The Effect of Granulometry of Carbonaceous Materials and Application Rates on the Availability of Soil-Bound Dichlorodiphenyltrichloroethane (DDT) and Its Metabolites
Abstract
:1. Introduction
2. Material and Methods
2.1. Preparation of Sargasso Biochar
2.2. Characterization of the Sequestering Matrices Particle Size
2.3. Characterization of the Sequestering Matrices Porous Surface
2.4. Soil Sampling and Amendment of Soil
2.5. Assessment of DDT and DDT Metabolites’ Environmental Availability
2.6. GC-MS/MS Analysis
2.7. Statistical Analysis
3. Results
3.1. Granulometry Results of Particles Biochar and Activated Carbon Samples
3.2. Improving a Sargasso Biochar-Based Sequestration Strategy
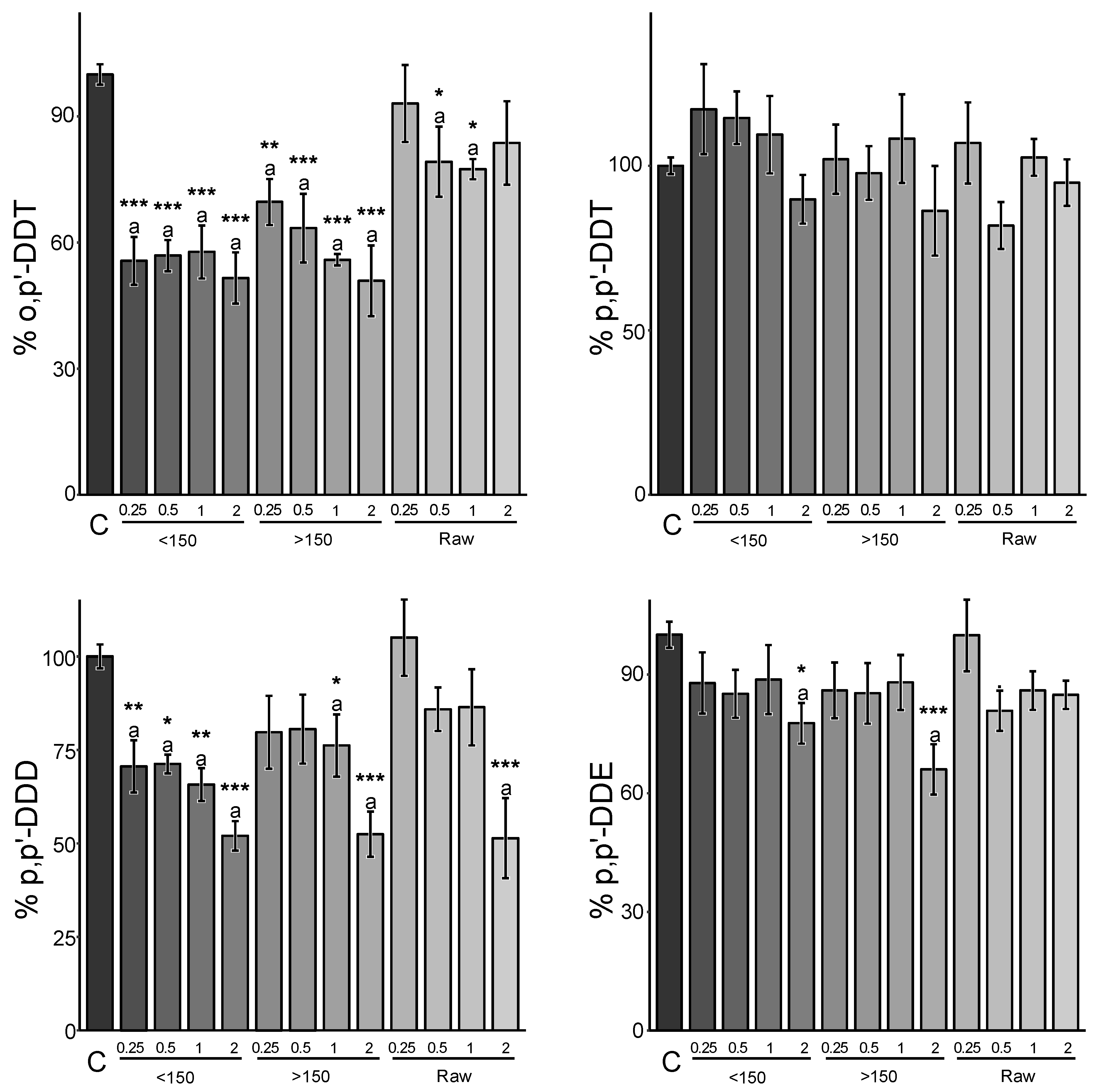
