Control of a Multi-Drug-Resistant Acinetobacter baumannii Outbreak after Orthopedics Department Relocation
Abstract
:1. Introduction
2. Outbreak Description and Laboratory Procedures
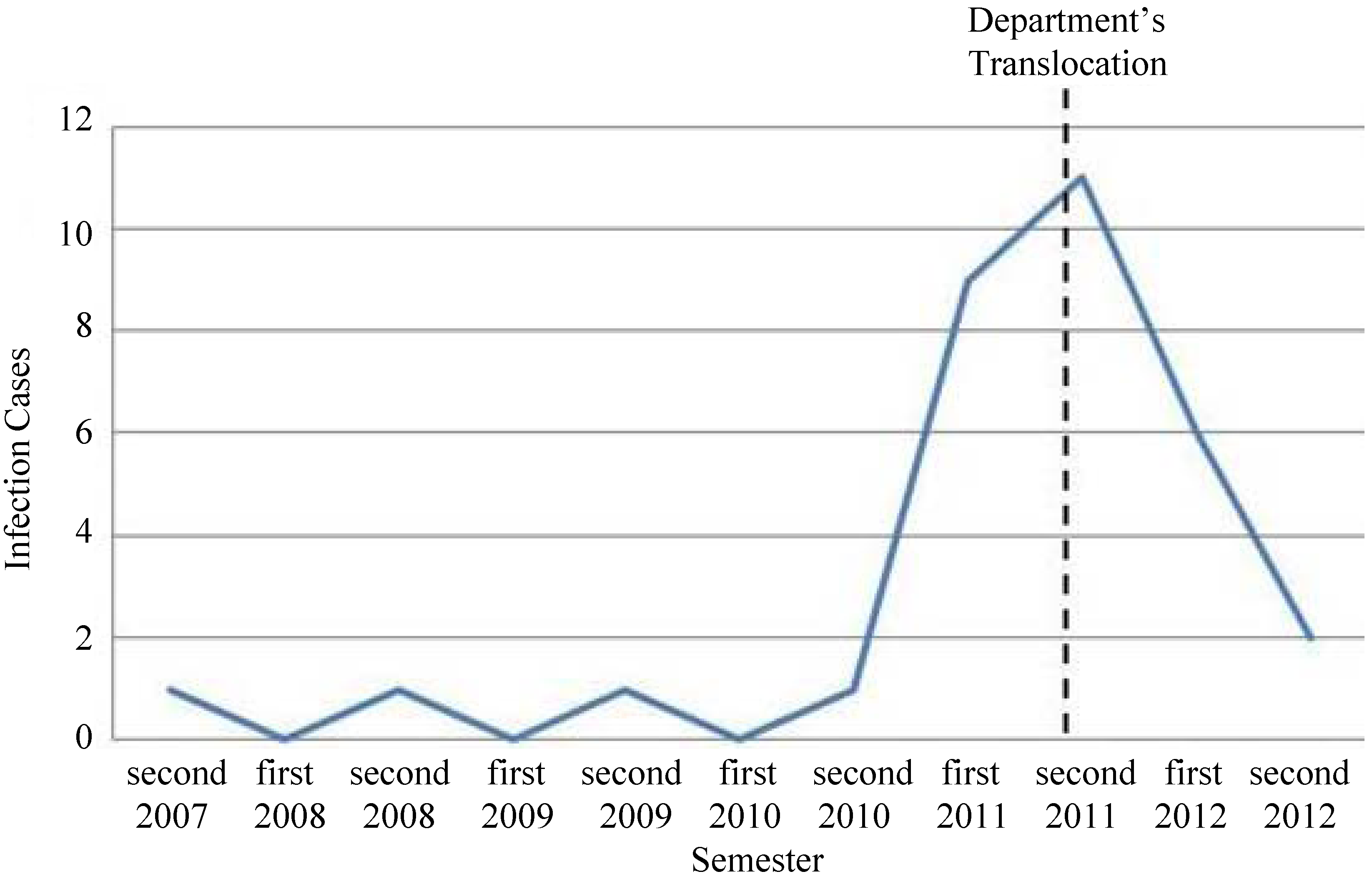
3. Conclusions
Conflicts of Interest
References
- Miyakis, S.; Pefanis, A.; Tsakris, A. The challenges of antimicrobial drug resistance in Greece. Clin. Infect. Dis. 2011, 53, 177–184. [Google Scholar]
- Pournaras, S.; Markogiannakis, A.; Ikonomidis, A.; Kondyli, L.; Bethimouti, K.; Maniatis, A.N.; Legakis, N.J.; Tsakris, A. Outbreak of multiple clones of imipenem-resistant Acinetobacter baumannii isolates expressing OXA-58 carbapenemase in an intensive care unit. J. Antimicrob. Chemother. 2006, 57, 557–561. [Google Scholar]
- Poirel, L.; Lebessi, E.; Héritier, C.; Patsoura, A.; Foustoukou, M.; Nordmann, P. Nosocomial spread of OXA-58-positive carbapenem-resistant Acinetobacter baumannii isolates in a paediatric hospital in Greece. Clin. Microbiol. Infect. 2006, 12, 1138–1141. [Google Scholar]
- Papa, A.; Koulourida, V.; Souliou, E. Molecular epidemiology of carbapenem-resistant Acinetobacter baumannii in a newly established Greek hospital. Microb. Drug Resist. 2009, 15, 257–260. [Google Scholar]
- Tsakris, A.; Ikonomidis, A.; Pournaras, S.; Tzouvelekis, L.S.; Sofianou, D.; Legakis, N.J.; Maniatis, A.N. VIM-1 metallo-β-lactamase in Acinetobacter baumannii. Emerg. Infect. Dis. 2006, 12, 981–983. [Google Scholar]
- Ikonomidis, A.; Ntokou, E.; Maniatis, A.N.; Tsakris, A.; Pournaras, S. Hidden VIM-1 metallo-β-lactamase phenotypes among Acinetobacter baumannii clinical isolates. J. Clin. Microbiol. 2008, 46, 346–349. [Google Scholar]
- Loli, A.; Tzouvelekis, L.S.; Gianneli, D.; Tzelepi, E.; Miriagou, V. Outbreak of Acinetobacter baumannii with chromosomally encoded VIM-1 undetectable by imipenem-EDTA synergy tests. Antimicrob. Agents Chemother. 2008, 52, 1894–1896. [Google Scholar]
