Drug-Eluting Intraocular Lenses
Abstract
:1. Intraocular Lenses (IOLs)
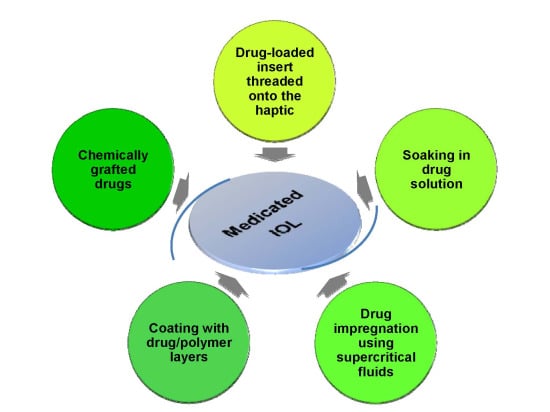
2. Drug Loading and Elution from IOLs
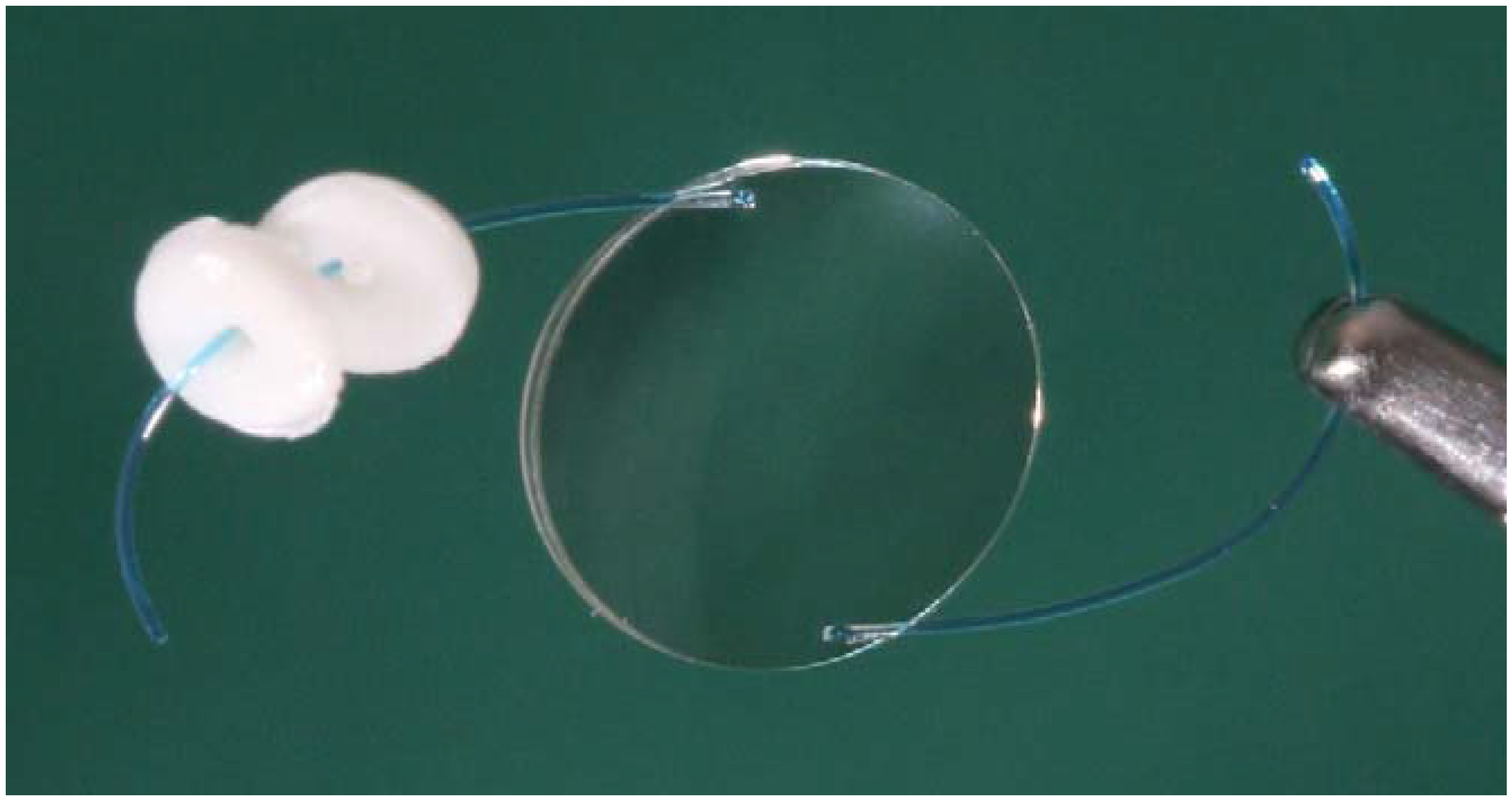
2.1. Pendant Solvent-Cast Drug Inserts
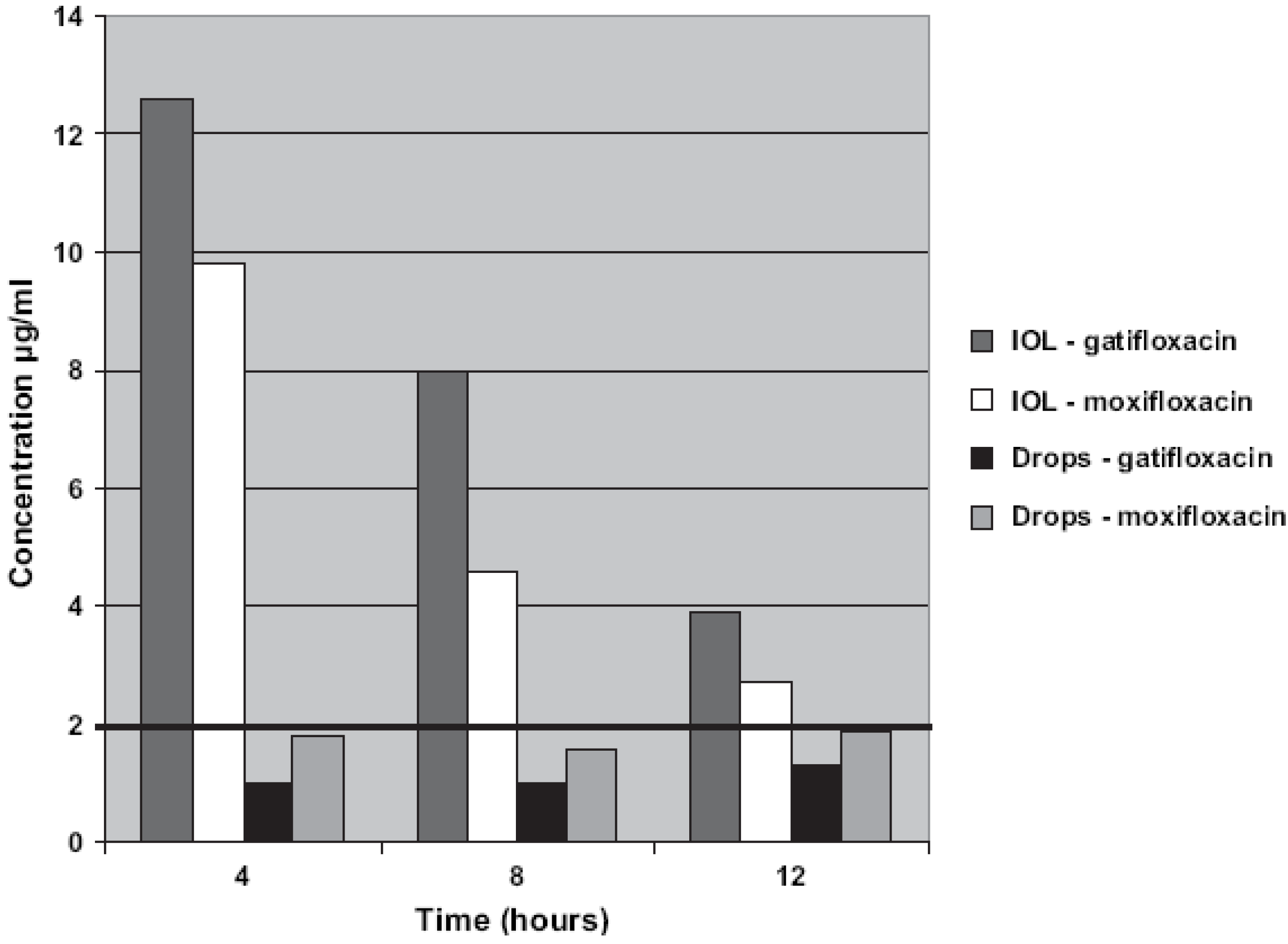
2.2. Drug Soaked IOLs
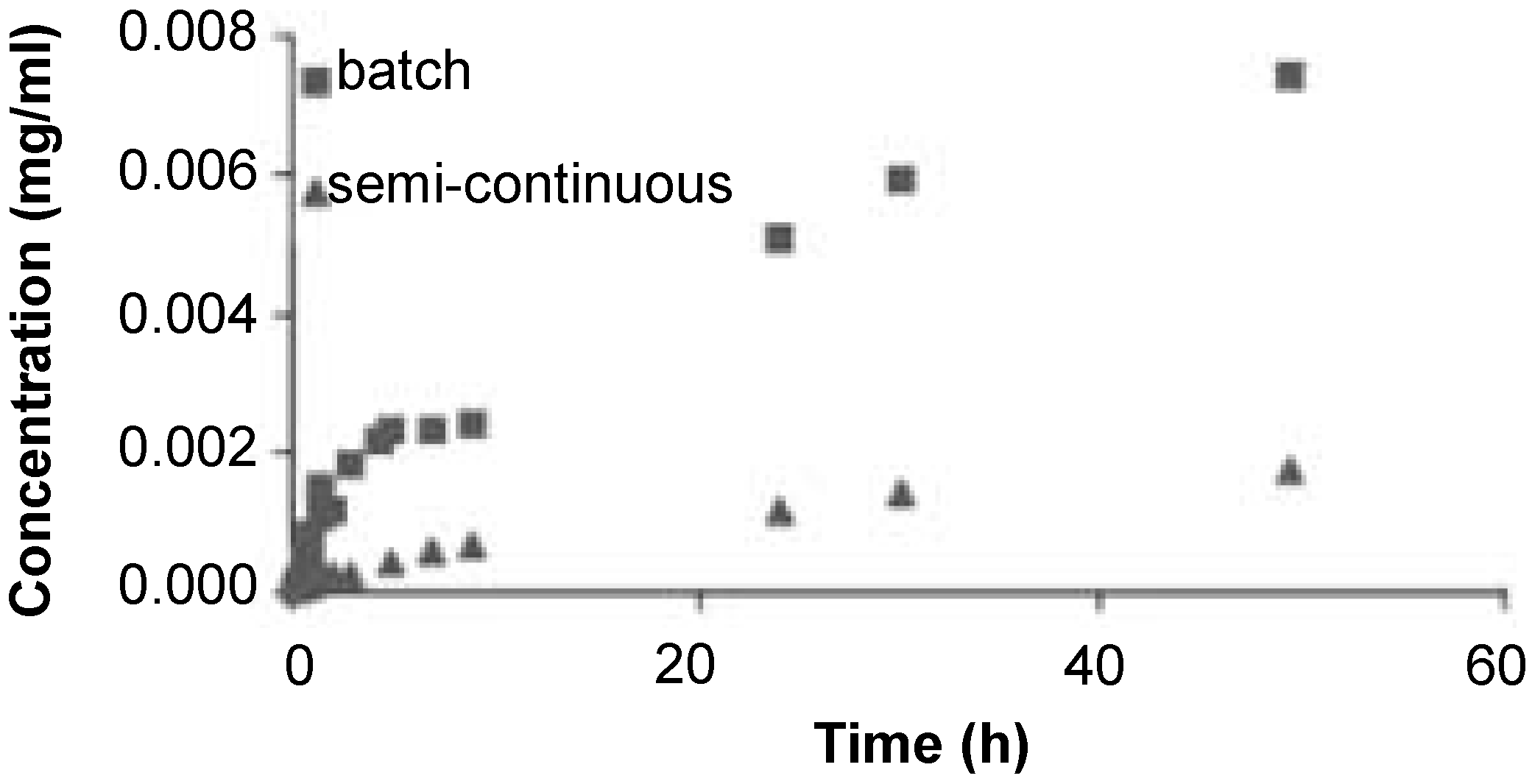
2.3. Supercritical Fluids Impregnation
2.4. Deposition of Drug/Coating Components
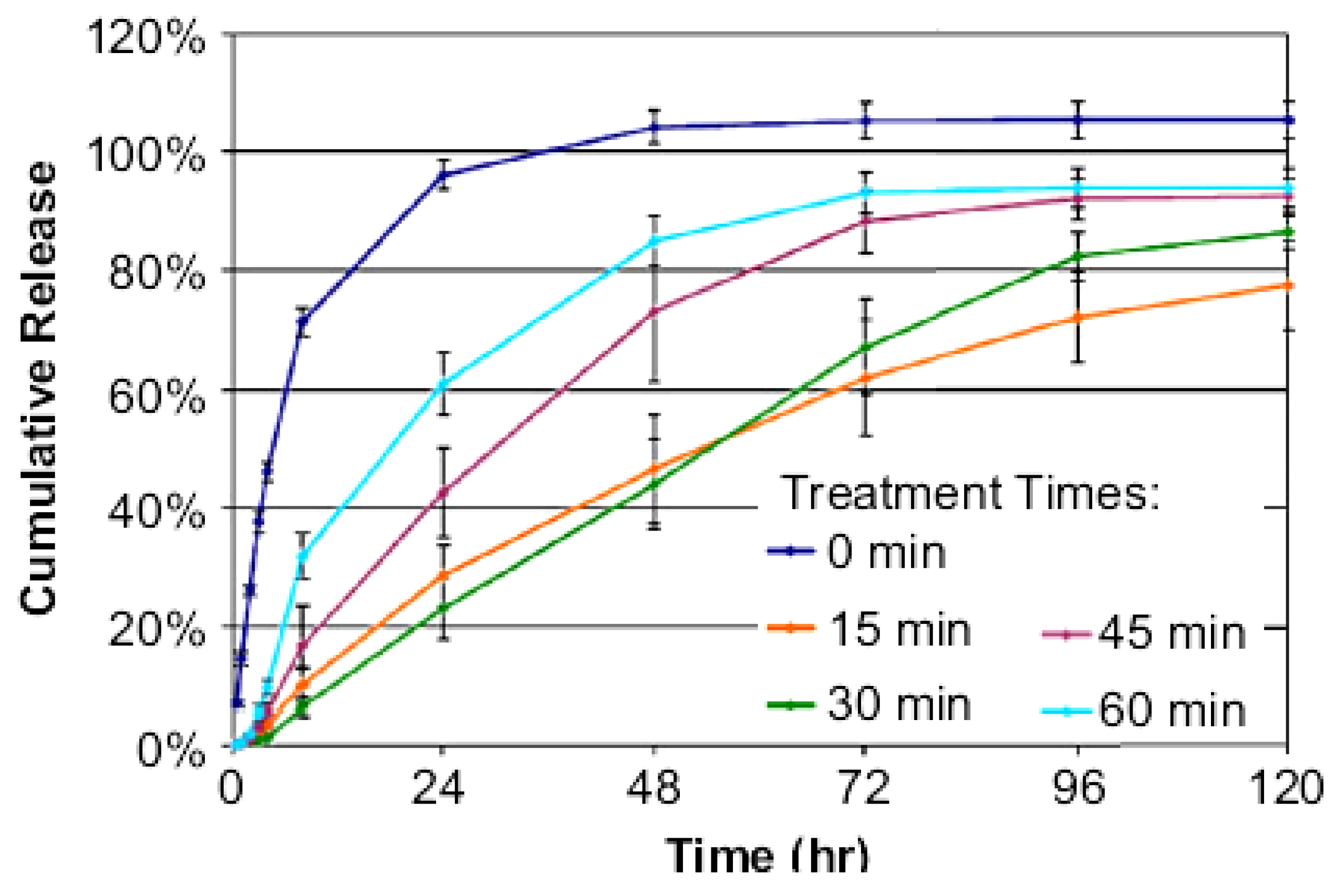

2.5. Chemically Grafted Drugs
| Polymer composition % w/w p(HEMA-co-MAA) | % Reduction in bacterial adherence ± SD relative to untreated control | ||
|---|---|---|---|
| Intense light (4300 lux) | Laboratory light (1260 lux) | Dark | |
| 60/40 | 93.90 ± 0.59 | 88.76 ± 2.57 | 90.09 ± 4.20 |
| 70/30 | 94.52 ± 0.77 | 90.01 ± 3.59 | 88.21 ± 6.07 |
| 80/20 | 96.17 ± 0.38 | 92.39 ± 5.38 | 84.70 ± 6.78 |
| 90/10 | 99.02 ± 0.42 | 90.99 ± 6.02 | 91.76 ± 5.99 |
| 100/0 | 98.88 ± 0.92 | 88.42 ± 2.82 | 86.22 ± 6.32 |

3. Conclusions
Acknowledgments
References
- Lloyd, A.W.; Faragher, R.G.A.; Denyer, S.P. Ocular biomaterials and implants. Biomaterials 2001, 22, 769–785. [Google Scholar] [CrossRef] [PubMed]
- Behndig, A.; Montan, P.; Stenevi, U.; Kugelberg, M.; Lundstrom, M. One million cataract surgeries: Swedish National Cataract Register 1992–2009. J. Cataract Refr. Surg. 2011, 37, 1539–1545. [Google Scholar] [CrossRef]
- Ellwein, L.B.; Kupfer, C. Strategic issues in preventing cataract blindness in developing countries. Bull. World Health Organ. 1995, 73, 681–690. [Google Scholar] [PubMed]
