The Viable but Non-Culturable (VBNC) State, a Poorly Explored Aspect of Beneficial Bacteria
Abstract
:1. Introduction
Conditions That Induce VBNC State
2. Bacterial Species Entering the VBNC State
3. The VBNC State in Beneficial Bacteria
3.1. Alphaproteobacteria and the VBNC State
3.2. Betaproteobacteria and the VBNC State
3.3. Gammaproteobacteria and the VBNC State
3.4. Actinobacteria and the VBNC State
3.5. Firmicutes and the VBNC State
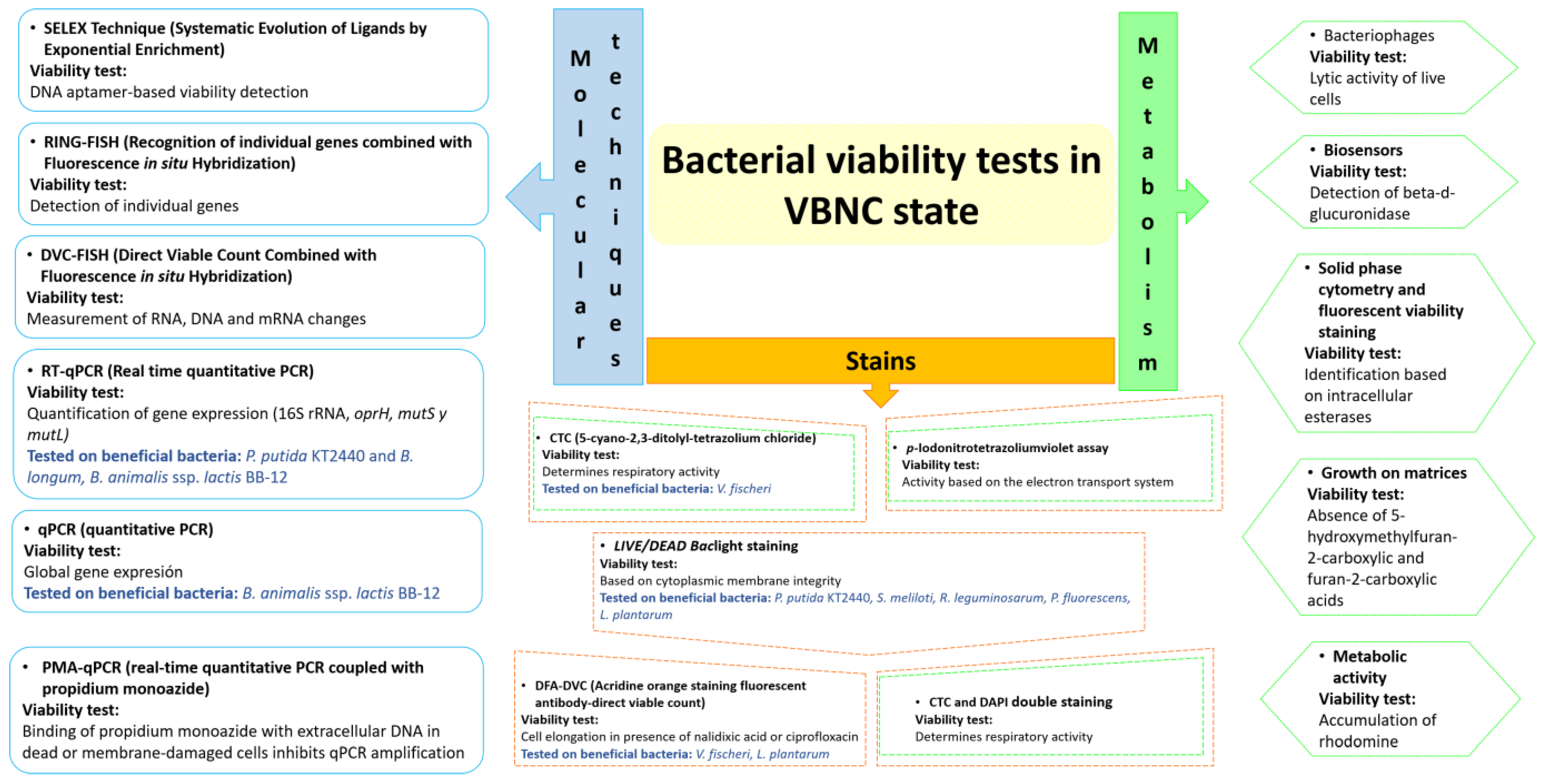
4. Techniques to Evaluate Bacterial Viability in the VBNC State
5. What Happens during the VBNC State?
6. Proteomics, Transcriptomics, and Metabolomics of Bacterial Cells in VBNC State
7. Resuscitation of Bacteria under VBNC State
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Oliver, J.D. The Viable but nonculturable state and cellular resuscitation. In Proceedings of the 8th International Symposium on Microbial Ecology, Halifax, NS, Canada, 9–14 August 1998. [Google Scholar]
- Oliver, J.D. The Viable but nonculturable state in bacteria. J. Microbiol. 2005, 43, 93–100. [Google Scholar] [PubMed]
- Trevors, J.T. Viable but Non-Culturable (VBNC) bacteria: Gene expression in planktonic and biofilm cells. J. Microbiol. Methods 2011, 86, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Pazos-Rojas, L.A.; Muñoz-Arenas, L.C.; Rodríguez-Andrade, O.; López-Cruz, L.E.; López-Ortega, O.; Lopes-Olivares, F.; Luna-Suarez, S.; Baez, A.; Morales-García, Y.E.; Quintero-Hernández, V.; et al. Desiccation-Induced viable but nonculturable state in Pseudomonas putida KT2440, a Survival Strategy. PLoS ONE 2019, 14, e0219554. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.-S.; Roberts, N.; Singleton, F.L.; Attwell, R.W.; Grimes, D.J.; Colwell, R.R. Survival and viability of nonculturable Escherichia coli and Vibrio cholerae in the estuarine and marine environment. Microb. Ecol. 1982, 8, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Gunasekera, T.S.; Sørensen, A.; Attfield, P.V.; Sørensen, S.J.; Veal, D.A. Inducible gene expression by nonculturable bacteria in milk after pasteurization. Appl. Environ. Microbiol. 2002, 68, 1988–1993. [Google Scholar] [CrossRef]
- Cunningham, E.; O’Byrne, C.; Oliver, J.D. Effect of weak acids on Listeria monocytogenes survival: Evidence for a viable but nonculturable state in response to low pH. Food Control 2009, 20, 1141–1144. [Google Scholar] [CrossRef]
- Magariños, B.; Romalde, J.L.; Barja, J.L.; Toranzo, A.E. Evidence of a dormant but infective state of the fish pathogen Pasteurella piscicida in seawater and sediment. Appl. Environ. Microbiol. 1994, 60, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Biosca, E.G.; Amaro, C.; Marco-Noales, E.; Oliver, J.D. Effect of low temperature on starvation-survival of the eel pathogen Vibrio vulnificus biotype 2. Appl. Environ. Microbiol. 1996, 62, 450–455. [Google Scholar] [CrossRef]
- Banin, E.; Israely, T.; Kushmaro, A.; Loya, Y.; Orr, E.; Rosenberg, E. Penetration of the coral-bleaching bacterium Vibrio shiloi into Oculina patagonica. Appl. Environ. Microbiol. 2000, 66, 3031–3036. [Google Scholar] [CrossRef]
