Human Antibodies against Herpes Simplex Virus 2 Glycoprotein G Do Not Neutralize but Mediate Antibody-Dependent Cellular Cytotoxicity
Abstract
1. Introduction
2. Material and Methods
2.1. Sera from Symptomatic HSV-1+2-Infected and Symptomatic HSV-2-Infected Subjects
2.2. Sera from HSV-1-Infected and HSV-Negative Subjects
2.3. Production of EXCT4-mgG-2 and Coupling to an Immunosorbent Column
2.4. Production of gD-2 and Coupling to an Immunosorbent Column
2.5. Production of Pools of Purified Human Antibodies Used for Standard Curves and for Functional Assays
2.6. The Specificity of Pools of Purified Antibodies
2.7. Measurement of Antibody Concentration in Serum in ELISA
2.8. Neutralization Assay without or with Complement
2.9. Antibody-Dependent Cellular Cytotoxicity (ADCC) and Complement-Dependent Cytotoxicity (CDC)
3. Statistics
4. Results
4.1. Specificity of Purified anti-EXCT4-mgG-2 and anti-gD-2 Antibodies
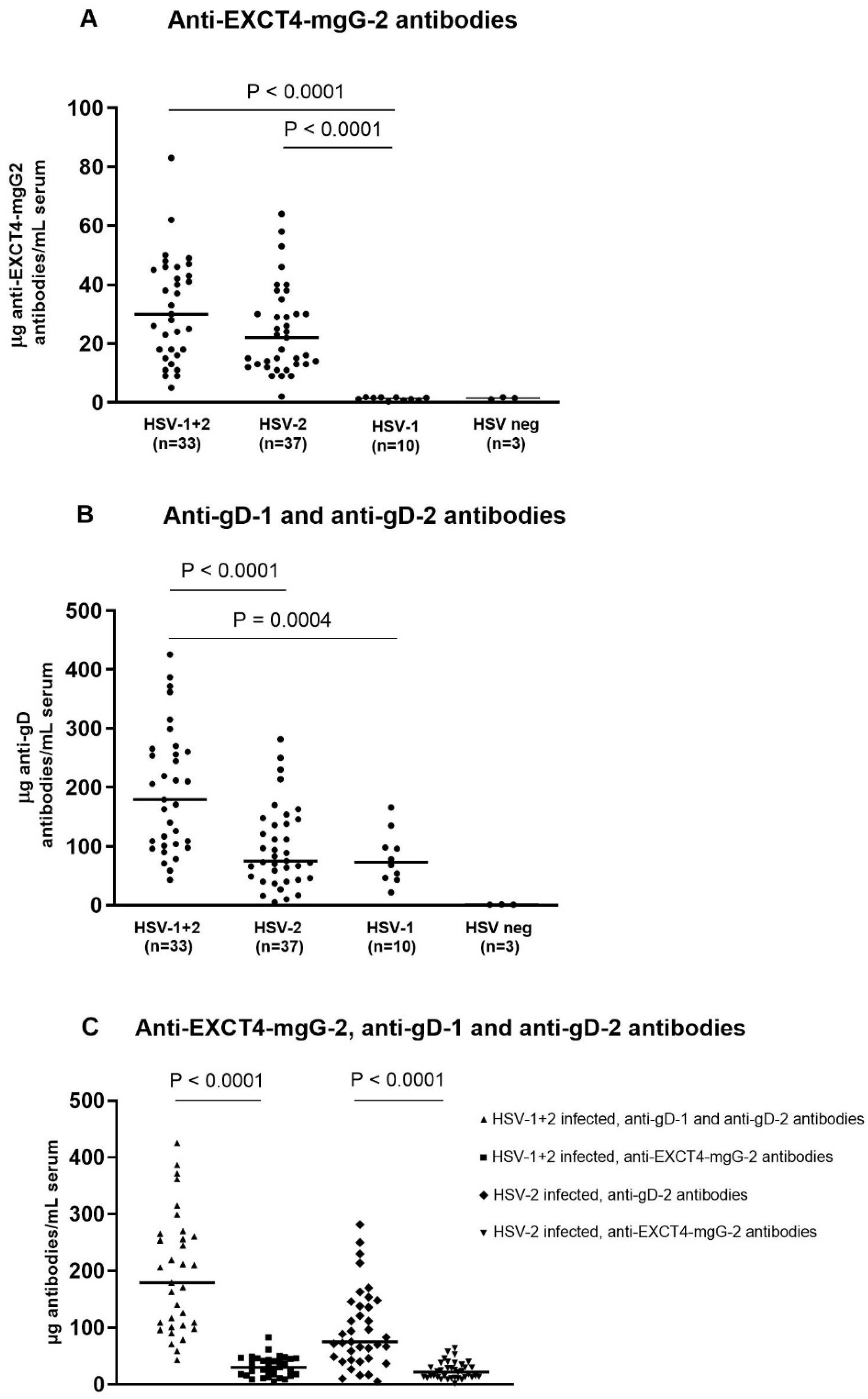
4.2. Concentrations of Antibodies in HSV-1+2-, HSV-2-, and HSV-1-Infected Subjects
4.3. Neutralization Activity
4.4. ADCC
4.5. CDC
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- James, C.; Harfouche, M.; Welton, N.J.; Turner, K.M.; Abu-Raddad, L.J.; Gottlieb, S.L.; Looker, K.J. Herpes simplex virus: Global infection prevalence and incidence estimates, 2016. Bull. World Health Organ. 2020, 98, 315. [Google Scholar] [CrossRef] [PubMed]
- Freeman, E.E.; Weiss, H.A.; Glynn, J.R.; Cross, P.L.; Whitworth, J.A.; Hayes, R.J. Herpes simplex virus 2 infection increases HIV acquisition in men and women: Systematic review and meta-analysis of longitudinal studies. Aids 2006, 20, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Corey, L.; Langenberg, A.G.; Ashley, R.; Sekulovich, R.E.; Izu, A.E.; Douglas, J.M., Jr.; Handsfield, H.H.; Warren, T.; Marr, L.; Tyring, S.; et al. Recombinant glycoprotein vaccine for the prevention of genital HSV-2 infection: Two randomized controlled trials. Chiron HSV Vaccine Study Group. JAMA 1999, 282, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Belshe, R.B.; Leone, P.A.; Bernstein, D.I.; Wald, A.; Levin, M.J.; Stapleton, J.T.; Gorfinkel, I.; Morrow, R.L.; Ewell, M.G.; Stokes-Riner, A.; et al. Efficacy results of a trial of a herpes simplex vaccine. N. Engl. J. Med. 2012, 366, 34–43. [Google Scholar] [CrossRef]
- Belshe, R.B.; Heineman, T.C.; Bernstein, D.I.; Bellamy, A.R.; Ewell, M.; van der Most, R.; Deal, C.D. Correlate of Immune Protection Against HSV-1 Genital Disease in Vaccinated Women. J. Infect. Dis. 2014, 209, 828–836. [Google Scholar] [CrossRef] [PubMed]
- Awasthi, S.; Belshe, R.B.; Friedman, H.M. Better neutralization of herpes simplex virus type 1 (HSV-1) than HSV-2 by antibody from recipients of GlaxoSmithKline HSV-2 glycoprotein D2 subunit vaccine. J. Infect. Dis. 2014, 210, 571–575. [Google Scholar] [CrossRef] [PubMed]
