- Review
Harnessing Poly(9,9-dialkylfluorene-alt-benzothiadiazole) for Circularly Polarized Electroluminescence: Advances and Perspectives
- Mariacecilia Pasini and
- Umberto Giovanella
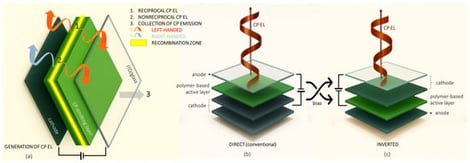
Circularly polarized (CP) organic light-emitting diodes (CP-OLEDs) have attracted considerable attention due to their promising applications in next-generation display systems, optical data transmission, and quantum computing, and their potential roles in medical devices. Achieving efficient and tunable CP emission remains a significant challenge, prompting the development of various strategies that leverage organic semiconductors. Notably, certain classes of materials now consistently deliver CP polarization at levels suitable for technological applications. Among these, conjugated polymers, particularly the copolymer poly(9,9-dialkylfluorene-alt-benzothiadiazole) (PFBT), stand out for their exceptional optoelectronic properties, ease of processing, and adaptability to produce CP emission. PFBT has played diverse roles within CP-OLED devices, enabling innovative architectural solutions. This review explores principal strategies for integrating PFBT into CP-OLED architectures, drawing upon findings from the recent scientific literature. By consolidating current knowledge and identifying unresolved issues, this work aims to inspire further research into the development of solution-processable, high-performance and tunable CP-OLEDs based on PFBT and conjugated polymers in general.
20 March 2026