1. Introduction
Head and neck squamous cell carcinoma (HNSCC) is the sixth most common cancer worldwide, with 878,348 new cases and 444,347 deaths estimated in 2020 [
1,
2]. Approximately 75% of patients with HNSCC present locally advanced disease at diagnosis, and the standard therapy for most cases involves chemoradiation or the induction of multi-agent chemotherapy, in which cisplatin (CDDP) is usually included [
3,
4]. Alternative treatments, such as carboplatin or cetuximab, were studied in the context of chemoradiation, though their equivalence regarding efficacy has yet to be validated by randomized trials [
5]. Additionally, in patients with treatment-naïve metastatic disease or platinum-sensitive relapse, CDDP-based regimens are commonly used in clinical practice, with benefits in progression-free survival (PFS) and overall survival (OS) [
6,
7]. Nonetheless, CDDP is related to significant adverse events, such as nausea, vomiting, nephrotoxicity, hypersensitivity reactions, and ototoxicity [
3,
8,
9]. Among these, hearing impairment is a current concern since there are, to date, no effective otoprotective measures, resulting in potentially permanent and quality-of-life-limiting damage [
10,
11].
Every year, one in five patients submitted to CDDP-based chemotherapy will suffer severe to profound hearing loss [
10,
12,
13]. Regarding chemoradiation for HNSCC, major losses are described in higher frequencies, with reported pure-tone median threshold increases ranging from 9.52 to 25 decibels (dB) at 4 kilohertz (kHz) and 18.57 to 27.14 dB at 8 kHz [
14,
15]. This event has a major negative impact on the quality of life [
16] and requires essential care regarding dosage management and the duration of therapy [
17]. Despite the association of cumulative CDDP dose, history of noise exposure, and smoking as independent risk factors, the prevalence and intensity of hearing impairment are remarkably heterogeneous among patients with similar characteristics and regimens [
18]. This finding indicates the involvement of unknown risk factors, with single-nucleotide variants (SNVs), on genes encoding proteins related to CDDP metabolism, being potential candidates for this risk [
19,
20].
Numerous proteins act in the mechanisms of CDDP cellular detoxification, as well as in the pathways of damage repair and apoptosis [
21,
22] (
Figure 1).
The detoxification of CDDP occurs mainly through its conjugation with glutathione, encoded by the Mu1 (
GSTM1), Theta1 (
GSTT1), and Pi1 (
GSTP1) genes [
23], in which the lack of functional proteins involved in this cascade may contribute to intracellular CDDP accumulation and cytotoxic effects [
24]. The cytotoxic activity of CDDP is also attributed to its DNA binding, leading to the activation of repair mechanisms. The DNA lesion induced by CDDP can be removed through the nucleotide excision repair (NER) pathway [
25], mediated by the xeroderma pigmentosum (
XPC, XPD, and
XPF) [
26,
27] and excision repair cross-complementation group 1 (
ERCC1) genes [
28], as well as by the mismatch repair (MMR) pathway, mediated through proteins encoded by MutL homolog 1 (
MLH1) [
29], MutS homolog 2 (
MSH2) [
30], MutS homolog 3 (
MSH3), and exonuclease 1 (
EXO1) genes [
29]. If the repair is ineffective, apoptosis is mediated by proteins encoded by
P53, Caspase 8 (
CASP8),
CASP9,
CASP3, Fas cell surface death receptor (
FAS), and Fas ligand (
FASL) tumor necrosis factors [
21,
31]. Defects in these pathways may promote increased DNA damage and/or apoptosis, with greater potential for toxicity [
32].
Genome-wide studies have described SNVs in acylphosphatase 2 (
ACYP2), involved in calcium homeostasis [
33,
34,
35] and Mendelian deafness
WFS1 genes [
20,
33,
36,
37], as predictors of CDDP-induced ototoxicity. Genes encoding thiopurine S- (
TPMT) and cathecol-O methyltransferases (
COMT) have also been described as potential risk factors [
35]. In CDDP-treated patients,
GSTM1,
GSTT1 [
18,
38,
39,
40,
41,
42], and
GSTP1 c.313A>G [
38,
39,
41,
43] were seen in pediatric solid or adult testicular tumors with controversial results in ototoxicity, while
XPC c.2815A>C SNV influenced ototoxicity in osteosarcoma patients [
44].