- Belshe, R.B.; Blevins, T.P.; Yu, Y.; Nethington, A.E.; Bellamy, A.; Bryant, C.; Morrison, L.A. Neutralizing Antibody Kinetics and Immune Protection Against Herpes Simplex Virus 1 Genital Disease in Vaccinated Women. J. Infect. Dis. 2023, 227, 522–527. [Google Scholar] [CrossRef]
- Blevins, T.P.; Yu, Y.; Belshe, R.B.; Bellamy, A.R.; Morrison, L.A. Correlation between herpes simplex virus neutralizing antibody titers determined by ELVIS cell and traditional plaque reduction assays. PLoS ONE 2019, 14, e0214467. [Google Scholar] [CrossRef]
- Dasgupta, G.; Chentoufi, A.A.; Kalantari, M.; Falatoonzadeh, P.; Chun, S.; Lim, C.H.; Felgner, P.L.; Davies, D.H.; BenMohamed, L. Immunodominant “asymptomatic” herpes simplex virus 1 and 2 protein antigens identified by probing whole-ORFome microarrays with serum antibodies from seropositive asymptomatic versus symptomatic individuals. J. Virol. 2012, 86, 4358–4369. [Google Scholar] [CrossRef]
- Cairns, T.M.; Huang, Z.Y.; Gallagher, J.R.; Lin, Y.; Lou, H.; Whitbeck, J.C.; Wald, A.; Cohen, G.H.; Eisenberg, R.J. Patient-Specific Neutralizing Antibody Responses to Herpes Simplex Virus Are Attributed to Epitopes on gD, gB, or Both and Can Be Type Specific. J. Virol. 2015, 89, 9213–9231. [Google Scholar] [CrossRef]
- Cairns, T.M.; Huang, Z.Y.; Whitbeck, J.C.; Ponce de Leon, M.; Lou, H.; Wald, A.; Krummenacher, C.; Eisenberg, R.J.; Cohen, G.H. Dissection of the antibody response against herpes simplex virus glycoproteins in naturally infected humans. J. Virol. 2014, 88, 12612–12622. [Google Scholar] [CrossRef] [PubMed]
- Lazear, E.; Whitbeck, J.C.; Ponce-de-Leon, M.; Cairns, T.M.; Willis, S.H.; Zuo, Y.; Krummenacher, C.; Cohen, G.H.; Eisenberg, R.J. Antibody-induced conformational changes in herpes simplex virus glycoprotein gD reveal new targets for virus neutralization. J. Virol. 2012, 86, 1563–1576. [Google Scholar] [CrossRef] [PubMed]
- Whitbeck, J.C.; Huang, Z.Y.; Cairns, T.M.; Gallagher, J.R.; Lou, H.; Ponce-de-Leon, M.; Belshe, R.B.; Eisenberg, R.J.; Cohen, G.H. Repertoire of epitopes recognized by serum IgG from humans vaccinated with herpes simplex virus 2 glycoprotein D. J. Virol. 2014, 88, 7786–7795. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Marsden, H.S.; MacAulay, K.; Murray, J.; Smith, I.W. Identification of an immunodominant sequential epitope in glycoprotein G of herpes simplex virus type 2 that is useful for serotype-specific diagnosis. J. Med. Virol. 1998, 56, 79–84. [Google Scholar] [CrossRef]
