Metal-Based Nanoparticles: Antibacterial Mechanisms and Biomedical Application
Abstract
:1. Introduction
2. Gram-Positive and Gram-Negative Bacteria and Biofilm
2.1. Gram-Positive and Gram-Negative Bacteria
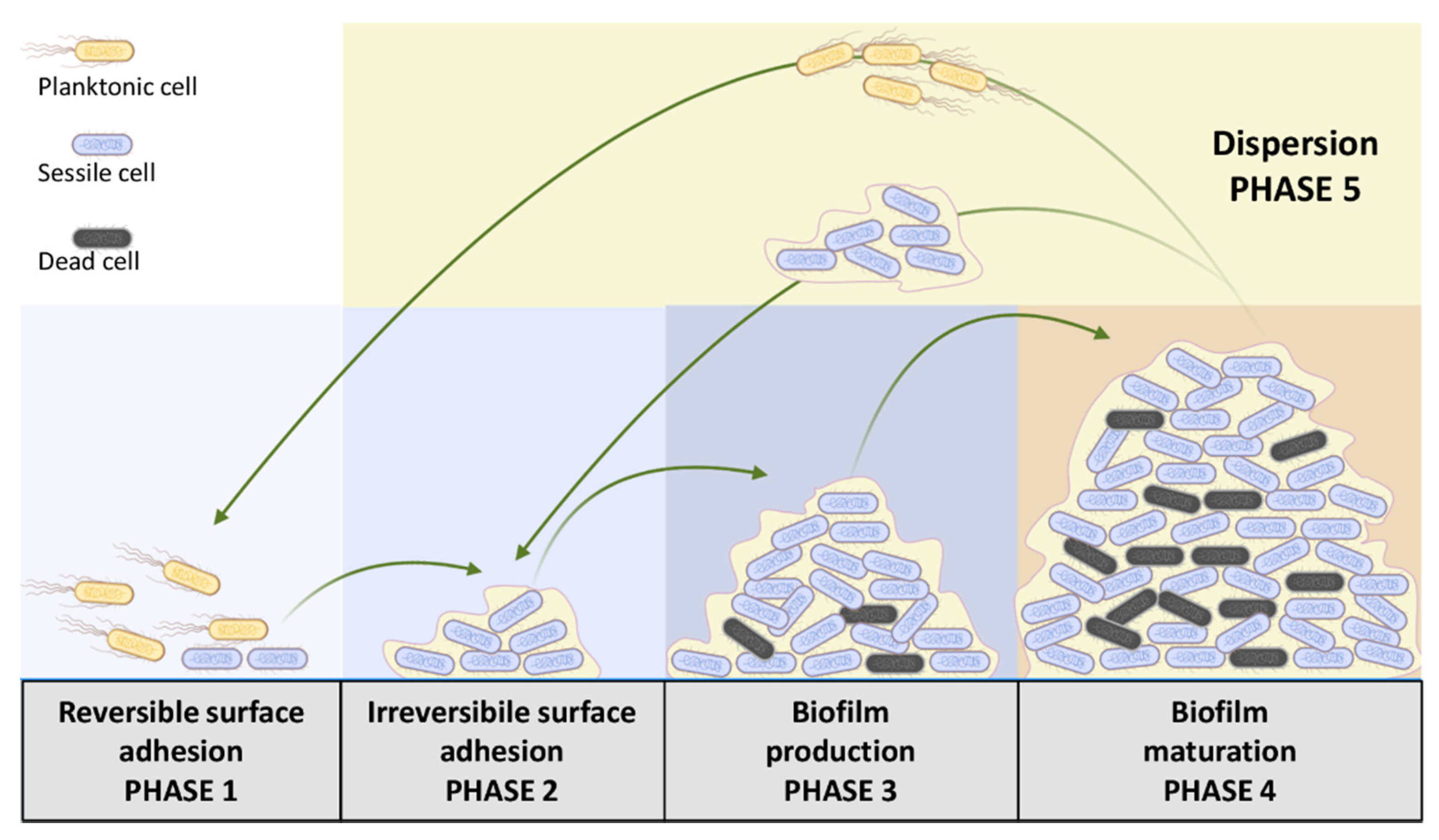
2.2. Biofilm
3. Influence of Metal NPs’ Physico-Chemical Properties on Antibacterial Activity
3.1. Size
3.2. Shape
3.3. Surface Charge
4. Antibacterial Mechanisms
4.1. Physical Interactions
4.2. Ion Leaching and Dissolution
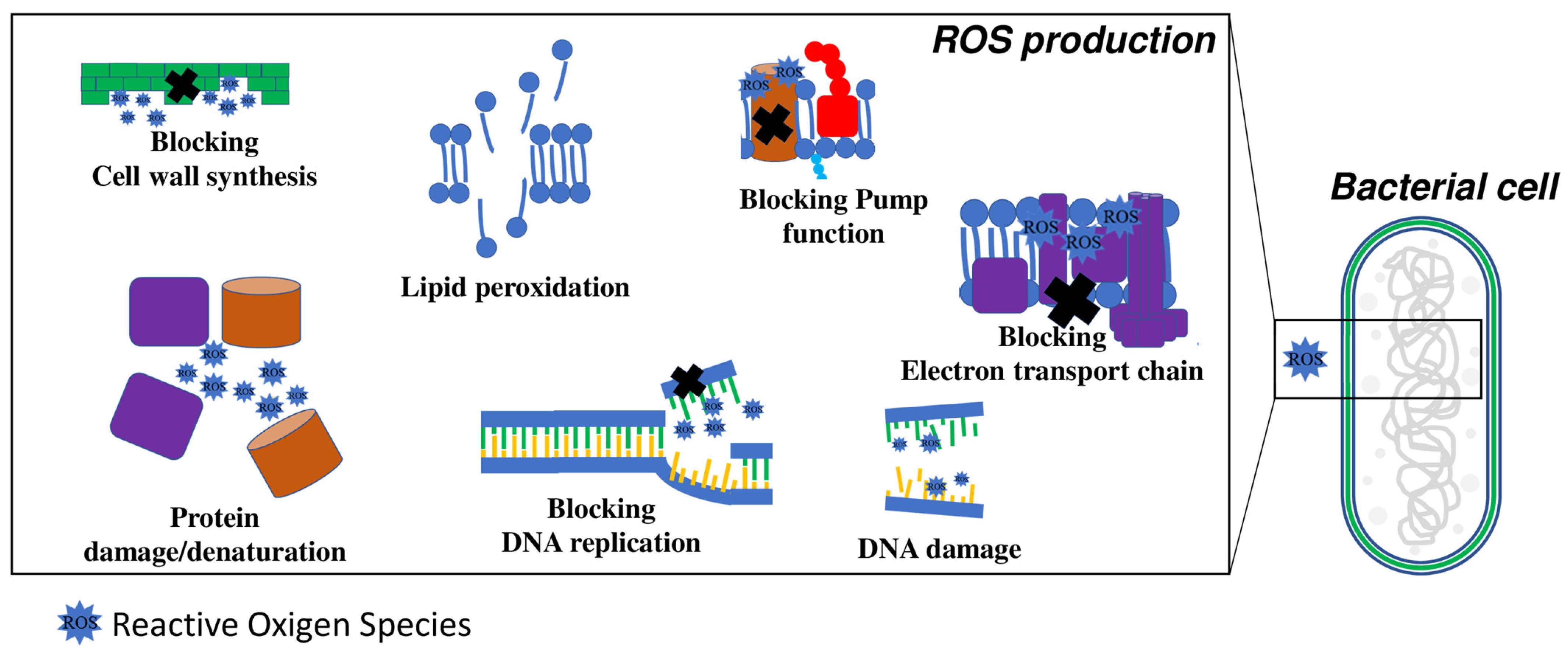
4.3. Production of Reactive Oxygen Species
4.4. Bacterial Resistance Strategy against NPs
5. Antimicrobial Application of Metal NPs
5.1. Biomedical Application
5.2. Recent Application of Metal NPs
6. Discussion and Final Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- CDC The Biggest Antibiotic-Resistant Threats in the U.S. Available online: https://www.cdc.gov/drugresistance/biggest-threats.html (accessed on 5 April 2022).
- Abraham, E.P.; Chain, E. An Enzyme from Bacteria Able to Destroy Penicillin. Nature 1940, 146, 837. [Google Scholar] [CrossRef]
- Khan, A.A.; Manzoor, K.N.; Sultan, A.; Saeed, M.; Rafique, M.; Noushad, S.; Talib, A.; Rentschler, S.; Deigner, H.-P. Pulling the Brakes on Fast and Furious Multiple Drug-Resistant (MDR) Bacteria. Int. J. Mol. Sci. 2021, 22, 859. [Google Scholar] [CrossRef]
- Santajit, S.; Indrawattana, N. Mechanisms of Antimicrobial Resistance in ESKAPE Pathogens. BioMed Res. Int. 2016, 2016, 2475067. [Google Scholar] [CrossRef] [PubMed]
- Rosini, R.; Nicchi, S.; Pizza, M.; Rappuoli, R. Vaccines Against Antimicrobial Resistance. Front. Immunol. 2020, 11, 1048. [Google Scholar] [CrossRef] [PubMed]
- Reygaert, W.C. An Overview of the Antimicrobial Resistance Mechanisms of Bacteria. AIMS Microbiol. 2018, 4, 482–501. [Google Scholar] [CrossRef] [PubMed]
- Chancey, S.T.; Zähner, D.; Stephens, D.S. Acquired Inducible Antimicrobial Resistance in Gram-Positive Bacteria. Future Microbiol. 2012, 7, 959–978. [Google Scholar] [CrossRef]
- Kapoor, G.; Saigal, S.; Elongavan, A. Action and Resistance Mechanisms of Antibiotics: A Guide for Clinicians. J. Anaesthesiol. Clin. Pharmacol. 2017, 33, 300–305. [Google Scholar] [CrossRef]
- Pacios, O.; Blasco, L.; Bleriot, I.; Fernandez-Garcia, L.; González Bardanca, M.; Ambroa, A.; López, M.; Bou, G.; Tomás, M. Strategies to Combat Multidrug-Resistant and Persistent Infectious Diseases. Antibiotics 2020, 9, 65. [Google Scholar] [CrossRef]
- Vimbela, G.V.; Ngo, S.M.; Fraze, C.; Yang, L.; Stout, D.A. Antibacterial Properties and Toxicity from Metallic Nanomaterials. Int. J. Nanomed. 2017, 12, 3941–3965. [Google Scholar] [CrossRef]
- Nocito, G.; Sciuto, E.L.; Franco, D.; Nastasi, F.; Pulvirenti, L.; Petralia, S.; Spinella, C.; Calabrese, G.; Guglielmino, S.; Conoci, S. Physicochemical Characterization and Antibacterial Properties of Carbon Dots from Two Mediterranean Olive Solid Waste Cultivars. Nanomaterials 2022, 12, 885. [Google Scholar] [CrossRef]
- Agnihotri, S.; Dhiman, N.K. Development of Nano-Antimicrobial Biomaterials for Biomedical Applications. In Advances in Biomaterials for Biomedical Applications; Tripathi, A., Melo, J.S., Eds.; Advanced Structured Materials; Springer: Singapore, 2017; pp. 479–545. ISBN 978-981-10-3328-5. [Google Scholar]
- Kittler, S.; Greulich, C.; Diendorf, J.; Köller, M.; Epple, M. Toxicity of Silver Nanoparticles Increases during Storage Because of Slow Dissolution under Release of Silver Ions. Available online: https://pubs.acs.org/doi/pdf/10.1021/cm100023p (accessed on 9 April 2022).
