Types of Xenogenic Olivine from Siberian Kimberlites
Abstract
:1. Introduction
2. Materials and Methods
3. Olivine in Kimberlites
3.1. Olivines Crystallized from Kimberlite Melt
3.2. Olivines of Cr-Poor Megacryst Assemblage
3.3. Peridotitic Olivines
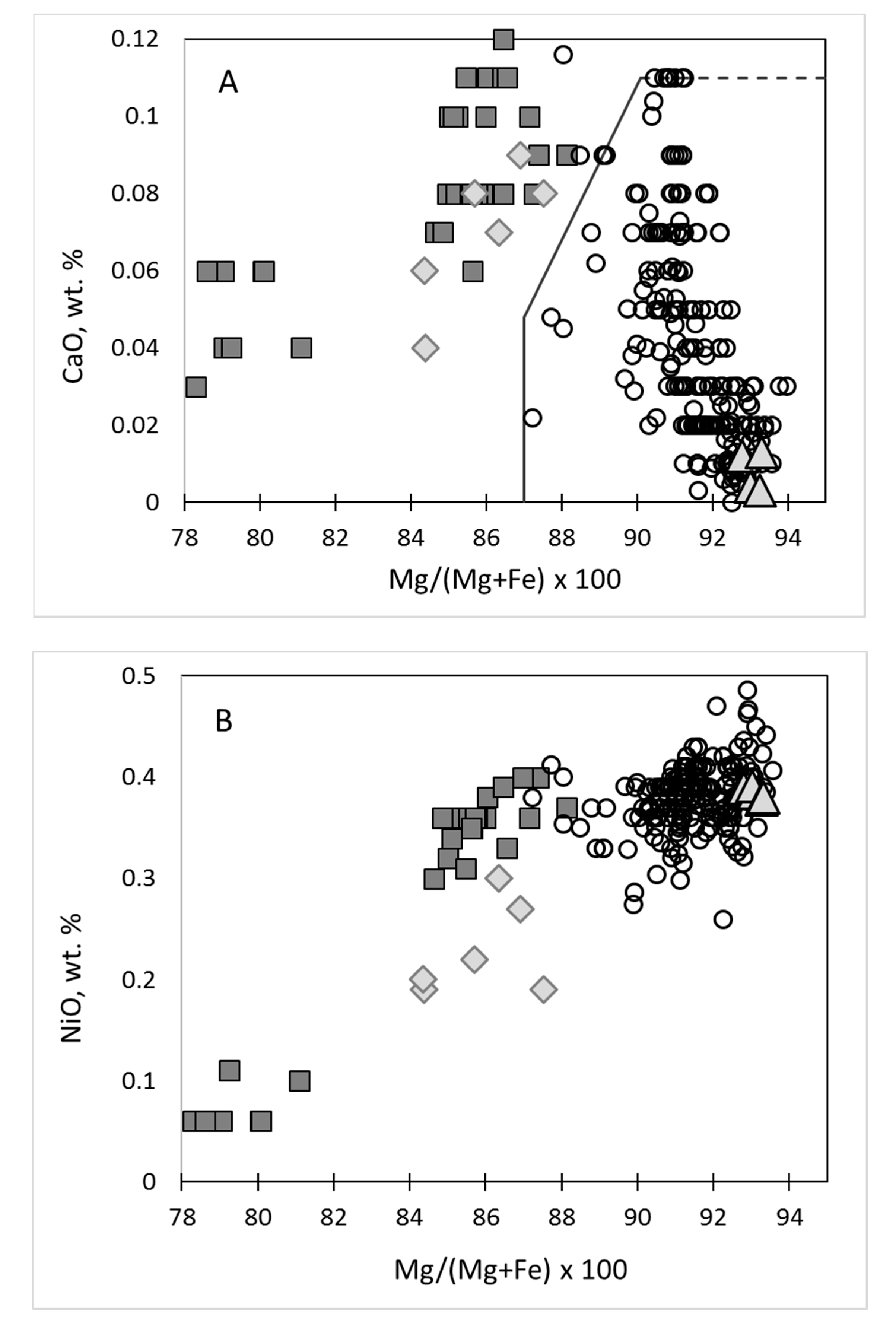
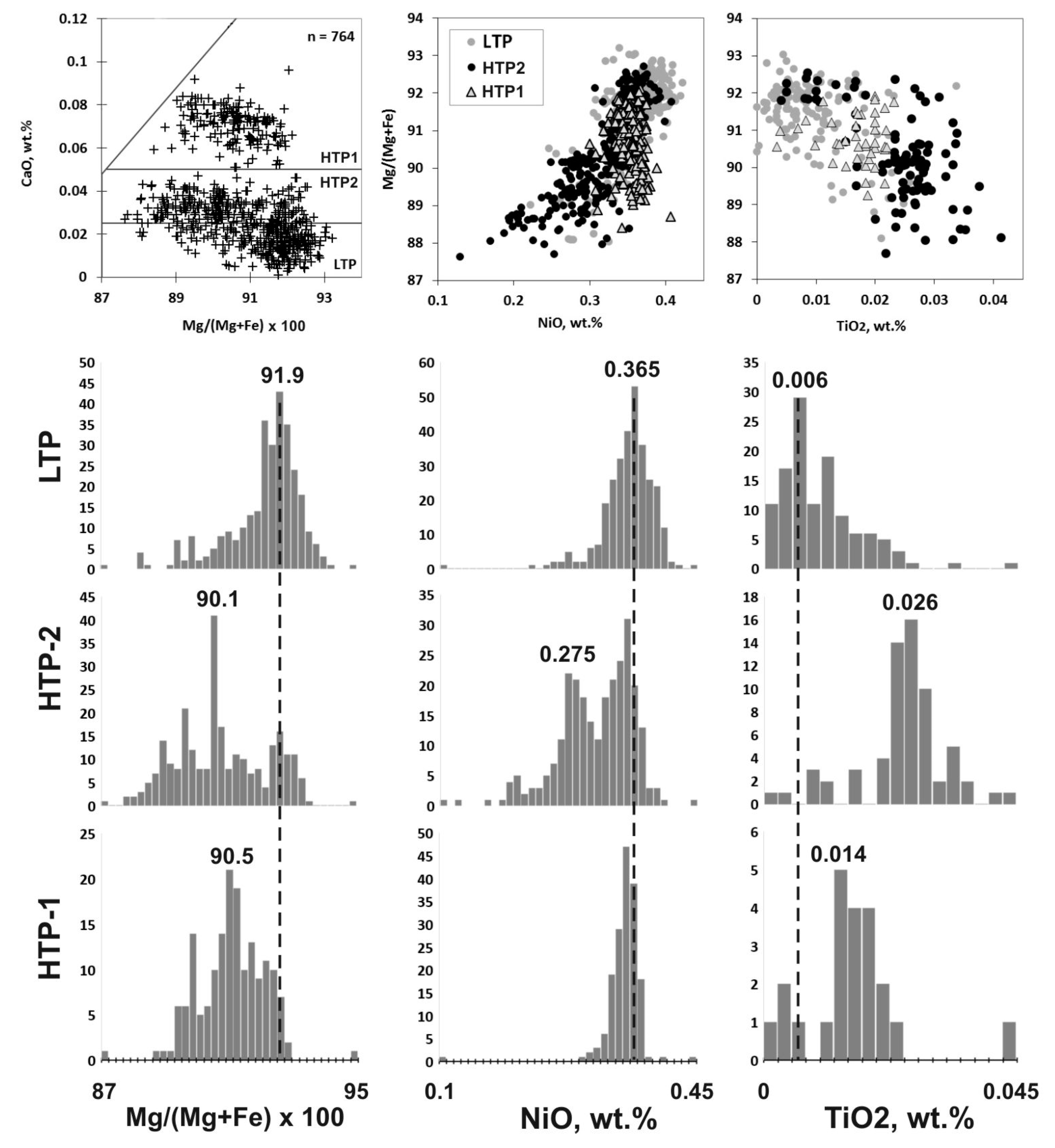
4. Classification Scheme for Mantle-Derived Olivines in Kimberlite
4.1. Craton Peridotitic, Megacrystic, and Magmatic Olivines
4.2. Different Olivines from Peridotites
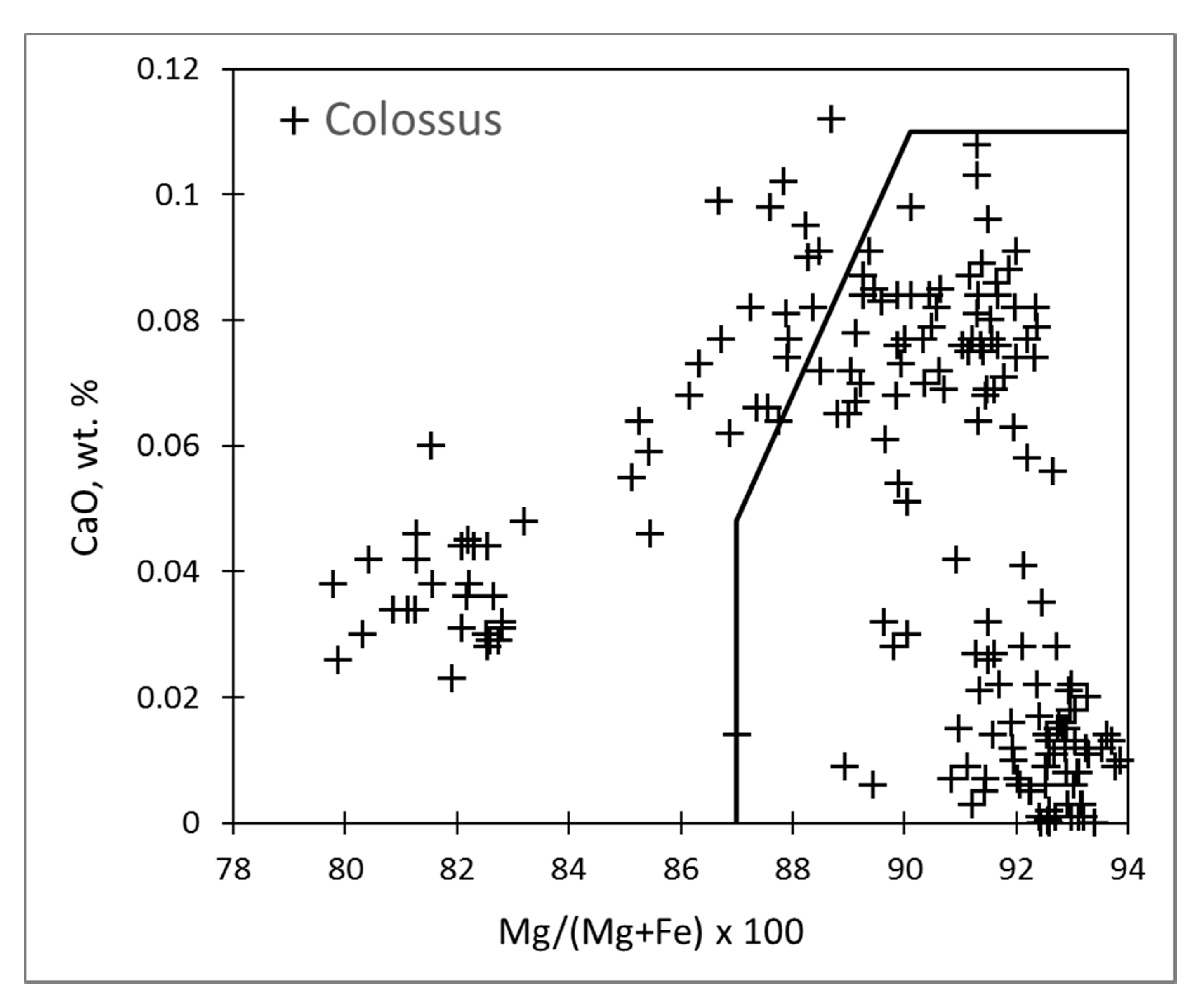
5. Distribution of Olivine Types in Sampled Kimberlites
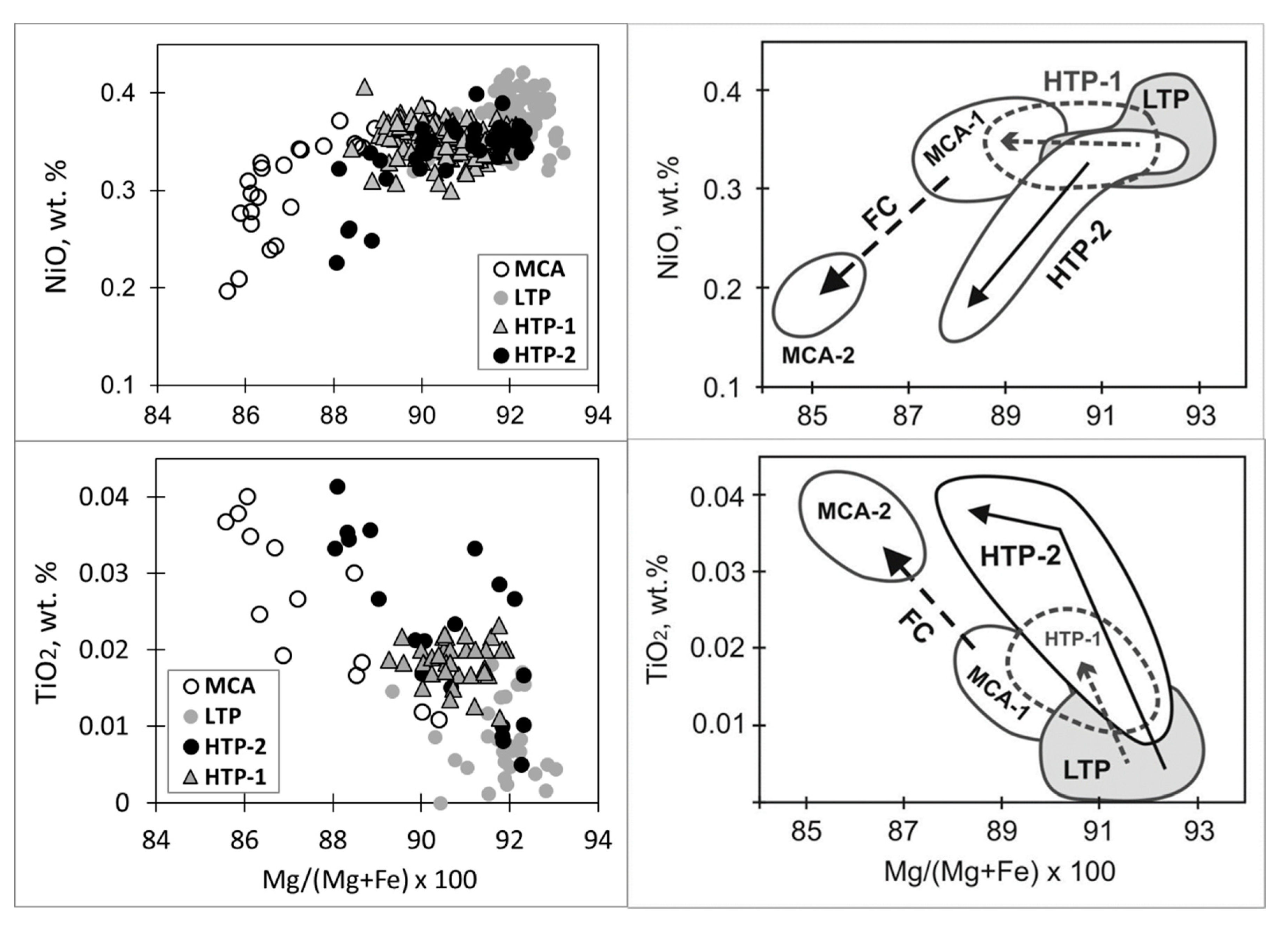
6. Mineral Chemistry of Olivines of Different Types
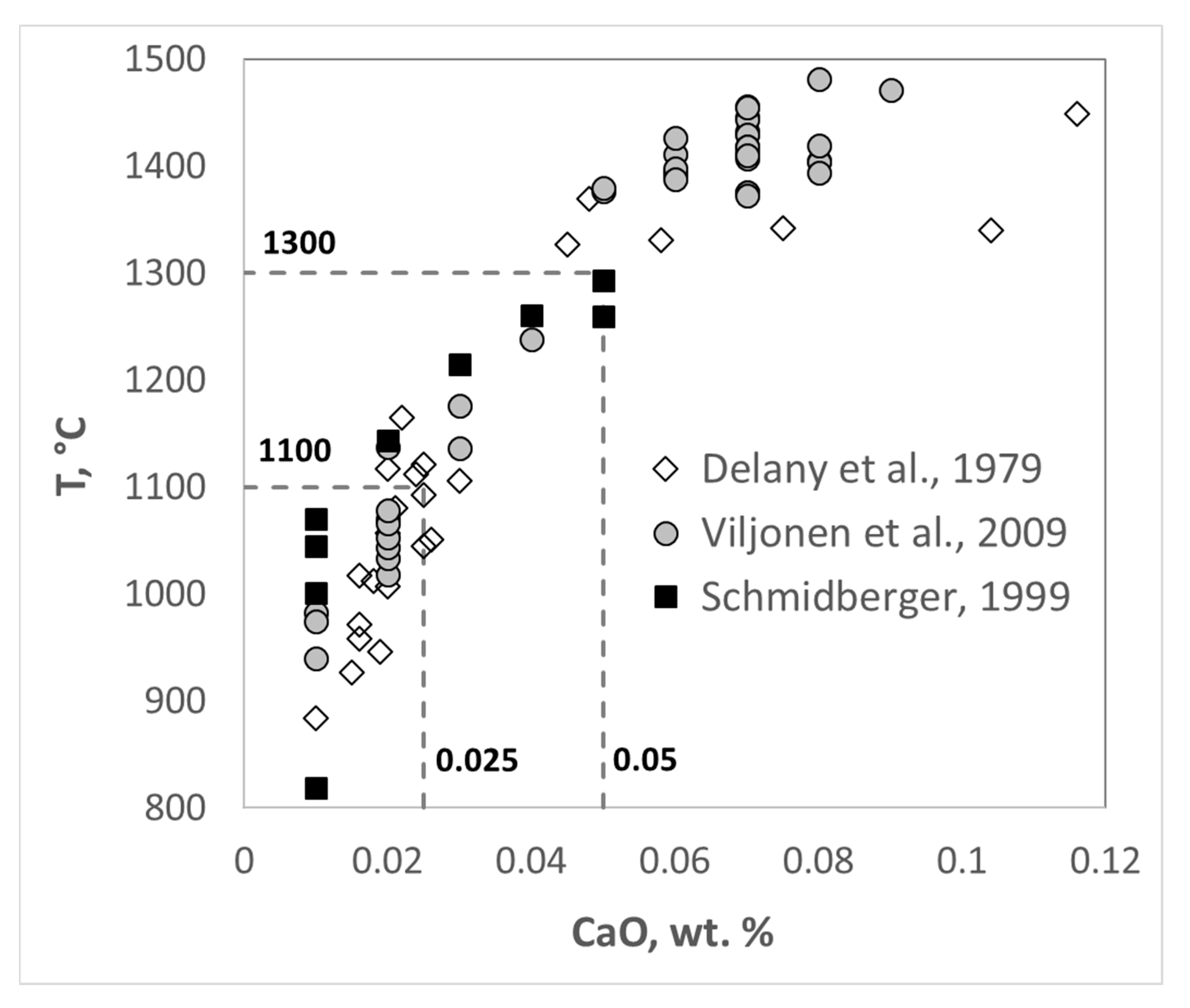
7. Origin of Different Olivine Types
8. Discussion and Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nixon, P.H. Mantle Xenoliths; Wiley: New York, NY, USA, 1987. [Google Scholar]
- Pearson, D.G.; Canil, D.; Shirey, B. Mantle Samples Included in Volcanic Rocks: Xenoliths and Diamonds. In The Mantle and Core: Treatise on Geochemistry; Elsevier: New York, NY, USA, 2003; Volume 2, pp. 171–275. [Google Scholar]
- Mitchell, R.H. Composition of olivine, silica activity and oxygen fugacity in kimberlite. Lithos 1973, 6, 65–81. [Google Scholar] [CrossRef]
- Mitchell, R.H. Kimberlites: Mineralogy, Geochemistry and Petrology; Plenum Press: New York, NY, USA, 1986; 442p. [Google Scholar]
- Arndt, N.T.; Guitreau, M.; Boullier, A.-M.; Le Roex, A.; Tommasi, A.; Cordier, P.; Sobolev, A. Olivine and the origin of kimberlite. J. Petrol. 2010, 51, 573–602. [Google Scholar] [CrossRef] [Green Version]
