Effect of CaO Content and Annealing Treatment on the Room-Temperature Mechanical Properties of AZ61 and AZ61-CaO Alloys
Abstract
:1. Introduction
2. Materials and Methods
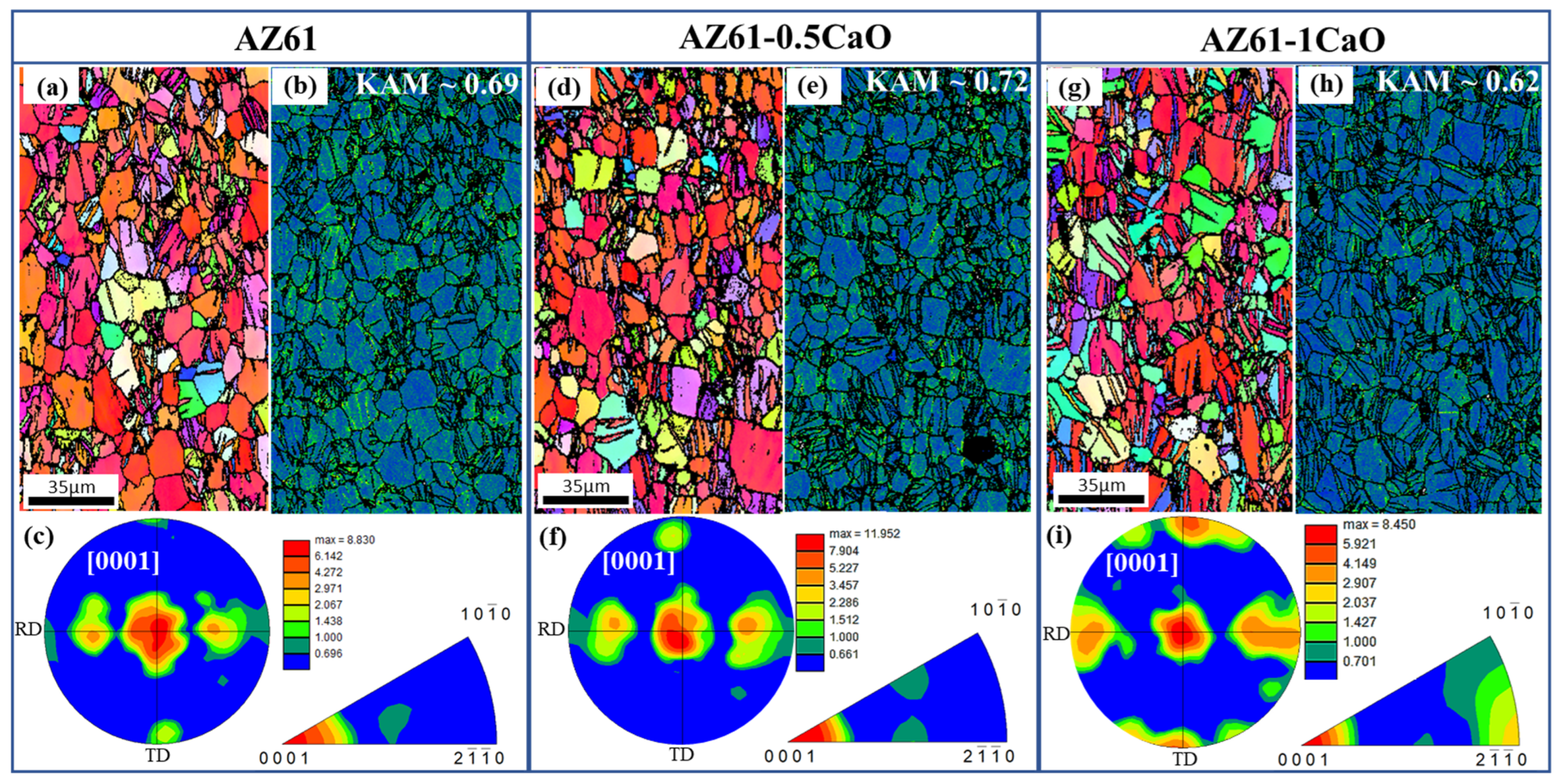
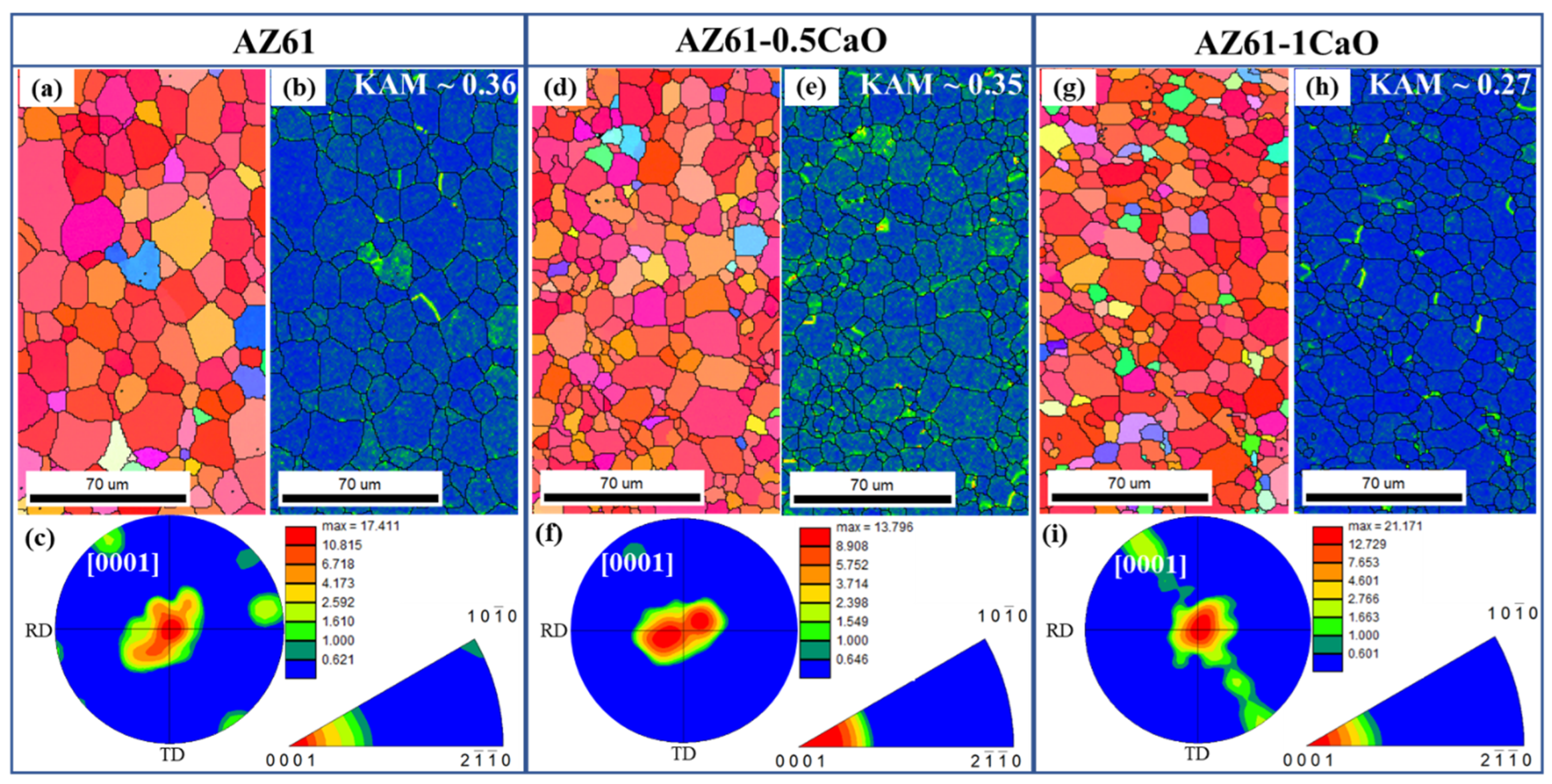
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Masood Chaudry, U.; Hamad, K.; Kim, J.-G. A further improvement in the room-temperature formability of magnesium alloy sheets by pre-stretching. Materials 2020, 13, 2633. [Google Scholar] [CrossRef] [PubMed]
- Atrens, A.; Song, G.L.; Liu, M.; Shi, Z.; Cao, F.; Dargusch, M.S. Review of recent developments in the field of magnesium corrosion. Adv. Eng. Mater. 2015, 17, 400–453. [Google Scholar] [CrossRef]
- Song, B.; Guo, N.; Liu, T.; Yang, Q. Improvement of formability and mechanical properties of magnesium alloys via pre-twinning: A review. Mater. Des. 2014, 62, 352–360. [Google Scholar] [CrossRef]
- Malik, A.; Wang, Y.; Huanwu, C.; Nazeer, F.; Khan, M.A. What is the major problem with wrought Mg alloys? Results Engineering 2020, 7, 100162. [Google Scholar] [CrossRef]
- Malik, A.; Wang, Y.; Huanwu, C.; Bhatti, T.M.; Nazeer, F. Superplastic behavior of fine-grained extruded ZK61 Mg alloy. Results Phys. 2021, 20, 103731. [Google Scholar] [CrossRef]
- Malik, A.; Wang, Y.; Cheng, H.; Nazeer, F.; Khan, M.A. Microstructure evolution of Mg-Zn-Zr magnesium alloy against soft steel core projectile. J. Mater. Sci. Technol. 2021, 79, 46–61. [Google Scholar] [CrossRef]
- Guan, D.; Rainforth, W.M.; Ma, L.; Wynne, B.; Gao, J. Twin recrystallization mechanisms and exceptional contribution to texture evolution during annealing in a magnesium alloy. Acta Mater. 2017, 126, 132–144. [Google Scholar] [CrossRef]
- Hadorn, J.P.; Mulay, R.P.; Hantzsche, K.; Yi, S.; Bohlen, J.; Letzig, D.; Agnew, S.R. Texture Weakening Effects in Ce-Containing Mg Alloys. Met. Mater. Trans. A 2013, 44, 1566–1576. [Google Scholar] [CrossRef]