3.3. Amendment Rate Effect
3.4. Granulometry Effect
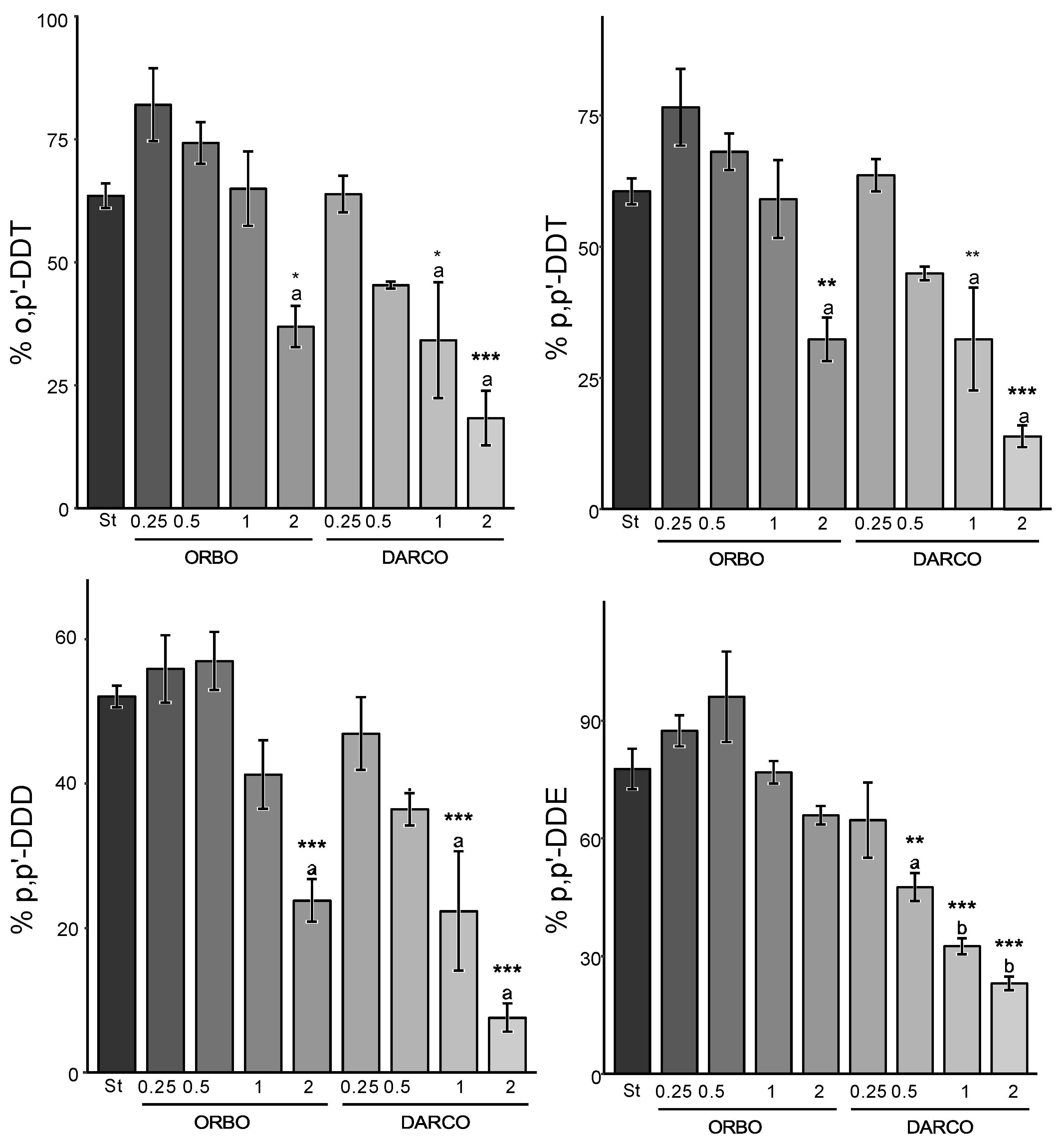
3.5. Comparing Sargasso Biochar to Activated Carbon-Based Sequestration Strategies
- -
- Carbonaceous matrix effect
- -
- Soil effect
- -
- Congener of DDT and DDT metabolites
- -
- Rate of amendment effect
- -
- Expected reduction in environmental availability using such strategies
4. Discussion
4.1. Particle Size and Amendment Rate Impact on DDT and DDT Metabolites Sequestration
4.2. Implement Such a Strategy on Contaminated Areas
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- ATSDR. Toxicological Profile for Mirex and Chlordecone; ATSDR: Atlanta, GA, USA, 2020. Available online: https://stacks.cdc.gov/view/cdc/78737 (accessed on 26 September 2023).