- Kamenetsky, V.S.; Kamenetsky, M.B.; Weiss, Y.; Navon, O.; Nielsen, T.F.D.; Mernagh, T.P. How unique is the Udachnaya-East kimberlite? Comparison with kimberlites from the Slave Craton (Canada) and SW Greenland. Lithos 2009, 112, 334–346. [Google Scholar] [CrossRef]
- Sobolev, N.V.; Sobolev, A.V.; Tomilenko, A.A.; Kovyazin, S.V.; Batanova, V.G.; Kuz’min, D.V. Paragenesis and complex zoning of olivine macrocrysts from unaltered kimberlite of the Udachnaya-East pipe, Yakutia: Relationship with the kimberlite formation conditions and evolution. Russ. Geol. Geophys. 2015, 56, 260–279. [Google Scholar] [CrossRef]
- Moore, A.; Belousova, E. Crystallization of Cr-poor and Cr-rich megacryst suites from the host kimberlite magma: Implications for mantle structure and the generation of kimberlite magmas. Contrib. Mineral. Petrol. 2005, 149, 462–481. [Google Scholar] [CrossRef]
- Hops, J.; Gurney, J.J.; Harte, B. The Jagersfontein Cr-poor megacryst suite—Towards a model for megacryst petrogenesis. J. Volcanol. Geotherm. Res. 1992, 50, 143–160. [Google Scholar] [CrossRef]
- Brett, R.C.; Russell, J.K.; Moss, S. Origin of olivine in kimberlite: Phenocryst or imposter. Lithos 2009, 1125, 20–212. [Google Scholar] [CrossRef]
- Giuliani, A. Insights into kimberlite petrogenesis and mantle metasomatism from a review of the compositional zoning of olivine in kimberlites worldwide. Lithos 2018, 312–313, 322–342. [Google Scholar] [CrossRef]
- Sobolev, N.V.; Sobolev, A.V.; Tomilenko, A.A. Unique compositional peculiarities of Olivine Phenocrysts from the post flood basalt diamondiferous Malokuonapskaya Kimberlite Pipe, Yakutia. Dokl. Earth. Sci. 2015, 463, 828–832. [Google Scholar] [CrossRef]
- Sobolev, V.S.; Nai, B.S.; Sobolev, N.V.; Lavrent’ev, Y.G.; Pospelova, L.N. Xenoliths of diamond-bearing pyrope serpentinites from the Aikhal kimberlite, Yakutia. Dokl. Akad. Nauk SSSR 1969, 188, 1141–1143. [Google Scholar]
