Mechanistic Study in Gold Nanoparticle Synthesis through Microchip Laser Ablation in Organic Solvents
Abstract
:1. Introduction
2. Materials and Methods
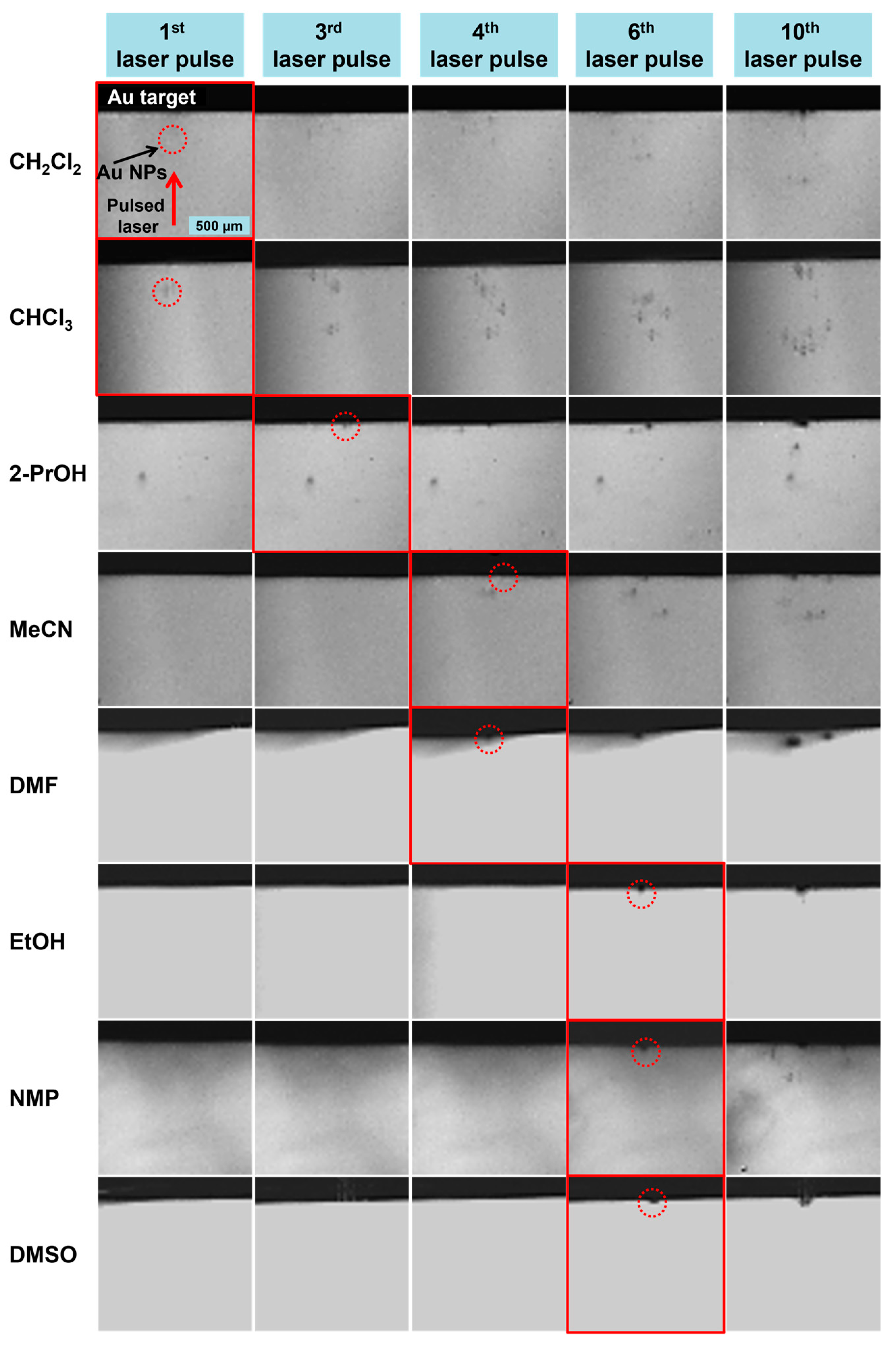
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, N.; Zhao, P.; Astruc, D. Anisotropic gold nanoparticles: Synthesis, properties, applications, and toxicity. Angew. Chem. Int. Ed. 2014, 53, 1756–1789. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Mou, L.; Jiang, X. Surface chemistry of gold nanoparticles for health-related applications. Chem. Sci. 2020, 11, 923–936. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Li, Z.; Sugioka, K. Laser ablation in liquids for nanomaterial synthesis: Diversities of targets and liquids. J. Phys. Photonics 2021, 3, 042002. [Google Scholar] [CrossRef]
- Zhang, D.; Gökce, B.; Barcikowski, S. Laser synthesis and processing of colloids: Fundamentals and applications. Chem. Rev. 2017, 5, 3990–4103. [Google Scholar] [CrossRef] [PubMed]
- Theerthagiri, J.; Karuppasamy, K.; Lee, S.J. Fundamentals and comprehensive insights on pulsed laser synthesis of advanced materials for diverse photo- and electrocatalytic applications. Light Sci. Appl. 2022, 11, 250. [Google Scholar] [CrossRef] [PubMed]
- Amendola, V.; Meneghetti, M. Laser ablation synthesis in solution and size manipulation of noble metal nanoparticles. Phis. Chem. Chem. Phys. 2009, 11, 3805–3821. [Google Scholar] [CrossRef] [PubMed]
- Amendola, V.; Polizzi, S.; Meneghetti, M. Free silver nanoparticles synthesized by laser ablation in organic solvents and their easy functionalization. Langmuir 2007, 23, 6766–6770. [Google Scholar] [CrossRef]
- Zhang, D.; Zhang, C.; Liu, J.; Cen, Q.; Zhu, X.; Liang, C. Carbon-Encapsulated Metal/Metal Carbide/Metal Oxide Core-Shell Nanostructures Generated by Laser Ablation of Metals in Organic Solvents. ACS Appl. Nano Mater. 2019, 2, 28–39. [Google Scholar] [CrossRef]
- Grass, R.N.; Athanassio, E.K.; Stark, W.J. Covalently Functionalized Cobalt Nanoparticles as a Platform for Magnetic Separations in Organic Synthesis. Angew. Chem. Int. Ed. 2007, 46, 4909–4912. [Google Scholar] [CrossRef]
- Amendola, V.; Polizzi, S.; Meneghetti, M.; Amendola, V. Laser ablation synthesis of gold nanoparticles in organic solvents. J. Phys. Chem. B 2006, 110, 7232–7237. [Google Scholar] [CrossRef]