- ATSDR. Toxicological Profile for DDT, DDE, and DDD; ATSDR: Atlanta, GA, USA, 2022. Available online: https://www.atsdr.cdc.gov/toxprofiles/tp35.pdf (accessed on 26 September 2023).
- ATSDR. Toxicological Profile for Hexachlorocyclohexane (HCH): Draft for Public Comment; ATSDR: Atlanta, GA, USA, 2023. Available online: https://www.atsdr.cdc.gov/Toxprofiles/tp43.pdf (accessed on 26 September 2023).
- Sharma, A.; Kumar, V.; Shahzad, B.; Tanveer, M.; Sidhu, G.P.S.; Handa, N.; Kohli, S.K.; Yadav, P.; Bali, A.S.; Parihar, R.D.; et al. Worldwide Pesticide Usage and Its Impacts on Ecosystem. SN Appl. Sci. 2019, 1, 1446. [Google Scholar] [CrossRef]
- Turusov, V.; Rakitsky, V.; Tomatis, L. Dichlorodiphenyltrichloroethane (DDT): Ubiquity, Persistence, and Risks. Environ Health Perspect 2002, 110, 125–128. [Google Scholar] [CrossRef] [PubMed]
- Amutova, F.; Delannoy, M.; Baubekova, A.; Konuspayeva, G.; Jurjanz, S. Transfer of Persistent Organic Pollutants in Food of Animal Origin—Meta-Analysis of Published Data. Chemosphere 2021, 262, 128351. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Gai, N.; Tang, H.; Chen, S.; Chen, D.; Lu, G.; Yang, Y. Organochlorine Pesticides and Polychlorinated Biphenyls in Grass, Yak Muscle, Liver, and Milk in Ruoergai High Altitude Prairie, the Eastern Edge of Qinghai-Tibet Plateau. Sci. Total Environ. 2014, 491–492, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Tao, S.; Liu, W.X.; Li, X.Q.; Zhou, D.X.; Li, X.; Yang, Y.F.; Yue, D.P.; Coveney, R.M. Organochlorine Pesticide Residuals in Chickens and Eggs at a Poultry Farm in Beijing, China. Environ. Pollut. 2009, 157, 497–502. [Google Scholar] [CrossRef]
- Shen, B.; Wu, J.; Zhan, S.; Jin, M. Residues of Organochlorine Pesticides (OCPs) and Polycyclic Aromatic Hydrocarbons (PAHs) in Waters of the Ili-Balkhash Basin, Arid Central Asia: Concentrations and Risk Assessment. Chemosphere 2021, 273, 129705. [Google Scholar] [CrossRef]
- El Wanny, N.; Le Roux, Y.; Fournier, A.; Baroudi, M.; Woignier, T.; Feidt, C.; Delannoy, M. Organochlorine POPs Sequestration Strategy by Carbonaceous Amendments of Contaminated Soils: Toward a Better Understanding of the Transfer Reduction to Laying Hens. J. Hazard. Mater. 2022, 434, 128871. [Google Scholar] [CrossRef]
- Hoogenboom, L.A.P.; Kan, C.A.; Zeilmaker, M.J.; Van Eijkeren, J.; Traag, W.A. Carry-over of Dioxins and PCBs from Feed and Soil to Eggs at Low Contamination Levels—Influence of Mycotoxin Binders on the Carry-over from Feed to Eggs. Food Addit. Contam. 2006, 23, 518–527. [Google Scholar] [CrossRef]
- Hoogenboom, R.L.A.P.; ten Dam, G.; van Bruggen, M.; Jeurissen, S.M.F.; van Leeuwen, S.P.J.; Theelen, R.M.C.; Zeilmaker, M.J. Polychlorinated Dibenzo-p-Dioxins and Dibenzofurans (PCDD/Fs) and Biphenyls (PCBs) in Home-Produced Eggs. Chemosphere 2016, 150, 311–319. [Google Scholar] [CrossRef]
- Hoogenboom, L.A.P.; Kan, C.A.; Bovee, T.F.H.; Van Der Weg, G.; Onstenk, C.; Traag, W.A. Residues of Dioxins and PCBs in Fat of Growing Pigs and Broilers Fed Contaminated Feed. Chemosphere 2004, 57, 35–42. [Google Scholar] [CrossRef]
- Costera, A.; Feidt, C.; Dziurla, M.A.; Monteau, F.; Le Bizec, B.; Rychen, G. Bioavailability of Polycyclic Aromatic Hydrocarbons (PAHs) from Soil and Hay Matrices in Lactating Goats. J. Agric. Food Chem. 2009, 57, 5352–5357. [Google Scholar] [CrossRef] [PubMed]
