1. Introduction
In the last few years, several reports have become available concerning the mutual or parallel utilization of silver nanoparticles (SN) and chlorhexidine (Cx) as a treatment/prophylactic strategy for specific infectious diseases. Both agents are widely studied and described in the literature as possessing distinct antimicrobial properties, which made a lot of scientists curious about a possible synergism, additive effects, or potentiation of their activities. Some interesting findings were reported by Zhou et al., who prepared a composite of poly(L-lactide) microcapsules of chlorhexidine with superficially adsorbed silver nanoparticles and found better antibacterial properties against
E. coli and
S. aureus with less usage of silver, compared to silver nanoparticles carrying non-drug loaded microcapsules [
1]. Elsewhere, Myronov et al. reported that adding silver nanoparticles to chlorhexidine treatment improved infected wound healing through the acceleration of bacteria elimination (
E. coli,
P. aeruginosa, and
S. aureus) and M2 macrophage polarization [
2]. Pernakov et al. established the accelerated wound healing effect of silver nano-architectures and chlorhexidine when combined, compared with single treatments with both agents, but did not observe any synergism in the antibacterial activity against
E. coli and
P. aeruginosa [
3]. Other authors reported synergism or the potentiation of SN and Cx against
Candida spp. [
4,
5,
6],
Enterococcus faecalis [
5,
7],
Klebsiella pneumonia [
5],
Pseudomonas aeruginosa [
6,
8],
Staphylococcus epidermidis [
6,
8], and
Streptococcus mutans [
6,
9]. To the best of our knowledge, yet to be cursorily addressed are the supramolecular interactions that may occur upon the addition of chlorhexidine to a nano-silver colloid, along with defining the nature of the resultant effects. It would be speculative to seek any controversy or agreement in all the findings above since the surface functionality of nano-silver is indeed the crucial factor for accessing their potential physicochemical and therapeutic properties. It depends highly on the preparation technique and the reducer that is used [
10,
11,
12,
13].
Today, due to their pronounced antibacterial, antifungal, antiviral, and anticancer properties, SN are considered highly potent multifunctional units with a broad application range (e.g., in nanomedicine, dentistry, ecology, the textile industry, cosmetics, etc.) [
14,
15,
16,
17]. Their antimicrobial activity is primarily owing to the silver ions (Ag
+) released from the critically increased silver surface and subsequent oxidation or bonding to essential for the microorganisms’ vitality, adhesion, and replication molecular factors [
18,
19,
20]. Ever since “green” synthetic methods were introduced, it has become a trend and a method of choice to obtain nano-silver colloids by reducing silver ions in dilute solutions of soluble silver salts with non-toxic reducers of natural origin—plant extracts, bacteria, yeast, and fungi [
11,
21]. Being a rich source of primary and secondary metabolic products, most of them serve not only as reducers but also as capping (stabilizing) agents [
11,
22]. However, for the same reason, the various biological methods lead to highly differentiating functionality (shape, size, charge, redox potential, and superficial organic load or “cap”) of the silver nanoparticles, affecting the efficiency of Ag
+ release. Thus, depending on the reduction process and the surface properties, SN possess varying antimicrobial strength, toxicity, and, last but not least, the ability to conjugate with other substrates [
11,
23].
Chlorhexidine (Cx) is a broad-spectrum topical antiseptic with a biguanide structure and the behavior of a cationic surfactant in neutral and alkaline media. The antifungal properties of the compound are slightly less pronounced than its antibacterial properties [
24,
25]. Cx was considered a suitable substrate for conjugation to SN for several reasons: (1) when ionized, the molecule produces quaternary ammonium functional groups, which can easily contribute to the electrostatic adsorption of Cx on a negatively charged substrate [
26,
27] (such as green-synthesized nano-silver in most cases [
28,
29]); (2) the presence of multiple basic N atoms sets a prerequisite for coordination with the silver nuclei [
30], whereas the acidic NH are expected to engage in hydrogen bonding with the available phenolic groups in the organic cap [
31,
32,
33,
34]. It is as yet unclear how these potential interactions may influence the therapeutic activity of SN and chlorhexidine itself, and whether they allow/determine potentiation or even synergistic action.