- Schulze, D.J. A classification scheme for mantle-derived garnets in kimberlite: A tool for investigating the mantle and exploring for diamonds. Lithos 2003, 71, 195–213. [Google Scholar] [CrossRef]
- Ramsay, R.R.; Tompkins, L.A.; Meyer, H.O.A.; Leonardos, O.H. The geology, heavy mineral concentrate mineralogy, and diamond prospectivity of the Boa Esperanca and Cana Verde pipes, Corrego D’anta, Minas, Brazil. In Proceedings of the 5th International Kimberlite Conference; Companhia de Pesquisa de Recursos Minerais: Brasilia, Brazil, 1994; Volume 1, pp. 329–345. [Google Scholar]
- Nimis, P.; Taylor, W.R. Single clinopyroxene thermobarometry for garnet peridotites. Part I. Calibration and testing of a Cr-in-Cpx barometer and an enstatite-in-Cpx thermometer. Contrib. Mineral. Petrol. 2000, 139, 541–554. [Google Scholar] [CrossRef]
- Tychkov, N.S.; Yudin, D.S.; Nikolenko, E.I.; Sobolev, N.V. Mesozoic lithospheric mantle of the north-eastern part of the Siberian Platform according to the data of minerals from kimberlites. Russ. Geol. Geophys. 2018, 59, 1254–1270. [Google Scholar] [CrossRef]
- Korolyuk, V.N.; Pokhilenko, L.N. Electron probe determination of trace elements in olivine. X-Ray Spectrom. 2014, 43, 353–358. [Google Scholar] [CrossRef]
- Korolyuk, V.N.; Lavrent’ev, Y.G.; Usova, L.V.; Nigmatulina, E.N. JXA-8100 microanalyzer: Accuracy of analysis of rock-forming minerals. Russ. Geol. Geophys. 2008, 49, 165–168. [Google Scholar] [CrossRef]
- Lavrent’ev, Y.G.; Korolyuk, V.N.; Usova, L.V.; Nigmatulina, E.N. Electron probe microanalysis of rock-forming minerals with JXA-8100 electron probe microanalyzer. Russ. Geol. Geophys. 2015, 56, 1428–1436. [Google Scholar]
