Robotic-Assisted Colonoscopy Platform with a Magnetically-Actuated Soft-Tethered Capsule
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Endoo Robotic System
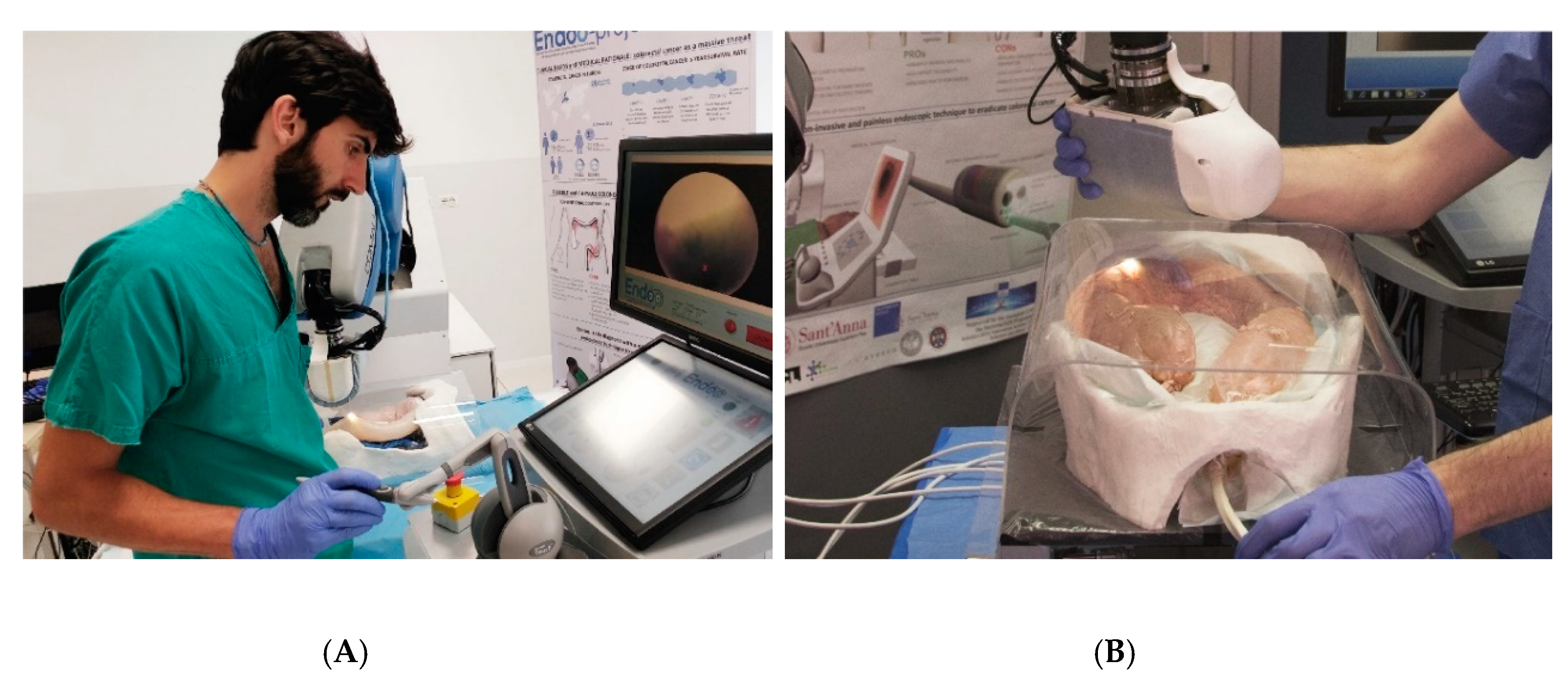
2.2. Experimental Setting
2.2.1. Operating Channel Tests
2.2.2. Target Approach Tests
2.2.3. Lumen Progression Tests
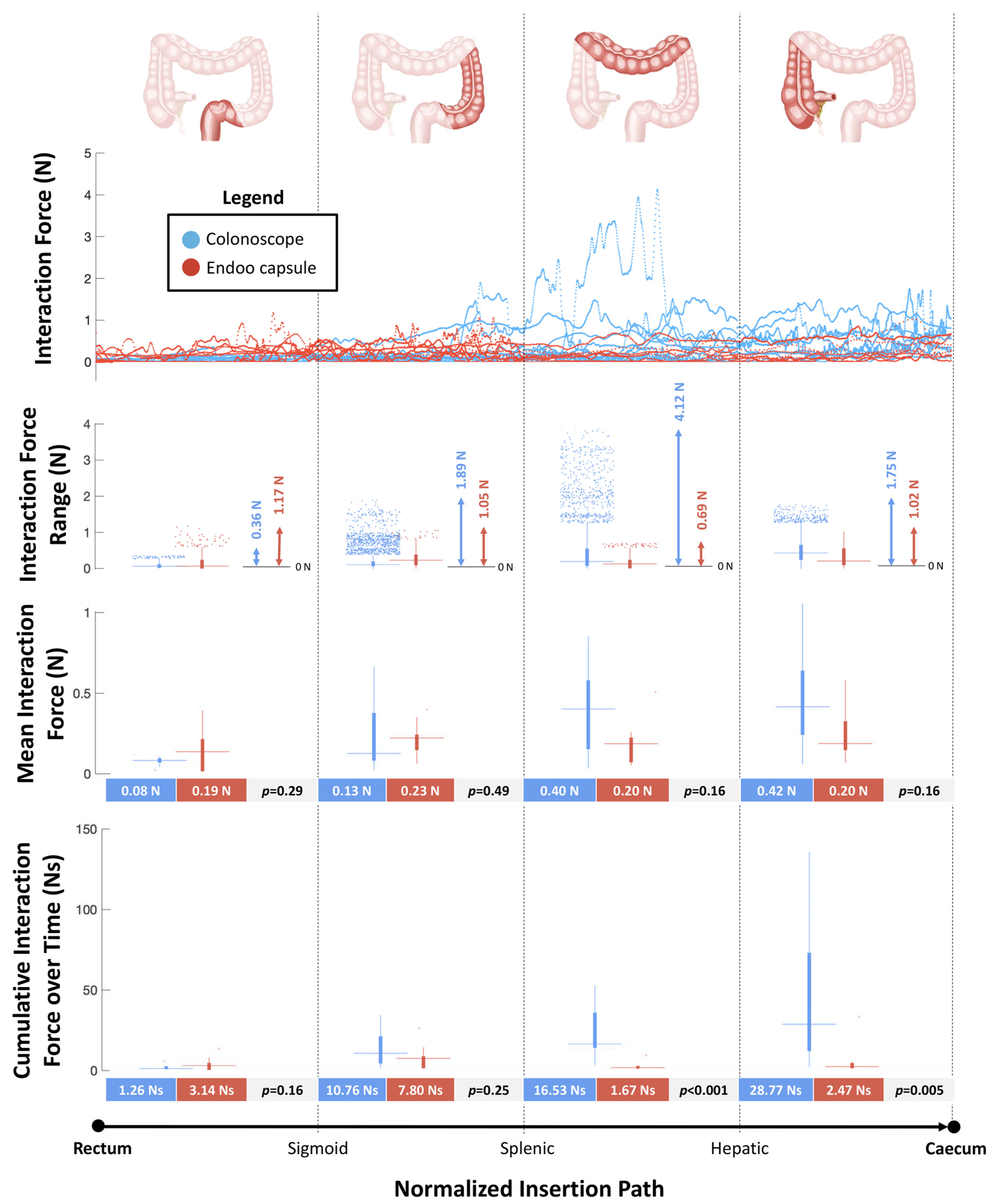
2.2.4. Interaction Force Tests
2.2.5. Polyps Detection Tests
2.2.6. Colonoscopy Simulation Tests
3. Results
3.1. Operating Channel Tests
3.2. Target Approach Tests
3.3. Lumen Progression Tests
3.4. Interaction Forces Tests
3.5. Polyps Detection Tests
3.6. Colonoscopy Simulation Tests
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CRC | Colorectal cancer |
| CC | Conventional Colonoscopy |
| CCE | Colon Capsule Endoscopy |
| CTC | Computed Tomography Colonoscopy |
| DoF | Degrees of Freedom |
| MIS | Minimally Invasive Surgery |
| CMOS | Complementary Metal Oxide Semiconductor |
| LEDs | Light Emitting Diodes |
| SD | Standard Deviation |
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Shergill, A.K.; McQuaid, K.R.; Rempel, D. Ergonomics and GI endoscopy. Gastrointest. Endosc. 2009, 70, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Niederreiter, M.; Niederreiter, L.; Schmiderer, A.; Tilg, H.; Djanani, A. Colorectal cancer screening and prevention—Pros and cons. In Memo-Magazine of European Medical Oncology; Springer: Wien, Austria, 2019; Volume 12, pp. 239–243. [Google Scholar]
