Artificial Spores: Immunoprotective Nanocoating of Red Blood Cells with Supramolecular Ferric Ion-Tannic Acid Complex
Abstract
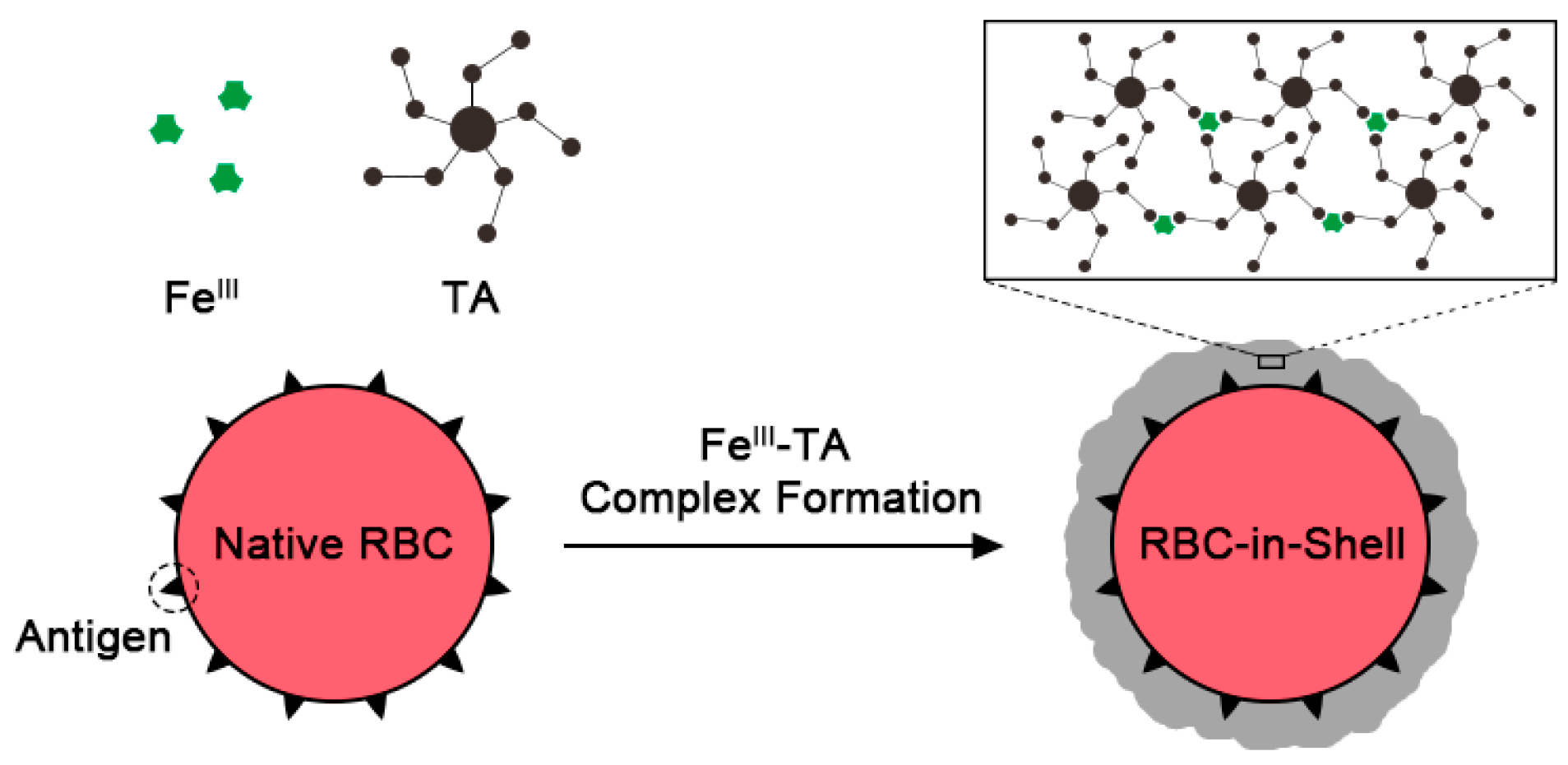
:1. Introduction
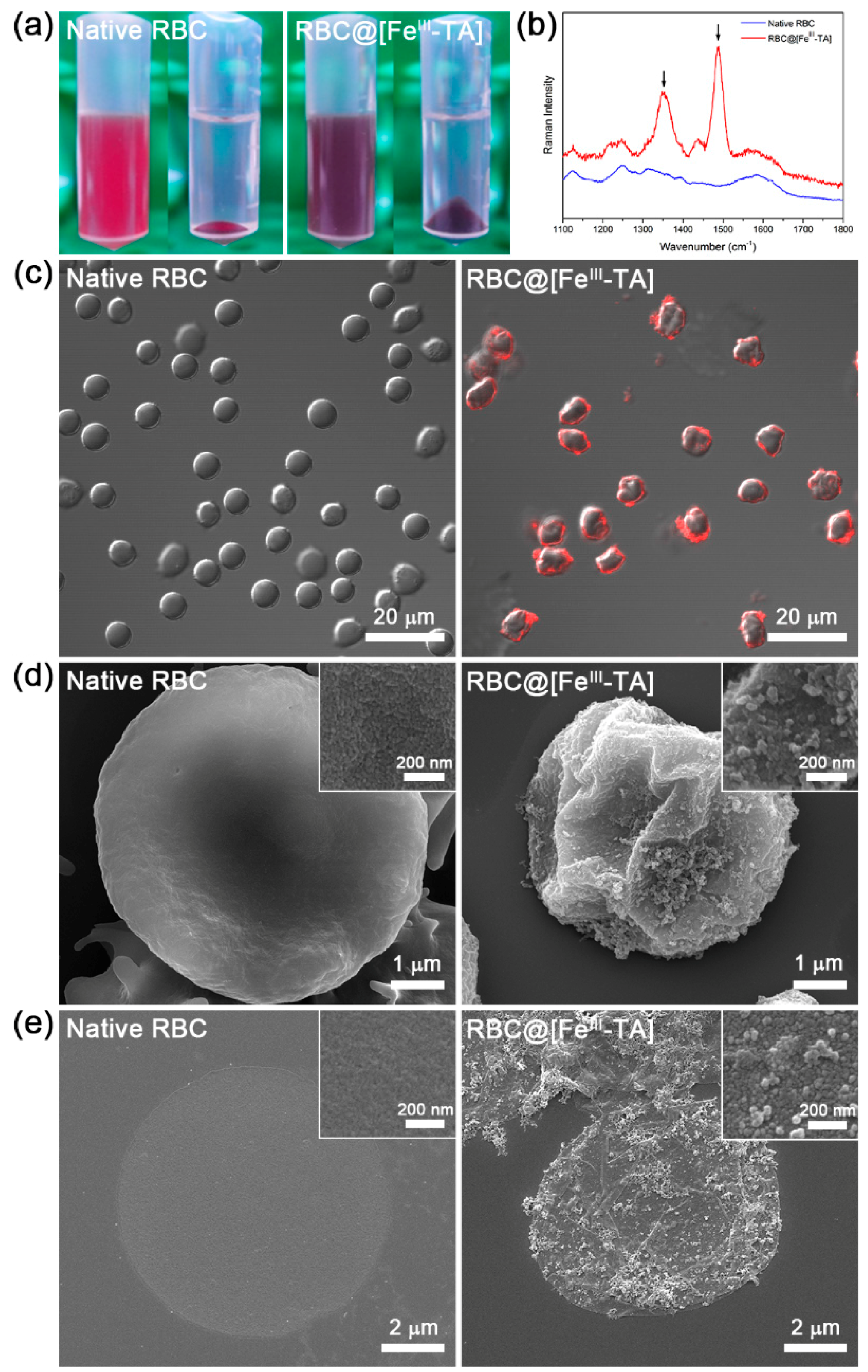
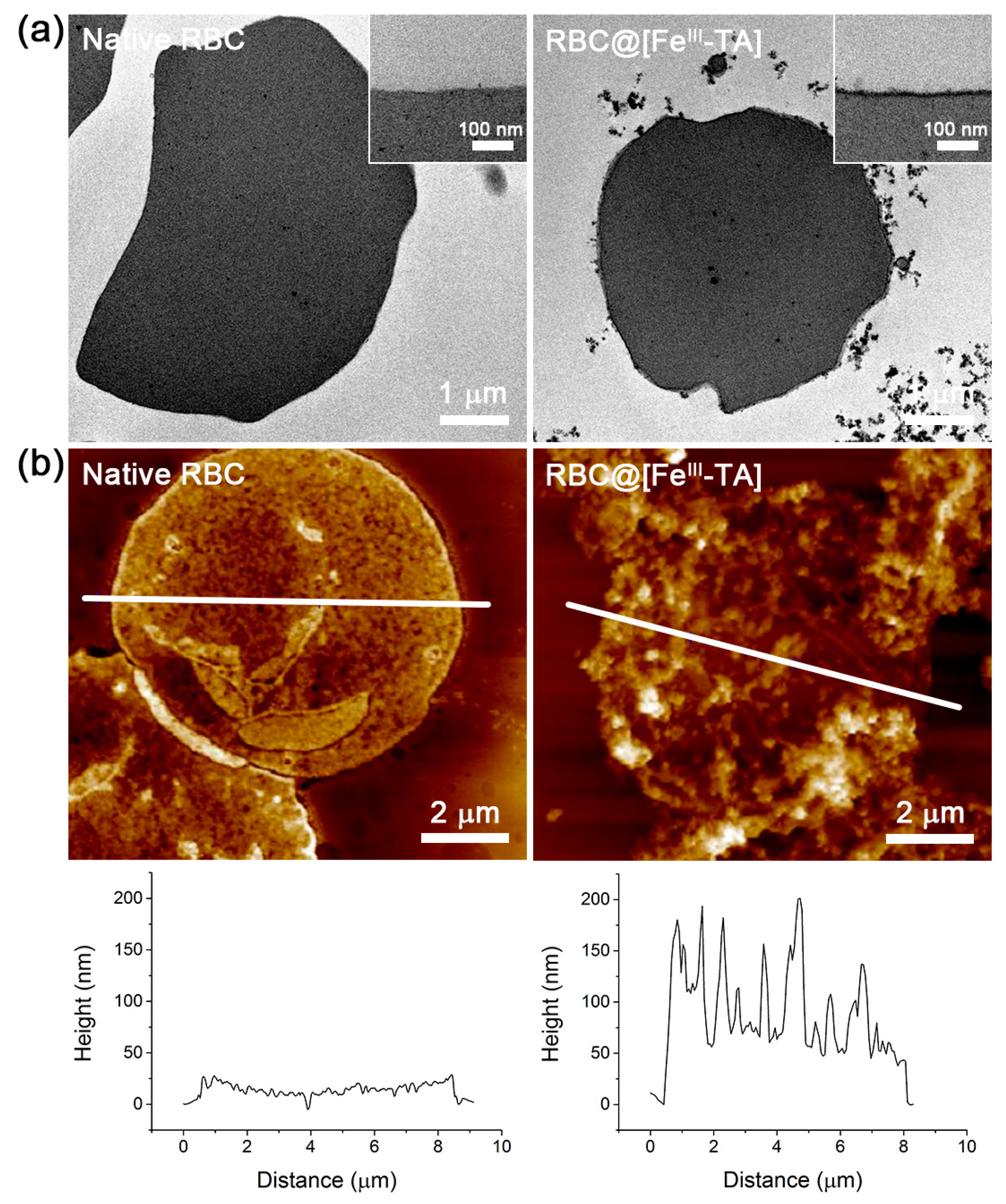
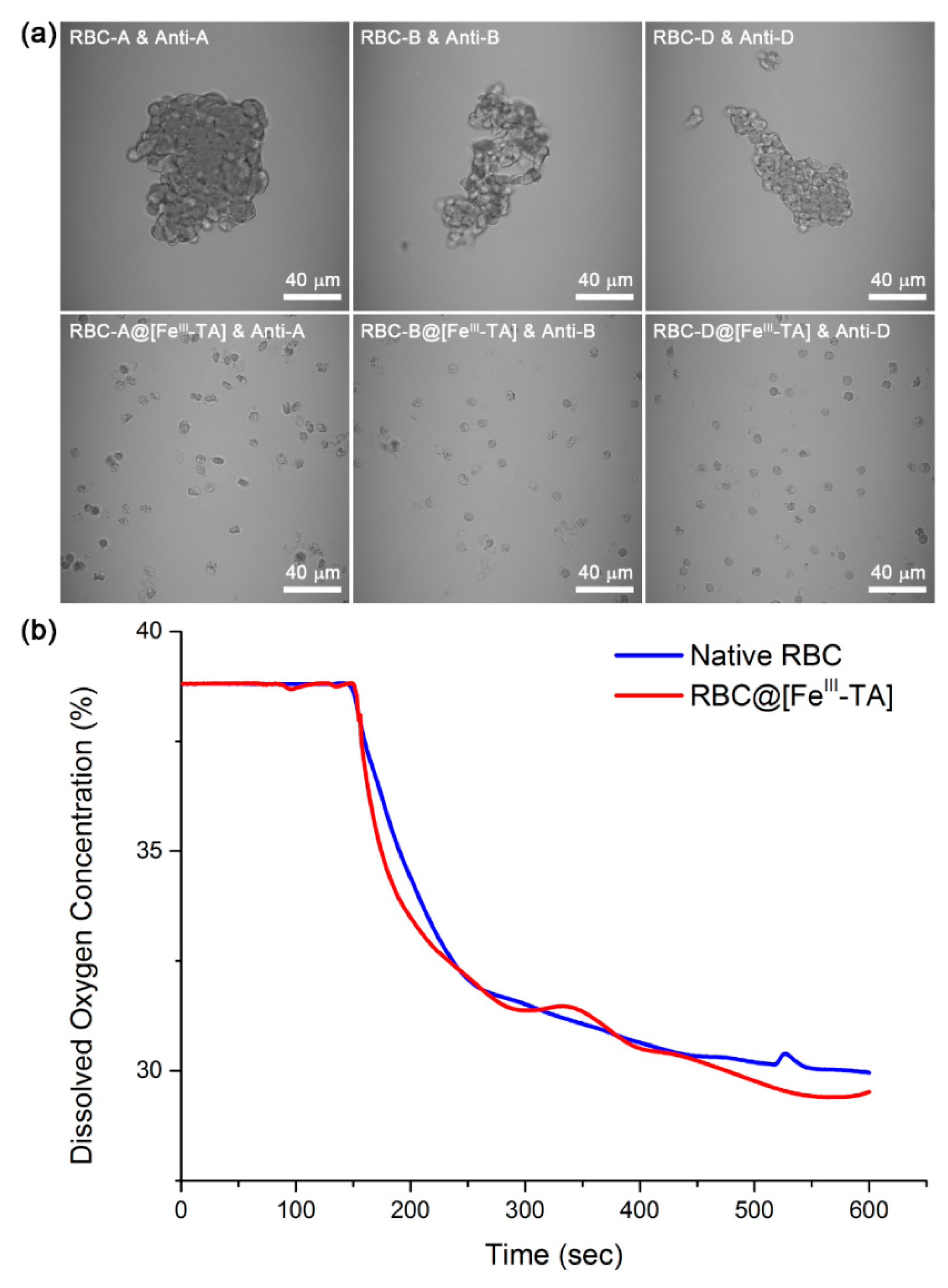
2. Results and Discussion
3. Experimental Section
3.1. Materials
3.2. Red Blood Cell (RBC) Samples
3.3. RBC Coating with FeIII-TA Complex
3.4. Effect of NaCl on Film Thickness: Gold Substrates
3.5. Characterizations
3.6. Antibody-Mediated Agglutination and Oxygen Consumption
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yang, S.H.; Hong, D.; Lee, J.; Ko, E.H.; Choi, I.S. Artificial Spores: Cytocompatible encapsulation of individual living cells within thin, tough artificial shells. Small 2013, 9, 178–186. [Google Scholar] [CrossRef] [PubMed]
- Hong, D.; Park, M.; Yang, S.H.; Lee, J.; Kim, Y.-G.; Choi, I.S. Artificial spores: Cytoprotective nanoencapsulation of living cells. Trends Biotechnol. 2013, 31, 442–447. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Yang, S.H.; Lee, J.; Ko, E.H.; Hong, D.; Choi, I.S. Nanocoating of single cells: From maintenance of cell viability to manipulation of cellular activities. Adv. Mater. 2014, 26, 2001–2010. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Hong, D.; Lee, J.; Choi, I.S. Cell-in-shell hybrids: Chemical nanoencapsulation of individual cells. Acc. Chem. Res. 2016, 49, 792–800. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.H.; Lee, K.-B.; Kong, B.; Kim, J.-H.; Kim, H.-S.; Choi, I.S. Biomimetic encapsulation of individual cells with silica. Angew. Chem. Int. Ed. 2009, 48, 9160–9163. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.H.; Kang, S.M.; Lee, K.-B.; Chung, T.D.; Lee, H.; Choi, I.S. Mussel-inspired encapsulation and functionalization of individual yeast cells. J. Am. Chem. Soc. 2011, 133, 2795–2797. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.H.; Ko, E.H.; Jung, Y.H.; Choi, I.S. Bio-inspired functionalization of silica-encapsulated yeast cells. Angew. Chem. Int. Ed. 2011, 50, 6115–6118. [Google Scholar] [CrossRef] [PubMed]
- Ko, E.H.; Yoon, Y.; Park, J.H.; Yang, S.H.; Hong, D.; Lee, K.-B.; Shon, H.K.; Lee, T.G.; Choi, I.S. Bioinspired, cytocompatible mineralization of silica-titania composites: Thermo-protective nanoshell formation for individual chlorella cells. Angew. Chem. Int. Ed. 2013, 52, 12279–12282. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Choi, J.; Park, J.H.; Kim, M.-H.; Hong, D.; Cho, H.; Yang, S.H.; Choi, I.S. Cytoprotective silica coating of individual mammalian cells through bioinspired silicification. Angew. Chem. Int. Ed. 2014, 53, 8056–8059. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.J.; Park, T.; Moon, H.C.; Park, S.-Y.; Hong, D.; Ko, E.H.; Kim, J.Y.; Hong, J.W.; Han, S.W.; Kim, Y.-G.; et al. Cytoprotective alginate/polydopamine core/shell microcapsules in microbial encapsulation. Angew. Chem. Int. Ed. 2014, 53, 14443–14446. [Google Scholar] [CrossRef] [PubMed]