- Liljeqvist, J.-A.; Trybala, E.; Svennerholm, B.; Jeansson, S.; Sjögren-Jansson, E.; Bergström, T. Localization of type-specific epitopes of herpes simplex virus type 2 glycoprotein G recognized by human and mouse antibodies. J. Gen. Virol. 1998, 79 Pt 5, 1215–1224. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Dropulic, L.; Bozekowski, J.; Pietz, H.L.; Jegaskanda, S.; Dowdell, K.; Vogel, J.S.; Garabedian, D.; Oestreich, M.; Nguyen, H.; et al. Serum and Cervicovaginal Fluid Antibody Profiling in Herpes Simplex Virus-Seronegative Recipients of the HSV529 Vaccine. J. Infect. Dis. 2021, 224, 1509–1519. [Google Scholar] [CrossRef] [PubMed]
- Görander, S.; Harandi, A.M.; Lindqvist, M.; Bergström, T.; Liljeqvist, J. Glycoprotein G of herpes simplex virus 2 as a novel vaccine antigen for immunity to genital and neurological disease. J. Virol. 2012, 86, 7544–7553. [Google Scholar] [CrossRef] [PubMed]
- Görander, S.; Honda-Okubo, Y.; Backström, M.; Baldwin, J.; Piplani, S.; Bergström, T.; Petrovsky, N.; Liljeqvist, J.-A. A truncated glycoprotein G vaccine formulated with Advax-CpG adjuvant provides protection of mice against genital herpes simplex virus 2 infection. Vaccine 2021, 24, 5866–5875. [Google Scholar] [CrossRef]
- Trybala, E.; Peerboom, N.; Adamiak, B.; Krzyzowska, M.; Liljeqvist, J.-A.; Bally, M.; Bergstrom, T. Herpes Simplex Virus Type 2 Mucin-Like Glycoprotein mgG Promotes Virus Release from the Surface of Infected Cells. Viruses 2021, 13, 887. [Google Scholar] [CrossRef] [PubMed]
- Jeansson, S.; Forsgren, M.; Svennerholm, B. Evaluation of solubilized herpes simplex virus membrane antigen by enzyme-linked immunosorbent assay. J. Clin. Microbiol. 1983, 18, 1160–1166. [Google Scholar] [CrossRef]
- Del Campo, J.; Lindqvist, M.; Cuello, M.; Backstrom, M.; Cabrerra, O.; Persson, J.; Perez, O.; Harandi, A.M. Intranasal immunization with a proteoliposome-derived cochleate containing recombinant gD protein confers protective immunity against genital herpes in mice. Vaccine 2010, 28, 1193–1200. [Google Scholar] [CrossRef]
- Trybala, E.; Liljeqvist, J.A.; Svennerholm, B.; Bergstrom, T. Herpes simplex virus types 1 and 2 differ in their interaction with heparan sulfate. J. Virol. 2000, 74, 9106–9114. [Google Scholar] [CrossRef] [PubMed]
- Thomsson, E.; Persson, L.; Grahn, A.; Snall, J.; Ekblad, M.; Brunhage, E.; Svensson, F.; Jern, C.; Hansson, G.C.; Backstrom, M.; et al. Recombinant glycoprotein E produced in mammalian cells in large-scale as an antigen for varicella-zoster-virus serology. J. Virol. Methods 2011, 175, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Bøyum, A.; Løvhaug, D.; Tresland, L.; Nordlie, E.M. Separation of leucocytes: Improved cell purity by fine adjustments of gradient medium density and osmolality. Scand. J. Immunol. 1991, 34, 697–712. [Google Scholar] [CrossRef]