- Israely, T.; Banin, E.; Rosenberg, E. Growth, Differentiation and death of Vibrio shiloi in coral tissue as a function of seawater temperature. Aquat. Microb. Ecol. 2001, 24, 1–8. [Google Scholar] [CrossRef]
- Grey, B.E.; Steck, T.R. The Viable But Nonculturable State of Ralstonia solanacearum may be involved in long-term survival and plant infection. Appl. Environ. Microbiol. 2001, 67, 3866–3872. [Google Scholar] [CrossRef] [PubMed]
- Del Campo, R.; Russi, P.; Mara, P.; Mara, H.; Peyrou, M.; De LeÃ3n, I.P.; Gaggero, C. Xanthomonas axonopodis pv. citri enters the vbnc state after copper treatment and retains its virulence. FEMS Microbiol. Lett. 2009, 298, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Ordax, M.; Biosca, E.G.; Wimalajeewa, S.C.; López, M.M.; Marco-Noales, E. Survival of Erwinia amylovora in mature apple fruit calyces through the Viable but Nonculturable (VBNC) state. J. Appl. Microbiol. 2009, 107, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Kan, Y.; Jiang, N.; Xu, X.; Lyu, Q.; Gopalakrishnan, V.; Walcott, R.; Burdman, S.; Li, J.; Luo, L. Induction and resuscitation of the Viable but Non-Culturable (VBNC) state in Acidovorax citrulli, the causal agent of bacterial fruit blotch of cucurbitaceous crops. Front. Microbiol. 2019, 10, 1081. [Google Scholar] [CrossRef] [PubMed]
- Vriezen, J.A.; De Bruijn, F.J.; Nüsslein, K.R. Desiccation Induces viable but non-culturable cells in Sinorhizobium meliloti 1021. AMB Expr. 2012, 2, 6. [Google Scholar] [CrossRef] [PubMed]
- Fakruddin, M.; Mannan, K.S.B.; Andrews, S. Viable but nonculturable bacteria: Food safety and public health perspective. ISRN Microbiol. 2013, 2013, 703813. [Google Scholar] [CrossRef]
- Pinto, D.; Santos, M.A.; Chambel, L. Thirty years of viable but nonculturable state research: Unsolved molecular mechanisms. Crit. Rev. Microbiol. 2015, 41, 61–76. [Google Scholar] [CrossRef]
- Liu, J.; Deng, Y.; Peters, B.M.; Li, L.; Li, B.; Chen, L.; Xu, Z.; Shirtliff, M.E. Transcriptomic analysis on the formation of the viable putative non-culturable state of beer-spoilage Lactobacillus acetotolerans. Sci. Rep. 2016, 6, 36753. [Google Scholar] [CrossRef]
- Liu, J.; Li, L.; Li, B.; Peters, B.M.; Deng, Y.; Xu, Z.; Shirtliff, M.E. Study on spoilage capability and VBNC state formation and recovery of Lactobacillus plantarum. Microb. Pathog. 2017, 110, 257–261. [Google Scholar] [CrossRef]
- Liu, J.; Deng, Y.; Li, L.; Li, B.; Li, Y.; Zhou, S.; Shirtliff, M.E.; Xu, Z.; Peters, B.M. Discovery and control of culturable and viable but non-culturable cells of a distinctive Lactobacillus harbinensis strain from spoiled beer. Sci. Rep. 2018, 8, 11446. [Google Scholar] [CrossRef]
- Li, Y.; Yang, L.; Fu, J.; Yan, M.; Chen, D.; Zhang, L. The novel loop-mediated isothermal amplification based confirmation methodology on the bacteria in Viable but Non-Culturable (VBNC) state. Microb. Pathog. 2017, 111, 280–284. [Google Scholar] [CrossRef] [PubMed]