- Kamenetsky, V.; Kamenetsky, M.; Sobolev, A.; Golovin, A.; Demouchy, S.; Faure, K.; Sharygin, V.; Kuzmin, D. Olivine in the Udachnaya-East kimberlite (Yakutia, Russia): Types, compositions and origins. J. Petrol. 2008, 49, 823–839. [Google Scholar] [CrossRef]
- Sobolev, N.V.; Logvinova, A.M.; Zedgenizov, D.A.; Pokhilenko, N.P.; Malygina, E.V.; Kuzmin, D.V.; Sobolev, A.V. Petrogenetic significance of minor elements in olivines from diamonds and peridotite xenoliths from kimberlites of Yakutia. Lithos 2009, 1125, 701–713. [Google Scholar] [CrossRef]
- Skinner, E.M.W. Contrasting Group I and Group II kimberlite petrology: Towards a genetic model for kimberlites. In Proceedings of The 4th International Kimberlite Conference: Extended Abstracts; Geology Society of Australia: Perth, Australia, 1989; pp. 528–544. [Google Scholar]
- Moore, A.E. Olivine: A monitor of magma evolutionary paths in kimberlite and olivine melilitites. Contrib. Mineral. Petrol. 1988, 99, 238–248. [Google Scholar] [CrossRef]
- Moore, A.E. The case for a cognate, polybaric origin for kimberlitic olivines. Lithos. 2012, 128–131, 1–10. [Google Scholar] [CrossRef]
- Nielsen, T.F.D.; Sand, K.K. The Majuagaa kimberlite dyke, Maniitsoq region, west Greenland: Constraints on an Mg-rich silicocarbonatitic melt composition from groundmass mineralogy and bulk composition. Can. Mineral. 2008, 46, 1043–1061. [Google Scholar] [CrossRef]
- Bussweiler, Y.; Foley, S.; Prelević, D.; Jakob, J.E. The olivine macrocryst problem: New insights from minor and trace element compositions of olivine from Lac de Gras kimberlites, Canada. Lithos. 2015, 220–223, 238–252. [Google Scholar] [CrossRef]
