind. Eng. Chem. Res. 2018, 57, 14607–14616. [Google Scholar] [CrossRef] [Green Version]
- Ikäheimo, J.; Kiviluoma, J.; Weiss, R.; Holttinen, H. Power-to-ammonia in future North European 100 % renewable power and heat system. Int. J. Hydrogen Energy 2018, 43, 17295–17308. [Google Scholar] [CrossRef]
- Osman, O.; Sgouridis, S.; Sleptchenko, A. Scaling the production of renewable ammonia: A techno-economic optimization applied in regions with high insolation. J. Clean. Prod. 2020, 271, 121627. [Google Scholar] [CrossRef]
- Sousa, J.; Waiblinger, W.; Friedrich, K.A. Techno-economic Study of an Electrolysis-Based Green Ammonia Production Plant. Ind. Eng. Chem. Res. 2022, 61, 14515–14530. [Google Scholar] [CrossRef]
- IEA. The Future of Hydrogen; Technical Report June; IEA: Paris, France, 2019. [Google Scholar] [CrossRef]
- IRENA. Renewable Power Generations Costs; IRENA: Masdar City, United Arab Emirates, 2018; p. 160. [Google Scholar]
- U.S. Energy Information Administration. Transportation sector energy consumption. In International Energy Outlook 2016; U.S. Energy Information Administration: Washington, DC, USA, 2016; Volume 2016, pp. 127–137. [Google Scholar]
- Office of Management and Bugdet. Budget of the U.S. Government Budget of the U.S. Government; Technical Report; Office of Management and Bugdet: Washington, DC, USA, 2022.
- Ivanic, M.; Martin, W.; Zaman, H. Estimating the Short-Run Poverty Impacts of the 2010–2011 Surge in Food Prices. World Dev. 2012, 40, 2302–2317. [Google Scholar] [CrossRef] [Green Version]
- Lomborg, B. False Alarm: How Climate Change Panic Costs Us Trillions, Hurts the Poor, and Fails to Fix the Planet; Basic Books: Hachette, UK, 2020. [Google Scholar]
- Patonia, A.; Poudineh, R. Ammonia as a Storage Solution for Future Decarbonized Energy Systems; Technical Report; OIES Paper: Oxford, UK, 2020. [Google Scholar]
- IEA. Ammonia Technology Roadmap: Towards More Sustainable Nitrogen Fertilizer Production; International Energy Agency: Paris, France, 2021; p. 163. [Google Scholar]
- Appl, M. Ammonia; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2006. [Google Scholar] [CrossRef]
- Engineering Toolbox: Fuels—Higher and Lower Calorific Values. Available online: https://www.engineeringtoolbox.com/fuels-higher-calorific-values-d_169.html (accessed on 15 May 2023).
- Rouwenhorst, K.H.; der Ham, A.G.V.; Mul, G.; Kersten, S.R. Islanded ammonia power systems: Technology review & conceptual process design. Renew. Sustain. Energy Rev. 2019, 114, 109339. [Google Scholar] [CrossRef]
- Richter, T.M.; Niewa, R. Chemistry of ammonothermal synthesis. Inorganics 2014, 2, 29–78. [Google Scholar] [CrossRef] [Green Version]
- Lemmon, E.W.; Bell, I.H.; Huber, M.L.; McLinden, M.O. NIST Standard Reference Database 23: Reference Fluid Thermodynamic and Transport Properties-REFPROP, Version 10.0; National Institute of Standards and Technology: Gaithersburg, MD, USA, 2018. [CrossRef]
- Christensen, C.H.; Johannessen, T.; Sørensen, R.Z.; Nørskov, J.K. Towards an ammonia-mediated hydrogen economy? Catal. Today 2006, 111, 140–144. [Google Scholar] [CrossRef]
- Sørensen, R.Z.; Hummelshøj, J.S.; Klerke, A.; Reves, J.B.; Vegge, T.; Nørskov, J.K.; Christensen, C.H. Indirect, reversible high-density hydrogen storage in compact metal ammine salts. J. Am. Chem. Soc. 2008, 130, 8660–8668. [Google Scholar] [CrossRef] [PubMed]
- Jensen, P.B.; Lysgaard, S.; Quaade, U.J.; Vegge, T. Designing mixed metal halide ammines for ammonia storage using density functional theory and genetic algorithms. Phys. Chem. Chem. Phys. 2014, 16, 19732–19740. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, T.; Miyaoka, H.; Miyaoka, H.; Ichikawa, T.; Kojima, Y. Review on Ammonia Absorption Materials: Metal Hydrides, Halides, and Borohydrides. Acs Appl. Energy Mater. 2018, 1, 232–242. [Google Scholar] [CrossRef]
- Malmali, M.; Le, G.; Hendrickson, J.; Prince, J.; McCormick, A.V.; Cussler, E.L. Better Absorbents for Ammonia Separation. ACS Sustain. Chem. Eng. 2018, 6, 6536–6546. [Google Scholar] [CrossRef]
- Langmi, H.W.; Engelbrecht, N.; Modisha, P.M.; Bessarabov, D. Hydrogen storage. In Electrochemical Power Sources: Fundamentals, Systems, and Applications; Elsevier: Amsterdam, The Netherlands, 2022; pp. 455–486. [Google Scholar] [CrossRef]
- Dimitriou, P.; Javaid, R. A review of ammonia as a compression ignition engine fuel. Int. J. Hydrogen Energy 2020, 45, 7098–7118. [Google Scholar] [CrossRef]