- Jia, W.; Hu, X.; Zhao, H.; Ju, D.; Chen, D. Texture evolution of AZ31 magnesium alloy sheets during warm rolling. J. Alloys Compd. 2015, 645, 70–77. [Google Scholar] [CrossRef]
- Chaudry, U.M.; Hamad, K.; Jun, T.-S. Investigating the Microstructure, Crystallographic Texture and Mechanical Behavior of Hot-Rolled Pure Mg and Mg-2Al-1Zn-1Ca Alloy. Crystals 2022, 12, 1330. [Google Scholar] [CrossRef]
- Park, S.H.; Lee, J.H.; Huh, Y.-H.; Hong, S.-G. Enhancing the effect of texture control using {1 0− 1 2} twins by retarding detwinning activity in rolled Mg–3Al–1Zn alloy. Scr. Mater. 2013, 69, 797–800. [Google Scholar] [CrossRef]
- Agnew, S.; Horton, J.; Lillo, T.; Brown, D. Enhanced ductility in strongly textured magnesium produced by equal channel angular processing. Scr. Mater. 2004, 50, 377–381. [Google Scholar] [CrossRef]
- Ansari, N.; Lee, S.Y.; Singh, S.S.; Jain, J. Effect of rolling routes on the microstructure, texture and mechanical properties of Mg-5Y binary alloy. Philos. Mag. 2023, 103, 840–859. [Google Scholar] [CrossRef]
- Wu, Z.; Curtin, W. The origins of high hardening and low ductility in magnesium. Nature 2015, 526, 62–67. [Google Scholar] [CrossRef]
- Barnett, M.R.; Nave, M.D.; Bettles, C.J. Deformation microstructures and textures of some cold rolled Mg alloys. Mater. Sci. Eng. A 2004, 386, 205–211. [Google Scholar] [CrossRef]
- Stanford, N.; Barnett, M. Effect of composition on the texture and deformation behaviour of wrought Mg alloys. Scr. Mater. 2008, 58, 179–182. [Google Scholar] [CrossRef]
- Watanabe, H.; Mukai, T.; Ishikawa, K. Effect of temperature of differential speed rolling on room temperature mechanical properties and texture in an AZ31 magnesium alloy. J. Mater. Process. Technol. 2007, 182, 644–647. [Google Scholar] [CrossRef]