- Parra, F.; Vázquez, B.; Benito, L.; Barcenilla, J.; San-Román, J. Foldable antibacterial acrylic intraocular lenses of high refractive index. Biomacromolecules 2009, 10, 3055–3061. [Google Scholar] [CrossRef] [PubMed]
- Takemoto, L.; Sorensen, C.M. Protein-protein interactions and lens transparency. Exp. Eye Res. 2008, 87, 496–501. [Google Scholar] [CrossRef] [PubMed]
- Bozukova, D.; Pagnoulle, C.; Jérôme, R.; Jérôme, C. Polymers in modern ophthalmic implants-historical background and recent advances. Mater. Sci. Eng. R-Rep. 2010, 69, 63–83. [Google Scholar] [CrossRef]
- Moore, D.B.; Harris, A.; Siesky, B. The world through a lens: The vision of Sir Harold Ridley. Br. J. Ophthalmol. 2010, 94, 1277–1280. [Google Scholar] [CrossRef] [PubMed]
- Werner, L. Biocompatibility of intraocular lens materials. Curr. Opin. Ophthalmol. 2008, 19, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Tripti, D.; Haldar, R.S.; Geetha, S.; Niyogi, U.K.; Khandal, R.K. Materials for intraocular lenses (IOLs): Review of developments to achieve biocompatibility. E-Polymers 2009, 124, 1–23. [Google Scholar]
- Riaz, Y.; Mehta, J.S.; Wormald, R.; Evans, J.R.; Foster, A.; Ravilla, T.; Snellingen, T. Surgical interventions for age-related cataract. Cochrane Database Syst. Rev. 2006, 4. Art. No. CD001323. [Google Scholar]
- Fernandes, P.; González-Méijome, J.M.; Madrid-Costa, D.; Ferrer-Blasco, T.; Jorge, J.; Montés-Micó, R. Implantable collamer posterior chamber intraocular lenses: A review of potential complications. J. Refractive Surg. 2011, 27, 765–776. [Google Scholar] [CrossRef] [Green Version]