- Wang, L.; He, H.; Yu, Y.; Sun, L.; Liu, S.; Zhang, C.; He, L. Morphology-Dependent Bactericidal Activities of Ag/CeO2 Catalysts against Escherichia Coli. J. Inorg. Biochem. 2014, 135, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Shkodenko, L.; Kassirov, I.; Koshel, E. Metal Oxide Nanoparticles Against Bacterial Biofilms: Perspectives and Limitations. Microorganisms 2020, 8, 1545. [Google Scholar] [CrossRef] [PubMed]
- Franco, D.; Calabrese, G.; Petralia, S.; Neri, G.; Corsaro, C.; Forte, L.; Squarzoni, S.; Guglielmino, S.; Traina, F.; Fazio, E.; et al. Antimicrobial Effect and Cytotoxic Evaluation of Mg-Doped Hydroxyapatite Functionalized with Au-Nano Rods. Molecules 2021, 26, 1099. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Liu, X.; Bao, S.; Xiao, B.; Fang, T. Evaluation of Nano-Specific Toxicity of Zinc Oxide, Copper Oxide, and Silver Nanoparticles through Toxic Ratio. J. Nanopart. Res. 2016, 18, 372. [Google Scholar] [CrossRef]
- Calabrese, G.; Petralia, S.; Franco, D.; Nocito, G.; Fabbi, C.; Forte, L.; Guglielmino, S.; Squarzoni, S.; Traina, F.; Conoci, S. A New Ag-Nanostructured Hydroxyapatite Porous Scaffold: Antibacterial Effect and Cytotoxicity Study. Mater. Sci. Eng. C 2021, 118, 111394. [Google Scholar] [CrossRef]
- Malanovic, N.; Lohner, K. Gram-Positive Bacterial Cell Envelopes: The Impact on the Activity of Antimicrobial Peptides. Biochim. Biophys. Acta Biomembr. 2016, 1858, 936–946. [Google Scholar] [CrossRef]
- Huang, K.C.; Mukhopadhyay, R.; Wen, B.; Gitai, Z.; Wingreen, N.S. Cell Shape and Cell-Wall Organization in Gram-Negative Bacteria. Proc. Natl. Acad. Sci. USA 2008, 105, 19282–19287. [Google Scholar] [CrossRef]
- Dorobantu, L.S.; Fallone, C.; Noble, A.J.; Veinot, J.; Ma, G.; Goss, G.G.; Burrell, R.E. Toxicity of Silver Nanoparticles against Bacteria, Yeast, and Algae. J. Nanopart. Res. 2015, 17, 172. [Google Scholar] [CrossRef]
- Tamayo, L.; Azócar, M.; Kogan, M.; Riveros, A.; Páez, M. Copper-Polymer Nanocomposites: An Excellent and Cost-Effective Biocide for Use on Antibacterial Surfaces. Mater. Sci. Eng. C 2016, 69, 1391–1409. [Google Scholar] [CrossRef]
- Morones-Ramirez, J.R.; Winkler, J.A.; Spina, C.S.; Collins, J.J. Silver Enhances Antibiotic Activity Against Gram-Negative Bacteria. Sci. Transl. Med. 2013, 5, 190ra81. [Google Scholar] [CrossRef] [Green Version]
- Feng, Q.L.; Wu, J.; Chen, G.Q.; Cui, F.Z.; Kim, T.N.; Kim, J.O. A Mechanistic Study of the Antibacterial Effect of Silver Ions on Escherichia Coli and Staphylococcus Aureus. J. Biomed. Mater. Res. 2000, 52, 662–668. [Google Scholar] [CrossRef]
- Cavassin, E.D.; de Figueiredo, L.F.P.; Otoch, J.P.; Seckler, M.M.; de Oliveira, R.A.; Franco, F.F.; Marangoni, V.S.; Zucolotto, V.; Levin, A.S.S.; Costa, S.F. Comparison of Methods to Detect the in Vitro Activity of Silver Nanoparticles (AgNP) against Multidrug Resistant Bacteria. J. Nanobiotechnology 2015, 13, 64. [Google Scholar] [CrossRef] [PubMed]
- The Effects of Interfacial Potential on Antimicrobial Propensity of ZnO Nanoparticle|Scientific Reports. Available online: https://www.nature.com/articles/srep09578 (accessed on 5 April 2022).
- Shinde, V.V.; Dalavi, D.S.; Mali, S.S.; Hong, C.K.; Kim, J.H.; Patil, P.S. Surfactant Free Microwave Assisted Synthesis of ZnO Microspheres: Study of Their Antibacterial Activity. Appl. Surf. Sci. 2014, 307, 495–502. [Google Scholar] [CrossRef]
- Rai, M.; Kon, K.; Ingle, A.; Duran, N.; Galdiero, S.; Galdiero, M. Broad-Spectrum Bioactivities of Silver Nanoparticles: The Emerging Trends and Future Prospects. Appl. Microbiol. Biotechnol. 2014, 98, 1951–1961. [Google Scholar] [CrossRef] [PubMed]
- Abbaszadegan, A.; Ghahramani, Y.; Gholami, A.; Hemmateenejad, B.; Dorostkar, S.; Nabavizadeh, M.; Sharghi, H. The Effect of Charge at the Surface of Silver Nanoparticles on Antimicrobial Activity against Gram-Positive and Gram-Negative Bacteria: A Preliminary Study. J. Nanomater. 2015, 2015, e720654. [Google Scholar] [CrossRef]
- Slavin, Y.N.; Asnis, J.; Häfeli, U.O.; Bach, H. Metal Nanoparticles: Understanding the Mechanisms behind Antibacterial Activity. J. Nanobiotechnol. 2017, 15, 65. [Google Scholar] [CrossRef] [PubMed]
- Padmavathy, N.; Vijayaraghavan, R. Enhanced Bioactivity of ZnO Nanoparticles-an Antimicrobial Study. Sci. Technol. Adv. Mater. 2008, 9, 035004. [Google Scholar] [CrossRef]
- Penesyan, A.; Paulsen, I.T.; Kjelleberg, S.; Gillings, M.R. Three Faces of Biofilms: A Microbial Lifestyle, a Nascent Multicellular Organism, and an Incubator for Diversity. NPJ Biofilms Microbiomes 2021, 7, 80. [Google Scholar] [CrossRef]
- Zheng, S.; Bawazir, M.; Dhall, A.; Kim, H.-E.; He, L.; Heo, J.; Hwang, G. Implication of Surface Properties, Bacterial Motility, and Hydrodynamic Conditions on Bacterial Surface Sensing and Their Initial Adhesion. Front. Bioeng. Biotechnol. 2021, 9, 643722. [Google Scholar] [CrossRef]
- Rumbaugh, K.P.; Sauer, K. Biofilm Dispersion. Nat. Rev. Microbiol. 2020, 18, 571–586. [Google Scholar] [CrossRef]
- Kragh, K.N.; Hutchison, J.B.; Melaugh, G.; Rodesney, C.; Roberts, A.E.L.; Irie, Y.; Jensen, P.Ø.; Diggle, S.P.; Allen, R.J.; Gordon, V.; et al. Role of Multicellular Aggregates in Biofilm Formation. mBio 2016, 7, e00237-16. [Google Scholar] [CrossRef] [PubMed]
- Choi, O.; Yu, C.-P.; Esteban Fernández, G.; Hu, Z. Interactions of Nanosilver with Escherichia Coli Cells in Planktonic and Biofilm Cultures. Water Res. 2010, 44, 6095–6103. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Kang, F.; Gao, Y.; Mao, X.; Hu, X. Sequestration of Nanoparticles by an EPS Matrix Reduces the Particle-Specific Bactericidal Activity. Sci. Rep. 2016, 6, 21379. [Google Scholar] [CrossRef] [PubMed]
- Gomes, I.B.; Simões, M.; Simões, L.C. Copper Surfaces in Biofilm Control. Nanomaterials 2020, 10, 2491. [Google Scholar] [CrossRef] [PubMed]
- Roe, D.; Karandikar, B.; Bonn-Savage, N.; Gibbins, B.; Roullet, J.-B. Antimicrobial Surface Functionalization of Plastic Catheters by Silver Nanoparticles. J. Antimicrob. Chemother. 2008, 61, 869–876. [Google Scholar] [CrossRef]
- Gomez-Carretero, S.; Nybom, R.; Richter-Dahlfors, A. Electroenhanced Antimicrobial Coating Based on Conjugated Polymers with Covalently Coupled Silver Nanoparticles Prevents Staphylococcus Aureus Biofilm Formation. Adv. Healthc. Mater. 2017, 6, 1700435. [Google Scholar] [CrossRef]
- Spirescu, V.A.; Șuhan, R.; Niculescu, A.-G.; Grumezescu, V.; Negut, I.; Holban, A.M.; Oprea, O.-C.; Bîrcă, A.C.; Vasile, B.Ș.; Grumezescu, A.M.; et al. Biofilm-Resistant Nanocoatings Based on ZnO Nanoparticles and Linalool. Nanomaterials 2021, 11, 2564. [Google Scholar] [CrossRef]
- Applerot, G.; Lellouche, J.; Perkas, N.; Nitzan, Y.; Gedanken, A.; Banin, E. ZnO Nanoparticle-Coated Surfaces Inhibit Bacterial Biofilm Formation and Increase Antibiotic Susceptibility. RSC Adv. 2012, 2, 2314–2321. [Google Scholar] [CrossRef]
- Bazzi, W.; Abou Fayad, A.G.; Nasser, A.; Haraoui, L.-P.; Dewachi, O.; Abou-Sitta, G.; Nguyen, V.-K.; Abara, A.; Karah, N.; Landecker, H.; et al. Heavy Metal Toxicity in Armed Conflicts Potentiates AMR in A. Baumannii by Selecting for Antibiotic and Heavy Metal Co-Resistance Mechanisms. Front. Microbiol. 2020, 11, 68. [Google Scholar] [CrossRef]
- Thambirajoo, M.; Maarof, M.; Lokanathan, Y.; Katas, H.; Ghazalli, N.F.; Tabata, Y.; Fauzi, M.B. Potential of Nanoparticles Integrated with Antibacterial Properties in Preventing Biofilm and Antibiotic Resistance. Antibiotics 2021, 10, 1338. [Google Scholar] [CrossRef]
- Chernousova, S.; Epple, M. Silver as Antibacterial Agent: Ion, Nanoparticle, and Metal. Angew. Chem. Int. Ed. 2013, 52, 1636–1653. [Google Scholar] [CrossRef]
- Guo, D.; Xie, G.; Luo, J. Mechanical Properties of Nanoparticles: Basics and Applications. J. Phys. D Appl. Phys. 2013, 47, 013001. [Google Scholar] [CrossRef]