- Harte, B. Rock nomenclature with particular reference to deformation and recrystallization textures in olivine-bearing xenoliths. J. Geol. 1977, 89, 749–753. [Google Scholar] [CrossRef]
- Moore, A.; Costin, G. Kimberlitic olivines derived from the Cr-poor and Cr-rich megacryst suites. Lithos 2016, 258–259, 215–227. [Google Scholar] [CrossRef]
- Delany, J.S.; Smith, J.V.; Dawson, J.B.; Nixon, P.H. Manganese thermometer for mantle peridotites. Contrib. Mineral Petrol. 1979, 71, 157–169. [Google Scholar] [CrossRef]
- MacKenzie, J.M.; Canil, D. Composition and thermal evolution of cratonic mantle beneath the central Archean Slave Province, NWT, Canada. Contrib. Mineral Petrol. 1999, 134, 313–324. [Google Scholar] [CrossRef]
- Schmidberger, S.S.; Francis, D. Nature of the mantle roots beneath the North American Craton: Mantle xenolith evidence from Somerset Island kimberlites. Lithos 1999, 48, 195–216. [Google Scholar] [CrossRef]
- Gregoire, M.; Bell, D.R.; Le Roex, A.P. Garnet lherzolites from the Kaapvaal Craton (South Africa): Trace element evidence for a metasomatic history. J. Petrol. 2003, 44, 629–657. [Google Scholar] [CrossRef] [Green Version]
- Boyd, F.R.; Pearson, D.G.; Hoal, K.O.; Hoal, B.G.; Nixon, P.H.; Kingston, M.J.; Mertzman, S.A. Garnet lherzolites from Louwrensia, Namibia: Bulk composition and P/T relations. Lithos 2004, 77, 573–592. [Google Scholar] [CrossRef]
- Viljoen, F.; Dobbe, R.; Smit, B. Geochemical processes in peridotite xenoliths from the Premier diamond mine, South Africa: Evidence for the depletion and refertilisation of subcratonic lithosphere. Lithos 2009, 112, 1133–1142. [Google Scholar] [CrossRef]