- Song, L.; Hu, W.; Wang, G.; Niu, G.; Zhang, H.; Cao, H.; Wang, K.; Yang, H.; Zhu, S. Tailored (meth)acrylate shape-memory polymer networks for ophthalmic applications. Macromol. Biosci. 2010, 10, 1194–1202. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Hu, W.; Zhang, H.; Wang, G.; Yang, H.; Zhu, S. In vitro evaluation of chemically cross-linked shape-memory acrylate-methacrylate copolymer networks as ocular implants. J. Phys. Chem. B 2010, 114, 7172–7178. [Google Scholar] [CrossRef] [PubMed]
- Sriram, R.D. Fluorine surface with square edge IOLs. Patent IN 2005CH01836, August 2007. [Google Scholar]
- Novack, G.D. Ophthalmic drug delivery: Development and regulatory considerations. Clin. Pharmacol. Ther. 2009, 85, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Morarescu, D.; West-Mays, J.A.; Sheardown, H.D. Effect of delivery of MMP inhibitors from PDMS as a model IOL material on PCO markers. Biomaterials 2010, 31, 2399–2407. [Google Scholar] [CrossRef] [PubMed]
- Behar-Cohen, F.F.; David, T.; Buechler, Y.; Nova, M.P.; Houston, L.L.; Pouliquen, Y.M.; Courtois, Y. Cytotoxic effects of FGF2-saporin on bovine epithelial lens cells in vitro. Invest. Ophth. Vis. Sci. 1995, 36, 2425–2433. [Google Scholar]
- Behar-Cohen, F.F.; David, T.; D´Hermies, F.; Pouliquen, Y.M.; Buechler, Y.; Nova, M.P.; Houston, L.L.; Courtois, Y. In vivo inhibition of lens regrowth by fibroblast growth factor 2-saporin. Invest. Ophth. Vis. Sci. 1995, 36, 2434–2448. [Google Scholar]