- Lue, J.-T. A Review of Characterization and Physical Property Studies of Metallic Nanoparticles. J. Phys. Chem. Solids 2001, 62, 1599–1612. [Google Scholar] [CrossRef]
- Khan, A.U.; Yuan, Q.; Wei, Y.; Khan, G.M.; Khan, Z.U.H.; Khan, S.; Ali, F.; Tahir, K.; Ahmad, A.; Khan, F.U. Photocatalytic and Antibacterial Response of Biosynthesized Gold Nanoparticles. J. Photochem. Photobiol. B Biol. 2016, 162, 273–277. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, X.; Yang, L.; Ren, L.; Wang, D.; Ye, J. Metal Nanoparticles Induced Photocatalysis. Natl. Sci. Rev. 2017, 4, 761–780. [Google Scholar] [CrossRef]
- Grazú, V.; Silber, A.; Moros, M.; Asín, L.; Torres, T.; Marquina, C.; Ibarra, M.; Goya, G. Application of Magnetically Induced Hyperthermia in the Model Protozoan Crithidia Fasciculata as a Potential Therapy against Parasitic Infections. Int. J. Nanomed. 2012, 7, 5351–5360. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, Y.; Wang, Y.; Zhu, W.; Li, G.; Ma, X.; Zhang, Y.; Chen, S.; Tiwari, S.; Shi, K.; et al. Comprehensive Understanding of Magnetic Hyperthermia for Improving Antitumor Therapeutic Efficacy. Theranostics 2020, 10, 3793–3815. [Google Scholar] [CrossRef]
- Cui, L.; Chen, P.; Chen, S.; Yuan, Z.; Yu, C.; Ren, B.; Zhang, K. In Situ Study of the Antibacterial Activity and Mechanism of Action of Silver Nanoparticles by Surface-Enhanced Raman Spectroscopy. Anal. Chem. 2013, 85, 5436–5443. [Google Scholar] [CrossRef]
- Pérez-Díaz, M.A.; Boegli, L.; James, G.; Velasquillo, C.; Sánchez-Sánchez, R.; Martínez-Martínez, R.-E.; Martínez-Castañón, G.A.; Martinez-Gutierrez, F. Silver Nanoparticles with Antimicrobial Activities against Streptococcus Mutans and Their Cytotoxic Effect. Mater. Sci. Eng. C 2015, 55, 360–366. [Google Scholar] [CrossRef]
- Wang, L.; Hu, C.; Shao, L. The Antimicrobial Activity of Nanoparticles: Present Situation and Prospects for the Future. Int. J. Nanomed. 2017, 12, 1227–1249. [Google Scholar] [CrossRef] [Green Version]
- Ivask, A.; ElBadawy, A.; Kaweeteerawat, C.; Boren, D.; Fischer, H.; Ji, Z.; Chang, C.H.; Liu, R.; Tolaymat, T.; Telesca, D.; et al. Toxicity Mechanisms in Escherichia Coli Vary for Silver Nanoparticles and Differ from Ionic Silver. ACS Nano 2014, 8, 374–386. [Google Scholar] [CrossRef] [PubMed]
- Thill, A.; Zeyons, O.; Spalla, O.; Chauvat, F.; Rose, J.; Auffan, M.; Flank, A.M. Cytotoxicity of CeO2 Nanoparticles for Escherichia Coli. Physico-Chemical Insight of the Cytotoxicity Mechanism. Environ. Sci. Technol. 2006, 40, 6151–6156. [Google Scholar] [CrossRef] [PubMed]
- Wang, E.C.; Wang, A.Z. Nanoparticles and Their Applications in Cell and Molecular Biology. Integr. Biol. 2014, 6, 9–26. [Google Scholar] [CrossRef] [PubMed]
- Shang, L.; Nienhaus, K.; Nienhaus, G.U. Engineered Nanoparticles Interacting with Cells: Size Matters. J. Nanobiotechnol. 2014, 12, 5. [Google Scholar] [CrossRef] [PubMed]
- Skomorokhova, E.A.; Sankova, T.P.; Orlov, I.A.; Savelev, A.N.; Magazenkova, D.N.; Pliss, M.G.; Skvortsov, A.N.; Sosnin, I.M.; Kirilenko, D.A.; Grishchuk, I.V.; et al. Size-Dependent Bioactivity of Silver Nanoparticles: Antibacterial Properties, Influence on Copper Status in Mice, and Whole-Body Turnover. Nanotechnol. Sci. Appl. 2020, 13, 137–157. [Google Scholar] [CrossRef]
- Korshed, P.; Li, L.; Liu, Z.; Mironov, A.; Wang, T. Size-Dependent Antibacterial Activity for Laser-Generated Silver Nanoparticles. J. Interdiscip. Nanomed. 2019, 4, 24–33. [Google Scholar] [CrossRef]
- Zare, M.; Namratha, K.; Byrappa, K.; Surendra, D.M.; Yallappa, S.; Hungund, B. Surfactant Assisted Solvothermal Synthesis of ZnO Nanoparticles and Study of Their Antimicrobial and Antioxidant Properties. J. Mater. Sci. Technol. 2018, 34, 1035–1043. [Google Scholar] [CrossRef]
- Adams, C.P.; Walker, K.A.; Obare, S.O.; Docherty, K.M. Size-Dependent Antimicrobial Effects of Novel Palladium Nanoparticles. PLoS ONE 2014, 9, e85981. [Google Scholar] [CrossRef]
- Hong, X.; Wen, J.; Xiong, X.; Hu, Y. Shape Effect on the Antibacterial Activity of Silver Nanoparticles Synthesized via a Microwave-Assisted Method. Environ. Sci. Pollut. Res. Int. 2016, 23, 4489–4497. [Google Scholar] [CrossRef]
- Huynh, P.T.; Nguyen, G.D.; Tran, K.T.L.; Ho, T.M.; Duong, B.T.; Lam, V.Q.; Ngo, T.V.K. One-Pot, Surfactant-Free Synthesis of Gold Nanostars and Evaluation of Their Antibacterial Effects against Propionibacterium Acnes. J. Nanomater. 2021, 2021, e6650661. [Google Scholar] [CrossRef]
- Li, X.; Ahmad, K.Z.; He, J.; Li, H.; Wang, X.; Feng, Z.; Wang, X.; Shen, G.; Ding, X. Silver Nanoflowers Coupled with Low Dose Antibiotics Enable the Highly Effective Eradication of Drug-Resistant Bacteria. J. Mater. Chem. B 2021, 9, 9839–9851. [Google Scholar] [CrossRef] [PubMed]
- Guo, B.-L.; Han, P.; Guo, L.-C.; Cao, Y.-Q.; Li, A.-D.; Kong, J.-Z.; Zhai, H.-F.; Wu, D. The Antibacterial Activity of Ta-Doped ZnO Nanoparticles. Nanoscale Res. Lett. 2015, 10, 336. [Google Scholar] [CrossRef] [PubMed]
- López-Heras, M.; Theodorou, I.G.; Leo, B.F.; Ryan, M.P.; Porter, A.E. Towards Understanding the Antibacterial Activity of Ag Nanoparticles: Electron Microscopy in the Analysis of the Materials-Biology Interface in the Lung. Environ. Sci. Nano 2015, 2, 312–326. [Google Scholar] [CrossRef]
- Li, Z.; Ma, J.; Ruan, J.; Zhuang, X. Using Positively Charged Magnetic Nanoparticles to Capture Bacteria at Ultralow Concentration. Nanoscale Res. Lett. 2019, 14, 195. [Google Scholar] [CrossRef]
- Mammari, N.; Lamouroux, E.; Boudier, A.; Duval, R.E. Current Knowledge on the Oxidative-Stress-Mediated Antimicrobial Properties of Metal-Based Nanoparticles. Microorganisms 2022, 10, 437. [Google Scholar] [CrossRef] [PubMed]
- El Badawy, A.M.; Silva, R.G.; Morris, B.; Scheckel, K.G.; Suidan, M.T.; Tolaymat, T.M. Surface Charge-Dependent Toxicity of Silver Nanoparticles. Environ. Sci. Technol. 2011, 45, 283–287. [Google Scholar] [CrossRef]
- Agnihotri, S.; Mukherji, S.; Mukherji, S. Size-Controlled Silver Nanoparticles Synthesized over the Range 5–100 Nm Using the Same Protocol and Their Antibacterial Efficacy. RSC Adv. 2013, 4, 3974–3983. [Google Scholar] [CrossRef]
- Dizaj, S.M.; Lotfipour, F.; Barzegar-Jalali, M.; Zarrintan, M.H.; Adibkia, K. Antimicrobial Activity of the Metals and Metal Oxide Nanoparticles. Mater. Sci. Eng. C 2014, 44, 278–284. [Google Scholar] [CrossRef]
- Rice, K.C.; Bayles, K.W. Molecular Control of Bacterial Death and Lysis. Microbiol. Mol. Biol. Rev. 2008, 72, 85–109. [Google Scholar] [CrossRef]
- Ramalingam, B.; Parandhaman, T.; Das, S.K. Antibacterial Effects of Biosynthesized Silver Nanoparticles on Surface Ultrastructure and Nanomechanical Properties of Gram-Negative Bacteria Viz. Escherichia Coli and Pseudomonas Aeruginosa. ACS Appl. Mater. Interfaces 2016, 8, 4963–4976. [Google Scholar] [CrossRef]
- Sukhanova, A.; Bozrova, S.; Sokolov, P.; Berestovoy, M.; Karaulov, A.; Nabiev, I. Dependence of Nanoparticle Toxicity on Their Physical and Chemical Properties. Nanoscale Res. Lett. 2018, 13, 44. [Google Scholar] [CrossRef] [PubMed]
- Ninganagouda, S.; Rathod, V.; Singh, D.; Hiremath, J.; Singh, A.K.; Mathew, J.; ul-Haq, M. Growth Kinetics and Mechanistic Action of Reactive Oxygen Species Released by Silver Nanoparticles from Aspergillus Niger on Escherichia Coli. BioMed Res. Int. 2014, 2014, 753419. [Google Scholar] [CrossRef] [PubMed]
- Leung, Y.H.; Ng, A.M.C.; Xu, X.; Shen, Z.; Gethings, L.A.; Wong, M.T.; Chan, C.M.N.; Guo, M.Y.; Ng, Y.H.; Djurišić, A.B.; et al. Mechanisms of Antibacterial Activity of MgO: Non-ROS Mediated Toxicity of MgO Nanoparticles towards Escherichia Coli. Small 2014, 10, 1171–1183. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Wang, Y.; Chen, Z.; Pan, D.; Cheng, Y.; Liu, Z.; Lin, Z.; Guan, X. Investigation of Antibacterial Activity and Related Mechanism of a Series of Nano-Mg(OH)2. ACS Appl. Mater. Interfaces 2013, 5, 1137–1142. [Google Scholar] [CrossRef] [PubMed]