- Hong, D.; Lee, H.; Ko, E.H.; Lee, J.; Cho, H.; Park, M.; Yang, S.H.; Choi, I.S. Organic/inorganic double-layered shells for multiple cytoprotection of individual living cells. Chem. Sci. 2015, 6, 203–208. [Google Scholar] [CrossRef]
- Liang, K.; Richardson, J.J.; Cui, J.; Caruso, F.; Doonan, C.J.; Falcaro, P. Metal-organic framework coatings as cytoprotective exoskeletons for living cells. Adv. Mater. 2016, 28, 7910–7914. [Google Scholar] [CrossRef] [PubMed]
- Chowdhuri, S.; Cole, C.M.; Devaraj, N.K. Encapsulation of living cells within giant phospholipid liposomes formed by the inverse-emulsion technique. ChemBioChem 2016, 17, 886–889. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Tam, U.C.; Czlapinski, J.L.; Lee, G.S.; Rabuka, D.; Zettl, A.; Bertozzi, C.R. Interfacing carbon nanotubes with living cells. J. Am. Chem. Soc. 2006, 128, 6292–6293. [Google Scholar] [CrossRef] [PubMed]
- Wojcik, M.; Hauser, M.; Li, W.; Moon, S.; Xu, K. Graphene-enabled electron microscopy and correlated super-resolution microscopy of wet cells. Nat. Commun. 2015, 6, 7384. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.H.; Lee, T.; Seo, E.; Ko, E.H.; Choi, I.S.; Kim, B.-S. Interfacing living yeast cells with graphene oxide nanosheaths. Macromol. Biosci. 2012, 12, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Kim, K.; Lee, J.; Choi, J.Y.; Hong, D.; Yang, S.H.; Caruso, F.; Lee, Y.; Choi, I.S. A cytoprotective and degradable metal-polyphenol nanoshell for single-cell encapsulation. Angew. Chem. Int. Ed. 2014, 53, 12420–12425. [Google Scholar] [CrossRef]