- Bartle, K.D. CHAPTER 3—Analysis and Occurrence of Polycyclic Aromatic Hydrocarbons in Food. In Food Contaminants: Sources and Surveillance; Creaser, C., Purchase, R., Eds.; Woodhead Publishing Series in Food Science, Technology and Nutrition; Woodhead Publishing: Sawston, UK, 2004; pp. 41–60. ISBN 978-1-85573-784-6. [Google Scholar]
- Purnomo, A.S.; Mori, T.; Kamei, I.; Kondo, R. Basic Studies and Applications on Bioremediation of DDT: A Review. Int. Biodeterior. Biodegrad. 2011, 65, 921–930. [Google Scholar] [CrossRef]
- Zeeb, B.A.; Amphlett, J.S.; Rutter, A.; Reimer, K.J. Potential for Phytoremediation of Polychlorinated Biphenyl-(PCB)-Contaminated Soil. Int. J. Phytoremediation 2006, 8, 199–221. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, S.D.; Anderson, T.A.; Paul Schwab, A.; Hsu, F.C. Phytoremediation of Soils Contaminated with Organic Pollutants. Adv. Agron. 1996, 56, 55–114. [Google Scholar] [CrossRef]
- Delannoy, M.; Yehya, S.; Techer, D.; Razafitianamaharavo, A.; Richard, A.; Caria, G.; Baroudi, M.; Montargès-Pelletier, E.; Rychen, G.; Feidt, C. Amendment of Soil by Biochars and Activated Carbons to Reduce Chlordecone Bioavailability in Piglets. Chemosphere 2018, 210, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Yehya, S.; Delannoy, M.; Fournier, A.; Baroudi, M.; Rychen, G.; Feidt, C. Activated Carbon, a Useful Medium to Bind Chlordecone in Soil and Limit Its Transfer to Growing Goat Kids. PLoS ONE 2017, 12, e0179548. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.; Rajapaksha, A.U.; Lim, J.E.; Zhang, M.; Bolan, N.; Mohan, D.; Vithanage, M.; Lee, S.S.; Ok, Y.S. Biochar as a Sorbent for Contaminant Management in Soil and Water: A Review. Chemosphere 2014, 99, 19–33. [Google Scholar] [CrossRef]
- Amutova, F.; Jurjanz, S.; Akhmetsadykov, N.; Kazankapova, M.; Razafitianamaharavo, A.; Renard, A.; Nurseitova, M.; Konuspayeva, G.; Delannoy, M. Adsorption of Organochlorinated Pesticides: Adsorption Kinetic and Adsorption Isotherm Study. Results Eng. 2023, 17, 100823. [Google Scholar] [CrossRef]
- Li, G.; Khan, S.; Ibrahim, M.; Sun, T.-R.; Tang, J.-F.; Cotner, J.B.; Xu, Y.-Y. Biochars Induced Modification of Dissolved Organic Matter (DOM) in Soil and Its Impact on Mobility and Bioaccumulation of Arsenic and Cadmium. J. Hazard. Mater. 2018, 348, 100–108. [Google Scholar] [CrossRef]
- Zheng, R.; Li, C.; Sun, G.; Xie, Z.; Chen, J.; Wu, J.; Wang, Q. The Influence of Particle Size and Feedstock of Biochar on the Accumulation of Cd, Zn, Pb, and As by Brassica Chinensis L. Environ. Sci. Pollut. Res. 2017, 24, 22340–22352. [Google Scholar] [CrossRef]
- Ranguin, R.; Delannoy, M.; Yacou, C.; Jean-Marius, C.; Feidt, C.; Rychen, G.; Gaspard, S. Biochar and Activated Carbons Preparation from Invasive Algae Sargassum Spp. for Chlordecone Availability Reduction in Contaminated Soils. J. Environ. Chem. Eng. 2021, 9, 105280. [Google Scholar] [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of Gases in Multimolecular Layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Lippens, B.C.; de Boer, J.H. Studies on Pore Systems in Catalysts: V. The t Method. J. Catal. 1965, 4, 319–323. [Google Scholar] [CrossRef]
- FAO World Reference Base for Soil Resources 2014—International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; World Soil Resources Reports; Update 2015; FAO Publications: Rome, Italy, 2015; Volume 201, ISBN 978-92-5-108369-7.