- McQuillan, J.S.; Shaw, A.M. Differential Gene Regulation in the Ag Nanoparticle and Ag(+)-Induced Silver Stress Response in Escherichia Coli: A Full Transcriptomic Profile. Nanotoxicology 2014, 8 (Suppl. 1), 177–184. [Google Scholar] [CrossRef]
- Mukha, I.P.; Eremenko, A.M.; Smirnova, N.P.; Mikhienkova, A.I.; Korchak, G.I.; Gorchev, V.F.; Chunikhin, A.Y. Antimicrobial Activity of Stable Silver Nanoparticles of a Certain Size. Appl. Biochem. Microbiol. 2013, 49, 199–206. [Google Scholar] [CrossRef]
- Dong, Y.; Zhu, H.; Shen, Y.; Zhang, W.; Zhang, L. Antibacterial Activity of Silver Nanoparticles of Different Particle Size against Vibrio Natriegens. PLoS ONE 2019, 14, e0222322. [Google Scholar] [CrossRef]
- Oves, M.; Rauf, M.A.; Hussain, A.; Qari, H.A.; Khan, A.A.P.; Muhammad, P.; Rehman, M.T.; Alajmi, M.F.; Ismail, I.I.M. Antibacterial Silver Nanomaterial Synthesis From Mesoflavibacter Zeaxanthinifaciens and Targeting Biofilm Formation. Front. Pharmacol. 2019, 10, 801. [Google Scholar] [CrossRef]
- Cui, Y.; Zhao, Y.; Tian, Y.; Zhang, W.; Lü, X.; Jiang, X. The Molecular Mechanism of Action of Bactericidal Gold Nanoparticles on Escherichia Coli. Biomaterials 2012, 33, 2327–2333. [Google Scholar] [CrossRef]
- Lemire, J.A.; Harrison, J.J.; Turner, R.J. Antimicrobial Activity of Metals: Mechanisms, Molecular Targets and Applications. Nat. Rev. Microbiol. 2013, 11, 371–384. [Google Scholar] [CrossRef]
- Chatterjee, A.K.; Chakraborty, R.; Basu, T. Mechanism of Antibacterial Activity of Copper Nanoparticles. Nanotechnology 2014, 25, 135101. [Google Scholar] [CrossRef] [PubMed]
- Godoy-Gallardo, M.; Eckhard, U.; Delgado, L.M.; de Roo Puente, Y.J.D.; Hoyos-Nogués, M.; Gil, F.J.; Perez, R.A. Antibacterial Approaches in Tissue Engineering Using Metal Ions and Nanoparticles: From Mechanisms to Applications. Bioact. Mater. 2021, 6, 4470–4490. [Google Scholar] [CrossRef] [PubMed]
- Ishida, T. Antibacterial Mechanism of Ag+ Ions for Bacteriolyses of Bacterial Cell Walls via Peptidoglycan Autolysins, and DNA Damages. MOJ Toxicol. 2018, 4, 345–350. [Google Scholar] [CrossRef]
- Saliani, M.; Jalal, R.; Goharshadi, E.K. Effects of PH and Temperature on Antibacterial Activity of Zinc Oxide Nanofluid Against Escherichia Coli O157: H7 and Staphylococcus Aureus. Jundishapur J. Microbiol. 2015, 8, e17115. [Google Scholar] [CrossRef] [PubMed]
- Moreau, J.W.; Weber, P.K.; Martin, M.C.; Gilbert, B.; Hutcheon, I.D.; Banfield, J.F. Extracellular Proteins Limit the Dispersal of Biogenic Nanoparticles. Science 2007, 316, 1600–1603. [Google Scholar] [CrossRef]
- Peretyazhko, T.S.; Zhang, Q.; Colvin, V.L. Size-Controlled Dissolution of Silver Nanoparticles at Neutral and Acidic PH Conditions: Kinetics and Size Changes. Environ. Sci. Technol. 2014, 48, 11954–11961. [Google Scholar] [CrossRef]
- Wigginton, N.S.; de Titta, A.; Piccapietra, F.; Dobias, J.; Nesatyy, V.J.; Suter, M.J.F.; Bernier-Latmani, R. Binding of Silver Nanoparticles to Bacterial Proteins Depends on Surface Modifications and Inhibits Enzymatic Activity. Environ. Sci. Technol. 2010, 44, 2163–2168. [Google Scholar] [CrossRef]
- Chatterjee, T.; Chatterjee, B.K.; Majumdar, D.; Chakrabarti, P. Antibacterial Effect of Silver Nanoparticles and the Modeling of Bacterial Growth Kinetics Using a Modified Gompertz Model. Biochim. Biophys. Acta Biomembr. 2015, 1850, 299–306. [Google Scholar] [CrossRef]
- Qiu, T.A.; Guidolin, V.; Hoang, K.N.L.; Pho, T.; Carra’, A.; Villalta, P.W.; He, J.; Yao, X.; Hamers, R.J.; Balbo, S.; et al. Nanoscale Battery Cathode Materials Induce DNA Damage in Bacteria. Chem. Sci. 2020, 11, 11244–11258. [Google Scholar] [CrossRef]
- Sharma, V.K.; Sayes, C.M.; Guo, B.; Pillai, S.; Parsons, J.G.; Wang, C.; Yan, B.; Ma, X. Interactions between Silver Nanoparticles and Other Metal Nanoparticles under Environmentally Relevant Conditions: A Review. Sci. Total Environ. 2019, 653, 1042–1051. [Google Scholar] [CrossRef]
- Levard, C.; Mitra, S.; Yang, T.; Jew, A.D.; Badireddy, A.R.; Lowry, G.V.; Brown, G.E. Effect of Chloride on the Dissolution Rate of Silver Nanoparticles and Toxicity to E. Coli. Environ. Sci. Technol. 2013, 47, 5738–5745. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Greden, K.; Alvarez, P.J.J.; Gregory, K.B.; Lowry, G.V. Adsorbed Polymer and NOM Limits Adhesion and Toxicity of Nano Scale Zerovalent Iron to E. Coli. Environ. Sci. Technol. 2010, 44, 3462–3467. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Kuk, E.; Yu, K.N.; Kim, J.-H.; Park, S.J.; Lee, H.J.; Kim, S.H.; Park, Y.K.; Park, Y.H.; Hwang, C.-Y.; et al. Antimicrobial Effects of Silver Nanoparticles. Nanomedicine 2007, 3, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Ranjan, S.; Ramalingam, C. Titanium Dioxide Nanoparticles Induce Bacterial Membrane Rupture by Reactive Oxygen Species Generation. Environ. Chem. Lett. 2016, 14, 487–494. [Google Scholar] [CrossRef]
- Sirelkhatim, A.; Mahmud, S.; Seeni, A.; Kaus, N.H.M.; Ann, L.C.; Bakhori, S.K.M.; Hasan, H.; Mohamad, D. Review on Zinc Oxide Nanoparticles: Antibacterial Activity and Toxicity Mechanism. Nano Micro Lett. 2015, 7, 219–242. [Google Scholar] [CrossRef]
- Jung, W.K.; Koo, H.C.; Kim, K.W.; Shin, S.; Kim, S.H.; Park, Y.H. Antibacterial Activity and Mechanism of Action of the Silver Ion in Staphylococcus Aureus and Escherichia Coli. Appl. Environ. Microbiol. 2008, 74, 2171–2178. [Google Scholar] [CrossRef]
- Jahnke, J.P.; Cornejo, J.A.; Sumner, J.J.; Schuler, A.J.; Atanassov, P.; Ista, L.K. Conjugated Gold Nanoparticles as a Tool for Probing the Bacterial Cell Envelope: The Case of Shewanella Oneidensis MR-1. Biointerphases 2016, 11, 011003. [Google Scholar] [CrossRef]
- Ezraty, B.; Gennaris, A.; Barras, F.; Collet, J.-F. Oxidative Stress, Protein Damage and Repair in Bacteria. Nat. Rev. Microbiol. 2017, 15, 385–396. [Google Scholar] [CrossRef]
- Xu, H.; Qu, F.; Xu, H.; Lai, W.; Andrew Wang, Y.; Aguilar, Z.P.; Wei, H. Role of Reactive Oxygen Species in the Antibacterial Mechanism of Silver Nanoparticles on Escherichia Coli O157:H7. Biometals 2012, 25, 45–53. [Google Scholar] [CrossRef]
- Choi, O.; Hu, Z. Size Dependent and Reactive Oxygen Species Related Nanosilver Toxicity to Nitrifying Bacteria. Environ. Sci. Technol. 2008, 42, 4583–4588. [Google Scholar] [CrossRef]
- Xie, Y.; He, Y.; Irwin, P.L.; Jin, T.; Shi, X. Antibacterial Activity and Mechanism of Action of Zinc Oxide Nanoparticles against Campylobacter Jejuni. Appl. Environ. Microbiol. 2011, 77, 2325–2331. [Google Scholar] [CrossRef] [PubMed]
- Mujeeb, A.A.; Khan, N.A.; Jamal, F.; Badre Alam, K.F.; Saeed, H.; Kazmi, S.; Alshameri, A.W.F.; Kashif, M.; Ghazi, I.; Owais, M. Olax Scandens Mediated Biogenic Synthesis of Ag-Cu Nanocomposites: Potential Against Inhibition of Drug-Resistant Microbes. Front. Chem. 2020, 8, 103. [Google Scholar] [CrossRef]
- Kumariya, R.; Sood, S.K.; Rajput, Y.S.; Saini, N.; Garsa, A.K. Increased Membrane Surface Positive Charge and Altered Membrane Fluidity Leads to Cationic Antimicrobial Peptide Resistance in Enterococcus Faecalis. Biochim. Biophys. Acta Biomembr. 2015, 1848, 1367–1375. [Google Scholar] [CrossRef]
- Jordan, S.; Hutchings, M.I.; Mascher, T. Cell Envelope Stress Response in Gram-Positive Bacteria. FEMS Microbiol. Rev. 2008, 32, 107–146. [Google Scholar] [CrossRef] [PubMed]
- Tzeng, Y.-L.; Ambrose, K.D.; Zughaier, S.; Zhou, X.; Miller, Y.K.; Shafer, W.M.; Stephens, D.S. Cationic Antimicrobial Peptide Resistance in Neisseria Meningitidis. J. Bacteriol. 2005, 187, 5387–5396. [Google Scholar] [CrossRef] [PubMed]