- Wang, L.; Mostaed, E.; Cao, X.; Huang, G.; Fabrizi, A.; Bonollo, F.; Chi, C.; Vedani, M. Effects of texture and grain size on mechanical properties of AZ80 magnesium alloys at lower temperatures. Mater. Des. 2016, 89, 1–8. [Google Scholar] [CrossRef]
- Lee, H.-J.; Lee, S.K.; Jung, K.H.; Lee, G.A.; Ahn, B.; Kawasaki, M.; Langdon, T.G. Evolution in hardness and texture of a ZK60A magnesium alloy processed by high-pressure torsion. Mater. Sci. Eng. A 2015, 630, 90–98. [Google Scholar] [CrossRef]
- Ali, S.; Karunanithi, R.; Prashanth, M.; Rahman, M.A. X-ray peak broadening on microstructure, and structural properties of titanium and Ti-6Al-4V alloys. Mater. Today Proc. 2020, 27, 2390–2393. [Google Scholar] [CrossRef]
- Dobatkin, S.; Rokhlin, L.; Lukyanova, E.; Murashkin, M.Y.; Dobatkina, T.; Tabachkova, N.Y. Structure and mechanical properties of the Mg-Y-Gd-Zr alloy after high pressure torsion. Mater. Sci. Eng. A 2016, 667, 217–223. [Google Scholar] [CrossRef]
- Lukyanova, E.A.; Martynenko, N.S.; Serebryany, V.N.; Belyakov, A.N.; Rokhlin, L.L.; Dobatkin, S.V.; Estrin, Y.Z. Structure and mechanical and corrosion properties of a magnesium Mg–Y–Nd–Zr alloy after high pressure torsion. Russ. Metall. 2017, 2017, 912–921. [Google Scholar] [CrossRef]
- Zhao, C.; Li, Z.; Shi, J.; Chen, X.; Tu, T.; Luo, Z.; Cheng, R.; Atrens, A.; Pan, F. Strain hardening behavior of Mg–Y alloys after extrusion process. J. Magnes. Alloys 2019, 7, 672–680. [Google Scholar] [CrossRef]
- Zhang, L.; Wu, X.; Yang, X.; Li, Y. Static recrystallization and precipitation behavior of forged and annealed Mg–8.7 Gd–4.18 Y–0.42 Zr magnesium alloy. Mater. Today Commun. 2023, 34, 105106. [Google Scholar] [CrossRef]
- Mackenzie, L.; Pekguleryuz, M. The recrystallization and texture of magnesium–zinc–cerium alloys. Scr. Mater. 2008, 59, 665–668. [Google Scholar] [CrossRef]