- Fazeli Sangani, M.; Abrishamkesh, S.; Owens, G. Physicochemical Characteristics of Biochars Can Be Beneficially Manipulated Using Post-Pyrolyzed Particle Size Modification. Bioresour. Technol. 2020, 306, 123157. [Google Scholar] [CrossRef] [PubMed]
- He, P.; Liu, Y.; Shao, L.; Zhang, H.; Lü, F. Particle Size Dependence of the Physicochemical Properties of Biochar. Chemosphere 2018, 212, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Cao, J.; Li, Y.; Howard, A.; Yu, K. Effect of Pyrolysis Temperature on Characteristics of Biochars Derived from Different Feedstocks: A Case Study on Ammonium Adsorption Capacity. Waste Manag. 2019, 87, 652–660. [Google Scholar] [CrossRef] [PubMed]
- Stephan, P.; Le Roux, Y.; Gaspard, S.; Michaux, F.; Feidt, C.; Soligot, C.; Rychen; Delannoy, M. Effects of Particle Size and Amendment Rates of Sargassum Biochar on Chlordecone Sequestration in West Indian Soils. Environ. Sci. Pollut. Res. 2023, 30, 5873–5880. [Google Scholar] [CrossRef]
- Ahn, S.; Werner, D.; Karapanagioti, H.K.; McGlothlin, D.R.; Zare, R.N.; Luthy, R.G. Phenanthrene and Pyrene Sorption and Intraparticle Diffusion in Polyoxymethylene, Coke, and Activated Carbon. Environ. Sci. Technol. 2005, 39, 6516–6526. [Google Scholar] [CrossRef]
- He, Z.; Cao, H.; Liang, J.; Hu, Q.; Zhang, Y.; Nan, X.; Li, Z. Effects of Biochar Particle Size on Sorption and Desorption Behavior of NH4+-N. Ind. Crops Prod. 2022, 189, 115837. [Google Scholar] [CrossRef]
- Zimmerman, J.R.; Werner, D.; Ghosh, U.; Millward, R.N.; Bridges, T.S.; Luthy, R.G. Effects of Dose and Particle Size on Activated Carbon Treatment to Sequester Polychlorinated Biphenyls and Polycyclic Aromatic Hydrocarbons in Marine Sediments. Environ. Toxicol. Chem. 2005, 24, 1594–1601. [Google Scholar] [CrossRef]
- Tomaszewski, J.E.; Werner, D.; Luthy, R.G. Activated Carbon Amendment as a Treatment for Residual Ddt in Sediment from a Superfund Site in San Francisco Bay, Richmond, California, USA. Environ. Toxicol. Chem. 2007, 26, 2143–2150. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-S.; Kim, K.-R.; Kim, H.-J.; Yoon, J.-H.; Yang, J.E.; Ok, Y.S.; Owens, G.; Kim, K.-H. Effect of Biochar on Heavy Metal Immobilization and Uptake by Lettuce (Lactuca Sativa L.) in Agricultural Soil. Environ Earth Sci 2015, 74, 1249–1259. [Google Scholar] [CrossRef]
- Beesley, L.; Moreno-Jiménez, E.; Gomez-Eyles, J.L.; Harris, E.; Robinson, B.; Sizmur, T. A Review of Biochars’ Potential Role in the Remediation, Revegetation and Restoration of Contaminated Soils. Environ. Pollut. 2011, 159, 3269–3282. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Song, W.; Tian, J. Biochar-Facilitated Soil Remediation: Mechanisms and Efficacy Variations. Front. Environ. Sci. 2020, 8. [Google Scholar] [CrossRef]