- Daranas, N.; Bonaterra, A.; Francés, J.; Cabrefiga, J.; Montesinos, E.; Badosa, E. Monitoring Viable cells of the biological control agent Lactobacillus plantarum PM411 in aerial plant surfaces by means of a strain-specific viability quantitative PCR method. Appl. Environ. Microbiol. 2018, 84, e00107-18. [Google Scholar] [CrossRef] [PubMed]
- Santander, R.D.; Figàs-Segura, À.; Biosca, E.G. Erwinia amylovora catalases KatA and KatG are virulence factors and delay the starvation-induced Viable but Non-culturable (VBNC) response. Mol. Plant Pathol. 2018, 19, 922–934. [Google Scholar] [CrossRef] [PubMed]
- Dong, K.; Pan, H.; Yang, D.; Rao, L.; Zhao, L.; Wang, Y.; Liao, X. Induction, detection, formation, and resuscitation of viable but non-culturable state microorganisms. Comp. Rev. Food Sci. Food Safe 2020, 19, 149–183. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-H.; Ahmad, W.; Zhu, X.-Y.; Chen, J.; Austin, B. Viable but nonculturable bacteria and their resuscitation: Implications for cultivating uncultured marine microorganisms. Mar. Life Sci. Technol. 2021, 3, 189–203. [Google Scholar] [CrossRef] [PubMed]
- Oliver, J.D. Recent Findings on the viable but nonculturable state in pathogenic bacteria. FEMS Microbiol. Rev. 2010, 34, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Dobereiner, J.; Urquiaga, S. Alternatives for nitrogen nutrition of crops in tropical agriculture. Fertil. Res. 1994, 42, 339–346. [Google Scholar] [CrossRef]
- Crowley, D.E.; Wang, Y.C.; Reid, C.P.P.; Szaniszlo, P.J. Mechanisms of iron acquisition from siderophores by microorganisms and plants. In Developments in Plant and Soil Sciences; Springer: Dordrecht, The Netherlands, 1991; pp. 179–198. ISBN 978-94-010-5455-3. [Google Scholar]
- Hoefman, S.; Van Hoorde, K.; Boon, N.; Vandamme, P.; De Vos, P.; Heylen, K. Survival or revival: Long-term preservation induces a reversible viable but non-culturable state in methane-oxidizing bacteria. PLoS ONE 2012, 7, e34196. [Google Scholar] [CrossRef]
- Babalola, O.O. Beneficial bacteria of agricultural importance. Biotechnol. Lett. 2010, 32, 1559–1570. [Google Scholar] [CrossRef]
- Lugtenberg, B.; Kamilova, F. Plant-Growth-Promoting Rhizobacteria. Annu. Rev. Microbiol. 2009, 63, 541–556. [Google Scholar] [CrossRef]
- Hayat, R.; Ali, S.; Amara, U.; Khalid, R.; Ahmed, I. Soil beneficial bacteria and their role in plant growth promotion: A review. Ann. Microbiol. 2010, 60, 579–598. [Google Scholar] [CrossRef]
- Pieterse, C.M.J.; Zamioudis, C.; Berendsen, R.L.; Weller, D.M.; Van Wees, S.C.M.; Bakker, P.A.H.M. Induced Systemic Resistance by beneficial microbes. Annu. Rev. Phytopathol. 2014, 52, 347–375. [Google Scholar] [CrossRef] [PubMed]
- Fira, D.; Dimkić, I.; Berić, T.; Lozo, J.; Stanković, S. Biological control of plant pathogens by Bacillus species. J. Biotechnol. 2018, 285, 44–55. [Google Scholar] [CrossRef] [PubMed]
- Brierley, C.L. Microbiological mining. Sci. Am. 1982, 247, 44–53. [Google Scholar] [CrossRef]