- Yu, Z.; Xu, X.; Mansoor, A.; Du, B.; Shi, K.; Liu, K.; Li, S.; Du, W. Precipitate characteristics and their effects on the mechanical properties of as-extruded Mg-Gd-Li-Y-Zn alloy. J. Mater. Sci. Technol. 2021, 88, 21–35. [Google Scholar] [CrossRef]
- Yu, Z.; Liu, L.; Mansoor, A.; Liu, K.; Li, S.; Du, W. Microstructures and mechanical properties of as-extruded Mg–8Gd–2Y–1Zn–6Li alloy. J. Alloys Compd. 2021, 864, 158826. [Google Scholar] [CrossRef]
- Mansoor, A.; Du, W.; Yu, Z.; Ding, N.; Fu, J.; Jia, L.; Liu, K.; Li, S. Effect of Secondary Extrusion on the Microstructure and Mechanical Properties of Mg-12Gd-2Er-0.4 Zr Alloy. J. Mater. Eng. Perform. 2021, 30, 8996–9007. [Google Scholar] [CrossRef]
- Kocich, R.; Kunčická, L.; Král, P.; Lowe, T.C. Texture, deformation twinning and hardening in a newly developed Mg–Dy–Al–Zn–Zr alloy processed with high pressure torsion. Mater. Des. 2016, 90, 1092–1099. [Google Scholar] [CrossRef]
- Chaudry, U.M.; Noh, Y.; Han, G.; Jaafreh, R.; Jun, T.-S.; Hamad, K. Effect of CaO on structure and properties of AZ61 magnesium alloy. Mater. Sci. Eng. A 2022, 844, 143189. [Google Scholar] [CrossRef]
- Bhattacharyya, J.J.; Agnew, S.; Muralidharan, G. Texture enhancement during grain growth of magnesium alloy AZ31B. Acta Mater. 2015, 86, 80–94. [Google Scholar] [CrossRef]
- Kim, S.-H.; You, B.-S.; Yim, C.D.; Seo, Y.-M. Texture and microstructure changes in asymmetrically hot rolled AZ31 magnesium alloy sheets. Mater. Lett. 2005, 59, 3876–3880. [Google Scholar] [CrossRef]
- Kim, H.; Lee, J.-H.; Lee, C.; Bang, W.; Ahn, S.; Chang, Y. Shear band formation during hot compression of AZ31 Mg alloy sheets. Mater. Sci. Eng. A 2012, 558, 431–438. [Google Scholar] [CrossRef]
- Pei, R.; Zou, Y.; Zubair, M.; Wei, D.; Al-Samman, T. Synergistic effect of Y and Ca addition on the texture modification in AZ31B magnesium alloy. Acta Mater. 2022, 233, 117990. [Google Scholar] [CrossRef]