- Neri, S.; Mariani, E.; Meneghetti, A.; Cattini, L.; Facchini, A. Calcein-acetyoxymethyl cytotoxicity assay: Standardization of a method allowing additional analyses on recovered effector cells and supernatants. Clin. Diagn. Lab. Immunol. 2001, 8, 1131–1135. [Google Scholar] [CrossRef] [PubMed]
- Awasthi, S.; Friedman, H.M. A paradigm shift: Vaccine-induced antibodies as an immune correlate of protection against herpes simplex virus type 1 genital herpes. J. Infect. Dis. 2014, 209, 813–815. [Google Scholar] [CrossRef] [PubMed]
- Laver, W.G.; Air, G.M.; Webster, R.G.; Smith-Gill, S.J. Epitopes on protein antigens: Misconceptions and realities. Cell 1990, 61, 553–556. [Google Scholar] [CrossRef] [PubMed]
- Grabowska, A.; Jameson, C.; Laing, P.; Jeansson, S.; Sjögren-Jansson, E.; Taylor, J.; Cunningham, A.; Irving, W.L. Identification of type-specific domains within glycoprotein G of herpes simplex virus type 2 (HSV-2) recognized by the majority of patients infected with HSV-2, but not by those infected with HSV-1. J. Gen. Virol. 1999, 80 Pt 7, 1789–1798. [Google Scholar] [CrossRef]
- van Rooijen, M.S.; Roest, W.; Hansen, G.; Kwa, D.; de Vries, H.J. False-negative type-specific glycoprotein G antibody responses in STI clinic patients with recurrent HSV-1 or HSV-2 DNA positive genital herpes, The Netherlands. Sex. Transm. Infect. 2016, 92, 257–260. [Google Scholar] [CrossRef]
- Balachandran, N.; Bacchetti, S.; Rawls, W. Protection against lethal challenge of BALB/c mice by passive transfer of monoclonal antibodies to five glycoproteins of herpes simplex virus type 2. Infect. Immun. 1982, 37, 1132–1137. [Google Scholar] [CrossRef]
- Para, M.F.; Parish, M.L.; Noble, A.G.; Spear, P.G. Potent neutralizing activity associated with anti-glycoprotein D specificity among monoclonal antibodies selected for binding to herpes simplex virions. J. Virol. 1985, 55, 483–488. [Google Scholar] [CrossRef]
- Balachandran, N.; Oba, D.E.; Hutt-Fletcher, L.M. Antigenic cross-reactions among herpes simplex virus types 1 and 2, Epstein-Barr virus, and cytomegalovirus. J. Virol. 1987, 61, 1125–1135. [Google Scholar] [CrossRef] [PubMed]
- Bruhns, P.; Jönsson, F. Mouse and human FcR effector functions. Immunol. Rev. 2015, 268, 25–51. [Google Scholar] [CrossRef]
- Kohl, S.; Charlebois, E.D.; Sigouroudinia, M.; Goldbeck, C.; Hartog, K.; Sekulovich, R.E.; Langenberg, A.G.; Burke, R.L. Limited antibody-dependent cellular cytotoxicity antibody response induced by a herpes simplex virus type 2 subunit vaccine. J. Infect. Dis. 2000, 181, 335–339. [Google Scholar] [CrossRef]