To our knowledge, the only cohort that evaluated SNVs in genes of distinct pathways of CDDP metabolism, damage repair, and apoptosis (
GSTM1,
GSTT1,
GSTP1 c.313A>G,
XPC c.2815A>C,
XPD c.934G>A,
XPD c.2251A>C,
XPF c.2505T>C,
ERCC1 c.354C>T,
MLH1 c.-93G>A,
MSH2 c.211 +9G>C,
MSH3 c.3133A>G,
EXO1 c.1762G>A,
P53 c.215G>C,
FAS c.-671A>G,
FAS c.-1378G>A,
FASL c.-844C>T,
CASP3 c.-1191A>G, and
CASP3 c.-182-247G>T) in the ototoxicity of HNSCC treated with CDDP chemoradiation was previously conducted by our group, and the functional roles of each SNV described in the literature are presented in
Table A1. We found that
GSTT1 [
45],
EXO1 [
19],
XPC [
46], and
FASL [
47] SNVs altered the occurrence of all-grade ototoxicity.
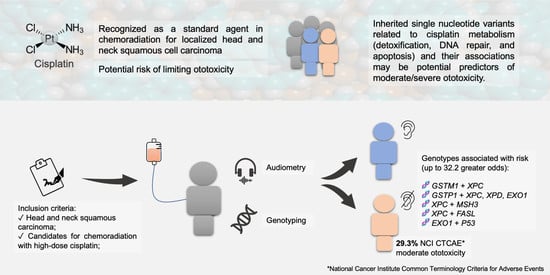
Since there is scarce information regarding pure tone and audiometric speech changes in patients under CDDP chemoradiation, considering that moderate/severe ototoxicity influences quality of life and the fact that patients may inherit defects in more than one pathway, we conducted a descriptive and pharmacogenetic study focusing isolated factors related to CDDP metabolism, aiming to contribute to the prompt recognition of patients at high risk of ototoxicity before treatment initiation and thus enabling treatment modifications.
4. Discussion
In this clinical and pharmacogenetic cohort, it was possible to reaffirm the clinical relevance of hearing loss induced by CDDP. CDDP induces ototoxicity through the promotion of oxidative stress and inflammation in the cochlea, with the increased generation of reactive oxygen species (ROS) [
81]. The long-term accumulation of CDDP in the cochlear endolymph was also described through plasma mass spectrometry in preclinical models, justifying the potential for permanent damage [
82].
Firstly, substantial hearing loss before treatment was observed in our cohort; high-frequency minimum thresholds were higher at baseline, ranging from 35 to 40 dB over 4 kHz. This may be attributable to the high proportion of smokers in our sample since smoking is a reported risk factor for loss at high frequencies [
18,
83,
84]. A history of noise exposure, not assessed in this cohort, could also explain this finding as well as uneven losses in left and right ears not associated with the tumor side [
85,
86,
87]. We were able to observe a meaningful change after CDDP exposure in regard to minimum hearing thresholds, particularly in higher frequencies in univariate analysis, as suggested by previous studies [
15], with limitations involving higher pitch sounds. Caballero and colleagues described similar findings in a cohort of 103 patients, with significantly meaningful changes after CDDP exposure (median change of 9.5 dB in the right and 18.75 dB in the left ears for 4 kHz; 18.6 dB in the right and 28.7 dB in the left, for 8 kHz). The limitations to quality of life related to hearing loss from CDDP have already been reported in a recent systematic review from Pearson and colleagues [
16]. Regarding additional clinical factors, cumulative CDDP dose was observed as a risk factor for greater change in high-frequency averages (3, 4, 6, and 8 kHz) for the right ear, which prompted the inclusion of this variable in multivariate analysis for both sides.
When accounting for the 0.25 to 4 kHz interval, there was also a significant relative increase in mild and moderate hearing loss after CDDP in our analysis, following GBD classification. The percentage of 64.1% with a threshold ≥ 20 dB after treatment is markedly superior to the overall prevalence reported in the literature for the general population (19.3%) [
88], pointing to the cytotoxic effects of CDDP on hearing impairment. To our knowledge, this is the first study to report this classification before and after CDDP in patients diagnosed with HNSCC [
89]. Unilateral hearing damage was observed in four patients (4.5%) after therapy under pure tone audiometry air conduction, from which two (2.2%) had reported losses in both conduction modalities (air and bone). It is worth commenting that all patients with unilateral damage had pharyngeal carcinoma located on the side of hearing loss, and most had changes at baseline. Even though the RT technique and CDDP dose did not differ amongst patients, the location of the tumor in relation to the inner ear could have influenced this finding.