- Lee, J.U.; Kim, Y.J.; Park, S.H. Microstructure evolution maps of AZ31 and AZ91–Ca–Y–MM alloys under precompression and subsequent annealing conditions. Mater. Sci. Eng. A 2022, 854, 143814. [Google Scholar] [CrossRef]
- Li, X.; Yang, P.; Wang, L.-N.; Meng, L.; Cui, F.-E. Orientational analysis of static recrystallization at compression twins in a magnesium alloy AZ31. Mater. Sci. Eng. A 2009, 517, 160–169. [Google Scholar] [CrossRef]
- Agnew, S. Plastic anisotropy of magnesium alloy AZ31B sheet. In Essential Readings in Magnesium Technology; Springer: Berlin/Heidelberg, Germany, 2016; pp. 351–356. [Google Scholar]
- Hu, L.; Lv, H.; Shi, L.; Chen, Y.; Chen, Q.; Zhou, T.; Li, M.; Yang, M. Research on deformation mechanism of AZ31 magnesium alloy sheet with non-basal texture during uniaxial tension at room temperature: A visco-plastic self-consistent analysis. J. Magnes. Alloys 2022, 10, 1994–2008. [Google Scholar] [CrossRef]
- He, S.M.; Zeng, X.Q.; Peng, L.M.; Gao X-q Nie, J.F.; Ding, W.J. Precipitation in a Mg–10Gd–3Y–0.4 Zr (wt.%) alloy during isothermal ageing at 250 C. J. Alloys Compd. 2006, 421, 309–313. [Google Scholar] [CrossRef]
- Bourezg, Y.I.; Azzeddine, H.; Baudin, T.; Helbert, A.-L.; Huang, Y.; Bradai, D.; Langdon, T.G. Texture and microhardness of Mg-Rare Earth (Nd and Ce) alloys processed by high-pressure torsion. Mater. Sci. Eng. A 2018, 724, 477–485. [Google Scholar] [CrossRef]
- Wu, B.; Zhao, Y.; Du, X.; Zhang, Y.; Wagner, F.; Esling, C. Ductility enhancement of extruded magnesium via yttrium addition. Mater. Sci. Eng. A 2010, 527, 4334–4340. [Google Scholar] [CrossRef]
- Pei, Z.; Li, R. The effect of yttrium on the generalized stacking fault energies in Mg. Comput. Mater. Sci. 2017, 133, 1–5. [Google Scholar] [CrossRef]