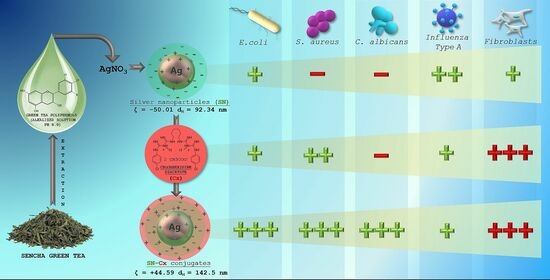
Our pilot study on “green” SN synthesis with green tea extracts confirmed the better stability and purity of nano-silver colloids when catechin-rich phenolic extract was used for the reduction of silver ions rather than using total aqueous green tea extract [
35]. Furthermore, we obtained a clue regarding the synthesis’ optimal parameters and the colloidal stability. In the present study, we attempted to explore the production variables in more detail and establish the physicochemical nature of the resultant nano-silver colloids with a view to their conjunction with chlorhexidine. We hypothesized that in a conjugated form, Cx and SN may present synergistic activity against common pathogens, such as the Influenza virus,
Staphylococcus aureus,
Escherichia coli, and
Candida albicans.
3. Results and Discussion
3.1. Silver Nanoparticle Synthesis, Optimization, and Conjunction
All test formulations resulted in the formation of nano-silver, recognized by the immediate change in color to the yellow-reddish-brown scale and the surface plasmon resonance (SPR) peak, manifesting in the range of 413–430 nm in all UV-Vis spectra. According to the spectral data (particularly, the absorption at the wavelength of the SPR extremum), the darkness of the color was found to correspond well with the concentration of the colloids. The highest nanosilver concentration with no visible signs of turbidity and/or sedimentation was established with sample 2C8R4.5, obtained in 2 mM of silver nitrate solution with green tea polyphenols (C8) at 4.5 mg/mL.
Table 1 summarizes the information for all test compositions and the obtained UV-Vis spectral data; in
Figure 1, the visual appearance of the samples over 7 days can be followed.
The colloidal stability of samples 2C8R [1.5–4.5] was confirmed via zeta potential analysis. With the zeta potential of sample 2C8R4.5 being the highest among all the samples according to absolute value [−50.01 mV], the initial observation was confirmed for it to be the composition of choice for further analyses and conjunction with Cx. Through evaporation to dryness, the nano-silver content (including the organic load) in the sample in question was established to be 1.005 ± 0.012 mg/mL.
As expected, upon the successful sorption of chlorhexidine diacetate (Cx) onto the silver nanoparticles’ surface, the colloids acquired a positive zeta potential; favorably for the purposes of this study, the latter was found to reach maximal absolute values in samples obtained with the highest investigated drug load, i.e., 1.5 mL of Cx 3.75 mg/mL solution. At such a dilution of the colloid, the nano-silver concentration drops to 0.7 mg/mL.
Table 2 summarizes the data from the zeta potential measurements.
The electron microscopy and the dynamic light scattering results confirmed a significant enlargement of the conjugated particles’ diameter and hydrodynamic diameter (d
H)m compared to the SN alone. These observations are in accordance with the shift in the SPR peak of the conjugates to higher values (SN λ
max = 421 nm < SN-Cx λ
max = 439 nm). Macroscopically, the conjugated nanosuspension generally retained the visual appearance of the unconjugated sample but presented slightly more pronounced opalescence. Negligible amounts of sediment were repeatedly observed after a few days of storage but the sediment was easily resuspended upon shaking.
Figure 2A,B shows the UV spectra, TEM micrographs, zeta potential distribution, and size analysis by the hydrodynamic diameter of SN sample 2C8R4.5, and the optimal SN-Cx conjugated form, namely, 2C8R4.5Cx+++.
3.2. FTIR Spectroscopy
In the infrared spectrum of the phenolic extract (
Figure 3), multiple bands of relatively high intensity, which are characteristic of the four catechin diastereoisomers, were observed at 1627, 1625, 1612, 1520, 1240, 1202, 1145, 1096, and 1037 cm
−1 [
39]. Bands falling in the range of 1400 to 1650 cm
−1 are considered to be characteristic of the catechin’s aromatic (benzenoid) residues [
40]. In the same spectrum, the remaining high-intensity bands (with maxima at 1037, 1145, 1240, and 1285 cm
−1) are associated with the inherent phenolic and alcohol groups of C–O stretching and δ(O–H) vibrations, and those registered at the higher wavelengths (at 766 and 823 cm
−1) are associated with the out-of-plane C
ar–H modes.