- Giorgetti, E.; Muniz-Miranda, M.; Marsili, P.; Scarpellini, D.; Giammanco, F. Stable gold nanoparticles obtained in pure acetone by laser ablation with different wavelengths. J. Nanopart. Res. 2012, 14, 648. [Google Scholar] [CrossRef]
- Compagnini, G.; Scalisi, A.A.; Puglisi, O.; Spinella, C. Synthesis of gold colloids by laser ablation in thiol-alkane solutions. J. Mater. Res. 2004, 19, 2795–2798. [Google Scholar] [CrossRef]
- Taira, T. RE3+-ion-doped YAG ceramic lasers. IEEE J. Sel. Top. Quantum Electron. 2007, 13, 798–809. [Google Scholar] [CrossRef]
- Sakai, H.; Kan, H.; Taira, T. >1 MW peak power single-mode high-brightness passively Q-switched Nd3+:YAG microchip laser. Opt. Express. 2008, 16, 19891–19899. [Google Scholar] [CrossRef]
- Dittrich, S.; Spellauge, M.; Barcikowski, S.; Huber, H.P.; Gökce, B. Time resolved studies reveal the origin of the unparalleled high efficiency of one nanosecond laser ablation in liquids. Opto-Electron. Adv. 2022, 5, 210053. [Google Scholar] [CrossRef]
- Hettiarachchi, B.S.; Takaoka, Y.; Uetake, Y.; Yakiyama, Y.; Lim, H.H.; Taira, T.; Maruyama, M.; Mori, Y.; Yoshikawa, H.Y.; Sakurai, H. Uncovering gold nanoparticle synthesis using microchip laser system through pulsed laser ablation in aqueous solution. Ind. Chem. Mater. 2024. [Google Scholar] [CrossRef]
- Amendola, V.; Rizzi, G.A.; Polizzi, S.; Meneghetti, M. Synthesis of gold nanoparticles by laser ablation in toluene: quenching and recovery of the surface plasmon absorption. J. Phys. Chem. B 2005, 109, 23125–23128. [Google Scholar] [CrossRef] [PubMed]
- Amendola, V.; Riello, P.; Meneghetti, M. Magnetic nanoparticles of iron carbide, iron oxide, iron@iron oxide, and metal iron synthesized by laser ablation in organic solvents. J. Phys. Chem. C 2011, 115, 5140–5146. [Google Scholar] [CrossRef]
- Begildayeva, T.; Ahn, A.; Naik, S.S.; Lee, S.J.; Theerthagiri, J.; Kim, T.H.; Choi, M.Y. Facile one-pot synthesis of CuCN by pulsed laser ablation in nitrile solvents and mechanistic studies using quantum chemical calculations. Sci. Rep. 2021, 11, 14389. [Google Scholar] [CrossRef] [PubMed]
- Long, J.; Eliceiri, M.H.; Wang, L.; Vangelatos, Z.; Ouyang, Y.; Xie, X.; Zhang, Y.; Grigoropoulos, C.P. Capturing the final stage of the collapse of cavitation bubbles generated during nanosecond laser ablation of submerged targets. Opt. Laser Technol. 2021, 134, 106647. [Google Scholar] [CrossRef]