- Barelli, L.; Bidini, G.; Perla, M.; Pilo, F.; Trombetti, L. Boil-off gas emission from the fuel tank of a LNG powered truck. Fuel 2022, 325, 124954. [Google Scholar] [CrossRef]
- Farzaneh-Gord, M.; Saadat-Targhi, M.; Khadem, J. Selecting optimal volume ratio of reservoir tanks in CNG refueling station with multi-line storage system. Int. J. Hydrogen Energy 2016, 41, 23109–23119. [Google Scholar] [CrossRef]
- Duijm, N.; Markert, F.; Paulsen, J.L. Safety Assessment of Ammonia as a Transport Fuel; Technical Report; Forskningscenter Risoe: Roskilde, Denmark, 2005. [Google Scholar]
- Amelina, L.; Biliaiev, M.; Berlov, O.; Verhun, O.; Rusakova, T. Modeling of Environmental Pollution by Ammonia Emission from a Damaged Pipeline. Sci. Transp. Prog. 2021, 1, 5–14. [Google Scholar] [CrossRef]
- Hang, T.; Wu, J.; Xiao, S.; Li, B.; Li, H.; Yang, C.; Hu, N.; Xu, Y.; Zhang, Y.; Xie, X. Anti-biofouling NH3 gas sensor based on reentrant thorny ZnO/graphene hybrid nanowalls. Microsystems Nanoeng. 2020, 6, 41. [Google Scholar] [CrossRef]
- Ezzat, M.F.; Dincer, I. Development and assessment of a new hybrid vehicle with ammonia and hydrogen. Appl. Energy 2018, 219, 226–239. [Google Scholar] [CrossRef]
- Willmann, M.; Berger, I.; Barrow, E. Woodward L’Orange’s New Injector Generation—An Ideal Platform for the Combustion of E-Fuels in Large Engines; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar] [CrossRef]
- Brohi, E.A. Ammonia as Fuel for Internal Combustion Engines? An Evaluation of the Feasibility of Using Nitrogen-Based Fuels in ICE; Chalmers University of Technology: Gothenburg, Sweden, 2014. [Google Scholar]
- Kurien, C.; Mittal, M. Review on the production and utilization of green ammonia as an alternate fuel in dual-fuel compression ignition engines. Energy Convers. Manag. 2022, 251, 114990. [Google Scholar] [CrossRef]
- Pielecha, I. Modeling of Fuel Cells Characteristics in Relation to Real Driving Conditions of FCHEV Vehicles. Energies 2022, 15, 6753. [Google Scholar] [CrossRef]
- JIVE 2 under way to accelerate commercial start of fuel cell buses. Fuel Cells Bull. 2018, 2018, 2. [CrossRef]
- Available online: https://cordis.europa.eu/project/id/826236 (accessed on 13 March 2023).
- Auditors, E.C. Rail Freight Transport in the EU: Still Not on the Right Track; Special Report No 08; Publications Office: Luxembourg, 2016. [Google Scholar]
- Ibrahim Ozigis, I.; Oche, J.; Lawal, N. Locomotive Engines and the Future of Railway Automotive Power in Africa: A Review. Niger. J. Technol. 2021, 40, 660–673. [Google Scholar] [CrossRef]
- García-Garre, A.; Gabaldón, A. Analysis, Evaluation and Simulation of Railway Diesel-Electric and Hybrid Units as Distributed Energy Resources. Appl. Sci. 2019, 9, 3605. [Google Scholar] [CrossRef] [Green Version]
- Grennfelt, P.; Engleryd, A.; Forsius, M.; Hov, Ø.; Rodhe, H.; Cowling, E. Acid rain and air pollution: 50 years of progress in environmental science and policy. Ambio 2020, 49, 849–864. [Google Scholar] [CrossRef] [Green Version]
- Valera-Medina, A.; Morris, S.; Runyon, J.; Pugh, D.G.; Marsh, R.; Beasley, P.; Hughes, T. Ammonia, Methane and Hydrogen for Gas Turbines. Energy Procedia 2015, 75, 118–123. [Google Scholar] [CrossRef] [Green Version]
- Cornelius, W.; Huellmantel, L.W.; Mitchell, H.R. Ammonia as an Engine Fuel. In Proceedings of the 1965 International Automotive Engineering Congress and Exposition; SAE International: Warrendale, PA, USA, 1965. [Google Scholar] [CrossRef]
- Lhuillier, C.; Brequigny, P.; Rousselle, C.; Contino, F. Performance and Emissions of an Ammonia-Fueled SI Engine with Hydrogen Enrichment. In Proceedings of the 14th International Conference on Engines & Vehicles; SAE International: Warrendale, PA, USA, 2019. [Google Scholar] [CrossRef]
- Grannell, S.; Assanis, D.; Bohac, S.; Gillespie, D. The Operating Features of a Stoichiometric, Ammonia and Gasoline Dual Fueled Spark Ignition Engine. In ASME International Mechanical Engineering Congress and Exposition; ASME: New York, NY, USA, 2006. [Google Scholar] [CrossRef] [Green Version]
- Haputhanthri, S.; Maxwell, T.; Fleming, J.; Austin, C. Ammonia Gasoline-Ethanol/Methanol Tertiary Fuel Blends as an Alternate Automotive Fuel. In ASME International Mechanical Engineering Congress and Exposition, Proceedings (IMECE); American Society of Mechanical Engineers: New York, NY, USA, 2014; Volume 6. [Google Scholar] [CrossRef]
- Comotti, M.; Frigo, S. Hydrogen generation system for ammonia–hydrogen fuelled internal combustion engines. Int. J. Hydrogen Energy 2015, 40, 10673–10686. [Google Scholar] [CrossRef]