- Knudsen, A.B.; Zauber, A.G.; Rutter, C.M.; Naber, S.K.; Doria-Rose, V.P.; Pabiniak, C.; Johanson, C.; Fischer, S.E.; Lansdorp-Vogelaar, I.; Kuntz, K.M. Estimation of benefits, burden, and harms of colorectal cancer screening strategies: Modeling study for the US preventive services Task Force. JAMA 2016, 315, 2595–2609. [Google Scholar] [CrossRef] [PubMed]
- Bynum, S.A.; Davis, J.L.; Green, B.L.; Katz, R.V. Unwillingness to participate in colorectal cancer screening: Examining fears, attitudes, and medical mistrust in an ethnically diverse sample of adults 50 years and older. Am. J. Health Promot. 2012, 26, 295–300. [Google Scholar] [CrossRef] [Green Version]
- Trevisani, L.; Zelante, A.; Sartori, S. Colonoscopy, pain and fears: Is it an indissoluble trinomial? World J. Gastrointest. Endosc. 2014, 6, 227. [Google Scholar] [CrossRef]
- van Rijn, J.C.; Reitsma, J.B.; Stoker, J.; Bossuyt, P.M.; van Deventer, S.J.; Dekker, E. Polyp Miss Rate Determined by Tandem Colonoscopy: A Systematic Review. Am. J. Gastroenterol. 2006, 101, 343–350. [Google Scholar] [CrossRef]
- Yeung, C.; Cheung, J.L.; Sreedhar, B. Emerging next-generation robotic colonoscopy systems towards painless colonoscopy. J. Dig. Dis. 2019, 20, 196–205. [Google Scholar] [CrossRef] [Green Version]
- Gluck, N.; Melhem, A.; Halpern, Z.; Mergener, K.; Santo, E. A novel self-propelled disposable colonoscope is effective for colonoscopy in humans (with video). Gastrointest. Endosc. 2016, 83, 998–1004.e1. [Google Scholar] [CrossRef] [Green Version]