- Zaidi, S.; Usmani, S.; Singh, B.R.; Musarrat, J. Significance of Bacillus subtilis Strain SJ-101 as a bioinoculant for concurrent plant growth promotion and nickel accumulation in Brassica juncea. Chemosphere 2006, 64, 991–997. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, X.; Chen, J.; Shim, H.; Bai, Z. New Advances in plant growth-promoting rhizobacteria for bioremediation. Environ. Int. 2007, 33, 406–413. [Google Scholar] [CrossRef]
- Leroy, F.; De Vuyst, L. Lactic acid bacteria as functional starter cultures for the food fermentation industry. Trends Food Sci. Technol. 2004, 15, 67–78. [Google Scholar] [CrossRef]
- Chemier, J.A.; Fowler, Z.L.; Koffas, M.A.G. Trends in microbial synthesis of natural products and biofuels. In Advances in Enzymology—And Related Areas of Molecular Biology; Toone, E.J., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2008; Volume 76, pp. 151–217. ISBN 978-0-470-39288-1. [Google Scholar]
- Tejero-Sariñena, S.; Barlow, J.; Costabile, A.; Gibson, G.R.; Rowland, I. In vitro evaluation of the antimicrobial activity of a range of probiotics against pathogens: Evidence for the effects of organic acids. Anaerobe 2012, 18, 530–538. [Google Scholar] [CrossRef]
- Liévin-Le Moal, V.; Servin, A.L. The front line of enteric host defense against unwelcome intrusion of harmful microorganisms: Mucins, antimicrobial peptides, and microbiota. Clin. Microbiol. Rev. 2006, 19, 315–337. [Google Scholar] [CrossRef]
- Pagnini, C.; Saeed, R.; Bamias, G.; Arseneau, K.O.; Pizarro, T.T.; Cominelli, F. Probiotics promote gut health through stimulation of epithelial innate immunity. Proc. Natl. Acad. Sci. USA 2010, 107, 454–459. [Google Scholar] [CrossRef]
- Lonvaud-Funel, A. Lactic acid bacteria in the quality improvement and depreciation of wine. In Lactic Acid Bacteria: Genetics, Metabolism and Applications; Konings, W.N., Kuipers, O.P., Huis Veld, J.H.J., Eds.; Springer: Dordrecht, The Netherlands, 1999; pp. 317–331. ISBN 978-90-481-5312-1. [Google Scholar]
- Virdis, C.; Sumby, K.; Bartowsky, E.; Jiranek, V. Lactic acid bacteria in wine: Technological advances and evaluation of their functional role. Front. Microbiol. 2021, 11, 612118. [Google Scholar] [CrossRef]
- Millet, V.; Lonvaud-Funel, A. The viable but non-culturable state of wine micro-organisms during storage. Lett. Appl. Microbiol. 2000, 30, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Xia, K.; Han, C.; Xu, J.; Liang, X. Toxin-antitoxin HicAB regulates the formation of persister cells responsible for the acid stress resistance in Acetobacter pasteurianus. Appl. Microbiol. Biotechnol. 2021, 105, 725–739. [Google Scholar] [CrossRef] [PubMed]
- Alexander, E.; Pham, D.; Steck, T.R. The Viable-but-nonculturable condition is induced by copper in Agrobacterium tumefaciens and Rhizobium leguminosarum. Appl. Environ. Microbiol. 1999, 65, 3754–3756. [Google Scholar] [CrossRef] [PubMed]