- Salusso, A.; Raimunda, D. Defining the Roles of the Cation Diffusion Facilitators in Fe2+/Zn2+ Homeostasis and Establishment of Their Participation in Virulence in Pseudomonas Aeruginosa. Front. Cell. Infect. Microbiol. 2017, 7, 84. [Google Scholar] [CrossRef]
- Niño-Martínez, N.; Salas Orozco, M.F.; Martínez-Castañón, G.-A.; Torres Méndez, F.; Ruiz, F. Molecular Mechanisms of Bacterial Resistance to Metal and Metal Oxide Nanoparticles. Int. J. Mol. Sci. 2019, 20, 2808. [Google Scholar] [CrossRef]
- Imran, M.; Das, K.R.; Naik, M.M. Co-Selection of Multi-Antibiotic Resistance in Bacterial Pathogens in Metal and Microplastic Contaminated Environments: An Emerging Health Threat. Chemosphere 2019, 215, 846–857. [Google Scholar] [CrossRef]
- Ellis, D.H.; Maurer-Gardner, E.I.; Sulentic, C.E.W.; Hussain, S.M. Silver Nanoparticle Antibacterial Efficacy and Resistance Development in Key Bacterial Species. Biomed. Phys. Eng. Express 2018, 5, 015013. [Google Scholar] [CrossRef]
- Randall, C.P.; Gupta, A.; Jackson, N.; Busse, D.; O’Neill, A.J. Silver Resistance in Gram-Negative Bacteria: A Dissection of Endogenous and Exogenous Mechanisms. J. Antimicrob. Chemother. 2015, 70, 1037–1046. [Google Scholar] [CrossRef] [Green Version]
- Chandrangsu, P.; Rensing, C.; Helmann, J.D. Metal Homeostasis and Resistance in Bacteria. Nat. Rev. Microbiol. 2017, 15, 338–350. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yang, F.; Zhao, J.; Xu, Y.; Mao, D.; Zhu, X.; Luo, Y.; Alvarez, P.J.J. Bacterial Exposure to ZnO Nanoparticles Facilitates Horizontal Transfer of Antibiotic Resistance Genes. NanoImpact 2018, 10, 61–67. [Google Scholar] [CrossRef]
- Qiu, Z.; Shen, Z.; Qian, D.; Jin, M.; Yang, D.; Wang, J.; Zhang, B.; Yang, Z.; Chen, Z.; Wang, X.; et al. Effects of Nano-TiO2 on Antibiotic Resistance Transfer Mediated by RP4 Plasmid. Nanotoxicology 2015, 9, 895–904. [Google Scholar] [CrossRef]
- Panáček, A.; Kvítek, L.; Smékalová, M.; Večeřová, R.; Kolář, M.; Röderová, M.; Dyčka, F.; Šebela, M.; Prucek, R.; Tomanec, O.; et al. Bacterial Resistance to Silver Nanoparticles and How to Overcome It. Nat. Nanotech. 2018, 13, 65–71. [Google Scholar] [CrossRef]
- Faghihzadeh, F.; Anaya, N.M.; Astudillo-Castro, C.; Oyanedel-Craver, V. Kinetic, Metabolic and Macromolecular Response of Bacteria to Chronic Nanoparticle Exposure in Continuous Culture. Environ. Sci. Nano 2018, 5, 1386–1396. [Google Scholar] [CrossRef]
- Sohm, B.; Immel, F.; Bauda, P.; Pagnout, C. Insight into the Primary Mode of Action of TiO2 Nanoparticles on Escherichia Coli in the Dark. Proteomics 2015, 15, 98–113. [Google Scholar] [CrossRef]
- Liu, J.; Liu, J.; Attarilar, S.; Wang, C.; Tamaddon, M.; Yang, C.; Xie, K.; Yao, J.; Wang, L.; Liu, C.; et al. Nano-Modified Titanium Implant Materials: A Way Toward Improved Antibacterial Properties. Front. Bioeng. Biotechnol. 2020, 8, 576969. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Yang, L.; Zhang, L.; Han, Y.; Lu, Z.; Qin, G.; Zhang, E. Effect of Nano/Micro-Ag Compound Particles on the Bio-Corrosion, Antibacterial Properties and Cell Biocompatibility of Ti-Ag Alloys. Mater. Sci. Eng. C 2017, 75, 906–917. [Google Scholar] [CrossRef]
- Bakhsheshi-Rad, H.R.; Hamzah, E.; Low, H.T.; Kasiri-Asgarani, M.; Farahany, S.; Akbari, E.; Cho, M.H. Fabrication of Biodegradable Zn-Al-Mg Alloy: Mechanical Properties, Corrosion Behavior, Cytotoxicity and Antibacterial Activities. Mater. Sci. Eng. C 2017, 73, 215–219. [Google Scholar] [CrossRef]
- Cao, H.; Zhang, W.; Meng, F.; Guo, J.; Wang, D.; Qian, S.; Jiang, X.; Liu, X.; Chu, P.K. Osteogenesis Catalyzed by Titanium-Supported Silver Nanoparticles. ACS Appl. Mater. Interfaces 2017, 9, 5149–5157. [Google Scholar] [CrossRef]
- Calabrese, G.; Franco, D.; Petralia, S.; Monforte, F.; Condorelli, G.G.; Squarzoni, S.; Traina, F.; Conoci, S. Dual-Functional Nano-Functionalized Titanium Scaffolds to Inhibit Bacterial Growth and Enhance Osteointegration. Nanomaterials 2021, 11, 2634. [Google Scholar] [CrossRef] [PubMed]
- Shahid, A.; Aslam, B.; Muzammil, S.; Aslam, N.; Shahid, M.; Almatroudi, A.; Allemailem, K.S.; Saqalein, M.; Nisar, M.A.; Rasool, M.H.; et al. The Prospects of Antimicrobial Coated Medical Implants. J. Appl. Biomater. Funct. Mater. 2021, 19, 22808000211040304. [Google Scholar] [CrossRef] [PubMed]
- Dube, T.; Freckmann, D.; Conoci, S.; Gambarotta, S.; Yap, G.P.A. Monomeric and Octameric Divalent Ytterbium Complexes of Diphenylmethyl Dipyrrolyl Dianion. Organometallics 2000, 19, 209–211. [Google Scholar] [CrossRef]
- Miola, M.; Bruno, M.; Maina, G.; Fucale, G.; Lucchetta, G.; Vernè, E. Antibiotic-Free Composite Bone Cements with Antibacterial and Bioactive Properties. A Preliminary Study. Mater. Sci. Eng. C 2014, 43, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Kose, N.; Çaylak, R.; Pekşen, C.; Kiremitçi, A.; Burukoglu, D.; Koparal, S.; Doğan, A. Silver Ion Doped Ceramic Nano-Powder Coated Nails Prevent Infection in Open Fractures: In Vivo Study. Injury 2016, 47, 320–324. [Google Scholar] [CrossRef]
- Samuel, U.; Guggenbichler, J.P. Prevention of Catheter-Related Infections: The Potential of a New Nano-Silver Impregnated Catheter. Int. J. Antimicrob. Agents 2004, 23 (Suppl. 1), 75–78. [Google Scholar] [CrossRef]
- Galiano, K.; Pleifer, C.; Engelhardt, K.; Brössner, G.; Lackner, P.; Huck, C.; Lass-Flörl, C.; Obwegeser, A. Silver Segregation and Bacterial Growth of Intraventricular Catheters Impregnated with Silver Nanoparticles in Cerebrospinal Fluid Drainages. Neurol. Res. 2008, 30, 285–287. [Google Scholar] [CrossRef]
- Chen, P.; Wu, Z.; Leung, A.; Chen, X.; Landao-Bassonga, E.; Gao, J.; Chen, L.; Zheng, M.; Yao, F.; Yang, H.; et al. Fabrication of a Silver Nanoparticle-Coated Collagen Membrane with Anti-Bacterial and Anti-Inflammatory Activities for Guided Bone Regeneration. Biomed. Mater. 2018, 13, 065014. [Google Scholar] [CrossRef]
- Ramazanzadeh, B.; Jahanbin, A.; Yaghoubi, M.; Shahtahmassbi, N.; Ghazvini, K.; Shakeri, M.; Shafaee, H. Comparison of Antibacterial Effects of ZnO and CuO Nanoparticles Coated Brackets against Streptococcus Mutans. J. Dent. 2015, 16, 200–205. [Google Scholar]
- Li, C.; Fu, R.; Yu, C.; Li, Z.; Guan, H.; Hu, D.; Zhao, D.; Lu, L. Silver Nanoparticle/Chitosan Oligosaccharide/Poly(Vinyl Alcohol) Nanofibers as Wound Dressings: A Preclinical Study. Int. J. Nanomed. 2013, 8, 4131–4145. [Google Scholar] [CrossRef]
- dos Santos, C.A.; Ingle, A.P.; Rai, M. The Emerging Role of Metallic Nanoparticles in Food. Appl. Microbiol. Biotechnol. 2020, 104, 2373–2383. [Google Scholar] [CrossRef] [PubMed]
- Couto, C.; Almeida, A. Metallic Nanoparticles in the Food Sector: A Mini-Review. Foods 2022, 11, 402. [Google Scholar] [CrossRef]
- Singh, S.P.; Bhargava, C.S.; Dubey, V.; Mishra, A.; Singh, Y. Silver Nanoparticles: Biomedical Applications, Toxicity, and Safety Issues. Int. J. Res. Pharm. Pharm. Sci. 2017, 4, 1–10. [Google Scholar]
- Alsammarraie, F.K.; Wang, W.; Zhou, P.; Mustapha, A.; Lin, M. Green Synthesis of Silver Nanoparticles Using Turmeric Extracts and Investigation of Their Antibacterial Activities. Colloids Surf. B Biointerfaces 2018, 171, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Makvandi, P.; Ashrafizadeh, M.; Ghomi, M.; Najafi, M.; Hossein, H.H.S.; Zarrabi, A.; Mattoli, V.; Varma, R.S. Injectable Hyaluronic Acid-Based Antibacterial Hydrogel Adorned with Biogenically Synthesized AgNPs-Decorated Multi-Walled Carbon Nanotubes. Prog. Biomater. 2021, 10, 77–89. [Google Scholar] [CrossRef]
- Makvandi, P.; Ali, G.W.; Della Sala, F.; Abdel-Fattah, W.I.; Borzacchiello, A. Hyaluronic Acid/Corn Silk Extract Based Injectable Nanocomposite: A Biomimetic Antibacterial Scaffold for Bone Tissue Regeneration. Mater. Sci. Eng. C 2020, 107, 110195. [Google Scholar] [CrossRef]