- Duarte, A.R.C.; Simplicio, A.L.; Vega-González, A.; Subra-Paternault, P.; Coimbra, P.; Gil, M.H.; de Sousa, H.C.; Duarte, C.M.M. Impregnation of an intraocular lens for ophthalmic drug delivery. Curr. Drug Del. 2008, 5, 102–107. [Google Scholar] [CrossRef]
- Nagaki, Y.; Hayasaka, S.; Kadoi, C.; Matsumoto, M.; Yanagisawa, S.; Watanabe, K.; Watanabe, K.; Hayasaka, Y.; Ikeda, N.; Sato, S.; Kataoka, Y.; Togashi, M.; Abe, T. Bacterial endophthalmitis after small-incision cataract surgery. Effect of incision placement and intraocular lens type. J. Cataract. Refract. Surg. 2003, 29, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Stein, J.D.; Grossman, D.S.; Mundy, K.M.; Sugar, A.; Sloan, F.A. Severe adverse events after cataract surgery among medicare beneficiaries. Ophthalmology 2011, 118, 1716–1723. [Google Scholar] [CrossRef] [PubMed]
- Edward, F.S.; Abdel, R.E.; Amilia, S.; Peter, D.B.; Issa, E. Antimicrobial activity of acrylic intraocular lenses soaked in fourth generation fluoroquinolones. J. Ocular Pharmacol. Ther. 2008, 24, 495–500. [Google Scholar] [CrossRef]
- Anderson, E.M.; Noble, M.L.; Garty, S.; Ma, H.; Bryers, J.D.; Shen, T.T.; Ratner, B.D. Sustained release of antibiotic from poly(2-hydroxyethyl methacrylate) to prevent blinding infections after cataract surgery. Biomaterials 2009, 30, 5675–5681. [Google Scholar] [CrossRef] [PubMed]