- Giagnoni, L.; Arenella, M.; Galardi, E.; Nannipieri, P.; Renella, G. Bacterial culturability and the Viable but Non-Culturable (VBNC) state studied by a proteomic approach using an artificial soil. Soil Biol. Biochem. 2018, 118, 51–58. [Google Scholar] [CrossRef]
- Furusawa, G.; Diyana, T.; Lau, N.-S. Metabolic strategies of dormancy of a marine bacterium Microbulbifer aggregans CCB-MM1: Its alternative electron transfer chain and sulfate-reducing pathway. Genomics 2022, 114, 443–455. [Google Scholar] [CrossRef] [PubMed]
- Arana, I.; Muela, A.; Orruño, M.; Seco, C.; Garaizabal, I.; Barcina, I. Effect of temperature and starvation upon survival strategies of Pseudomonas fluorescens CHA0: Comparison with Escherichia coli. FEMS Microbiol. Ecol. 2010, 74, 500–509. [Google Scholar] [CrossRef] [PubMed]
- Ramaiah, N.; Ravel, J.; Straube, W.L.; Hill, R.T.; Colwell, R.R. Entry of Vibrio harveyi and Vibrio fischeri into the viable but nonculturable state. J. Appl. Microbiol. 2002, 93, 108–116. [Google Scholar] [CrossRef]
- Verruck, S.; Silva, K.J.; De Oliveira Santeli, H.; Scariot, M.C.; Venturelli, G.L.; Prudencio, E.S.; Arisi, A.C.M. Bifidobacterium animalis ssp. lactis BB-12 enumeration by quantitative PCR assay in microcapsules with full-fat goat milk and inulin-type fructans. Food Res. Int. 2020, 133, 109131. [Google Scholar] [CrossRef]
- Lahtinen, S.J.; Ahokoski, H.; Reinikainen, J.P.; Gueimonde, M.; Nurmi, J.; Ouwehand, A.C.; Salminen, S.J. Degradation of 16S rRNA and Attributes of viability of viable but nonculturable probiotic bacteria. Lett. Appl. Microbiol. 2008, 46, 693–698. [Google Scholar] [CrossRef]
- Su, X.; Shen, X.; Ding, L.; Yokota, A. Study on the flocculability of the Arthrobacter sp., an actinomycete resuscitated from the VBNC state. World J. Microbiol. Biotechnol. 2012, 28, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Chen, X.; Zhang, Y.; Wang, W.; Xu, J. Description of Sinomonas soli sp. nov., reclassification of Arthrobacter echigonensis and Arthrobacter albidus (Ding et al. 2009) as Sinomonas echigonensis comb. nov. and Sinomonas albida comb. nov., respectively, and emended description of the genus Sinomonas. Int. J. Syst. Evol. Microbiol. 2012, 62, 764–769. [Google Scholar] [CrossRef] [PubMed]
- Majeed, M.; Majeed, S.; Nagabhushanam, K.; Punnapuzha, A.; Philip, S.; Mundkur, L. Rapid Assessment of Viable but non-culturable Bacillus coagulans MTCC 5856 in commercial formulations using flow cytometry. PLoS ONE 2018, 13, e0192836. [Google Scholar] [CrossRef] [PubMed]
- Saitou, N.; Nei, M. The Neighbor-Joining Method: A New method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar] [CrossRef] [PubMed]
- Felsenstein, J. Phylogenies and the comparative method. Am. Soc. Nat. 1985, 125, 1–15. [Google Scholar] [CrossRef]
- Andrés-Barrao, C.; Saad, M.M.; Chappuis, M.-L.; Boffa, M.; Perret, X.; Ortega Pérez, R.; Barja, F. Proteome analysis of Acetobacter pasteurianus during Acetic acid fermentation. J. Proteom. 2012, 75, 1701–1717. [Google Scholar] [CrossRef]
- Semrau, J.D.; DiSpirito, A.A.; Yoon, S. Methanotrophs and copper. FEMS Microbiol. Rev. 2010, 34, 496–531. [Google Scholar] [CrossRef]