- Spokas, K.A. Review of the Stability of Biochar in Soils: Predictability of O:C Molar Ratios. Carbon Manag. 2010, 1, 289–303. [Google Scholar] [CrossRef]
- Fu, Q.; Zhao, H.; Li, T.; Hou, R.; Liu, D.; Ji, Y.; Zhou, Z.; Yang, L. Effects of Biochar Addition on Soil Hydraulic Properties before and after Freezing-Thawing. CATENA 2019, 176, 112–124. [Google Scholar] [CrossRef]
- Liu, P.; Zhu, M.; Zhang, Z.; Leong, Y.-K.; Zhang, Y.; Zhang, D. Rheological Behaviour and Stability Characteristics of Biochar-Water Slurry Fuels: Effect of Biochar Particle Size and Size Distribution. Fuel Process. Technol. 2017, 156, 27–32. [Google Scholar] [CrossRef]
- Yu, Z.; Ling, L.; Singh, B.P.; Luo, Y.; Xu, J. Gain in Carbon: Deciphering the Abiotic and Biotic Mechanisms of Biochar-Induced Negative Priming Effects in Contrasting Soils. Sci. Total Environ. 2020, 746, 141057. [Google Scholar] [CrossRef]
- Fungo, B.; Lehmann, J.; Kalbitz, K.; Thionģo, M.; Okeyo, I.; Tenywa, M.; Neufeldt, H. Aggregate Size Distribution in a Biochar-Amended Tropical Ultisol under Conventional Hand-Hoe Tillage. Soil Tillage Res. 2017, 165, 190–197. [Google Scholar] [CrossRef]



| Feedstock | Pyrolysis | Activation | Grinding–Sieving | Textural Parameters | ||
|---|---|---|---|---|---|---|
| BET Surface Area (m2·g−1) | VMicropores (D-R) (cm3·g−1) | |||||
| Sarg | Sargassum spp. | 700 °C for 3 h | None | 582 | 0.15 | |
| Sarg< | Sargassum spp. | 700 °C for 3 h | 2 min grinding and sieved < 150 µm | 497.6 | 0.12 | |
| Sarg> | Sargassum spp. | 700 °C for 3 h | 2 min grinding fraction > 150 µm | 567.0 | 0.15 | |
| ORBOTM | Coconut | N/A | N/A | 1127 | 0.34 | |
| DARCO© | Lignite | N/A | Physical (water) | 793.8 | 0.17 | |
| Kyzylkairat | Beskainar | |
|---|---|---|
| DDD-4,4′ (p,p’-DDD) | 852 | 8.47 |
| DDE-4,4′ (p,p’-DDE) | 6035 | 276 |
| DDT-2,4′ (o,p’-DDT) | 906 | 48.3 |
| DDT-4,4′ (p,p’-DDT) | 2277 | 10.6 |
| Congener | Retention Time (min) | Mass | Product Mass | Collision Energy (eV) | Quantification (Q) or Qualification (-) |
|---|---|---|---|---|---|
| p,p’-DDE | 27.01 | 246 | 176.1 | 28 | Q |
| 27.01 | 317.8 | 246 | 20 | ||
| 27.01 | 317.8 | 248 | 18 | ||
| o,p’-DDT | 29.66 | 235 | 165.1 | 22 | Q |
| 29.66 | 235 | 199.5 | 10 | ||
| 29.66 | 236.8 | 165 | 22 | ||
| p,p’-DDD | 29.73 | 235 | 165.1 | 20 | Q |
| 29.73 | 235 | 199 | 14 | ||
| 29.73 | 236.8 | 165 | 20 | ||
| p,p’-DDT | 30.45 | 235 | 165.1 | 22 | Q |
| 30.45 | 235 | 199.5 | 10 | ||
| 30.45 | 236.8 | 165 | 22 | ||
| 13C o,p’-DDT | 29.66 | 247 | 177.1 | 22 | Q |
| 29.66 | 177.1 | 176.1 | 22 | ||
| 29.66 | 249 | 177.1 | 20 |
| Amendment Material | Amendment Rate | 90th Confidence Interval | Reduction Factor | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| p,p’-DDD | p,p’-DDE | o,p’-DDT | p,p’-DDT | p,p’-DDD | p,p’-DDE | o,p’-DDT | p,p’-DDT | |||||||