- Mahant, A.M.; Guerguis, S.; Blevins, T.P.; Cheshenko, N.; Gao, W.; Anastos, K.; Belshe, R.B.; Herold, B.C. Failure of Herpes Simplex Virus Glycoprotein D Antibodies to Elicit Antibody-Dependent Cell-Mediated Cytotoxicity: Implications for Future Vaccines. J. Infect. Dis. 2022, 226, 1489–1498. [Google Scholar] [CrossRef]
- Kohl, S.; West, M.S.; Prober, C.G.; Sullender, W.M.; Loo, L.S.; Arvin, A.M. Neonatal antibody-dependent cellular cytotoxic antibody levels are associated with the clinical presentation of neonatal herpes simplex virus infection. J. Infect. Dis. 1989, 160, 770–776. [Google Scholar] [CrossRef] [PubMed]
- Burn Aschner, C.; Knipe, D.M.; Herold, B.C. Model of vaccine efficacy against HSV-2 superinfection of HSV-1 seropositive mice demonstrates protection by antibodies mediating cellular cytotoxicity. NPJ Vaccines 2020, 5, 35. [Google Scholar] [CrossRef] [PubMed]
- Burn Aschner, C.; Pierce, C.; Knipe, D.M.; Herold, B.C. Vaccination Route as a Determinant of Protective Antibody Responses against Herpes Simplex Virus. Vaccines 2020, 8, 277. [Google Scholar] [CrossRef]
- Petro, C.D.; Weinrick, B.; Khajoueinejad, N.; Burn, C.; Sellers, R.; Jacobs, W.R., Jr.; Herold, B.C. HSV-2 ΔgD elicits FcγR-effector antibodies that protect against clinical isolates. JCI Insight 2016, 1, e88529. [Google Scholar] [CrossRef]
- Kao, C.M.; Goymer, J.; Loh, L.N.; Mahant, A.; Burn Aschner, C.; Herold, B.C. Murine Model of Maternal Immunization Demonstrates Protective Role for Antibodies That Mediate Antibody-Dependent Cellular Cytotoxicity in Protecting Neonates From Herpes Simplex Virus Type 1 and Type 2. J. Infect. Dis. 2020, 221, 729–738. [Google Scholar] [CrossRef]
- Eisenberg, R.J.; Long, D.; Pereira, L.; Hampar, B.; Zweig, M.; Cohen, G.H. Effect of monoclonal antibodies on limited proteolysis of native glycoprotein gD of herpes simplex virus type 1. J. Virol. 1982, 41, 478–488. [Google Scholar] [CrossRef] [PubMed]
- Muggeridge, M.I.; Wilcox, W.C.; Cohen, G.H.; Eisenberg, R.J. Identification of a site on herpes simplex virus type 1 glycoprotein D that is essential for infectivity. J. Virol. 1990, 64, 3617–3626. [Google Scholar] [CrossRef] [PubMed]
- Seigneurin, J.M.; Desgranges, C.; Seigneurin, D.; Paire, J.; Renversez, J.C.; Jacquemont, B.; Micouin, C. Herpes simplex virus glycoprotein D: Human monoclonal antibody produced by bone marrow cell line. Science 1983, 221, 173–175. [Google Scholar] [CrossRef] [PubMed]
- Zarnitsyna, V.I.; Lavine, J.; Ellebedy, A.; Ahmed, R.; Antia, R. Multi-epitope Models Explain How Pre-existing Antibodies Affect the Generation of Broadly Protective Responses to Influenza. PLoS Pathog. 2016, 12, e1005692. [Google Scholar] [CrossRef]