- Frigo, S.; Gentili, R.; Doveri, N. Ammonia Plus Hydrogen as Fuel in a S.I. Engine: Experimental Results. SAE Tech. Pap. 2012, 4. [Google Scholar] [CrossRef]
- Ryu, K.; Zacharakis-Jutz, G.E.; Kong, S.C. Effects of gaseous ammonia direct injection on performance characteristics of a spark-ignition engine. Appl. Energy 2014, 116, 206–215. [Google Scholar] [CrossRef]
- Niki, Y.; Nitta, Y.; Sekiguchi, H.; Hirata, K. Diesel Fuel Multiple Injection Effects on Emission Characteristics of Diesel Engine Mixed Ammonia Gas Into Intake Air. J. Eng. Gas Turbines Power 2019, 141, 061020. [Google Scholar] [CrossRef]
- Tay, K.L.; Yang, W.; Li, J.; Zhou, D.; Yu, W.; Zhao, F.; Chou, S.K.; Mohan, B. Numerical investigation on the combustion and emissions of a kerosene-diesel fueled compression ignition engine assisted by ammonia fumigation. Appl. Energy 2017, 204, 1476–1488. [Google Scholar] [CrossRef]
- Lamas Galdo, M.I.; Castro-Santos, L.; Rodriguez Vidal, C.G. Numerical Analysis of NOx Reduction Using Ammonia Injection and Comparison with Water Injection. J. Mar. Sci. Eng. 2020, 8, 109. [Google Scholar] [CrossRef] [Green Version]
- Lamas, M.; Rodriguez, C. Numerical model to analyze Nox reduction by ammonia injection in diesel-hydrogen engines. Int. J. Hydrogen Energy 2017, 42, 26132–26141. [Google Scholar] [CrossRef]
- Pyrc, M.; Gruca, M.; Jamrozik, A.; Tutak, W.; Juknelevičius, R. An experimental investigation of the performance, emission and combustion stability of compression ignition engine powered by diesel and ammonia solution (NH4OH). Int. J. Engine Res. 2021, 22, 2639–2653. [Google Scholar] [CrossRef]
- Schönborn, A. Aqueous solution of ammonia as marine fuel. Proc. Inst. Mech. Eng. J. Eng. Marit. Environ. 2021, 235, 142–151. [Google Scholar] [CrossRef]
- Şahin, Z.; Akcanca, İ.Z.; Durgun, O. Experimental investigation of the effects of ammonia solution (NH3OH) on engine performance and exhaust emissions of a small diesel engine. Fuel 2018, 214, 330–341. [Google Scholar] [CrossRef]
- Sivasubramanian, R.; Sajin, J.; Omanakuttan Pillai, G. Effect of ammonia to reduce emission from biodiesel fuelled diesel engine. Int. J. Ambient. Energy 2022, 43, 661–665. [Google Scholar] [CrossRef]
- Lin, C.Y.; Lin, H.A. Engine performance and emission characteristics of a three-phase emulsion of biodiesel produced by peroxidation. Fuel Process. Technol. 2007, 88, 35–41. [Google Scholar] [CrossRef]
- Zacharakis-Jutz, G.; Kong, S.C.; Ryu, K.H. Effects of Fuel Compositions on Diesel Engine Performance Using Ammonia-DME Mixtures. In Proceedings of the SAE 2013 World Congress & Exhibition; SAE International: Warrendale, PA, USA, 2013. [Google Scholar] [CrossRef]
- Gross, C.W.; Kong, S.C. Performance characteristics of a compression-ignition engine using direct-injection ammonia–DME mixtures. Fuel 2013, 103, 1069–1079. [Google Scholar] [CrossRef]
- Wang, W.; Herreros, J.M.; Tsolakis, A.; York, A.P. Ammonia as hydrogen carrier for transportation; investigation of the ammonia exhaust gas fuel reforming. Int. J. Hydrogen Energy 2013, 38, 9907–9917. [Google Scholar] [CrossRef] [Green Version]
- Gill, S.; Chatha, G.; Tsolakis, A.; Golunski, S.; York, A. Assessing the effects of partially decarbonising a diesel engine by co-fuelling with dissociated ammonia. Int. J. Hydrogen Energy 2012, 37, 6074–6083. [Google Scholar] [CrossRef]
- Pochet, M.; Jeanmart, H.; Contino, F. A 22:1 Compression Ratio Ammonia-Hydrogen HCCI Engine: Combustion, Load, and Emission Performances. Front. Mech. Eng. 2020, 6, 43. [Google Scholar] [CrossRef]
- Zhu, J.; Zhou, D.; Yang, W.; Qian, Y.; Mao, Y.; Lu, X. Investigation on the potential of using carbon-free ammonia in large two-stroke marine engines by dual-fuel combustion strategy. Energy 2023, 263, 125748. [Google Scholar] [CrossRef]
- World’s First Tractor Powered by Renewable Fuels with Zero Carbon Emissions Debuts on IOWA Farm. Available online: https://solarhydrogensystem.com/2016/01/06/worlds-first-tractor-powered-by-renewable-fuels-with-zero-carbon-emissions-debuts-on-iowa-farm/ (accessed on 13 April 2023).
- Toyota GT86-R Marangoni Eco Explorer: Sport and Technology in the New 2013 Show Car. Available online: https://marangonipress.com/2013/02/28/toyota-gt86-r-marangoni-eco-explorer-sport-and-technology-in-the-new-2013-show-car-fitted-with-marangoni-m-power-evored-tyres/?cookie-state-change=1670785787773 (accessed on 28 February 2023).