- Jiang, H.; Chen, Y.; Jiang, P.; Zhang, C.; Smith, T.J.; Murrell, J.C.; Xing, X.-H. Methanotrophs: Multifunctional bacteria with promising applications in environmental bioengineering. Biochem. Eng. J. 2010, 49, 277–288. [Google Scholar] [CrossRef]
- Wendlandt, K.-D.; Stottmeister, U.; Helm, J.; Soltmann, B.; Jechorek, M.; Beck, M. The Potential of methane-oxidizing bacteria for applications in environmental biotechnology. Eng. Life Sci. 2010, 10, 87–102. [Google Scholar] [CrossRef]
- Coba De La Peña, T.; Fedorova, E.; Pueyo, J.J.; Lucas, M.M. The symbiosome: Legume and rhizobia co-evolution toward a nitrogen-fixing organelle? Front. Plant Sci. 2018, 8, 2229. [Google Scholar] [CrossRef]
- Moawad, H.; Abd el-Rahim, W.M.; Abd el-Aleem, D.; Abo Sedera, S.A. Persistence of two Rhizobium etli inoculant strains in clay and silty loam soils. J. Basic Microbiol. 2005, 45, 438–446. [Google Scholar] [CrossRef] [PubMed]
- Abdelkhalek, A.; Yassin, Y.; Abdel-Megeed, A.; Abd-Elsalam, K.; Moawad, H.; Behiry, S. Rhizobium leguminosarum bv. viciae-mediated silver nanoparticles for controlling Bean Yellow Mosaic Virus (BYMV) infection in faba bean plants. Plants 2022, 12, 45. [Google Scholar] [CrossRef] [PubMed]
- Mergeay, M.; Monchy, S.; Vallaeys, T.; Auquier, V.; Benotmane, A.; Bertin, P.; Taghavi, S.; Dunn, J.; Van Der Lelie, D.; Wattiez, R. Ralstonia metallidurans, a bacterium specifically adapted to toxic metals: Towards a catalogue of metal-responsive genes. FEMS Microbiol. Rev. 2003, 27, 385–410. [Google Scholar] [CrossRef] [PubMed]
- Diels, L.; Van Roy, S.; Taghavi, S.; Van Houdt, R. From industrial sites to environmental applications with Cupriavidus metallidurans. Antonie Leeuwenhoek 2009, 96, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Lal, D.; Nayyar, N.; Kohli, P.; Lal, R. Cupriavidus metallidurans: A modern alchemist. Indian J. Microbiol. 2013, 53, 114–115. [Google Scholar] [CrossRef] [PubMed]
- Reith, F.; Etschmann, B.; Grosse, C.; Moors, H.; Benotmane, M.A.; Monsieurs, P.; Grass, G.; Doonan, C.; Vogt, S.; Lai, B.; et al. Mechanisms of gold biomineralization in the bacterium Cupriavidus metallidurans. Proc. Natl. Acad. Sci. USA 2009, 106, 17757–17762. [Google Scholar] [CrossRef] [PubMed]
- Iguchi, H.; Yurimoto, H.; Sakai, Y. Interactions of methylotrophs with plants and other heterotrophic bacteria. Microorganisms 2015, 3, 137–151. [Google Scholar] [CrossRef]
- Semrau, J.D.; DiSpirito, A.A.; Vuilleumier, S. Facultative methanotrophy: False leads, true results, and suggestions for future research: Facultative methanotrophy. FEMS Microbiol. Lett. 2011, 323, 1–12. [Google Scholar] [CrossRef]
- Stock, M.; Hoefman, S.; Kerckhof, F.-M.; Boon, N.; De Vos, P.; De Baets, B.; Heylen, K.; Waegeman, W. Exploration and prediction of interactions between methanotrophs and heterotrophs. Res. Microbiol. 2013, 164, 1045–1054. [Google Scholar] [CrossRef]