In the spectrum of the SN sample, the presence was observed of all the absorption bands mentioned above, which are characteristic of the phenolic extract used. Regarding the reason for the preserved IR catechin imprint in the spectrum of the SN sample, we would suggest the tendency of catechins to sorb on the surface of metal nanoparticles that were formed with their participation [
29]. It should be noted that in the spectrum of the sample in question, the appearance of bands characteristic of the asymmetric and symmetric stretching vibrations of NO
2 groups (at 1558 and 1384 cm
−1) was also registered. As an additional but debatable indication of the presence of the latter, we would point to the presence of the low-intensity band at 833 cm
−1, which represents a band characteristic of C
ar–NO
2 oscillations. The reason for the existence of this functional group will not be discussed here, but it is assumed that in the course of the imposed redox process, the nitration of the organic substrate also takes place.
Moreover, as shown when comparing the spectra of the Cx and SN-Cx samples, the presence of the used anti-infective drug in the composition of the target nanoparticles was established.
3.3. X-ray Diffraction (XRD)
The X-ray diffractograms of the silver nanoparticles (SN), their conjugates with chlorhexidine (SN-Cx), and chlorhexidine acetate (Cx) are shown in
Figure 4. Recognizable in the conjugated and non-conjugated nanoparticles’ spectra is the standard diffraction pattern of elemental silver (Ag), with face-centered cubic symmetry. The latter is distinguished by reflections at 2θ 38°, 44°, 64°, and 77°, corresponding to the (111), (200), (220), and (311) planes [
41,
42,
43,
44]. The broad scattering and the lack of sharp, intense peaks in the SN and SN-Cx spectra reveal the substantial presence of amorphous matter and the small crystallite size [
41,
43]. None of the intrinsic chlorhexidine diffraction peaks (e.g., 2θ 19.8°, 20.4°, and 25.1°) [
45,
46,
47] was evident in the conjugated nano-silver form; this observation confirms the amorphization of the drug upon loading onto the metal delivery platforms. The presence of crystalline silver oxide—Ag
2O, the recognized standard being intense reflections at 28° and 32° of the (110) and (111) planes [
48,
49,
50]—was not detected in the nano samples. However, an extra peak was recorded in the X-ray spectrum of SN at 29°, which might be a result of catechin crystalline segments in the sample [
51], which are spectrally disguised or amorphized in the presence of chlorhexidine.
3.4. Antibacterial and Antifungal Activity
3.4.1. Minimal Inhibitory Concentration (MIC)
The inhibitory activity of SN alone against E. coli was recorded at relatively high concentrations of 175 µg/mL (e.g., dilution 1:4); antimicrobial activity against the other two pathogens—S. aureus and C. albicans—did not manifest at the investigated concentrations. It is worth mentioning that a slight opalescence was observed even within the negative controls with SN, which directed our attention to the MBC/MFC results. Such an opalescence is likely a result of the colloidal instability of the SN samples in the electrolyte-rich fluid growth medium.
When Cx was tested alone, MICs of 35.3 µg/mL (dilution 1:32) against E. coli and 17.7 µg/mL (dilution 1:64) against S. aureus and C. albicans were established. The SN-Cx conjugates exceeded these results by demonstrating MIC against all strains at SN 5.5 µg/mL + Cx 8.8 µg/mL (dilution 1:128). Only vials from the last in-row tested concentrations, corresponding to the 1:256 dilutions, remained turbid. The negative controls here were clear and transparent.
3.4.2. Minimal Bactericidal/Fungicidal Concentration (MBC/MFC)
The MBC/MFC study confirmed the lack of antimicrobial activity of SN alone against
S. aureus and
C. albicans, whereas the established MIC against
E. coli, 175 µg/mL, was also shown to correspond to the MBC. The results from the MBC/MFC tests of the Cx and SN-Cx conjugates are presented in
Figure 5 and are summarized in
Table 3.
According to these results, the conjugates (SN-Cx) demonstrate synergism against
E. coli,
S. aureus, and
C. albicans. Due to the lack of established activity of SN alone against
S. aureus and
C. albicans, the interaction therein could be further defined as potentiation [
52]. Additionally, we have confirmed the observations of other authors that Gram-negative microorganisms are, in general, more susceptible to the presence of silver nanoparticles than Gram-positive bacteria, which is likely due to the thicker cellular wall–peptidoglycan layer of the latter and the consequently restricted penetration of Ag
+ [
53,
54].