- Kim, K.-H.; Jeon, J.B.; Kim, N.J.; Lee, B.-J. Role of yttrium in activation of< c+ a> slip in magnesium: An atomistic approach. Scr. Mater. 2015, 108, 104–108. [Google Scholar]
- Chaudry, U.M.; Noh, Y.; Hamad, K.; Jun, T.-S. Effect of deformation temperature on the slip activity in pure Mg and AZX211. J. Mater. Res. Technol. 2022, 19, 3406–3420. [Google Scholar] [CrossRef]
- Ha, C.; Bohlen, J.; Zhou, X.; Brokmeier, H.-G.; Kainer, K.; Schell, N.; Letzig, D.; Yi, S. Texture development and dislocation activities in Mg-Nd and Mg-Ca alloy sheets. Mater. Charact. 2021, 175, 111044. [Google Scholar] [CrossRef]
- Guo, F.; Jiang, L.; Yang, M.; Ma, Y.; Deng, Y.; Zhang, D.; Pan, F. Tailoring the rolling texture of AZ31 Mg Alloy with Calcium and Tin Addition. Adv. Eng. Mater. 2019, 21, 1800920. [Google Scholar] [CrossRef]
- Fu, J.; Du, W.; Liu, K.; Du, X.; Zhao, C.; Liang, H.; Mansoor, A.; Li, S.; Wang, Z. Effect of the Ca2Mg6Zn3 Phase on the Corrosion Behavior of Biodegradable Mg-4.0 Zn-0.2 Mn-x Ca Alloys in Hank’s Solution. Materials 2022, 15, 2079. [Google Scholar] [CrossRef]
- Liu, B.-Y.; Liu, F.; Yang, N.; Zhai, X.-B.; Zhang, L.; Yang, Y.; Li, B.; Li, J.; Ma, E.; Nie, J.-F.; et al. Large plasticity in magnesium mediated by pyramidal dislocations. Science 2019, 365, 73–75. [Google Scholar] [CrossRef]
- Razavi, S.; Foley, D.; Karaman, I.; Hartwig, K.; Duygulu, O.; Kecskes, L.; Mathaudhu, S.N.; Hammond, V.H. Effect of grain size on prismatic slip in Mg–3Al–1Zn alloy. Scr. Mater. 2012, 67, 439–442. [Google Scholar] [CrossRef]
- Akhtar, A.; Teghtsoonian, E. Solid solution strengthening of magnesium single crystals—ii the effect of solute on the ease of prismatic slip. Acta Metall. 1969, 17, 1351–1356. [Google Scholar] [CrossRef]