- Liebner, S.; Zeyer, J.; Wagner, D.; Schubert, C.; Pfeiffer, E.-M.; Knoblauch, C. Methane oxidation associated with submerged brown mosses reduces methane emissions from siberian polygonal tundra: Moss-associated methane oxidation. J. Ecol. 2011, 99, 914–922. [Google Scholar] [CrossRef]
- Moh, T.H.; Furusawa, G.; Amirul, A.A.-A. Microbulbifer aggregans sp. nov., isolated from estuarine sediment from a mangrove forest. Int. J. Syst. Evol. Microbiol. 2017, 67, 4089–4094. [Google Scholar] [CrossRef] [PubMed]
- Komarnisky, L.A.; Christopherson, R.J.; Basu, T.K. Sulfur: Its clinical and toxicologic aspects. Nutrition 2003, 19, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Spiers, A.J.; Buckling, A.; Rainey, P.B. The causes of Pseudomonas diversity. Microbiology 2000, 146, 2345–2350. [Google Scholar] [CrossRef] [PubMed]
- Preston, G.M. Plant Perceptions of plant growth-promoting Pseudomonas. Phil. Trans. R. Soc. Lond. B 2004, 359, 907–918. [Google Scholar] [CrossRef] [PubMed]
- Bunker, S.T.; Bates, T.C.; Oliver, J.D. Effects of temperature on detection of plasmid or chromosomally encoded gfp and lux labeled Pseudomonas fluorescens in Soil. Environ. Biosaf. Res. 2004, 3, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Wallace, R.L.; Hirkala, D.L.; Nelson, L.M. Efficacy of Pseudomonas fluorescens for control of mucor rot of apple during commercial storage and potential modes of action. Can. J. Microbiol. 2018, 64, 420–431. [Google Scholar] [CrossRef]
- Van Der Ent, S.; Van Wees, S.C.M.; Pieterse, C.M.J. Jasmonate signaling in plant interactions with resistance-inducing beneficial microbes. Phytochemistry 2009, 70, 1581–1588. [Google Scholar] [CrossRef]
- Wang, Z.; Zhong, T.; Chen, K.; Du, M.; Chen, G.; Chen, X.; Wang, K.; Zalán, Z.; Takács, K.; Kan, J. Antifungal activity of volatile organic compounds produced by Pseudomonas fluorescens ZX and potential biocontrol of blue mold decay on postharvest citrus. Food Control 2021, 120, 107499. [Google Scholar] [CrossRef]
- Ramos-González, M.I.; Ramos-Díaz, M.A.; Ramos, J.L. Chromosomal gene capture mediated by the Pseudomonas putida TOL catabolic plasmid. J. Bacteriol. 1994, 176, 4635–4641. [Google Scholar] [CrossRef]
- Espinosa-Urgel, M.; Kolter, R.; Ramos, J.-L. Root colonization by Pseudomonas putida: Love at first sight. Microbiology 2002, 148, 341–343. [Google Scholar] [CrossRef]
- Matilla, M.A.; Ramos, J.L.; Bakker, P.A.H.M.; Doornbos, R.; Badri, D.V.; Vivanco, J.M.; Ramos-González, M.I. Pseudomonas putida KT2440 causes induced systemic resistance and changes in Arabidopsis root exudation. Environ. Microbiol. Rep. 2010, 2, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Molina-Romero, D.; Morales-García, Y.-E.; Hernández-Tenorio, A.-L.; Netzahuatl-Muñoz, A.-R. Pseudomonas putida estimula el crecimiento de maíz en función de la temperatura. Rev. Iberoam. Cienc. 2017, 4, 80–88. [Google Scholar]
- Morales-García, Y.E.; Baez, A.; Quintero-Hernández, V.; Molina-Romero, D.; Rivera-Urbalejo, A.P.; Pazos-Rojas, L.A.; Muñoz-Rojas, J. Bacterial Mixtures, the future generation of inoculants for sustainable crop production. In Field Crops: Sustainable Management by PGPR, Sustainable Development and Biodiversity; Springer Nature: Cham, Switzerland, 2019; Volume 23, pp. 11–44. ISBN 978-3-030-30925-1. [Google Scholar]