- Shike, M.; Fireman, Z.; Eliakim, R.; Segol, O.; Sloyer, A.; Cohen, L.B.; Goldfarb-Albak, S.; Repici, A. Sightline Colono Sight system for a disposable, power-assisted, non-fiber-optic colonoscopy (with video). Gastrointest. Endosc. 2008, 68, 701–710. [Google Scholar] [CrossRef]
- Lucarini, G.; Ciuti, G.; Mura, M.; Rizzo, R.; Menciassi, A. A new concept for magnetic capsule colonoscopy based on an electromagnetic system regular paper. Int. J. Adv. Robot. Syst. 2015, 12, 70–80. [Google Scholar] [CrossRef] [Green Version]
- Ciuti, G.; Menciassi, A.; Dario, P. Capsule endoscopy: From current achievements to open challenges. IEEE Rev. Biomed. Eng. 2011, 4, 59–72. [Google Scholar]
- Ciuti, G.; Caliò, R.; Camboni, D.; Neri, L.; Bianchi, F.; Arezzo, A.; Koulaouzidis, A.; Schostek, S.; Stoyanov, D.; Oddo, C.M.; et al. Frontiers of robotic endoscopic capsules: A review. J. Micro-Bio. Robot. 2016, 11, 1–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ciuti, G.; Skonieczna-Żydecka, K.; Marlicz, W.; Iacovacci, V.; Liu, H.; Stoyanov, D.; Arezzo, A.; Chiurazzi, M.; Toth, E.; Thorlacius, H.; et al. Frontiers of Robotic Colonoscopy: A Comprehensive Review of Robotic Colonoscopes and Technologies. J. Clin. Med. 2020, 9, 1648. [Google Scholar] [CrossRef]
- Endoo EU Project. Available online: https://cordis.europa.eu/project/id/688592 (accessed on 8 October 2017).