- Commission, E.; Centre, J.R.; Ortiz Cebolla, R.; Davies, J.; Weidner, E. Global Deployment of Large Capacity Stationary Fuel Cells: Drivers of, and Barriers to, Stationary Fuel Cell Deployment; Publications Office: Luxembourg, 2019. [Google Scholar] [CrossRef]
- Fan, L.; Tu, Z.; Chan, S.H. Recent development of hydrogen and fuel cell technologies: A review. Energy Rep. 2021, 7, 8421–8446. [Google Scholar] [CrossRef]
- The Comparison of Different Technologies for On-Board Power Supply of Cruise Ships; Nautilius Technical Report; Fuel Cells and Hydrogen Observatory: Brussels, Belgium, 2020; Available online: https://www.fchobservatory.eu/sites/default/files/reports/Chapter_1_Technology_and_Market_070920.pdf (accessed on 15 May 2023).
- Ministry of Ecnomic Affairs and Climate Policy. Hydrogen Economy Development in Korea; Ministry of Economy and Affairs and Climate Policy: Seoul, Republic of Korea, 2020.
- E4tech. Fuel Cell Industry Review 2019; E4tech: London, UK, 2019. [Google Scholar]
- Hydrogen Fuel Cell Vehicle Market. Available online: https://www.polarismarketresearch.com/industry-analysis/hydrogen-fuel-cell-vehicle-market (accessed on 13 February 2023).
- Hoelzen, J.; Silberhorn, D.; Zill, T.; Bensmann, B.; Hanke-Rauschenbach, R. Hydrogen-powered aviation and its reliance on green hydrogen infrastructure—Review and research gaps. Int. J. Hydrogen Energy 2022, 47, 3108–3130. [Google Scholar] [CrossRef]
- Lucentini, I.; Garcia, X.; Vendrell, X.; Llorca, J. Review of the Decomposition of Ammonia to Generate Hydrogen. Ind. Eng. Chem. Res. 2021, 60, 18560–18611. [Google Scholar] [CrossRef]
- Mekhilef, S.; Saidur, R.; Safari, A. Comparative study of different fuel cell technologies. Renew. Sustain. Energy Rev. 2012, 16, 981–989. [Google Scholar] [CrossRef]
- Shabani, B.; Hafttananian, M.; Khamani, S.; Ramiar, A.; Ranjbar, A. Poisoning of proton exchange membrane fuel cells by contaminants and impurities: Review of mechanisms, effects, and mitigation strategies. J. Power Sources 2019, 427, 21–48. [Google Scholar] [CrossRef]
- Zarabi Golkhatmi, S.; Asghar, M.I.; Lund, P.D. A review on solid oxide fuel cell durability: Latest progress, mechanisms, and study tools. Renew. Sustain. Energy Rev. 2022, 161, 112339. [Google Scholar] [CrossRef]
- Hagen, A.; Langnickel, H.; Sun, X. Operation of solid oxide fuel cells with alternative hydrogen carriers. Int. J. Hydrogen Energy 2019, 44, 18382–18392. [Google Scholar] [CrossRef]
- Kishimoto, M.; Muroyama, H.; Suzuki, S.; Saito, M.; Koide, T.; Takahashi, Y.; Horiuchi, T.; Yamasaki, H.; Matsumoto, S.; Kubo, H.; et al. Development of 1 kW-class Ammonia-fueled Solid Oxide Fuel Cell Stack. Fuel Cells 2020, 20, 80–88. [Google Scholar] [CrossRef]
- Wang, Y.; Gu, Y.; Zhang, H.; Yang, J.; Wang, J.; Guan, W.; Chen, J.; Chi, B.; Jia, L.; Muroyama, H.; et al. Efficient and durable ammonia power generation by symmetric flat-tube solid oxide fuel cells. Appl. Energy 2020, 270, 115185. [Google Scholar] [CrossRef]
- Fuerte, A.; Valenzuela, R.; Escudero, M.; Daza, L. Ammonia as efficient fuel for SOFC. J. Power Sources 2009, 192, 170–174. [Google Scholar] [CrossRef]
- Hung, Y.; Shy, S. A pressurized ammonia-fed planar anode-supported solid oxide fuel cell at 1–5 atm and 750–850 °C and its loaded short stability test. Int. J. Hydrogen Energy 2020, 45, 27597–27610. [Google Scholar] [CrossRef]
- Lan, R.; Tao, S. Ammonia as a Suitable Fuel for Fuel Cells. Front. Energy Res. 2014, 2, 35. [Google Scholar] [CrossRef] [Green Version]
- Zhao, J.F.; Liang, Q.C.; Liang, Y.F. Simulation and Study of PEMFC System Directly Fueled by Ammonia Decomposition Gas. Front. Energy Res. 2022, 10, 819939. [Google Scholar] [CrossRef]
- IEA. ETP Clean Energy Technology Guide. Available online: https://www.iea.org/data-and-statistics/data-tools/etp-clean-energy-technology-guide?search=ammonia (accessed on 13 February 2023).
- Mukerjee, S.; Leah, R.; Selby, M.; Stevenson, G.; Brandon, N.P. Chapter 9—Life and Reliability of Solid Oxide Fuel Cell-Based Products: A Review. In Solid Oxide Fuel Cell Lifetime and Reliability; Brandon, N.P., Ruiz-Trejo, E., Boldrin, P., Eds.; Academic Press: New York, NY, USA, 2017; pp. 173–191. [Google Scholar] [CrossRef]
- Ghezel-Ayagh, H.; Borglum, B.P. Review of Progress in Solid Oxide Fuel Cells at FuelCell Energy. ECS Trans. 2017, 78, 77. [Google Scholar] [CrossRef]
- Bloom completes first international project, for SoftBank in Japan. Fuel Cells Bull. 2013, 2013, 6. [CrossRef]
- Corigliano, O.; Pagnotta, L.; Fragiacomo, P. On the Technology of Solid Oxide Fuel Cell (SOFC) Energy Systems for Stationary Power Generation: A Review. Sustainability 2022, 14, 15276. [Google Scholar] [CrossRef]
- Kishimoto, M.; Furukawa, N.; Kume, T.; Iwai, H.; Yoshida, H. Formulation of ammonia decomposition rate in Ni-YSZ anode of solid oxide fuel cells. Int. J. Hydrogen Energy 2017, 42, 2370–2380. [Google Scholar] [CrossRef]
- Engines, M. On-Road Diesel Engines, Axles and Transfer Cases for Trucks. Available online: https://pdf.directindustry.com/pdf/man-engines-division-man-truck-bus/brochure-truck/81737-302605.html (accessed on 13 February 2023).