- Li, W.; Bing, W.; Huang, S.; Ren, J.; Qu, X. Mussel byssus-like reversible metal-chelated supramolecular complex used for dynamic cellular surface engineering and imaging. Adv. Funct. Mater. 2015, 24, 3775–3784. [Google Scholar] [CrossRef]
- Yang, S.H.; Choi, J.; Palanikumar, L.; Choi, E.S.; Lee, J.; Kim, J.; Choi, I.S.; Ryu, J.-H. Cytocompatible in situ cross-linking of degradable lbl films based on thiol-exchange reaction. Chem. Sci. 2015, 6, 4698–4703. [Google Scholar] [CrossRef]
- Lee, J.; Cho, H.; Choi, J.; Kim, D.; Hong, D.; Park, J.H.; Yang, S.H.; Choi, I.S. Chemical sporulation and germination: Cytoprotective nanocoating of individual mammalian cells with a degradable tannic acid-FeIII complex. Nanoscale 2015, 7, 18918–18922. [Google Scholar] [CrossRef] [PubMed]
- Villa, C.H.; Anselmo, A.C.; Mitragotri, S.; Muzykantov, V. Red blood cells: Supercarriers for drugs, biologicals, and nanoparticles and inspiration for advanced delivery systems. Adv. Drug Deliv. Rev. 2016, 106, 88–103. [Google Scholar] [CrossRef] [PubMed]
- Carson, J.L.; Grossman, B.J.; Kleinman, S.; Tinmouth, A.T.; Marques, M.B.; Fung, M.K.; Holcomb, J.B.; Illoh, O.; Kaplan, L.J.; Katz, L.M.; et al. Red blood cell transfusion: A clinical practice guideline from the Aabb. Ann. Intern. Med. 2012, 157, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Mujahid, A.; Dickert, F.L. Blood group typing: From classical strategies to the application of synthetic antibodies generated by molecular imprinting. Sensors 2016, 16, 51. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.P.; Sulzenbacher, G.; Yuan, H.; Bennett, E.P.; Pietz, G.; Saunders, K.; Spence, J.; Nudelman, E.; Levery, S.B.; White, T.; et al. Bacterial glycosidases for the production of universal red blood cells. Nat. Biotechnol. 2007, 25, 454–464. [Google Scholar] [CrossRef] [PubMed]
- Giarratana, M.-C.; Rouard, H.; Dumont, A.; Kiger, L.; Safeukui, I.; Le Pennec, P.-Y.; François, S.; Trugnan, G.; Peyrard, T.; Marie, T.; et al. Proof of principle for transfusion of in vitro-generated red blood cells. Blood 2011, 118, 5071–5079. [Google Scholar] [CrossRef] [PubMed]
- Douay, L.; Andreu, G. Ex vivo production of human red blood cells from hematopoietic stem cells: What is the future in transfusion? Transfus. Med. Rev. 2007, 21, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Reid, M.E.; Mohandas, N. Red blood cell blood group antigens: Structure and function. Semin. Hematol. 2004, 41, 93–117. [Google Scholar] [CrossRef] [PubMed]
- Timmins, N.E.; Nielsen, L.K. Blood cell manufacture: Current methods and future challenges. Trends Biotechnol. 2009, 27, 415–422. [Google Scholar] [CrossRef] [PubMed]