- Menzies, A.H.; Westerlund, K.; Grutter, H.; Gurney, J.J.; Carlson, J.A.; Fung, A.; Nowicki, T. Peridotitic mantle xenoliths from kimberlites on the Ekati diamond mine property, NWT, Canada: Major element compositions and implications for the lithosphere beneath the central Slave craton. Lithos 2004, 77, 395–412. [Google Scholar] [CrossRef]
- Jakob, W.R.O. Geochemical Aspects of the Megacryst Suite from the Monastery Kimberlite Pipe. Master’s Thesis, University of Cape Town, Cape Town, South Africa, 1977; p. 81. [Google Scholar]
- Kopylova, M.G.; Russell, J.K.; Cookenboo, H. Petrology of peridotite and pyroxenite xenoliths from the Jericho kimberlite: Implications for the thermal state of the mantle beneath the Slave Craton, northern Canada. J. Petrol. 1999, 40, 79–104. [Google Scholar] [CrossRef]
- Bizzarro, M.; Stevenson, R. Major element composition of the lithospheric mantle under the North Atlantic craton: Evidence from peridotite xenoliths of the Sarfartoq area, southwestern Greenland. Contrib. Mineral. Petrol. 2003, 146, 223–240. [Google Scholar] [CrossRef]
- Pokhilenko, L.N.; Mal’kovets, V.G.; Kuz’min, D.V.; Pokhilenko, N.P. New data on the mineralogy of megacrystalline pyrope peridotite from the Udachnaya kimberlite pipe, Siberian Craton, Yakutian diamondiferous province. Dok. Earth Sci. 2014, 454, 179–184. [Google Scholar] [CrossRef]
- Foley, S.F.; Prelevic, D.; Rehfeldt, T.; Jacob, D.E. Minor and trace elements in olivines as probes into early igneous and mantle melting processes, Earth Planet. Sci. Lett. 2013, 363, 181–191. [Google Scholar]
- Pokhilenko, N.P.; Sobolev, N.V.; Boyd, F.R.; Pearson, D.J.; Shimizu, N. Megacrystalline pyrope peridotites in the lithosfere of the Siberian Platform: Mineralogy, geochemical peculiarities and the problem of their origin. Russ. Geol. Geophys. 1993 34, 56–67.