On the other hand, median outcomes from speech audiometry (SRT and SDS) were practically unchanged after platinum exposure. One possible explanation for this may be related to the fact that human speech usually ranges from 0.25 to 4 kHz [
90], while CDDP-related hearing loss involves more relevant changes beyond 3 kHz. In an isolated acoustic environment, frequencies related to speech may be unaltered, though it is possible to expect a greater extent of limitation in terms of communication with background noise, which was not assessed in this cohort. The largest study analyzing speech audiometry after CDDP exposure was performed by Shahbazi and colleagues [
91], evaluating the prevalence of speech recognition disability, defined as SRT greater than 15 dB, in testicular cancer survivors. In 1347 patients, speech recognition disability was identified in 10.4%, and the association of the cumulative CDDP dose could also not be confirmed. Those findings are distinct from our analysis, where 51.6% could be classified as speech-disabled before therapy and 60.7% after therapy. The study populations are markedly different since the Platinum Study [
91] included younger patients not bearing primary tumors in the head and neck and without risk factors such as tobacco and alcohol consumption. There are, to date, only scarce amounts of the literature data on speech audiometry for HNSCC, thus limiting further comparisons.
When considering the
NCI CTCAE criteria for the classification of hearing loss, the proportion of 29.5% moderate/severe ototoxicity was marginally higher than previously reported literature data, ranging from 20 to 25% in adults [
10,
37]. Except for race and BMI, other clinical variables such as age, sex, tumor location, and staging could not be identified as prognostic factors in this analysis, and although cumulative CDDP is recognized as a risk factor for hearing damage [
37], this association could not be observed in the present data for this outcome specifically. Some recent studies have suggested the presence of emotional stress as a possible risk factor for enhanced tumorigenesis and neurotoxicity induced by chemotherapy in general [
92]. A cross-sectional analysis of 623 cancer survivors described a higher association of tinnitus (
p = 0.029) and hearing loss (
p = 0.007) amongst patients with higher distress scores [
93]. Due to the characteristics of the study design, it is not possible to differentiate stress as an independent risk factor, as opposed to a consequence of long-term toxicity. Even though a longitudinal study of the current analysis could potentially assess this variable, distress scores were not previously planned and included in this cohort.
In this study,
GSTP1 c313AG or GG and
XPC c.2815 AA genotypes increased the odds of moderate/severe ototoxicity 4.20- and 3.13-fold, respectively. Preclinical studies have demonstrated that
GSTP1 c313 A>G encodes a change from isoleucine to valine in codon 105, leading to reduced protein activity and detoxification [
63], while the XPC c.2815 C allele promotes the change from lysine to glutamine in codon 939, also diminishing protein activity and, consequently, DNA repair (
Table A1) [
64]. There is, however, marked heterogeneity of clinical effects in terms of the currently available literature. For instance,
GSTP1 c313AG or GG was associated with an increased risk of moderate/severe hearing impairment in 106 [
41] and 64 children [
43], respectively, treated with platinum agents, using the Brock hearing loss classification of equal or greater than 2 [
20]. The association between cumulative CDDP and ototoxicity was found in the study conducted by Lui and colleagues [
41] but not in the study by Sherief and colleagues [
43]. Even though our findings in a previous analysis of the data [
45] are similar and in agreement with the functional roles of
GSTP1 c313A>G [
94], this SNV was not related to CDDP-induced ototoxicity in an additional cohort that recruited 71 children and young patients with various solid tumors [
38], while in 173 patients with testicular carcinoma, post-treatment audiometric evaluations prompted divergent results, even though baseline assessments were not retrievable [
39]. Reported results were also conflicting for isolated
XPC c.2815A>G [
35]. The
XPC c.2815AA genotype was associated with an increased risk of any grade of toxicity [
46] in a previous analysis of the data conducted by our group, and the same effect was observed in a smaller subset of patients with osteosarcoma [
44]. Nonetheless, Lui and colleagues [
41] did not present a significant association among 106 pediatric patients treated with platin analogs. Functional analyses performed for this variant [
64] suggest the presence of the C allele reduces DNA repair, which would theoretically increase the risk of toxicity in contrast to what is currently reported, though an additional assay from Khan and colleagues [
26] did not demonstrate a clear difference for the rate of nucleotide excision repair. Differences in the results obtained from the studies are not easily explained and may have originated from limitations related to sample size, patient baseline characteristics, tumor types, and treatment administered. There are also markedly distinct hearing loss classifications applied in previous cohorts, hampering proper direct comparisons with NCI CTCAE v4.03. Larger cohorts, in addition to further functional assays, would be ideal to better confront these findings.