- Mansouri, S.; Merhi, Y.; Winnik, F.M.; Tabrizian, M. Investigation of layer-by-layer assembly of polyelectrolytes on fully functional human red blood cells in suspension for attenuated immune response. Biomacromolecules 2011, 12, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Wang, G.; Zhao, B.; Chen, J.; Zhang, X.; Tang, R. Antigenically shielded universal red blood cells by polydopamine-based cell surface engineering. Chem. Sci. 2014, 5, 3463–3468. [Google Scholar] [CrossRef]
- Kim, J.Y.; Lee, H.; Park, T.; Park, J.; Kim, M.-H.; Cho, H.; Youn, W.; Kang, S.M.; Choi, I.S. Artificial spores: Cytocompatible coating of living cells with plant-derived pyrogallol. Chem. Asian J. 2016, 11, 3183–3187. [Google Scholar] [CrossRef] [PubMed]
- Ejima, H.; Richardson, J.J.; Liang, K.; Best, J.P.; van Koeverden, M.P.; Such, G.K.; Cui, J.; Caruso, F. One-step assembly of coordination complexes for versatile film and particle engineering. Science 2013, 341, 154–157. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, D.S.; Kang, S.M. Reversible layer-by-layer deposition on solid substrates inspired by mussel byssus cuticle. Chem. Asian J. 2014, 9, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Jandl, J.H.; Simmons, R.L. The agglutination and sensitization of red blood cells by metallic cations: Interactions between multivalent metals and the red-cell membrane. Br. J. Haematol. 1957, 3, 19–38. [Google Scholar] [CrossRef] [PubMed]
- Kozlovskaya, V.; Kharlampieva, E.; Drachuk, I.; Cheng, D.; Tsukruk, V.V. Responsive microcapsule reactors based on hydrogen-bonded tannic acid layer-by-layer assemblies. Soft Matter 2010, 6, 3596–3608. [Google Scholar] [CrossRef]