- Tsakris, A.; Poulou, A.; Pournaras, S.; Voulgari, E.; Vrioni, G.; Themeli-Digalaki, K.; Petropoulou, D.; Sofianou, D. A simple phenotypic method for the differentiation of metallo-β-lactamases and class A KPC carbapenemases in Enterobacteriaceae clinical isolates. J. Antimicrob. Chemother. 2010, 65, 1664–1671. [Google Scholar]
- Diancourt, L.; Passet, V.; Nemec, A.; Dijkshoorn, L.; Brisse, S. The population structure of Acinetobacter baumannii: Expanding multiresistant clones from an ancestral susceptible genetic pool. PLoS One 2010, 5, e10034. [Google Scholar]
- Tsakris, A.; Tsioni, C.; Pournaras, S.; Polyzos, S.; Maniatis, A.N.; Sofianou, D. Spread of low-level carbapenem-resistant Acinetobacter baumannii clones in a tertiary care Greek hospital. J. Antimicrob. Chemother. 2003, 52, 1046–1047. [Google Scholar]
- Gogou, V.; Pournaras, S.; Giannouli, M.; Voulgari, E.; Piperaki, E.T.; Zarrilli, R.; Tsakris, A. Evolution of multidrug-resistant Acinetobacter baumannii clonal lineages: A 10 year study in Greece (2000–09). J. Antimicrob. Chemother. 2011, 66, 2767–2772. [Google Scholar]
- Liakopoulos, A.; Miriagou, V.; Katsifas, E.; Karagouni, A.D.; Daikos, G.L.; Tzouvelekis, L.S.; Petinaki, E. Identification of OXA-23-producing Acinetobacter baumannii in Greece, 2010 to 2011. Euro Surveill. 2012, 17, p. 20117. Available online: http://www.eurosurveillance.org/ViewArticle.aspx?ArticleId=20117 (accessed on 22 November 2013).
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gogou, V.; Meletis, G.; Tsitouras, D. Control of a Multi-Drug-Resistant Acinetobacter baumannii Outbreak after Orthopedics Department Relocation. Microorganisms 2013, 1, 158-161. https://doi.org/10.3390/microorganisms1010158
Gogou V, Meletis G, Tsitouras D. Control of a Multi-Drug-Resistant Acinetobacter baumannii Outbreak after Orthopedics Department Relocation. Microorganisms. 2013; 1(1):158-161. https://doi.org/10.3390/microorganisms1010158
Chicago/Turabian StyleGogou, Vasiliki, Georgios Meletis, and Dimosthenis Tsitouras. 2013. "Control of a Multi-Drug-Resistant Acinetobacter baumannii Outbreak after Orthopedics Department Relocation" Microorganisms 1, no. 1: 158-161. https://doi.org/10.3390/microorganisms1010158