| Sargasso biochar | <150 | 0.25% | 108% | [89, 128] | 84% | [80, 88] | 92% | [84, 100] | 98% | [85, 112] | - | 12% | - | - |
| 0.5% | 92% | [72, 112] | 81% | [68, 95] | 91% | [75, 106] | 93% | [73, 113] | - | 5% | - | - | ||
| 1% | 91% | [66, 116] | 65% | [52, 79] | 81% | [68, 94] | 88% | [63, 112] | - | 21% | 6% | - | ||
| 2% | 52% | [49, 56] | 42% | [34, 51] | 64% | [58, 69] | 61% | [55, 66] | 44% | 49% | 31% | 34% | ||
| >150 | 0.25% | 105% | [75, 135] | 88% | [75, 101] | 92% | [80, 103] | 97% | [86, 108] | - | - | - | - | |
| 0.5% | 99% | [80, 117] | 86% | [76, 97] | 96% | [85, 106] | 93% | [82, 105] | - | 3% | - | - | ||
| 1% | 86% | [69, 103] | 72% | [58, 85] | 86% | [67, 105] | 87% | [71, 103] | - | 15% | - | - | ||
| 2% | 68% | [55, 80] | 55% | [47, 63] | 75% | [65, 85] | 75% | [67, 84] | 20% | 37% | 15% | 16% | ||
| All | 0.25% | 93% | [69, 116] | 89% | [73, 106] | 89% | [74, 105] | 87% | [70, 103] | - | - | - | - | |
| 0.5% | 63% | [46, 80] | 76% | [58, 94] | 78% | [62, 95] | 78% | [62, 93] | 20% | 6% | 5% | 7% | ||
| 1% | 71% | [62, 80] | 76% | [70, 82] | 80% | [73, 87] | 80% | [75, 86] | 20% | 18% | 13% | 14% | ||
| 2% | 67% | [56, 79] | 63% | [59, 67] | 73% | [65, 81] | 74% | [66, 83] | 21% | 33% | 19% | 17% | ||
| ORBO | 0.25% | 56% | [45, 67] | 66% | [49, 84] | 82% | [65, 99] | 77% | [59, 94] | 33% | 16% | 1% | 6% | |
| 0.5% | 57% | [47, 66] | 51% | [41, 62] | 74% | [64, 84] | 68% | [60, 76] | 34% | 38% | 16% | 24% | ||
| 1% | 41% | [30, 52] | 36% | [19, 53] | 65% | [47, 83] | 59% | [42, 76] | 48% | 47% | 17% | 24% | ||
| 2% | 24% | [17, 31] | 19% | [13, 25] | 37% | [27, 47] | 32% | [22, 42] | 69% | 75% | 53% | 58% | ||
| DARCO | 0.25% | 47% | [35, 59] | 56% | [46, 66] | 64% | [55, 73] | 64% | [56, 71] | 41% | 34% | 27% | 29% | |
| 0.5% | 36% | [31, 42] | 38% | [36, 39] | 45% | [44, 47] | 45% | [42, 48] | 58% | 61% | 53% | 52% | ||
| 1% | 22% | [3.0, 42] | 23% | [10, 35] | 34% | [6.5, 62] | 32% | [9.3, 55] | 58% | 65% | 38% | 45% | ||
| 2% | 7.6% | [3.6, 12] | 15% | [5.2, 24] | 18% | [5.3, 31] | 14% | [9.6, 18] | 88% | 76% | 69% | 82% | ||
| Amendment Material | Amendment Rate | 90th Confidence Interval | Reduction Factor | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| p,p’-DDD | p,p’-DDE | o,p’-DDT | p,p’-DDT | p,p’-DDD | p,p’-DDE | o,p’-DDT | p,p’-DDT | |||||||
| Sargasso biochar | <150 | 0.25% | 71% | [54, 87] | 88% | [70, 106] | 56% | [42, 69] | 117% | [85, 149] | 13% | - | 31% | - |
| 0.5% | 71% | [65, 77] | 85% | [71, 99] | 57% | [48, 66] | 115% | [91, 138] | 23% | 1% | 34% | - | ||
| 1% | 66% | [55, 76] | 89% | [68, 109] | 58% | [43, 73] | 109% | [82, 137] | 24% | - | 27% | - | ||
| 2% | 52% | [43, 61] | 78% | [66, 90] | 52% | [34, 69] | 90% | [72, 107] | 39% | 10% | 31% | - | ||
| >150 | 0.25% | 80% | [57, 103] | 86% | [69, 103] | 70% | [57, 83] | 102% | [77, 127] | - | - | 17% | - | |