- Shaw, J.; Smith, E.F.; Desai, R.U.; Enriquez, B.; Schrier, A. Can intraocular lenses deliver antibiotics intracamerally? J. Ocular Pharmacol. Ther. 2010, 26, 587–589. [Google Scholar] [CrossRef]
- Garty, S.; Shirakawa, R.; Warsen, A.; Anderson, E.M.; Noble, M.L.; Bryers, J.D.; Ratner, B.D.; Shen, T.T. Sustained antibiotic release from an intraocular lens-hydrogel assembly for cataract surgery. Invest. Ophthalmol. Vis. Sci. 2011, in press. [Google Scholar]
- Siqueira, R.C.; Filho, E.R.; Fialho, S.L.; Lucena, L.R.; Filho, A.M.; Haddad, A. Pharmacokinetic and toxicity investigations of a new intraocular lens with a dexamethasone drug delivery system: A pilot study. Ophthalmologica 2006, 220, 338–342. [Google Scholar] [CrossRef] [PubMed]
- Eperon, S.; Bossy-Nobs, L.; Petropoulos, I.K.; Gurny, R.; Guex-Crosier, Y. A biodegradable drug delivery system for the treatment of postoperative inflammation. Int. J. Pharm. 2008, 352, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Choonara, Y.E.; Pillay, V.; Danckwerts, M.P.; Carmichael, T.R.; Toit, L.C.D. A review of implantable intravitreal drug delivery technologies for the treatment of posterior segment eye diseases. J. Pharm. Sci. 2010, 99, 2219–2239. [Google Scholar] [CrossRef] [PubMed]
- Yasukawa, T.; Tabata, Y.; Kimura, H.; Ogura, Y. Recent advances in intraocular drug delivery systems. Recent Pat. Drug Deliv. Formul. 2011, 5, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Chieh, J.J.; Carlson, A.N.; Jaffe, G.J. Combined fluocinolone acetonide intraocular delivery system insertion, phacoemulsification, and intraocular lens implantation for severe uveitis. Am. J. Ophthalmol. 