- Kalus, M.-R.; Lanyumba, R.; Barcikowski, S.; Gökce, B. Discrimination of ablation, shielding, and interface layer effects on the steady-state formation of persistent bubbles under liquid flow conditions during laser synthesis of colloids. J. Flow Chem. 2021, 11, 773–792. [Google Scholar] [CrossRef]
- Forte, G.; D’Urso, L.; Fazio, E.; Patane, S.; Neri, F.; Puglisi, O.; Compagnini, G. The effects of liquid environments on the optical properties of linear carbon chains prepared by laser ablation generated plasmas. Appl. Surf. Sci. 2013, 272, 76–81. [Google Scholar] [CrossRef]
- Rawat, R.; Tiwari, A.; Arun, N.; Rao, S.V.S.N.; Pathak, A.P.; Shadangi, Y.; Mukhopadhyay, N.K.; Rao, S.V.; Tripathi, A. Nanosecond pulsed laser ablation of Al–Cu–Fe quasicrystalline material: Effects of solvent and fluence. J. Alloys Compd. 2021, 859, 157871. [Google Scholar] [CrossRef]
- Zhang, G.-R.; Xu, B.-Q. Surprisingly strong effect of stabilizer on the properties of Au nanoparticles and PtAu nanostructures in electrocatalysis. Nanoscale 2010, 2, 2798–2804. [Google Scholar] [CrossRef] [PubMed]
- Tsunoyama, H.; Ichikuni, N.; Sakurai, H.; Tsukuda, T. Effect of Electronic Structures of Au Clusters Stabilized by Poly(N-vinyl-2-pyrrolidone) on Aerobic Oxidation Catalysis. J. Am. Chem. Soc. 2009, 131, 7086–7093. [Google Scholar] [CrossRef] [PubMed]
- Okumura, M.; Kitagawa, Y.; Kawakami, T.; Haruta, M. Theoretical investigation of the hetero-junction effect in PVP stabilized Au13 clusters. The role of PVP in their catalytic activities. Chem. Phys. Lett. 2008, 459, 133–136. [Google Scholar] [CrossRef]
- Haesuwannakij, S.; Kimura, T.; Furutani, Y.; Okumura, K.; Kokubo, K.; Sakata, T.; Yasuda, H.; Yakiyama, Y.; Sakurai, H. The Impact of the Polymer Chain Length on the Catalytic Activity of Poly(N-vinyl-2-pyrrolidone)-supported Gold Nanoclusters. Sci. Rep. 2017, 7, 9579. [Google Scholar] [CrossRef]
- Vinsen; Uetake, Y.; Sakurai, H. Selective Oxidative Hydroxylation of Arylboronic Acids by Colloidal Nanogold Catalyzed in Situ Generation of H2O2 from Alcohols Under Aerobic Conditions. Bull. Chem. Soc. Jpn. 2020, 93, 299–301. [Google Scholar] [CrossRef]
- Dhital, R.N.; Nomura, K.; Sato, Y.; Haesuwannakij, S.; Ehara, M.; Sakurai, H. Pt–Pd Nanoalloy for the Unprecedented Activation of Carbon–Fluorine Bond at Low Temperature. Bull. Chem. Soc. Jpn. 2020, 93, 1180–1185. [Google Scholar] [CrossRef]
- Uetake, Y.; Mouri, S.; Haesuwannakij, S.; Okumura, K.; Sakurai, H. Volcano-Type Correlation between Particle Size and Catalytic Activity on Hydrodechlorination Catalyzed by AuPd Nanoalloy. Nanoscale Adv. 2021, 3, 1496–1501. [Google Scholar] [CrossRef]