- Ravindran, D.; Ramanathan, S.; Arunachalam, K.; Jeyaraj, G.P.; Shunmugiah, K.P.; Arumugam, V.R. Phytosynthesized Silver Nanoparticles as Antiquorum Sensing and Antibiofilm Agent against the Nosocomial Pathogen Serratia Marcescens: An in Vitro Study. J. Appl. Microbiol. 2018, 124, 1425–1440. [Google Scholar] [CrossRef]
- Thomas, R.; Snigdha, S.; Bhavitha, K.B.; Babu, S.; Ajith, A.; Radhakrishnan, E.K. Biofabricated Silver Nanoparticles Incorporated Polymethyl Methacrylate as a Dental Adhesive Material with Antibacterial and Antibiofilm Activity against Streptococcus Mutans. 3 Biotech 2018, 8, 404. [Google Scholar] [CrossRef]
- Kim, M.H.; Park, H.; Nam, H.C.; Park, S.R.; Jung, J.-Y.; Park, W.H. Injectable Methylcellulose Hydrogel Containing Silver Oxide Nanoparticles for Burn Wound Healing. Carbohydr. Polym. 2018, 181, 579–586. [Google Scholar] [CrossRef]
- Hasan, A.; Morshed, M.; Memic, A.; Hassan, S.; Webster, T.J.; Marei, H.E.-S. Nanoparticles in Tissue Engineering: Applications, Challenges and Prospects. Int. J. Nanomed. 2018, 13, 5637–5655. [Google Scholar] [CrossRef]
- Jamróz, E.; Cabaj, A.; Juszczak, L.; Tkaczewska, J.; Zimowska, M.; Cholewa-Wójcik, A.; Krzyściak, P.; Kopel, P. Active Double-Layered Films Enriched with AgNPs in Great Water Dock Root and Pu-Erh Extracts. Materials 2021, 14, 6925. [Google Scholar] [CrossRef] [PubMed]
- Myakinin, A.; Turlybekuly, A.; Pogrebnjak, A.; Mirek, A.; Bechelany, M.; Liubchak, I.; Oleshko, O.; Husak, Y.; Korniienko, V.; Leśniak-Ziółkowska, K.; et al. In Vitro Evaluation of Electrochemically Bioactivated Ti6Al4V 3D Porous Scaffolds. Mater. Sci. Eng. C 2021, 121, 111870. [Google Scholar] [CrossRef] [PubMed]
- Cochis, A.; Azzimonti, B.; Della Valle, C.; De Giglio, E.; Bloise, N.; Visai, L.; Cometa, S.; Rimondini, L.; Chiesa, R. The Effect of Silver or Gallium Doped Titanium against the Multidrug Resistant Acinetobacter Baumannii. Biomaterials 2016, 80, 80–95. [Google Scholar] [CrossRef] [PubMed]
- Felix, W.P.; Muthu, P. Bioscaffolds Impregnated with Ormocarpum Cochinchinense Mediated Ag Nanoparticles. In Proceedings of the 2016 International Conference on Electrical, Electronics, and Optimization Techniques (ICEEOT), Chennai, India, 3–5 March 2016; pp. 3533–3535. [Google Scholar]
- Shimazaki, T.; Miyamoto, H.; Ando, Y.; Noda, I.; Yonekura, Y.; Kawano, S.; Miyazaki, M.; Mawatari, M.; Hotokebuchi, T. In Vivo Antibacterial and Silver-Releasing Properties of Novel Thermal Sprayed Silver-Containing Hydroxyapatite Coating. J. Biomed. Mater. Res. B Appl. Biomater. 2010, 92, 386–389. [Google Scholar] [CrossRef]
- Saravanan, S.; Nethala, S.; Pattnaik, S.; Tripathi, A.; Moorthi, A.; Selvamurugan, N. Preparation, Characterization and Antimicrobial Activity of a Bio-Composite Scaffold Containing Chitosan/Nano-Hydroxyapatite/Nano-Silver for Bone Tissue Engineering. Int. J. Biol. Macromol. 2011, 49, 188–193. [Google Scholar] [CrossRef]
- Alt, V.; Bechert, T.; Steinrücke, P.; Wagener, M.; Seidel, P.; Dingeldein, E.; Domann, E.; Schnettler, R. An in Vitro Assessment of the Antibacterial Properties and Cytotoxicity of Nanoparticulate Silver Bone Cement. Biomaterials 2004, 25, 4383–4391. [Google Scholar] [CrossRef]
- Halkai, K.R.; Mudda, J.A.; Shivanna, V.; Rathod, V.; Halkai, R.S. Biosynthesis, Characterization and Antibacterial Efficacy of Silver Nanoparticles Derived from Endophytic Fungi against P. Gingivalis. J. Clin. Diagn. Res. 2017, 11, ZC92–ZC96. [Google Scholar] [CrossRef]
- Halkai, K.R.; Mudda, J.A.; Shivanna, V.; Rathod, V.; Halkai, R.S. Evaluation of Antibacterial Efficacy of Biosynthesized Silver Nanoparticles Derived from Fungi against Endo-Perio Pathogens Porphyromonas Gingivalis, Bacillus Pumilus, and Enterococcus Faecalis. J. Conserv. Dent. 2017, 20, 398–404. [Google Scholar] [CrossRef]
- Science (IJDOS). Effectiveness of a Novel Nano-Silver Fluoride with Green Tea Extract Compared with Silver Diamine Fluoride: A Randomized, Controlled, Non-Inferiority Trial; Research Article; SciDoc Publishers: Lewes, DE, USA, 2020. [Google Scholar]
- Sundeep, D.; Vijaya Kumar, T.; Rao, P.S.S.; Ravikumar, R.V.S.S.N.; Gopala Krishna, A. Green Synthesis and Characterization of Ag Nanoparticles from Mangifera Indica Leaves for Dental Restoration and Antibacterial Applications. Prog. Biomater. 2017, 6, 57–66. [Google Scholar] [CrossRef]
- Mala, R.; Annie Aglin, A.; Ruby Celsia, A.S.; Geerthika, S.; Kiruthika, N.; VazagaPriya, C.; Srinivasa Kumar, K. Foley Catheters Functionalised with a Synergistic Combination of Antibiotics and Silver Nanoparticles Resist Biofilm Formation. IET Nanobiotechnol. 2017, 11, 612–620. [Google Scholar] [CrossRef]
- Wu, K.; Yang, Y.; Zhang, Y.; Deng, J.; Lin, C. Antimicrobial Activity and Cytocompatibility of Silver Nanoparticles Coated Catheters via a Biomimetic Surface Functionalization Strategy. Int. J. Nanomed. 2015, 10, 7241–7252. [Google Scholar] [CrossRef]
- Fufă, O.; Andronescu, E.; Grumezescu, V.; Holban, A.; Mogoanta, L.; Mogoșanu, G.; Socol, G.; Iordache, F.; Chifiriuc, M.; Grumezescu, A. Silver Nanostructurated Surfaces Prepared by MAPLE for Biofilm Prevention. Biointerface Res. Appl. Chem. 2015, 5, 1011–1017. [Google Scholar]
- Walker, J.N.; Flores-Mireles, A.L.; Pinkner, C.L.; Schreiber, H.L.; Joens, M.S.; Park, A.M.; Potretzke, A.M.; Bauman, T.M.; Pinkner, J.S.; Fitzpatrick, J.A.J.; et al. Catheterization Alters Bladder Ecology to Potentiate Staphylococcus Aureus Infection of the Urinary Tract. Proc. Natl. Acad. Sci. USA 2017, 114, E8721–E8730. [Google Scholar] [CrossRef] [Green Version]
- Wilkinson, L.J.; White, R.J.; Chipman, J.K. Silver and Nanoparticles of Silver in Wound Dressings: A Review of Efficacy and Safety. J. Wound Care 2011, 20, 543–549. [Google Scholar] [CrossRef]
- Thomas, R.; Nair, A.P.; Kr, S.; Mathew, J.; Ek, R. Antibacterial Activity and Synergistic Effect of Biosynthesized AgNPs with Antibiotics against Multidrug-Resistant Biofilm-Forming Coagulase-Negative Staphylococci Isolated from Clinical Samples. Appl. Biochem. Biotechnol. 2014, 173, 449–460. [Google Scholar] [CrossRef]
- Seth, D.; Choudhury, S.R.; Pradhan, S.; Gupta, S.; Palit, D.; Das, S.; Debnath, N.; Goswami, A. Nature-Inspired Novel Drug Design Paradigm Using Nanosilver: Efficacy on Multi-Drug-Resistant Clinical Isolates of Tuberculosis. Curr. Microbiol. 2010, 62, 715–726. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, K.; De Gusseme, B.; Verstraete, W.; Field, R. The Antibacterial and Anti-Biofouling Performance of Biogenic Silver Nanoparticles by Lactobacillus Fermentum. Biofouling 2014, 30, 347–357. [Google Scholar] [CrossRef]
- Russo, T.; Gloria, A.; De Santis, R.; D’Amora, U.; Balato, G.; Vollaro, A.; Oliviero, O.; Improta, G.; Triassi, M.; Ambrosio, L. Preliminary Focus on the Mechanical and Antibacterial Activity of a PMMA-Based Bone Cement Loaded with Gold Nanoparticles. Bioact. Mater. 2017, 2, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Zheng, K.; Setyawati, M.I.; Leong, D.T.; Xie, J. Antimicrobial Gold Nanoclusters. ACS Nano 2017, 11, 6904–6910. [Google Scholar] [CrossRef]
- Han, S.; Han, K.; Hong, J.; Yoon, D.-Y.; Park, C.; Kim, Y. Photothermal Cellulose-Patch with Gold-Spiked Silica Microrods Based on Escherichia Coli. ACS Omega 2018, 3, 5244–5251. [Google Scholar] [CrossRef]