- Cinti, G.; Desideri, U. SOFC fuelled with reformed urea. Appl. Energy 2015, 154, 242–253. [Google Scholar] [CrossRef]
- Okanishi, T.; Okura, K.; Srifa, A.; Muroyama, H.; Matsui, T.; Kishimoto, M.; Saito, M.; Iwai, H.; Yoshida, H.; Saito, M.; et al. Comparative Study of Ammonia-fueled Solid Oxide Fuel Cell Systems. Fuel Cells 2017, 17, 383–390. [Google Scholar] [CrossRef]
- Tan, W.C.; Iwai, H.; Kishimoto, M.; Brus, G.; Szmyd, J.S.; Yoshida, H. Numerical analysis on effect of aspect ratio of planar solid oxide fuel cell fueled with decomposed ammonia. J. Power Sources 2018, 384, 367–378. [Google Scholar] [CrossRef]
- Quach, T.Q.; Giap, V.T.; Keun Lee, D.; Pineda Israel, T.; Young Ahn, K. High-efficiency ammonia-fed solid oxide fuel cell systems for distributed power generation. Appl. Energy 2022, 324, 119718. [Google Scholar] [CrossRef]
- Cinti, G.; Discepoli, G.; Sisani, E.; Desideri, U. SOFC operating with ammonia: Stack test and system analysis. Int. J. Hydrogen Energy 2016, 41, 13583–13590. [Google Scholar] [CrossRef]
- Barelli, L.; Bidini, G.; Cinti, G. Operation of a Solid Oxide Fuel Cell Based Power System with Ammonia as a Fuel: Experimental Test and System Design. Energies 2020, 13, 6173. [Google Scholar] [CrossRef]
- Stoeckl, B.; Preininger, M.; Subotić, V.; Megel, S.; Folgner, C.; Hochenauer, C. Towards a wastewater energy recovery system: The utilization of humidified ammonia by a solid oxide fuel cell stack. J. Power Sources 2020, 450, 227608. [Google Scholar] [CrossRef]
- WHO Air Pollution. 2019. Available online: https://www.who.int/health-topics/air-pollution (accessed on 7 March 2023).
- Mulholland, E.; Miller, J.; Bernard, Y.; Lee, K.; Rodríguez, F. The role of NOx emission reductions in Euro 7/VII vehicle emission standards to reduce adverse health impacts in the EU27 through 2050. Transp. Eng. 2022, 9, 100133. [Google Scholar] [CrossRef]
- European Environment Agency. Report No. 15/2021: Air Quality in Europe—2021 Report; Technical Report; EEA: Copenhagen, Denmark, 2021. [Google Scholar]
- Calderón-Garcidueñas, L.; Villarreal-Ríos, R. Living close to heavy traffic roads, air pollution, and dementia. Lancet 2017, 389, 675–677. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Batterman, S. Air pollution and health risks due to vehicle traffic. Sci. Total Environ. 2013, 450, 307–316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weagle, C.L.; Snider, G.; Li, C.; Donkelaar, A.V.; Philip, S.; Bissonnette, P.; Burke, J.; Jackson, J.; Latimer, R.; Stone, E.; et al. Global Sources of Fine Particulate Matter: Interpretation of PM2.5 Chemical Composition Observed by SPARTAN using a Global Chemical Transport Model. Environ. Sci. Technol. 2018, 52, 11670–11681. [Google Scholar] [CrossRef] [PubMed]
- Anenberg, S.C.; Miller, J.; Henze, D.K.; Minjares, R.; Achakulwisut, P. The global burden of transportation tailpipe emissions on air pollution-related mortality in 2010 and 2015. Environ. Res. Lett. 2019, 14, 094012. [Google Scholar] [CrossRef]