- Zhao, J.; You, C.; Chen, M.; Lyu, S.; Tie, D.; Liu, H. Effect of calcium oxide particle size on microstructure and properties of AZ91 Mg alloy. J. Alloys Compd. 2021, 886, 160970. [Google Scholar] [CrossRef]
- Lee, T.W.; Park, H.W.; Lim, H.; Kim, S.K.; Lim, S.H. Microstructural characterization of oxide layers in CaO added AZ31 Mg alloy. J. Alloys Compd. 2017, 714, 397–408. [Google Scholar] [CrossRef]
- Liu, Y.H.; Li, X.G.; Li, Y.J.; Shi, G.L.; Yuan, J.W.; Zhang, K. Homogenization treatment of Mg-6Zn-3Sn (wt.%) alloy and its effects on microstructure and mechanical properties. Trans. Nonferrous Met. Soc. China 2023, 33, 67–78. [Google Scholar] [CrossRef]
- Chaudry, U.M.; Tariq, H.M.R.; Ansari, N.; Kim, C.-S.; Lee, S.Y.; Jun, T.-S. Exceptional improvement in the yield strength of AZ61 magnesium alloy via cryo-stretching and its implications on the grain growth during annealing. J. Alloys Compd. 2024, 970, 172630. [Google Scholar] [CrossRef]
- Jeong, J.; Im, J.; Song, K.; Kwon, M.; Kim, S.K.; Kang, Y.B.; Oh, S.H. Transmission electron microscopy and thermodynamic studies of CaO-added AZ31 Mg alloys. Acta Mater. 2013, 61, 3267–3277. [Google Scholar] [CrossRef]
- Amberger, D.; Eisenlohr, P.; Göken, M. On the importance of a connected hard-phase skeleton for the creep resistance of Mg alloys. Acta Mater. 2012, 60, 2277–2289. [Google Scholar] [CrossRef]
- Park, J.; Kim, M.; Yoon, U.; Kim, W. Microstructures and mechanical properties of Mg–Al–Zn–Ca alloys fabricated by high frequency electromagnetic casting method. J. Mater. Sci. 2009, 44, 47–54. [Google Scholar] [CrossRef]
- Chaudry, U.M.; Tariq, H.M.R.; Zubair, M.; Ansari, N.; Jun, T.-S. Implications of twinning on the microstructure development, crystallographic texture and mechanical performance of Mg alloys-a critical review. J. Magnes. Alloys 2023, in press. [Google Scholar] [CrossRef]