- Calabrese, G.; Petralia, S.; Fabbi, C.; Forte, S.; Franco, D.; Guglielmino, S.; Esposito, E.; Cuzzocrea, S.; Traina, F.; Conoci, S. Au, Pd and Maghemite Nanofunctionalized Hydroxyapatite Scaffolds for Bone Regeneration. Regen. Biomater. 2020, 7, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Bucharskaya, A.; Maslyakova, G.; Terentyuk, G.; Yakunin, A.; Avetisyan, Y.; Bibikova, O.; Tuchina, E.; Khlebtsov, B.; Khlebtsov, N.; Tuchin, V. Towards Effective Photothermal/Photodynamic Treatment Using Plasmonic Gold Nanoparticles. Int. J. Mol. Sci. 2016, 17, 1295. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Li, Z.; Xue, N.; Cheng, Z.; Miao, X. A Gold Nanoparticle Based Fluorescent Probe for Simultaneous Recognition of Single-Stranded DNA and Double-Stranded DNA. Mikrochim. Acta 2018, 185, 93. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, C.; Xu, C.; Wang, X.; Liu, C.; Waterhouse, G.I.N.; Wang, Y.; Yin, H. Ultrasmall Au Nanoclusters for Biomedical and Biosensing Applications: A Mini-Review. Talanta 2019, 200, 432–442. [Google Scholar] [CrossRef]
- Kailasa, S.K.; Park, T.-J.; Rohit, J.V.; Koduru, J.R. Chapter 14—Antimicrobial Activity of Silver Nanoparticles. In Nanoparticles in Pharmacotherapy; Grumezescu, A.M., Ed.; William Andrew Publishing: Norwich, NY, USA, 2019; pp. 461–484. ISBN 978-0-12-816504-1. [Google Scholar]
- Salah, I.; Parkin, I.P.; Allan, E. Copper as an Antimicrobial Agent: Recent Advances. RSC Adv. 2021, 11, 18179–18186. [Google Scholar] [CrossRef]
- Müller, D.W.; Lößlein, S.; Terriac, E.; Brix, K.; Siems, K.; Moeller, R.; Kautenburger, R.; Mücklich, F. Increasing Antibacterial Efficiency of Cu Surfaces by Targeted Surface Functionalization via Ultrashort Pulsed Direct Laser Interference Patterning. Adv. Mater. Interfaces 2021, 8, 2001656. [Google Scholar] [CrossRef]
- Wang, P.; Yuan, Y.; Xu, K.; Zhong, H.; Yang, Y.; Jin, S.; Yang, K.; Qi, X. Biological Applications of Copper-Containing Materials. Bioact. Mater. 2020, 6, 916–927. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Gao, L.; Schlaich, C.; Zhang, J.; Donskyi, I.S.; Yu, G.; Li, W.; Tu, Z.; Rolff, J.; Schwerdtle, T.; et al. Construction of Functional Coatings with Durable and Broad-Spectrum Antibacterial Potential Based on Mussel-Inspired Dendritic Polyglycerol and in Situ-Formed Copper Nanoparticles. ACS Appl. Mater. Interfaces 2017, 9, 35411–35418. [Google Scholar] [CrossRef]
- Harikumar, P.S.; Aravind, A. Antibacterial Activity of Copper Nanoparticles and Copper Nanocomposites against Escherichia Coli Bacteria. Int. J. Sci. 2016, 5, 83–90. [Google Scholar] [CrossRef]
- Dadi, R.; Azouani, R.; Traore, M.; Mielcarek, C.; Kanaev, A. Antibacterial Activity of ZnO and CuO Nanoparticles against Gram Positive and Gram Negative Strains. Mater. Sci. Eng. C 2019, 104, 109968. [Google Scholar] [CrossRef]
- Khan, Z.; Al-Thabaiti, S.A. Green Synthesis of Zero-Valent Fe-Nanoparticles: Catalytic Degradation of Rhodamine B, Interactions with Bovine Serum Albumin and Their Enhanced Antimicrobial Activities. J. Photochem. Photobiol. B Biol. 2018, 180, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H.; Kang, Y.-G.; Chang, Y.-S.; Kim, J.-H. Effects of Zerovalent Iron Nanoparticles on Photosynthesis and Biochemical Adaptation of Soil-Grown Arabidopsis Thaliana. Nanomaterials 2019, 9, 1543. [Google Scholar] [CrossRef]
- Nehra, P.; Chauhan, R.P.; Garg, N.; Verma, K. Antibacterial and Antifungal Activity of Chitosan Coated Iron Oxide Nanoparticles. Br. J. Biomed. Sci. 2018, 75, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Jiang, C.Z.; Roy, V.A.L. Designed Synthesis and Surface Engineering Strategies of Magnetic Iron Oxide Nanoparticles for Biomedical Applications. Nanoscale 2016, 8, 19421–19474. [Google Scholar] [CrossRef] [PubMed]
- Hasanova, U.; Ramazanov, M.; Maharramov, A.; Eyvazova, Q.; Aghamaliyev, Z.; Parfyonova, Y.; Hajiyeva, S.; Hajiyeva, F.; Veliyeva, S. Nano-Coupling of Cephalosporin Antibiotics with Fe3O4 Nanoparticles: Trojan Horse Approach in Antimicrobial Chemotherapy of Infections Caused by Klebsiella spp. J. Biomater. Nanobiotechnol. 2015, 06, 225–235. [Google Scholar] [CrossRef]
- Ali, K.; Ahmed, B.; Khan, M.S.; Musarrat, J. Differential Surface Contact Killing of Pristine and Low EPS Pseudomonas Aeruginosa with Aloe Vera Capped Hematite (α-Fe2O3) Nanoparticles. J. Photochem. Photobiol. B Biol. 2018, 188, 146–158. [Google Scholar] [CrossRef]
- Bukhari, S.; Kim, D.; Liu, Y.; Karabucak, B.; Koo, H. Novel Endodontic Disinfection Approach Using Catalytic Nanoparticles. J. Endod. 2018, 44, 806–812. [Google Scholar] [CrossRef]
- Parveen, S.; Wani, A.H.; Shah, M.A.; Devi, H.S.; Bhat, M.Y.; Koka, J.A. Preparation, Characterization and Antifungal Activity of Iron Oxide Nanoparticles. Microb. Pathog. 2018, 115, 287–292. [Google Scholar] [CrossRef]
- Rafi, M.M.; Ahmed, K.S.Z.; Premnazeer, K.; Kumar, D.S. Antibacterial Activity of Iron Oxide Nanoparticles on Polysaccharide Templates: Synthesis, Characterization and Magnetic Studies. Malays. Polym. J. 2015, 10, 16–22. [Google Scholar]
- Ranmadugala, D.; Ebrahiminezhad, A.; Manley-Harris, M.; Ghasemi, Y.; Berenjian, A. Impact of 3–Aminopropyltriethoxysilane-Coated Iron Oxide Nanoparticles on Menaquinone-7 Production Using B. Subtilis. Nanomaterials 2017, 7, 350. [Google Scholar] [CrossRef]
- El Nahrawy, A.M.; Abou Hammad, A.B.; Mansour, A.M. Preparation and Characterization of Transparent Semiconducting Silica Nanocomposites Doped with P2O5 and Al2O3. Silicon 2021, 13, 3733–3739. [Google Scholar] [CrossRef]
- Liakos, I.L.; Abdellatif, M.H.; Innocenti, C.; Scarpellini, A.; Carzino, R.; Brunetti, V.; Marras, S.; Brescia, R.; Drago, F.; Pompa, P.P. Antimicrobial Lemongrass Essential Oil—Copper Ferrite Cellulose Acetate Nanocapsules. Molecules 2016, 21, 520. [Google Scholar] [CrossRef] [PubMed]
- Samavati, A.; Ismail, A.F. Antibacterial Properties of Copper-Substituted Cobalt Ferrite Nanoparticles Synthesized by Co-Precipitation Method. Particuology 2017, 30, 158–163. [Google Scholar] [CrossRef]
- Biswas, K.; De, D.; Bandyopadhyay, J.; Dutta, N.; Rana, S.; Sen, P.; Bandyopadhyay, S.K.; Chakraborty, P.K. Enhanced Polarization, Magnetic Response and Pronounced Antibacterial Activity of Bismuth Ferrite Nanorods. Mater. Chem. Phys. 2017, 195, 207–212. [Google Scholar] [CrossRef]
- Žalnėravičius, R.; Paškevičius, A.; Mažeika, K.; Jagminas, A. Fe(II)-Substituted Cobalt Ferrite Nanoparticles against Multidrug Resistant Microorganisms. Appl. Surf. Sci. 2018, 435, 141–148. [Google Scholar] [CrossRef]
- Wang, C.; Gu, B.; Liu, Q.; Pang, Y.; Xiao, R.; Wang, S. Combined Use of Vancomycin-Modified Ag-Coated Magnetic Nanoparticles and Secondary Enhanced Nanoparticles for Rapid Surface-Enhanced Raman Scattering Detection of Bacteria. Int. J. Nanomed. 2018, 13, 1159–1178. [Google Scholar] [CrossRef] [PubMed]
- Esteban Florez, F.L.; Hiers, R.D.; Larson, P.; Johnson, M.; O’Rear, E.; Rondinone, A.J.; Khajotia, S.S. Antibacterial Dental Adhesive Resins Containing Nitrogen-Doped Titanium Dioxide Nanoparticles. Mater. Sci. Eng. C 2018, 93, 931–943. [Google Scholar] [CrossRef]
- Chen, R.; Han, Z.; Huang, Z.; Karki, J.; Wang, C.; Zhu, B.; Zhang, X. Antibacterial Activity, Cytotoxicity and Mechanical Behavior of Nano-Enhanced Denture Base Resin with Different Kinds of Inorganic Antibacterial Agents. Dent. Mater. J. 2017, 36, 693–699. [Google Scholar] [CrossRef]
- Ono, Y.; Iwahashi, H. Titanium Dioxide Nanoparticles Impart Protection from Ultraviolet Irradiation to Fermenting Yeast Cells. Biochem. Biophys. Rep. 2022, 30, 101221. [Google Scholar] [CrossRef]
- Rodríguez-González, V.; Obregón, S.; Patrón-Soberano, O.A.; Terashima, C.; Fujishima, A. An Approach to the Photocatalytic Mechanism in the TiO2-Nanomaterials Microorganism Interface for the Control of Infectious Processes. Appl. Catal. B Environ. 2020, 270, 118853. [Google Scholar] [CrossRef]
- Viet, P.V.; Phan, B.T.; Mott, D.; Maenosono, S.; Sang, T.T.; Thi, C.M.; Hieu, L.V. Silver Nanoparticle Loaded TiO2 Nanotubes with High Photocatalytic and Antibacterial Activity Synthesized by Photoreduction Method. J. Photochem. Photobiol. A Chem. 2018, 352, 106–112. [Google Scholar] [CrossRef]