- Timmers, V.R.; Achten, P.A. Non-exhaust PM emissions from electric vehicles. Atmos. Environ. 2016, 134, 10–17. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, H.; Gao, J.; Li, Y.; Dave, K.; Chen, J.; Federici, M.; Perricone, G. Comparative analysis of non-exhaust airborne particles from electric and internal combustion engine vehicles. J. Hazard. Mater. 2021, 420, 126626. [Google Scholar] [CrossRef]
- Beddows, D.C.; Harrison, R.M. PM10 and PM2.5 emission factors for non-exhaust particles from road vehicles: Dependence upon vehicle mass and implications for battery electric vehicles. Atmos. Environ. 2021, 244, 117886. [Google Scholar] [CrossRef]
- Shaw, S.; Heyst, B.V. Nitrogen Oxide (NOx) emissions as an indicator for sustainability. Environ. Sustain. Indic. 2022, 15, 100188. [Google Scholar] [CrossRef]
- Lozhkina, O.V.; Lozhkin, V.N. Estimation of nitrogen oxides emissions from petrol and diesel passenger cars by means of on-board monitoring: Effect of vehicle speed, vehicle technology, engine type on emission rates. Transp. Res. Transp. Environ. 2016, 47, 251–264. [Google Scholar] [CrossRef]
- Hinokuma, S.; Kiritoshi, S.; Kawabata, Y.; Araki, K.; Matsuki, S.; Sato, T.; Machida, M. Catalytic ammonia combustion properties and operando characterization of copper oxides supported on aluminum silicates and silicon oxides. J. Catal. 2018, 361, 267–277. [Google Scholar] [CrossRef]
- Weissenberger, T.; Zapf, R.; Pennemann, H.; Kolb, G. Effect of the Active Metal on the NOx Formation during Catalytic Combustion of Ammonia SOFC Off-Gas. Catalysts 2022, 12, 1186. [Google Scholar] [CrossRef]
- Farren, N.J.; Davison, J.; Rose, R.A.; Wagner, R.L.; Carslaw, D.C. Characterisation of ammonia emissions from gasoline and gasoline hybrid passenger cars. Atmos. Environ. 2021, 11, 100117. [Google Scholar] [CrossRef]
- Giannakis, E.; Kushta, J.; Bruggeman, A.; Lelieveld, J. Costs and benefits of agricultural ammonia emission abatement options for compliance with European air quality regulations. Environ. Sci. Eur. 2019, 31, 93. [Google Scholar] [CrossRef] [Green Version]
- Wyer, K.E.; Kelleghan, D.B.; Blanes-Vidal, V.; Schauberger, G.; Curran, T.P. Ammonia emissions from agriculture and their contribution to fine particulate matter: A review of implications for human health. J. Environ. Manag. 2022, 323, 116285. [Google Scholar] [CrossRef]
- Werner, M.; Kryza, M.; Dore, A.J. Differences in the Spatial Distribution and Chemical Composition of PM10 Between the UK and Poland. Environ. Model. Assess. 2014, 19, 179–192. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Wei, N.; Zou, C.; Mao, H. Evaluating the ammonia emission from in-use vehicles using on-road remote sensing test. Environ. Pollut. 2021, 271, 116384. [Google Scholar] [CrossRef] [PubMed]
- Nadimi, E.; Przybyła, G.; Lewandowski, M.T.; Adamczyk, W. Effects of ammonia on combustion, emissions, and performance of the ammonia/diesel dual-fuel compression ignition engine. J. Energy Inst. 2023, 107, 101158. [Google Scholar] [CrossRef]