| 0.5% | 81% | [59, 102] | 85% | [67, 103] | 63% | [44, 83] | 98% | [79, 117] | - | - | 17% | - | ||
| 1% | 76% | [57, 96] | 88% | [72, 104] | 56% | [53, 59] | 108% | [77, 140] | 4% | - | 41% | - | ||
| 2% | 53% | [38, 67] | 66% | [51, 81] | 51% | [31, 71] | 86% | [54, 118] | 33% | 19% | 29% | - | ||
| All | 0.25% | 105% | [81, 129] | 100% | [79, 121] | 93% | [72, 115] | 107% | [78, 136] | - | - | - | - | |
| 0.5% | 86% | [72, 100] | 81% | [69, 93] | 79% | [60, 99] | 82% | [65, 99] | 0% | 7% | 1% | 1% | ||
| 1% | 86% | [63, 110] | 86% | [74, 97] | 77% | [72, 83] | 103% | [89, 116] | - | 3% | 17% | - | ||
| 2% | 51% | [20, 83] | 85% | [76, 93] | 84% | [21, 146] | 95% | [78, 112] | 17% | 7% | - | - | ||
| ORBO | 0.25% | 79% | [67, 92] | 87% | [78, 97] | 60% | [51, 70] | 95% | [79, 112] | 8% | 3% | 30% | - | |
| 0.5% | 71% | [49, 94] | 96% | [69, 123] | 79% | [61, 97] | 105% | [79, 132] | 6% | - | 3% | - | ||
| 1% | 59% | [45, 73] | 77% | [68, 85] | 66% | [56, 76] | 89% | [75, 104] | 27% | 15% | 24% | - | ||
| 2% | 45% | [34, 57] | 66% | [60, 71] | 46% | [41, 51] | 64% | [48, 79] | 43% | 29% | 49% | 21% | ||
| DARCO | 0.25% | 60% | [29, 91] | 65% | [42, 87] | 54% | [29, 79] | 67% | [49, 84] | 9% | 13% | 21% | 16% | |
| 0.5% | 39% | [27, 51] | 48% | [39, 56] | 37% | [27, 48] | 63% | [48, 78] | 49% | 44% | 52% | 22% | ||
| 1% | 26% | [22, 30] | 33% | [28, 37] | 28% | [23, 33] | 43% | [35, 52] | 70% | 63% | 67% | 48% | ||
| 2% | 21% | [15, 27] | 23% | [18, 28] | 21% | [15, 27] | 33% | [17, 49] | 73% | 72% | 73% | 51% | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amutova, F.; Turganova, R.; Konuspayeva, G.; Gaspard, S.; Mamirova, A.; Michaux, F.; Hartmeyer, P.; Soligot, C.; Djansugurova, L.; Jurjanz, S.; et al. The Effect of Granulometry of Carbonaceous Materials and Application Rates on the Availability of Soil-Bound Dichlorodiphenyltrichloroethane (DDT) and Its Metabolites. J. Xenobiot. 2024, 14, 267-284. https://doi.org/10.3390/jox14010016
Amutova F, Turganova R, Konuspayeva G, Gaspard S, Mamirova A, Michaux F, Hartmeyer P, Soligot C, Djansugurova L, Jurjanz S, et al. The Effect of Granulometry of Carbonaceous Materials and Application Rates on the Availability of Soil-Bound Dichlorodiphenyltrichloroethane (DDT) and Its Metabolites. Journal of Xenobiotics. 2024; 14(1):267-284. https://doi.org/10.3390/jox14010016
Chicago/Turabian StyleAmutova, Farida, Ronagul Turganova, Gaukhar Konuspayeva, Sarra Gaspard, Aigerim Mamirova, Florentin Michaux, Pamela Hartmeyer, Claire Soligot, Leyla Djansugurova, Stefan Jurjanz, and et al. 2024. "The Effect of Granulometry of Carbonaceous Materials and Application Rates on the Availability of Soil-Bound Dichlorodiphenyltrichloroethane (DDT) and Its Metabolites" Journal of Xenobiotics 14, no. 1: 267-284. https://doi.org/10.3390/jox14010016