- Anaya-Esparza, L.M.; Villagrán-de la Mora, Z.; Ruvalcaba-Gómez, J.M.; Romero-Toledo, R.; Sandoval-Contreras, T.; Aguilera-Aguirre, S.; Montalvo-González, E.; Pérez-Larios, A. Use of Titanium Dioxide (TiO2) Nanoparticles as Reinforcement Agent of Polysaccharide-Based Materials. Processes 2020, 8, 1395. [Google Scholar] [CrossRef]
- Jin, S.-E.; Jin, J.E.; Hwang, W.; Hong, S.W. Photocatalytic Antibacterial Application of Zinc Oxide Nanoparticles and Self-Assembled Networks under Dual UV Irradiation for Enhanced Disinfection. Int. J. Nanomed. 2019, 14, 1737–1751. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.-J.; Huang, S.-C.; Chen, Y.-P.; Chiueh, L.-C.; Shih, D.Y.-C. Analysis of Titanium Dioxide and Zinc Oxide Nanoparticles in Cosmetics. J. Food Drug Anal. 2015, 23, 587–594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baek, S.; Joo, S.H.; Kumar, N.; Toborek, M. Antibacterial Effect and Toxicity Pathways of Industrial and Sunscreen ZnO Nanoparticles on Escherichia Coli. J. Environ. Chem. Eng. 2017, 5, 3024–3032. [Google Scholar] [CrossRef]
- Lu, P.J.; Fang, S.W.; Cheng, W.L.; Huang, S.C.; Huang, M.C.; Cheng, H.F. Characterization of Titanium Dioxide and Zinc Oxide Nanoparticles in Sunscreen Powder by Comparing Different Measurement Methods. J. Food Drug Anal. 2018, 26, 1192–1200. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Mudai, A.; Roy, B.; Basumatary, I.B.; Mukherjee, A.; Dutta, J. Biodegradable Hybrid Nanocomposite of Chitosan/Gelatin and Green Synthesized Zinc Oxide Nanoparticles for Food Packaging. Foods 2020, 9, 1143. [Google Scholar] [CrossRef]
- Valencia, G.A.; Zare, E.N.; Makvandi, P.; Gutiérrez, T.J. Self-Assembled Carbohydrate Polymers for Food Applications: A Review. Compr. Rev. Food Sci. Food Saf. 2019, 18, 2009–2024. [Google Scholar] [CrossRef]
- Zare, E.N.; Makvandi, P.; Borzacchiello, A.; Tay, F.R.; Ashtari, B.; Padil, V.V.T. Antimicrobial Gum Bio-Based Nanocomposites and Their Industrial and Biomedical Applications. Chem. Commun. 2019, 55, 14871–14885. [Google Scholar] [CrossRef]
- Tüzüner, T.; Güçlü, Z.A.; Hurt, A.; Coleman, N.; Nicholson, J. Release of Antimicrobial Compounds from a Zinc Oxide-Chelate Cement. J. Oral Sci. 2018, 60, 24–28. [Google Scholar] [CrossRef]
- Andrade, V.; Martínez, A.; Rojas, N.; Bello-Toledo, H.; Flores, P.; Sánchez-Sanhueza, G.; Catalán, A. Antibacterial Activity against Streptococcus Mutans and Diametrical Tensile Strength of an Interim Cement Modified with Zinc Oxide Nanoparticles and Terpenes: An in Vitro Study. J. Prosthet. Dent. 2018, 119, 862.e1–862.e7. [Google Scholar] [CrossRef] [PubMed]
- Garcia, I.M.; Balhaddad, A.A.; Ibrahim, M.S.; Weir, M.D.; Xu, H.H.K.; Collares, F.M.; Melo, M.A.S. Antibacterial Response of Oral Microcosm Biofilm to Nano-Zinc Oxide in Adhesive Resin. Dent. Mater. 2021, 37, e182–e193. [Google Scholar] [CrossRef] [PubMed]
- Mishra, P.K.; Mishra, H.; Ekielski, A.; Talegaonkar, S.; Vaidya, B. Zinc Oxide Nanoparticles: A Promising Nanomaterial for Biomedical Applications. Drug Discov. Today 2017, 22, 1825–1834. [Google Scholar] [CrossRef]
- Barma, M.D.; Muthupandiyan, I.; Samuel, S.R.; Amaechi, B.T. Inhibition of Streptococcus Mutans, Antioxidant Property and Cytotoxicity of Novel Nano-Zinc Oxide Varnish. Arch. Oral Biol. 2021, 126, 105132. [Google Scholar] [CrossRef]
- Kachoei, M.; Divband, B.; Rahbar, M.; Esmaeilzadeh, M.; Ghanizadeh, M.; Alam, M. A Novel Developed Bioactive Composite Resin Containing Silver/Zinc Oxide (Ag/ZnO) Nanoparticles as an Antimicrobial Material against Streptococcus Mutans, Lactobacillus, and Candida Albicans. Evid. Based Complement. Alternat. Med. 2021, 2021, 4743411. [Google Scholar] [CrossRef]
- Shimabukuro, M.; Tsutsumi, Y.; Nozaki, K.; Chen, P.; Yamada, R.; Ashida, M.; Doi, H.; Nagai, A.; Hanawa, T. Chemical and Biological Roles of Zinc in a Porous Titanium Dioxide Layer Formed by Micro-Arc Oxidation. Coatings 2019, 9, 705. [Google Scholar] [CrossRef]
- Chouirfa, H.; Bouloussa, H.; Migonney, V.; Falentin-Daudré, C. Review of Titanium Surface Modification Techniques and Coatings for Antibacterial Applications. Acta Biomater. 2019, 83, 37–54. [Google Scholar] [CrossRef]
- Maleki-Ghaleh, H.; Siadati, M.H.; Fallah, A.; Koc, B.; Kavanlouei, M.; Khademi-Azandehi, P.; Moradpur-Tari, E.; Omidi, Y.; Barar, J.; Beygi-Khosrowshahi, Y.; et al. Antibacterial and Cellular Behaviors of Novel Zinc-Doped Hydroxyapatite/Graphene Nanocomposite for Bone Tissue Engineering. Int. J. Mol. Sci. 2021, 22, 9564. [Google Scholar] [CrossRef]
- He, J.; Feng, W.; Zhao, B.-H.; Zhang, W.; Lin, Z. In Vivo Effect of Titanium Implants with Porous Zinc-Containing Coatings Prepared by Plasma Electrolytic Oxidation Method on Osseointegration in Rabbits. Int. J. Oral Maxillofac. Implants 2018, 33, 298–310. [Google Scholar] [CrossRef]
- Hemeg, H.A. Nanomaterials for Alternative Antibacterial Therapy. Int. J. Nanomed. 2017, 12, 8211–8225. [Google Scholar] [CrossRef]
- Spirescu, V.A.; Chircov, C.; Grumezescu, A.M.; Vasile, B.Ș.; Andronescu, E. Inorganic Nanoparticles and Composite Films for Antimicrobial Therapies. Int. J. Mol. Sci. 2021, 22, 4595. [Google Scholar] [CrossRef] [PubMed]
- Maliszewska, I.; Sadowski, Z. Synthesis and Antibacterial Activity of of Silver Nanoparticles. J. Phys. Conf. Ser. 2009, 146, 012024. [Google Scholar] [CrossRef]
- AlMasoud, N.; Alhaik, H.; Almutairi, M.; Houjak, A.; Hazazi, K.; Alhayek, F.; Aljanoubi, S.; Alkhaibari, A.; Alghamdi, A.; Soliman, D.A.; et al. Green Nanotechnology Synthesized Silver Nanoparticles: Characterization and Testing Its Antibacterial Activity. Green Process. Synth. 2021, 10, 518–528. [Google Scholar] [CrossRef]
- Huh, A.J.; Kwon, Y.J. “Nanoantibiotics”: A New Paradigm for Treating Infectious Diseases Using Nanomaterials in the Antibiotics Resistant Era. J. Control. Release 2011, 156, 128–145. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-F.; Liu, Z.-G.; Shen, W.; Gurunathan, S. Silver Nanoparticles: Synthesis, Characterization, Properties, Applications, and Therapeutic Approaches. Int. J. Mol. Sci. 2016, 17, 1534. [Google Scholar] [CrossRef]
- Mitra, D.; Kang, E.-T.; Neoh, K.G. Antimicrobial Copper-Based Materials and Coatings: Potential Multifaceted Biomedical Applications. ACS Appl. Mater. Interfaces 2020, 12, 21159–21182. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Kareem, M.M.; Zohri, A.A. Extracellular Mycosynthesis of Gold Nanoparticles Using Trichoderma Hamatum: Optimization, Characterization and Antimicrobial Activity. Lett. Appl. Microbiol. 2018, 67, 465–475. [Google Scholar] [CrossRef]
- Vijayakumar, S.; Krishnakumar, C.; Arulmozhi, P.; Mahadevan, S.; Parameswari, N. Biosynthesis, Characterization and Antimicrobial Activities of Zinc Oxide Nanoparticles from Leaf Extract of Glycosmis Pentaphylla (Retz.) DC. Microb. Pathog. 2018, 116, 44–48. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Franco, D.; Calabrese, G.; Guglielmino, S.P.P.; Conoci, S. Metal-Based Nanoparticles: Antibacterial Mechanisms and Biomedical Application. Microorganisms 2022, 10, 1778. https://doi.org/10.3390/microorganisms10091778
Franco D, Calabrese G, Guglielmino SPP, Conoci S. Metal-Based Nanoparticles: Antibacterial Mechanisms and Biomedical Application. Microorganisms. 2022; 10(9):1778. https://doi.org/10.3390/microorganisms10091778
Chicago/Turabian StyleFranco, Domenico, Giovanna Calabrese, Salvatore Pietro Paolo Guglielmino, and Sabrina Conoci. 2022. "Metal-Based Nanoparticles: Antibacterial Mechanisms and Biomedical Application" Microorganisms 10, no. 9: 1778. https://doi.org/10.3390/microorganisms10091778
APA StyleFranco, D., Calabrese, G., Guglielmino, S. P. P., & Conoci, S. (2022). Metal-Based Nanoparticles: Antibacterial Mechanisms and Biomedical Application. Microorganisms, 10(9), 1778. https://doi.org/10.3390/microorganisms10091778