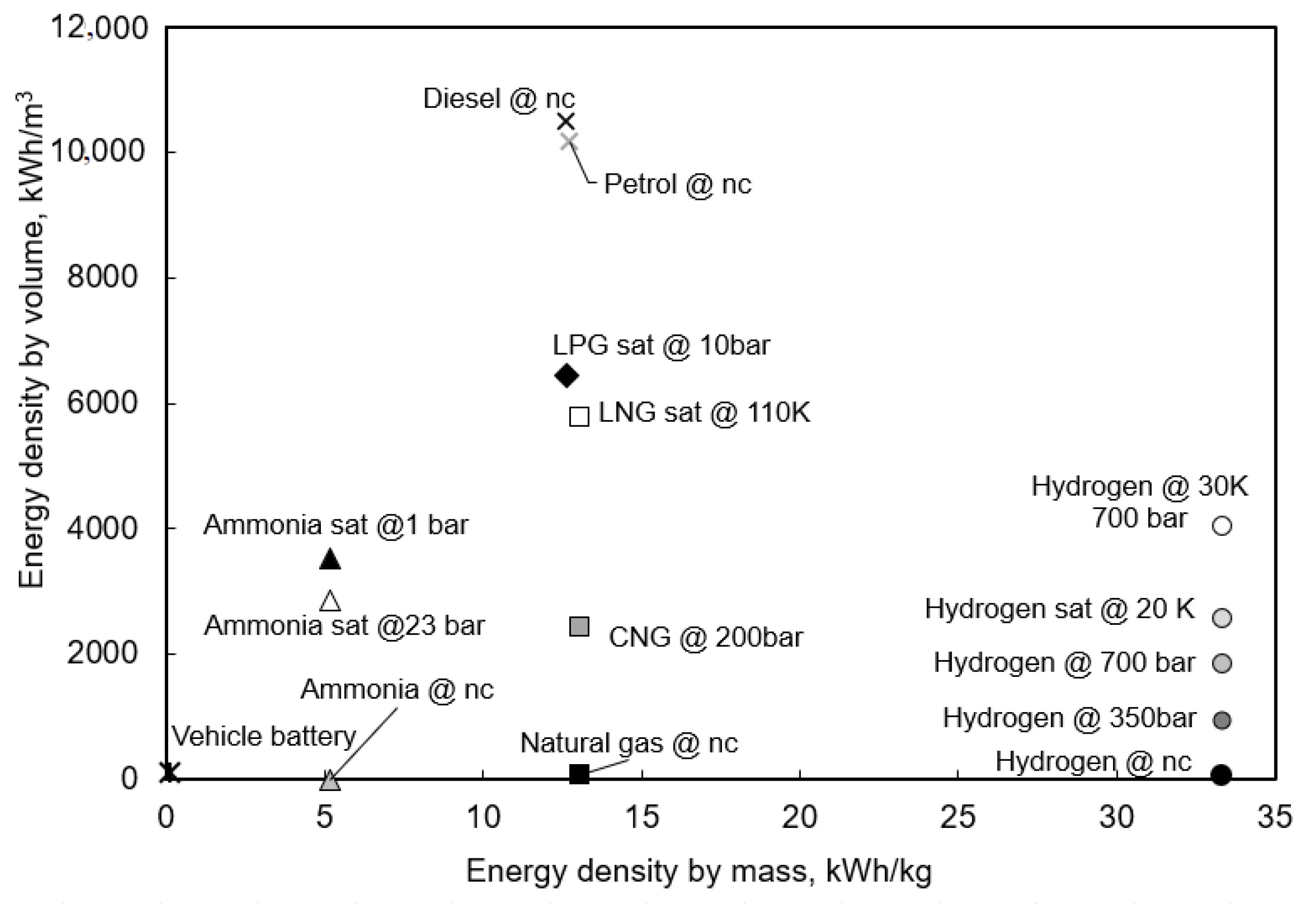
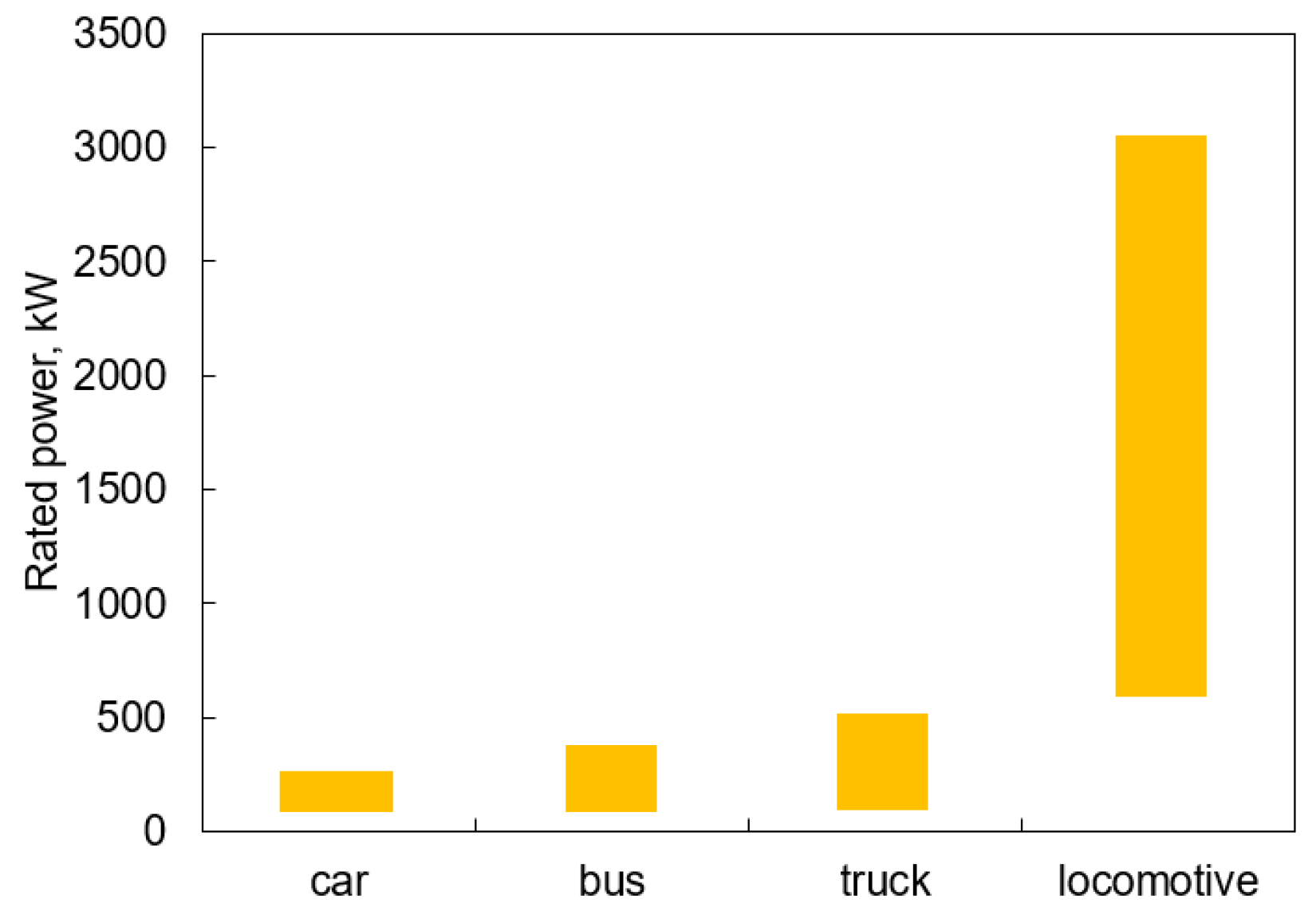

| Concentration ppm | Symptoms |
|---|---|
| 5 | Felt in the air |
| 10 | Road vehicles emission limit in acc. to [26,27] |
| 25 | Permissible concentration in air for exposure up to 8 h |
| 35 | Permissible concentration with exposure up to 15 min, perceptible in water |
| 50 | Irritating to eyes, nose and throat |
| 100 | Limit concentration at which without personal protective equipment exposure for up to 1 h does not cause irreversible damage to health |
| 300 | Concentration at which, during short-term exposure, disturbances of consciousness and irreversible health effects are not observed |
| 500 | Limit concentration at which, without personal protective equipment, exposure for up to 30 min does not cause irreversible damage to health |
| 700–1700 | Coughing, bronchospasm, significant tearing and irritation of the eyes |
| 5000 | Chemical bronchitis, pulmonary edema, chemical burns to the skin, high risk of death |
| Species | LC50, ppm | LD50, ppm |
|---|---|---|
| Rat [33] | 7000 | 40,000 |
| Mouse [33] | 4000 | 10,000 |
| Cat [33] | 1000 | 3000 |
| Daphnia magna (an aquatic organism from the plankton group) [34] | 0.66 | - |
| Lepomis cyanellus (Freshwater fish) [34] | 140 | - |
| Scophthalmus maximus [34] | 11 | - |
| Fuels | Octane [RON] | Flame Velocity (m/s) | Flammability Limits (vol%) | Minimum Ignition Energy (mJ) | Autoignition [AIT] (°C) |
|---|---|---|---|---|---|
| NH 1 bar/−33 °C | >130 | 0.07 | 15–28 | 680 | 651 |
| NH 300 bar/25 °C | |||||
| H 1 bar/−253 °C | >130 | 3.25 | 4.7–75 | −0.02 | 500 |
| H 300 bar/25 °C | |||||
| Diesel 1 bar/25 °C | <20 | −0.8 | 0.43–0.6 | −0.02 | 210–350 |
| Gasoline 1 bar/25 °C | 100 | 0.41 | 0.6–8 | −0.14 | 247–280 |
| Methanol 1 bar/25 °C | 109 | 0.56 | 6.7–36 | 0.14 | 470 |
| Ethanol 1 bar/25 °C | 109 | 0.58 | 3.3–19 | 0.6 | 365 |
| Description | Advantages | Disadvantages |
|---|---|---|
| neat ammonia [89]: - air mixing | - better engine stability and better efficiency | - efficiency lower than gasoline engines - can only operate up to 2400 RPM |
| - turbocharging | - output power comparable with gasoline engines | |
| - increased CR | - extends engine operating speed to 4000 RPM | |
| fuel blending: - with H [89,90] | - reduces unburned NH emission; - efficiency competitive to neat gasoline engine | - efficiency declined below 3000 RPM. - NO emissions increased |
| - with gasoline [91] | - satisfactory engine stability and performances | |
| - with gasoline & methanol/ethanol [92] | - performance at >3500 RPM better than baseline fuel | |
| dissociating NH by hot exhaust gas [16] | Compared to engine without NH dissociation: - engine brake power elevated - NO emission lowered - NH slip and CO emissions lowered. | - additional NH dissiociating installation |