- Ciuti, G.; Valdastri, P.; Menciassi, A.; Dario, P. Robotic magnetic steering and locomotion of capsule endoscope for diagnostic and surgical endoluminal procedures. Robotica 2010, 28, 7–15. [Google Scholar] [CrossRef] [Green Version]
- Ciuti, G.; Donlin, R.; Valdastri, P.; Arezzo, A.; Menciassi, A.; Morino, M.; Dario, P. Robotic versus manual control in magnetic steering of an endoscopic capsule. Endoscopy 2010, 42, 148–152. [Google Scholar] [CrossRef] [Green Version]
- Valdastri, P.; Ciuti, G.; Verbeni, A.; Menciassi, A.; Dario, P.; Arezzo, A.; Morino, M. Magnetic air capsule robotic system: Proof of concept of a novel approach for painless colonoscopy. Surg. Endosc. 2012, 26, 1238–1246. [Google Scholar] [CrossRef]
- Arezzo, A.; Menciassi, A.; Valdastri, P.; Ciuti, G.; Lucarini, G.; Salerno, M.; di Natali, C.; Verra, M.; Dario, P.; Morino, M. Experimental assessment of a novel robotically-driven endoscopic capsule compared to traditional colonoscopy. Dig. Liver Dis. 2013, 45, 657–662. [Google Scholar] [CrossRef]
- Ciuti, G.; Lucarini, G.; Valdastri, P.; Arezzo, A.; Menciassi, A.; Morino, M.; Dario, P. A comparative evaluation of control interfaces for a robotic-aided endoscopic capsule platform. IEEE Trans. Robot. 2012, 28, 534–538. [Google Scholar]
- Matthias, B. ISO/TS 15066-Collaborative Robots Present Status. In Proceedings of the European Robotics Forum 2015, Vienna, Austria, 15 October 2015. [Google Scholar]
- ISO-ISO/TS 15066: 2016-Robots and Robotic Devices—Collaborative Robots. Available online: https://www.iso.org/standard/62996.html (accessed on 30 June 2020).