- De Hoog, J.C.M.; Gall, L.; Cornell, D.H. Trace-element geochemistry of mantle olivine and application to mantle petrogenesis and geothermobarometry. Chem. Geol. 2010, 270, 196–215. [Google Scholar] [CrossRef] [Green Version]
- Köhler, T.P.; Brey, G.P. Calcium exchange between olivine and clinopyroxene calibrated as a geothermobarometer for natural peridotites from 2 to 60 kb with applications. Geochim. Cosmochim. Acta 1990, 54, 2375–2388. [Google Scholar]
- Brey, G.P.; Kohler, T. Geothermobarometry in four-phase lherzolites II. New thermobarometers, and practical assessment of existing thermobarometers. J. Geol. 1990, 31, 1353–1378. [Google Scholar] [CrossRef]
- O’Neill, H.S.C.; Wood, B.J. An experimental study of Fe–Mg partitioning between garnet and olivine and its calibration as a geothermometer. Contrib. Mineral. Petrol. 1979, 70, 59–70. [Google Scholar]
- Gibsher, A.A.; Malkovets, V.G.; Litasov, K.D.; Litasov, Y.D.; Pokhilenko, N.P. Composition of the ordovician lithospheric mantle: Evidence from the study of peridotite xenoliths from camptonite of the Sangilen highland, Central Asian Fold Belt. Dokl. Earth Sci. 2010, 433, 957–961. [Google Scholar] [CrossRef]
- Amundsen, H.E.F.; Griffin, W.L.; O’Reilly, S.Y. The lower crust and upper mantle beneath northwestern Spitsbergen: Evidence from xenoliths and geophysics. Tectonophysics 1987, 139, 169–185. [Google Scholar] [CrossRef]
- Pokhilenko, N.P.; Sobolev, N.V.; Kuligin, S.S.; Shimizu, N. Peculiarities of distribution of pyroxenite paragenesis garnet in Yakutian kimberlites and some aspects of the Siberian craton lithospheric mantle. In Proceedings of the 7th International Kimberlite Conference; Red Roof Design: Cape Town, South Africa, 1999; Volume 2, pp. 689–698. [Google Scholar]
- Sobolev, N.V.; Yefimova, E.S.; Koptil, V.I. Mineral inclusions in diamonds in the Northeast of the Yakutian diamondiferous province. In Proceedings of the 7th Kimberlite Conference, Cape Town, South Africa, 11–17 April 1999; pp. 816–823. [Google Scholar]
- Zinchuk, N.I.; Koptil, V.I. Typomorphism of Diamonds in the Siberian Craton; Nedra: Moscow, Russia, 2003. [Google Scholar]
- Sobolev, N.V.; Sobolev, A.V.; Tomilenko, A.A.; Kuz’min, D.V.; Grakhanov, S.A.; Batanova, V.G.; Logvinova, A.M.; Bul’bak, T.A.; Kostrovitskii, S.I.; Yakovlev, D.A.; et al. Prospects of search for diamondiferous kimberlites in the northeastern Siberian Platform. Russ. Geol. Geophys. 2018, 59, 1365–1379. [Google Scholar] [CrossRef]
- Sun, J.; Liu, C.Z.; Kostrovisky, S.I.; Wu, F.Y.; Yang, J.H.; Chu, Z.Y.; Yang, Y.H.; Kalashnikova, T.; Fan, S. Composition of the lithospheric mantle in the northern part of Siberian craton: Constraints from peridotites in the Obnazhennaya kimberlite. Lithos 2017, 294, 383–396. [Google Scholar] [CrossRef]
- Howarth, G.H.; Barry, P.H.; Pernet-Fisher, J.F.; Baziotis, I.P.; Pokhilenko, N.P.; Pokhilenko, L.N.; Bodnar, R.J.; Taylor, L.A. Superplume metasomatism: Evidence from Siberian mantle xenoliths. Lithos. 2014, 184–187, 209–224. [Google Scholar] [CrossRef]
- Ionov, D.A.; Doucet, L.S.; Ashchepkov, I.V. Composition of the lithospheric mantle in the Siberian craton: New constraints from fresh peridotites in the Udachnaya-East kimberlite. J. Petrol. 2010, 51, 2177–2210. [Google Scholar] [CrossRef] [Green Version]