- Tsuji, T.; Thang, D.-H.; Okazaki, Y.; Nakanishi, M.; Tsuboi, Y.; Tsuji, M. Preparation of silver nanoparticles by laser ablation in polyvinylpyrrolidone solutions. Appl. Surf. Sci. 2008, 254, 5224–5230. [Google Scholar] [CrossRef]
- Letzel, A.; Reich, S.; dos Santos Rolo, T.; Kanitz, A.; Hoppius, J.; Rack, A.; Olbinado, M.P.; Ostendorf, A.; Gökce, B.; Plech, A.; et al. Time and mechanism of nanoparticle functionalization by macromolecular ligands during pulsed laser ablation in liquids. Langmuir 2019, 35, 3038–3047. [Google Scholar] [CrossRef] [PubMed]
- Kassavetis, S.; Kaziannis, S.; Pliatsikas, N.; Avgeropoulos, A.; Karantzalis, A.E.; Kosmidis, C.; Lidorikis, E.; Patsalas, P. Formation of plasmonic colloidal silver for flexible and printed electronics using laser ablation. Appl. Surf. Sci. 2015, 336, 262–266. [Google Scholar] [CrossRef]
- Kalus, M.R.; Barsch, N.; Streubel, R.; Gökce, E.; Barcikowski, S.; Gökce, B. How persistent microbubbles shield nanoparticle productivity in laser synthesis of colloids-quantification of their volume, dwell dynamics, and gas composition. Phys. Chem. Chem. Phys. 2017, 19, 7112–7123. [Google Scholar] [CrossRef] [PubMed]
- Fromme, T.; Tintrop, L.K.; Reichenberger, S.; Schmidt, T.C.; Barcikowski, S. Impact of chemical and physical properties of organic solvents on the gas and hydrogen formation during laser synthesis of gold nanoparticles. ChemPhysChem 2023, 24, e202300089. [Google Scholar] [CrossRef] [PubMed]
- Chickos, J.S. Heat of Sublimation Data. In NIST Chemistry WebBook; NIST Standard Reference Database Number 69; Linstrom, P.J., Mallard, W.G., Eds.; National Institute of Standards and Technology: Gaithersburg, MD, USA, 2020; p. 20899. [Google Scholar]
- Sperling, L.H. Introduction to Physical Polymer Science; Wiley-Inter Science: New York, NY, USA, 1992; p. 87. [Google Scholar]
- Reich, S.; Letzel, A.; Gökce, B.; Menzel, A.; Barcikowski, S.; Plech, A. Incubation effect of pre-irradiation on bubble formation and ablation in laser ablation in liquids. ChemPhysChem 2019, 20, 1036. [Google Scholar] [CrossRef]
- Lei, Q.-F.; Lin, R.-S.; Ni, D.-Y.; Hou, Y.-C. Thermal Conductivities of Some Organic Solvents and Their Binary Mixtures. J. Chem. Eng. Data 1997, 42, 971–974. [Google Scholar]