- Ju, Y.; Cui, J.; Müllner, M.; Suma, T.; Hu, M.; Caruso, F. Engineering low-fouling and pH-degradable capsules through the assembly of metal-phenolic networks. Biomacromolecules 2015, 16, 807–814. [Google Scholar] [CrossRef] [PubMed]
- Ju, Y.; Cui, J.; Sun, H.; Müllner, M.; Dai, Y.; Guo, J.; Bertleff-Zieschang, N.; Suma, T.; Richardson, J.J.; Caruso, F. Engineered metal-phenolic capsules show tunable targeted delivery to cancer cells. Biomacromolecules 2016, 17, 2268–2276. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, T.; Kim, J.Y.; Cho, H.; Moon, H.C.; Kim, B.J.; Park, J.H.; Hong, D.; Park, J.; Choi, I.S. Artificial Spores: Immunoprotective Nanocoating of Red Blood Cells with Supramolecular Ferric Ion-Tannic Acid Complex. Polymers 2017, 9, 140. https://doi.org/10.3390/polym9040140
Park T, Kim JY, Cho H, Moon HC, Kim BJ, Park JH, Hong D, Park J, Choi IS. Artificial Spores: Immunoprotective Nanocoating of Red Blood Cells with Supramolecular Ferric Ion-Tannic Acid Complex. Polymers. 2017; 9(4):140. https://doi.org/10.3390/polym9040140
Chicago/Turabian StylePark, Taegyun, Ji Yup Kim, Hyeoncheol Cho, Hee Chul Moon, Beom Jin Kim, Ji Hun Park, Daewha Hong, Joonhong Park, and Insung S. Choi. 2017. "Artificial Spores: Immunoprotective Nanocoating of Red Blood Cells with Supramolecular Ferric Ion-Tannic Acid Complex" Polymers 9, no. 4: 140. https://doi.org/10.3390/polym9040140