2008, 146, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Bossy-Nobs, L.; Felt-Baeyens, O.; Gurny, R.; Eperon, S.; Guex-Crosier, Y. Intraocular lens with drug delivery system attached thereto. Patent US2009/0130176, May 2009. [Google Scholar]
- Alvarez-Lorenzo, C.; Hiratani, H.; Concheiro, A. Contact lenses for drug delivery: Achieving sustained release with novel systems. Am. J. Drug Del. 2006, 4, 131–151. [Google Scholar] [CrossRef]
- Xinming, L.; Yingde, C.; Lloyd, A.W.; Mikhalovsky, S.V.; Sandeman, S.R.; Howel, C.A.; Liewen, L. Polymeric hydrogels for novel contact lens-based ophthalmic drug delivery systems: A review. Cont. Lens. Anterior. Eye. 2008, 31, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Kugelberg, M.; Shafiei, K.; Ploeg, I.; Zetterström, C. Intraocular lens as a drug delivery system for dexamethasone. Acta Ophthalmol. 2010, 88, 241–244. [Google Scholar] [CrossRef] [PubMed]
- Kleinmann, G.; Apple, D.J.; Chew, J.; Hunter, B.; Stevens, S.; Larson, S.; Mamalis, N.; Olson, R.J. Hydrophilic acrylic intraocular lens as a drug-delivery system for fourth-generation fluoroquinolones. J. Cataract. Refract. Surg. 2006, 32, 1717–1721. [Google Scholar] [CrossRef] [PubMed]
- Davis, L.T.; Kumar, N.; Nijm, L.M.; Ulanski II, L.J.; Tu, E.Y.; Fiscella, R.G.; Peterson, R.J.; Glickman, R.D. An adaptable HPLC method for the analysis of frequently used antibiotics in ocular samples. J. Chromatogr. B 2010, 878, 2421–2426. [Google Scholar] [CrossRef]