- Daubert, T.E.; Danner, R.P. Physical and Thermodynamic Properties of Pure Chemicals: Data Compilation; Taylor & Francis: Washington, DC, USA, 1989. [Google Scholar]
- Shih, C.-Y.; Shugaev, M.V.; Wu, C.; Zhigilei, L.V. Generation of subsurface voids, incubation effect, and formation of nanoparticles in short pulse laser interactions with bulk metal targets in liquid: Molecular dynamics study. J. Phys. Chem. C 2017, 121, 16549–16567. [Google Scholar] [CrossRef]
- Ouyang, P.; Li, P.; Leksina, E.; Michurin, S.; He, L. Effect of liquid properties on laser ablation of aluminum and titanium alloys. Appl. Surf. Sci. 2016, 360, 880–888. [Google Scholar] [CrossRef]



| Solvent a | Particle Size (nm) | Au Productivity (µg/h) | Viscosity (mPa·s) | Thermal Conductivity (W/m·K) [34,35] | Required Number of the Pulse |
|---|---|---|---|---|---|
| CH2Cl2 | 3.2 ± 0.7 | 79.3 | 0.68 ± 0.02 | 0.13 | 1 |
| CHCl3 | 3.5 ± 1.0 | 109.1 | 0.67 ± 0.02 | 0.12 | 1 |
| 2-PrOH | 4.4 ± 1.1 | 134.7 | 2.92 ± 0.02 | 0.16 | 3 |
| MeCN | 3.5 ± 0.7 | 147.5 | 0.45 ± 0.02 | 0.15 | 4 |
| DMF | 3.4 ± 0.8 | 155.2 | 1.12 ± 0.02 | 0.16 | 4 |
| EtOH | 3.4 ± 0.8 | 166.8 | 1.81 ± 0.02 | 0.17 | 6 |
| NMP | 3.3 ± 0.4 | 168.6 | 2.16 ± 0.02 | 0.18 | 6 |
| DMSO | 3.0 ± 0.8 | 173.1 | 2.72 ± 0.02 | 0.20 | 6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hettiarachchi, B.S.; Takaoka, Y.; Uetake, Y.; Yakiyama, Y.; Yoshikawa, H.Y.; Maruyama, M.; Sakurai, H. Mechanistic Study in Gold Nanoparticle Synthesis through Microchip Laser Ablation in Organic Solvents. Metals 2024, 14, 155. https://doi.org/10.3390/met14020155
Hettiarachchi BS, Takaoka Y, Uetake Y, Yakiyama Y, Yoshikawa HY, Maruyama M, Sakurai H. Mechanistic Study in Gold Nanoparticle Synthesis through Microchip Laser Ablation in Organic Solvents. Metals. 2024; 14(2):155. https://doi.org/10.3390/met14020155
Chicago/Turabian StyleHettiarachchi, Barana Sandakelum, Yusuke Takaoka, Yuta Uetake, Yumi Yakiyama, Hiroshi Y. Yoshikawa, Mihoko Maruyama, and Hidehiro Sakurai. 2024. "Mechanistic Study in Gold Nanoparticle Synthesis through Microchip Laser Ablation in Organic Solvents" Metals 14, no. 2: 155. https://doi.org/10.3390/met14020155
APA StyleHettiarachchi, B. S., Takaoka, Y., Uetake, Y., Yakiyama, Y., Yoshikawa, H. Y., Maruyama, M., & Sakurai, H. (2024). Mechanistic Study in Gold Nanoparticle Synthesis through Microchip Laser Ablation in Organic Solvents. Metals, 14(2), 155. https://doi.org/10.3390/met14020155