- Dropulic, L.K.; Oestreich, M.C.; Pietz, H.L.; Laing, K.J.; Hunsberger, S.; Lumbard, K.; Garabedian, D.; Turk, S.P.; Chen, A.; Hornung, R.L.; et al. A Randomized, Double-Blinded, Placebo-Controlled, Phase 1 Study of a Replication-Defective Herpes Simplex Virus (HSV) Type 2 Vaccine, HSV529, in Adults with or without HSV Infection. J. Infect. Dis. 2019, 220, 990–1000. [Google Scholar] [CrossRef]

| Cohorts | Median Age, Year (Range) | Number of Subjects (f, Female; m, Men) |
|---|---|---|
| HSV-1+2-infected | 40 (22–69) | n = 33, (19 f, 14 m) |
| HSV-2-infected | 36 (26–70) | n = 37, (18 f, 19 m) |
| HSV-1-infected | 40 (16–65) | n = 10, (5 f, 5 m) |
| HSV-negative | 26 (21–32) | n = 3, (2 f, 1 m) |
| Antigens and Assays | HSV-Negative Sera a | Anti-EXCT4-mgG-2 Abs | Anti-gD-2 Abs |
|---|---|---|---|
| HPA-mgG-2 b,c | − | + | − |
| HSV-1 d | − | − | ++ |
| HerpeSelect1 IgG (gG1 antigen, Focus) | neg | neg | neg |
| HerpeSelect2 IgG (gG2 antigen, Focus) | neg | pos | neg |
| LiaisonXL HSV1 (gG1 antigen, Diasorin) | neg | neg | neg |
| LiaisonXL HSV2 (gG2 antigen, Diasorin) | neg | pos | neg |
| EXCT4-mgG-2 (Rec e) | − | + | − |
| gD-2 (Rec e, EC f) | − | − | ++ |
| gB-2 b | ND | − | − |
| gC-2 b | ND | − | − |
| gC-2 (Rec e, EC f, GenScript) | ND | − | − |
| mgG-2-peptide g (LifeTein) | ND | + | − |
| Concentration of Abs, µg/mL | PFU with Anti-EXCT4-mgG-2 Abs | PFU with Anti-gD-2 Abs | ||
|---|---|---|---|---|
| C− | C+ | C− | C+ | |
| 10 | 65 | 60 | 0 | 0 |
| 5.0 | 69 | 66 | 0 | 0 |
| 2.5 | 70 | 72 | 0 | 0 |
| 1.25 | 68 | 75 | 0 | 0 |
| 0.63 | 72 | 70 | 0 | 0 |
| 0.31 | 75 | 73 | 0 | 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liljeqvist, J.-Å.; Önnheim, K.; Tunbäck, P.; Eriksson, K.; Görander, S.; Bäckström, M.; Bergström, T. Human Antibodies against Herpes Simplex Virus 2 Glycoprotein G Do Not Neutralize but Mediate Antibody-Dependent Cellular Cytotoxicity. Antibodies 2024, 13, 40. https://doi.org/10.3390/antib13020040
Liljeqvist J-Å, Önnheim K, Tunbäck P, Eriksson K, Görander S, Bäckström M, Bergström T. Human Antibodies against Herpes Simplex Virus 2 Glycoprotein G Do Not Neutralize but Mediate Antibody-Dependent Cellular Cytotoxicity. Antibodies. 2024; 13(2):40. https://doi.org/10.3390/antib13020040
Chicago/Turabian StyleLiljeqvist, Jan-Åke, Karin Önnheim, Petra Tunbäck, Kristina Eriksson, Staffan Görander, Malin Bäckström, and Tomas Bergström. 2024. "Human Antibodies against Herpes Simplex Virus 2 Glycoprotein G Do Not Neutralize but Mediate Antibody-Dependent Cellular Cytotoxicity" Antibodies 13, no. 2: 40. https://doi.org/10.3390/antib13020040
APA StyleLiljeqvist, J.-Å., Önnheim, K., Tunbäck, P., Eriksson, K., Görander, S., Bäckström, M., & Bergström, T. (2024). Human Antibodies against Herpes Simplex Virus 2 Glycoprotein G Do Not Neutralize but Mediate Antibody-Dependent Cellular Cytotoxicity. Antibodies, 13(2), 40. https://doi.org/10.3390/antib13020040