The metabolism of CDDP is known to involve cellular efflux, NER, and MMR damage repair, as well as apoptosis [
37]. We were able to observe meaningful interactions between variants encoding those distinct pathways, suggesting that toxicity may be enhanced by the coexistence of more than one mutation in the different stages of CDDP metabolism and cytotoxic effect. The combination of
GSTM1 null plus
XPC c.2815AA,
GSTP1 c.313AG or GG plus
XPC c.2815AA,
XPD c.934AA or
EXO1 c.1762AA, or
XPC c.2815AA plus
MSH3 c.3133AA or
FASL c.-844CC (
Table A1) intensified the odds of moderate/severe ototoxicity up to 32.22-fold. The variant alleles from SNVs
XPD c.934 (A) [
65],
EXO1 c.1762 (A) [
71], and
MSH3 c.3133 (A) [
70] have been shown to reduce DNA repair activity by encoding amino acid replacements with the consequent loss of protein function or expression (
Table A1). Additionally, the SNV
FASL c.-844 is located within the enhancer-biding region of
FASL, and luciferase assays have described the variant genotype TT to promote protein affinity twice lower than wild CC donors, leading to less protein expression and, as such, reduced apoptosis [
74]. Hence, the combination of genotypes enhancing CDDP accumulation and reducing repair or activating apoptosis could potentiate the risk of ototoxicity, as observed in this analysis. To our knowledge, no studies focusing on the effects of the combinations of SNVs on the genes of distinct pathways of CDDP metabolism have been conducted to date.
An association with MMR and apoptosis mechanisms was also noted in this study, as the combination of
EXO1 c.1762GA or AA and
P53 c.215CC genotypes increased the risk for events with OR 20.97. The
P53 c.215 wild allele encoding arginine (G) was described as more efficient in inducing apoptosis than the proline (C) variant [
72]. The P53 protein signaling pathway promotes cell death triggered by the generation of ROS [
35]. In addition to apoptosis, P53 is related to cell cycle arrest, cell senescence, and DNA repair [
95]. Cellular senescence is a state of permanent cell cycle arrest that is able to promote local inflammation and tissue damage [
96]. In vitro studies have suggested that early senescence in response to genotoxic stress was P53-dependent and EXO1-depleted [
97,
98]. Moreover, Benkafadar and colleagues [
99] observed that the response to ROS-induced DNA damage leads to cochlear cell senescence by activating the P53 pathway and hence contributing to age-related hearing loss. To date, there is a lack of evidence for a direct association between ototoxicity by CDDP and cell senescence. However, the accumulation of senescent neuronal cells is associated with CDDP-induced peripheral neuropathy in mice [
100]. Thus, we may infer that patients with
EXO1 c.1792GA or AA and
P53 c.215CC combined genotypes were more efficient in promoting cell cycle arrest and senescence of sensory cells after injury by CDDP and, consequently, were at greater risk of hearing loss when compared to patients carrying other genotypes.
We are aware that this study is limited for its sample size; thus, similarly to previous studies, lacking power for further SNVs combinations or polygenic evaluations and correction for other possible confounders. Though statistical tools were used to stabilize risk, such as bootstrap and power post hoc calculations, there may still be unknown influential factors not identifiable in this sample. It must also be considered that not all SNVs in the genes related to CDDP detoxification, DNA repair, and the apoptosis of damaged cells were evaluated in this study; only those recognized with a greater potential to induce ototoxicity were evaluated here. Thus, it is possible that other SNVs with equal or even greater importance in CDDP ototoxicity will be identified in future studies. Furthermore, other known SNVs for CDDP-induced ototoxicity unrelated to stages regarding drug absorption, distribution, metabolism, and excretion were not assessed and could be additional confounders. There is evidence supporting additional SNVs as risk factors for ototoxicity induced by CDDP related to
ACYP2 [
33,
34],
TPMT [
35],
COMT [
35], and
WFS1 [
36] genes.
ACYP2 is known to influence ATP-dependent calcium signaling, which may play a role in sensorineural hearing loss [
33]. TPMT and COMT are methyltransferases that may inactivate CDDP and purine compounds. The Mendelian deafness genes, amongst which
WSF1 is included, encode proteins reported to control endothelial reticulum stress response, thus influencing inner ear cellular damage [
36]. Apart from known and unknown genetic risk factors for toxicity and hearing loss, clinical variables, such as a history of noise exposure [
18] and distress scores [
93], were also not collected from this cohort. We believe, however, that the exclusion of patients with reported hearing loss and hearing impairment in audiometry before treatment could, to some extent, attenuate these limitations.
It is also important to consider distinct patient characteristics when assessing the generalizability of this study for other tumors since the population was predominantly male, with a high frequency of smokers and alcohol users, as well as locally advanced stages of HNSCC. Treatment approaches in the field of RT may also be distinct and could influence the prevalence and severity of adverse events, mainly in institutions with more frequent use of intensity-modulated RT. Though prespecified treatment protocols were strictly followed, therefore preventing confounding to some extent, heterogeneity in therapy protocols could affect the generalizability of these results.