3.5. Cytotoxicity, Antiviral, and Virucidal Activity
The results for CC
50, IC
50, SI, and virucidal activity (Δlg) of the SN, Cx, and SN-Cx samples are presented in
Table 4,
Table 5 and
Table 6.
The as-determined CC50 values of SN in MDCK, A549, and BJ cell cultures—56.4, 60.7, and 16.9 μg/mL, respectively—reveal SN as an active agent with relatively low to moderate toxicity. In MDCK cells, the CC50 of Cx alone was 2.6 and was 2.5 μg/mL in the conjugated form of SN-Cx. In A549 cell cultures, the same respective values were 5.3 and 5.8 μg/mL, while in human BJ fibroblasts, they were 2.2 and 2.4 μg/mL. According to these results, Cx and the Cx-bearing conjugated form (SN-Cx) could be defined as agents with high toxicity. SN showed a weak effect on the replication of the A/Aichi/2/68 virus, with a mean IC50 = 43.6 μg/mL and SI = 1.29. In the applied concentrations, the Cx and SN-Cx samples did not affect the replication of the Influenza virus A/Aichi/2/68 (H3N2) in MDCK cell cultures in a dose-response experimental design.
A strong virucidal effect was established at an SN MTC of 35 μg/mL with a duration from 5 to 60 min. The absence of a virucidal effect of Cx and SN-Cx against the investigated Influenza strain A/Aichi/2/68 (H3N2) is probably due to the low working concentrations of 2 μg/mL for Cx and 1.2/2 μg/mL for SN-Cx, respectively, which were applied because of the substrates’ high toxicity.
The distinguished time-dependent virucidal effect of SN was also confirmed against the Influenza strain A/Panama/2007/99 (H3N2), at a working concentration of 350 μg/mL upon contact and following ten-fold dilution prior to titration in the cell monolayer. The virucidal activity of Cx and SN-Cx against the Influenza A/Panama/2007/99 (H3N2) virus was assessed as weak (5 and 15 min) to moderate (30 and 60 min) when higher concentrations (than those in the former experiment with the A/Aichi/2/68 (H3N2) strain) were in contact with the virus, namely, Cx 113.0 μg/mL and SN-Cx 70.0/113.0 μg/mL, respectively. Before titration in the cell monolayer, additional ten-fold dilutions were still performed so that non-toxic concentrations were applied. However, the residual toxicity of the Cx and SN-Cx preparations in the greater dilutions and respective interference with the CPE could explain the established lower virucidal activity compared to that in SN alone.
4. Conclusions
Chlorhexidine diacetate was successfully adsorbed onto the “green”-synthesized silver nanoparticles’ negatively charged surface. The thus-obtained colloidal solution retained satisfactory physical stability for the duration of the survey. The resultant conjugates presented enhanced antimicrobial activity, judging by the results for common pathogens such as Escherichia coli, Staphylococcus aureus, and Candida albicans. The addition of nano-silver to chlorhexidine solution significantly decreased the minimal bactericidal/fungicidal concentrations for all tested bacteria and fungi. Moreover, the fungicidal activity of chlorhexidine alone against C. albicans was not established at the investigated concentrations, whereas the unconjugated silver nanoparticles were not shown to inhibit or kill S. aureus and C. albicans independently. As expected, the cationic surfactant chlorhexidine exhibited high toxicity on the Madine–Darby canine kidney line, human fibroblasts, and lung carcinoma cells; this was despite the much lower applied active concentrations (0.00004–0.11%) compared to those in commercial topical antiseptic products (0.02–0.2%). The effect did not attenuate upon the conjugation of the drug to nano-silver vehicles, which limited the active concentration range to be tested for antiviral and virucidal activity on Influenza type A. The unconjugated silver nanoparticles, on the other hand, demonstrated good tolerability and low toxicity. Therefore, a relatively high concentration of 35 μg/mL was applied and showed a weak antiviral effect but distinct virucidal properties. Regardless of the latter ascertainments, the chlorhexidine–silver nanoparticle conjugate exhibited a pronounced synergistic action and an extended spectrum of antimicrobial activity. Therefore, it is relevant to test the applicability and efficacy of this promising newly synthesized agent in the form of pharmaceutical dosage for dermal, nasal, oromucosal, and ocular administration. This prospect will require more specific and bio-relevant tolerability tests to be conducted on 3D and organoid cultures.