- Zhang, K.; Shao, Z.; Daniel, C.S.; Turski, M.; Pruncu, C.; Lang, L.; Robson, J.; Jiang, J. A comparative study of plastic deformation mechanisms in room-temperature and cryogenically deformed magnesium alloy AZ31. Mater. Sci. Eng. A 2021, 807, 140821. [Google Scholar] [CrossRef]




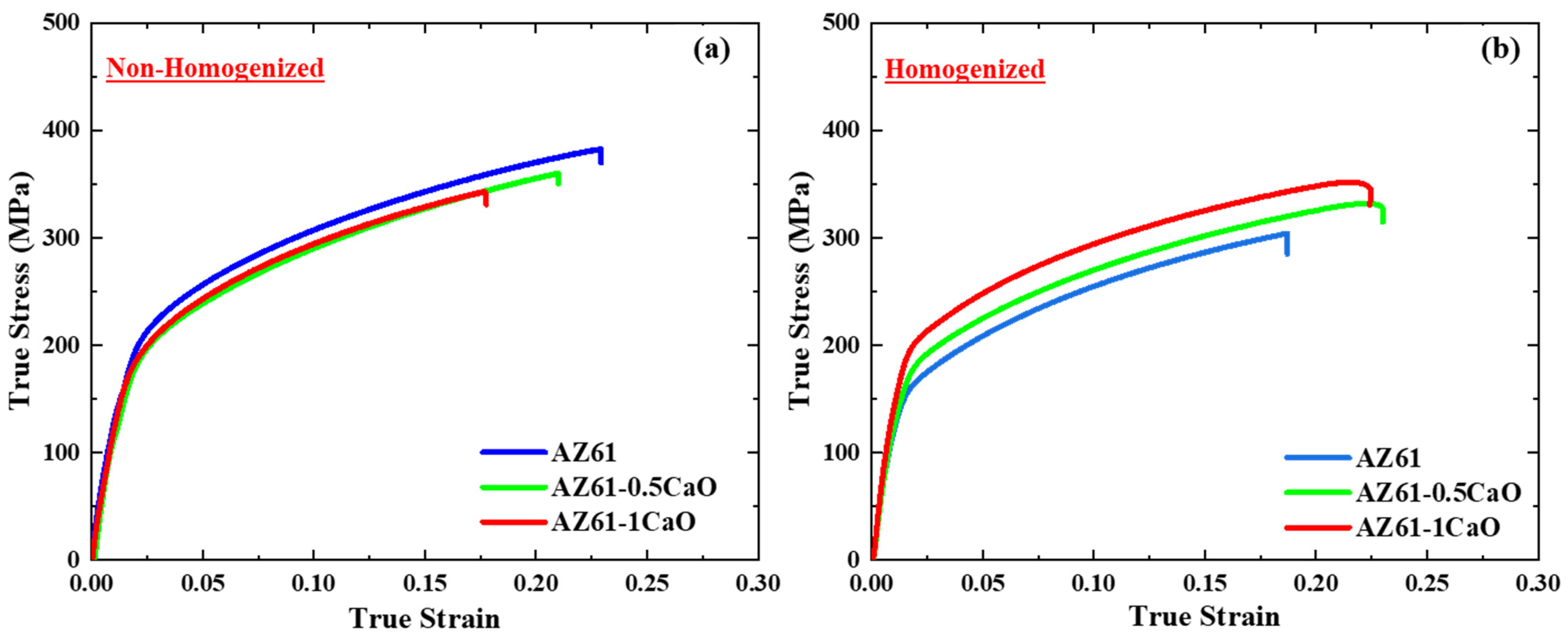
| Condition | Material | YS (MPa) | UTS (MPa) | True Strain |
|---|---|---|---|---|
| As-fabricated | AZ61 | 195 ± 1.1 | 382 ± 1.5 | 0.22 |
| AZ61-0.5CaO | 180 ± 2.1 | 350 ± 2.6 | 0.21 | |
| AZ61-1CaO | 184 ± 1.6 | 331 ± 2.2 | 0.17 | |
| Annealed | AZ61 | 166 ± 1.4 | 304 ± 2.7 | 0.18 |
| AZ61-0.5CaO | 181 ± 1.7 | 324 ± 2.1 | 0.23 | |
| AZ61-1CaO | 203 ± 3.5 | 330 ± 3.8 | 0.22 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaudry, U.M.; Tariq, H.M.R.; Ansari, N.; Mansoor, A.; Khan, M.K.; Hamad, K.; Jun, T.-S. Effect of CaO Content and Annealing Treatment on the Room-Temperature Mechanical Properties of AZ61 and AZ61-CaO Alloys. Metals 2023, 13, 1962. https://doi.org/10.3390/met13121962
Chaudry UM, Tariq HMR, Ansari N, Mansoor A, Khan MK, Hamad K, Jun T-S. Effect of CaO Content and Annealing Treatment on the Room-Temperature Mechanical Properties of AZ61 and AZ61-CaO Alloys. Metals. 2023; 13(12):1962. https://doi.org/10.3390/met13121962
Chicago/Turabian StyleChaudry, Umer Masood, Hafiz Muhammad Rehan Tariq, Nooruddin Ansari, Adil Mansoor, Muhammad Kashif Khan, Kotiba Hamad, and Tea-Sung Jun. 2023. "Effect of CaO Content and Annealing Treatment on the Room-Temperature Mechanical Properties of AZ61 and AZ61-CaO Alloys" Metals 13, no. 12: 1962. https://doi.org/10.3390/met13121962
APA StyleChaudry, U. M., Tariq, H. M. R., Ansari, N., Mansoor, A., Khan, M. K., Hamad, K., & Jun, T.-S. (2023). Effect of CaO Content and Annealing Treatment on the Room-Temperature Mechanical Properties of AZ61 and AZ61-CaO Alloys. Metals, 13(12), 1962. https://doi.org/10.3390/met13121962