- Degani, A.; Choset, H.; Wolf, A.; Zenati, M.A. Highly articulated robotic probe for minimally invasive surgery. In Proceedings of the IEEE International Conference on Robotics and Automation, Orlando, FL, USA, 15–19 May 2006; Volume 2006, pp. 4167–4172. [Google Scholar]
- Kwok, K.W.; Tsoi, K.H.; Vitiello, V.; Clark, J.; Chow, G.C.T.; Luk, W.; Yang, G.-Z. Dimensionality reduction in controlling articulated snake robot for endoscopy under dynamic active constraints. IEEE Trans. Robot. 2013, 29, 15–31. [Google Scholar] [CrossRef] [Green Version]
- Ding, J.; Goldman, R.E.; Xu, K.; Allen, P.K.; Fowler, D.L.; Simaan, N. Design and coordination kinematics of an insertable robotic effectors platform for single-port access surgery. IEEE/ASME Trans. Mechatron. 2013, 18, 1612–1624. [Google Scholar] [CrossRef] [Green Version]
- Xu, K.; Zhao, J.; Fu, M. Development of the SJTU Unfoldable Robotic System (SURS) for Single Port Laparoscopy. IEEE/ASME Trans. Mechatron. 2015, 20, 2133–2145. [Google Scholar] [CrossRef]
- Tortora, G.; Valdastri, P.; Susilo, E.; Menciassi, A.; Dario, P.; Rieber, F.; Schurr, M.O. Propeller-based wireless device for active capsular endoscopy in the gastric district. Minim. Invasive Ther. Allied Technol. 2009, 18, 280–290. [Google Scholar] [CrossRef] [PubMed]
- Kósa, G.; Jakab, P.; Székely, G.; Hata, N. MRI driven magnetic microswimmers. Biomed. Microdevices 2012, 14, 165–178. [Google Scholar] [CrossRef] [Green Version]
- Caprara, R.; Obstein, K.L.; Scozzarro, G.; di Natali, C.; Beccani, M.; Morgan, D.R.; Valdastri, P. A platform for gastric cancer screening in low- and middle-income countries. IEEE Trans. Biomed. Eng. 2015, 62, 1324–1332. [Google Scholar] [CrossRef] [PubMed] [Green Version]




| Tests Category | Tests ID | Platforms | Experimental Condition | Sensorized Simulator | Participants (Expert) | Participants (Trainees) | Outcomes |
|---|---|---|---|---|---|---|---|
| Endoo basic key functionalities | Operating Channel | Endoo | Ex-vivo | NO | 10 | 0 | Success rate (%) and qualitative score (1–5) |
| Target Approach | Endoo | Ex-vivo | NO | 10 | 0 | Success rate (%) and qualitative score (1–5) | |
| Lumen Progression | Endoo | Ex-vivo | NO | 10 | 0 | Success rate (%), time (mm:ss), qualitative scores for navigation (1–5), number of capsule losses (#), and success rate for restoring link (%) | |
| Comparative pre-compliance | Interaction Forces | Endoo vs. Colonoscopy | Ex-vivo | YES | 5 | 5 | Interaction force (IF), IF range, mean force (N), and cumulative interaction force overtime (Ns) |
| Polyps Detection | Endoo vs. Colonoscopy | Ex-vivo | NO | 5 | 5 | Total and mean numbers of detected polyps (#) and overall rate (#/total) | |
| Usability and comparative compliance | Colonoscopic Simulation | Endoo vs. Colonoscopy | Ex-vivo | NO | 15 (15 procedures with Endoo—5 procedures with colonoscopes) | 8 (6 procedures with Endoo—8 procedures with colonoscopes) | Procedural times to cecum/rectum (mm:ss), success rates (%), number of capsule losses (#), success rate for restoring link (%) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Verra, M.; Firrincieli, A.; Chiurazzi, M.; Mariani, A.; Lo Secco, G.; Forcignanò, E.; Koulaouzidis, A.; Menciassi, A.; Dario, P.; Ciuti, G.; et al. Robotic-Assisted Colonoscopy Platform with a Magnetically-Actuated Soft-Tethered Capsule. Cancers 2020, 12, 2485. https://doi.org/10.3390/cancers12092485
Verra M, Firrincieli A, Chiurazzi M, Mariani A, Lo Secco G, Forcignanò E, Koulaouzidis A, Menciassi A, Dario P, Ciuti G, et al. Robotic-Assisted Colonoscopy Platform with a Magnetically-Actuated Soft-Tethered Capsule. Cancers. 2020; 12(9):2485. https://doi.org/10.3390/cancers12092485
Chicago/Turabian StyleVerra, Mauro, Andrea Firrincieli, Marcello Chiurazzi, Andrea Mariani, Giacomo Lo Secco, Edoardo Forcignanò, Anastasios Koulaouzidis, Arianna Menciassi, Paolo Dario, Gastone Ciuti, and et al. 2020. "Robotic-Assisted Colonoscopy Platform with a Magnetically-Actuated Soft-Tethered Capsule" Cancers 12, no. 9: 2485. https://doi.org/10.3390/cancers12092485