- Matsushima, H.; Mukai, K.; Gotoo, N.; Yoshida, S.; Yoshida, T.; Sawano, M.; Senoo, T.; Obara, Y.; Clark, J.I. The effects of drug delivery via hydrophilic acrylic (hydrogel) intraocular lens systems on the epithelial cells in culture. Ophthalmic Surg. Lasers Imaging 2005, 36, 386–392. [Google Scholar] [PubMed]
- Yañez, F.; Martikainen, L.; Braga, M.E.M.; Alvarez-Lorenzo, C.; Concheiro, A.; Duarte, C.M.M.; Gil, M.H.; de Sousa, H.C. Supercritical fluid-assisted preparation of imprinted contact lenses for drug delivery. Acta Biomater. 2011, 7, 1019–1030. [Google Scholar] [CrossRef] [PubMed]
- Costa, V.P.; Braga, M.E.M.; Guerra, J.P.; Duarte, A.R.C.; Duarte, C.M.M.; Leite, E.O.B.; Gil, M.H.; de Sousa, H.C. Development of therapeutic contact lenses using a supercritical solvent impregnation method. J. Supercrit. Fluids 2010, 52, 306–316. [Google Scholar] [CrossRef]
- Masmoudi, Y.; Azzouk, L.B.; Forzano, O.; Andre, J.M.; Badens, E. Supercritical impregnation of intraocular lenses. J. Supercrit. Fluids 2011, in press. [Google Scholar]
- Manju, S.; Kunnatheeri, S. Layer-by-layer modification of poly(methyl methacrylate) intra ocular lens: Drug delivery applications. Pharm. Dev. Technol. 2010, 15, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wu, L.; Fu, S.; Hou, Y.; Liu, P.; Cui, H.; Liu, J.; Xing, L.; Zhang, X. Polylactide-glycolic acid and rapamycin coating intraocular lens prevent posterior capsular opacification in rabbit eyes. Graef. Arch. Clin. Exp. 2009, 247, 801–807. [Google Scholar] [CrossRef]