| dissociating NH by Hydrogen Generation System [93] | - at 3000 RPM nearly identical efficiency to gasoline - at >3000 RPM efficiency engine remained - NO emission lowered to the gasoline engine | - additional NH Hydrogen Generation System installation |
| decomposed NH mixing with gasoline [89] | - increasing flame speed and combustion stability | - additional NH decomposing installation |
| separate injection NH and H [94] | - good power response to a neat gasoline engine | - NO emissions increased |
| NH direct-injection blended with gasoline [16,95] | - higher engine power. | - fuel efficiency lower to gasoline/NH |
| Description | Advantages | Disadvantages |
|---|---|---|
| advanced Start of Ignition (SOI) [96,97] | - enhanced engine heat release rate - increased cylinder pressure | - higher NO emissions |
| retarded fuel injection timing [98,99] | - NO emission reduction | - increase of unburned NH |
| aqueous ammonia [100,101,102] | - improved performance - reduced HC, CO, NO emissions | - increase noise level - higher CR for stable operation |
| blending NH with fossil fuels | - enhanced engine heat release rate | - increased NO emissions |
| oxygenated fuels [103,104,105,106] | - HC, CO emissions reduction | - lower efficiency |
| ammonia decomposition [107,108,109] | - HC, CO emissions reduction | - lower efficiency |
| Material | Stack Power Class, W | Stack Electrical Efficiency, % | Noted Power Density, mW/cm | Anode Support Thickness, m | Electrolyte Thickness, m | Temperature, C | Number of Cells, - | Cell Active Area, cm | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Ni-YSZ/YSZ/LSM | 75 | 30–65 | 240 | 1500 | 8–10 | 800 | 4 | 80 | [138] |
| Ni-YSZ/YSZ/LSM | 75 | 30–65 | 240 | 1500 | 8–10 | 800 | 4 | 80 | [142] |
| NiO-YSZ/YSZ/not specified | 200 | - | 210 | 1000–1100 | 7–13 | 770 | 10 | 95 | [139] |
| Ni-YSZ/YSZ/GDC/LSCF-GDC | 100 | 67 | 375 | 240 | 8 | 800 | 6 | 48 | [140] * |
| Ni-YSZ/YSZ/GDC/LSCF-GDC | 100 | 52.1 | 360 | 240 | 8 | 750 | 6 | 80 | [143] |
| Ni/GDC/10Sc1CeSZ/LSM/ScSZ | 200 | - | 180 | 40 ** | 165 | 840 | 10 | 127 | [144] |
| Ni-YSZ/YSZ/LaNiO-based perovskite material | 1000 | - | 368 | 1000–1100 | 7–13 | 750 | 30 | 95 | [125] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chorowski, M.; Lepszy, M.; Machaj, K.; Malecha, Z.; Porwisiak, D.; Porwisiak, P.; Rogala, Z.; Stanclik, M. Challenges of Application of Green Ammonia as Fuel in Onshore Transportation. Energies 2023, 16, 4898. https://doi.org/10.3390/en16134898
Chorowski M, Lepszy M, Machaj K, Malecha Z, Porwisiak D, Porwisiak P, Rogala Z, Stanclik M. Challenges of Application of Green Ammonia as Fuel in Onshore Transportation. Energies. 2023; 16(13):4898. https://doi.org/10.3390/en16134898
Chicago/Turabian StyleChorowski, Maciej, Michał Lepszy, Krystian Machaj, Ziemowit Malecha, Dominika Porwisiak, Paweł Porwisiak, Zbigniew Rogala, and Michał Stanclik. 2023. "Challenges of Application of Green Ammonia as Fuel in Onshore Transportation" Energies 16, no. 13: 4898. https://doi.org/10.3390/en16134898