- Solovyeva, L.V.; Yasnygina, T.A.; Egorov., K.N. Metasomatic parageneses in deep-seated xenoliths from Udachnaya and Komsomolskaya-Magnitnaya pipes as indicators of fluid transfer through the mantle lithosphere of the Siberian craton. Russ. Geol. Geophys. 2012, 53, 1304–1323. [Google Scholar] [CrossRef]
- Tychkov, N.S.; Agashev, A.M.; Malygina, E.V.; Nikolenko, E.I.; Pokhilenko, N.P. Thermal perturbations in the lithospheric mantle: Case study of PT equilibrium conditions of xenoliths from the Udachnaya pipe. Dokl. Earth Sci. 2014, 454, 328–333. [Google Scholar] [CrossRef]
- Harte, B.; Hunter, R.H.; Kinny, P.D. Melt geometry, movement and crystallization in relation to mantle dykes, veins and metasomatism. Philos. Trans. R. Soc. Lond. 1993, 342, 1–21. [Google Scholar]
- Moore, R.O.; Griffin, W.L.; Gurney, J.J.; Ryan, C.G.; Cousens, D.R.; Sie, S.H.; Suter, G.F. Trace element geochemistry of ilmenite megacrysts from the Monastery kimberlite, South Africa. Lithos 1992, 29, 1–18. [Google Scholar] [CrossRef]
- Agashev, A.M. Geochemistry of Garnet Megacrysts from the Mir Kimberlite Pipe (Yakutia) and the Nature of Protokimberlite Melts. Dokl. Earth Sc. 2019, 486, 675–678. [Google Scholar] [CrossRef]
- Burgess, S.R.; Harte, B. Tracing lithosphere evolution through the analysis of heterogeneous G9/G10 garnet in peridotite xenoliths. I: Major element chemistry. In Proceedings of the 7th Kimberlite Conference (Dawson volume), Cape Town, South Africa, 11–17 April 1999. [Google Scholar]
- Burgess, S.R.; Harte, B. Tracing lithosphere evolution through the analysis of heterogeneous G9–G10 garnets in peridotite xenoliths, II: REE chemistry. J. Petrol. 2004, 45, 609–633. [Google Scholar] [CrossRef]
- Cox, K.G.; Bell, J.D.; Pankhurst, R.J. The Interpretation of Igneous Rocks; George Allen Unwin: London, UK, 1979. [Google Scholar]
- Cox, K.G.; Jamieson, B.G. The Olivine-rich Lavas of Nuanetsi: A Study of Polybaric Magmatic Evolution. J. Petrol. 1974, 15, 269–301. [Google Scholar] [CrossRef]
- Brey, G.P.; Köhler, T.; Nickel, K.G. Geothermobarometry in four-phase lherzolites I. Experimental results from 10 to 60 kb. J. Petrol. 1990, 31, 1313–1352. [Google Scholar] [CrossRef]





| Kimberlite | MCP | HTP-1 | HTP-2 | LTP | n |
|---|---|---|---|---|---|
| Obnajennaya | 0 | 0 | 0 | 100 | 110 |
| Vtorogodnitsa | 1 | 1 | 56 | 42 | 342 |
| Olivinovaya | 1 | 5 | 42 | 52 | 88 |
| Dianga | 10 | 46 | 12 | 32 | 334 |
| Colossus | 29 | 30 | 6 | 34 | 209 |
| Types | Mg# | NiO | CaO | TiO2 |
|---|---|---|---|---|
| Granular | 87.8–94.0 | 0.26–0.48 | 0–0.092 | 0–0.008 |
| LTP | 89.8–93.1 | 0.34–0.42 | 0–0.025 | 0–0.022 |
| Sheared | 86.0–92.2 | 0.22–0.42 | 0.032–0.091 | 0.017–0.050 |
| HTP-1 + HTP-2 | 88.0–92.5 | 0.19–0.40 | 0.025–0.089 | 0.008–0.041 |
| MCA | 80.0–90.0 | 0.10–0.38 | 0.022–0.10 | 0.011–0.040 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tychkov, N.S.; Agashev, A.M.; Pokhilenko, N.P.; Tsykh, V.A.; Sobolev, N.V. Types of Xenogenic Olivine from Siberian Kimberlites. Minerals 2020, 10, 302. https://doi.org/10.3390/min10040302
Tychkov NS, Agashev AM, Pokhilenko NP, Tsykh VA, Sobolev NV. Types of Xenogenic Olivine from Siberian Kimberlites. Minerals. 2020; 10(4):302. https://doi.org/10.3390/min10040302
Chicago/Turabian StyleTychkov, Nikolay S., Alexey M. Agashev, Nikolay P. Pokhilenko, Vladimir A. Tsykh, and Nikolay V. Sobolev. 2020. "Types of Xenogenic Olivine from Siberian Kimberlites" Minerals 10, no. 4: 302. https://doi.org/10.3390/min10040302