- Pot, S.A.; Chandler, H.L.; Colitz, C.M.H.; Bentley, E.; Dubielzig, R.R.; Mosley, T.S.; Reid, T.W.; Murphy, C.J. Selenium functionalized intraocular lenses inhibit posterior capsule opacification in an ex vivo canine lens capsular bag assay. Exp. Eye Res. 2009, 89, 728–734. [Google Scholar] [CrossRef] [PubMed]
- Parsons, C.; McCoy, C.P.; Gorman, S.P.; Jones, D.S.; Bell, S.E.J.; Brady, C.; McGlinchey, M. Anti-infective photodynamic biomaterials for the prevention of intraocular lens-associated infectious endophthalmitis. Biomaterials 2009, 30, 597–602. [Google Scholar] [CrossRef] [PubMed]
- Sinkel, C.; Greiner, A.; Agarwal, S. A polymeric drug depot based on 7-(2’-methacryloyloxyethoxy)-4-methylcoumarin copolymers for photoinduced release of 5-fluorouracil designed for the treatment of secondary cataracts. Macromol. Chem. Phys. 2010, 211, 1857–1867. [Google Scholar] [CrossRef]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
González-Chomón, C.; Concheiro, A.; Alvarez-Lorenzo, C. Drug-Eluting Intraocular Lenses. Materials 2011, 4, 1927-1940. https://doi.org/10.3390/ma4111927
González-Chomón C, Concheiro A, Alvarez-Lorenzo C. Drug-Eluting Intraocular Lenses. Materials. 2011; 4(11):1927-1940. https://doi.org/10.3390/ma4111927
Chicago/Turabian StyleGonzález-Chomón, Clara, Angel Concheiro, and Carmen Alvarez-Lorenzo. 2011. "Drug-Eluting Intraocular Lenses" Materials 4, no. 11: 1927-1940. https://doi.org/10.3390/ma4111927