- Pazos-Rojas, L.A.; Marín-Cevada, V.; García, Y.E.M.; Baez, A.; Villalobos-López, M.A.; Pérez-Santos, M. Uso de microorganismos benéficos para reducir los daños causados por la revolución verde. Rev. Iberoam. Cienc. 2016, 3, 72–85. [Google Scholar]
- Meighen, E.A. Molecular biology of bacterial bioluminescence. Microbiol. Rev. 1991, 55, 123–142. [Google Scholar] [CrossRef] [PubMed]
- Jones, B.W.; Nishiguchi, M.K. Counterillumination in the Hawaiian Bobtail squid, Euprymna scolopes berry (Mollusca: Cephalopoda). Mar. Biol. 2004, 144, 1151–1155. [Google Scholar] [CrossRef]
- Lee, K.-H.; Ruby, E.G. Effect of the squid host on the abundance and distribution of symbiotic Vibrio fischeri in Nature. Appl. Environ. Microbiol. 1994, 60, 1565–1571. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Hirose, T.; Yokota, A. Four novel Arthrobacter species Isolated from filtration substrate. Int. J. Syst. Evol. Microbiol. 2009, 59, 856–862. [Google Scholar] [CrossRef] [PubMed]
- Soomro, A.H.; Masud, T.; Anwaar, K. Role of Lactic Acid Bacteria (LAB) in food preservation and human health—A Review. Pak. J. Nutr. 2001, 1, 20–24. [Google Scholar] [CrossRef]
- Mousavi Khaneghah, A.; Abhari, K.; Eş, I.; Soares, M.B.; Oliveira, R.B.A.; Hosseini, H.; Rezaei, M.; Balthazar, C.F.; Silva, R.; Cruz, A.G.; et al. Interactions between probiotics and pathogenic microorganisms in Hosts and foods: A review. Trends Food Sci. Technol. 2020, 95, 205–218. [Google Scholar] [CrossRef]
- Majeed, M.; Nagabhushanam, K.; Natarajan, S.; Sivakumar, A.; Eshuis-de Ruiter, T.; Booij-Veurink, J.; De Vries, Y.P.; Ali, F. Evaluation of genetic and phenotypic consistency of Bacillus coagulans MTCC 5856: A commercial probiotic strain. World J. Microbiol. Biotechnol. 2016, 32, 60. [Google Scholar] [CrossRef]
- Saw, C.-Y.; Chang, T.-J.; Chen, P.-Y.; Dai, F.-J.; Lau, Y.-Q.; Chen, T.-Y.; Chau, C.-F. Presence of Bacillus coagulans spores and vegetative cells in rat intestine and feces and their physiological effects. Biosci. Biotechnol. Biochem. 2019, 83, 2327–2333. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.K.; Srivastava, R. Resuscitation Promoting Factors: A Family of microbial proteins in survival and resuscitation of dormant mycobacteria. Indian J. Microbiol. 2012, 52, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Seddik, H.A.; Bendali, F.; Gancel, F.; Fliss, I.; Spano, G.; Drider, D. Lactobacillus plantarum and its probiotic and food potentialities. Probiotics Antimicro. Prot. 2017, 9, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Todorov, S.D.; Franco, B.D.G.D.M. Lactobacillus plantarum: Characterization of the species and application in food production. Food Rev. Int. 2010, 26, 205–229. [Google Scholar] [CrossRef]
- Barbosa, M.S.; Todorov, S.D.; Ivanova, I.V.; Belguesmia, Y.; Choiset, Y.; Rabesona, H.; Chobert, J.-M.; Haertlé, T.; Franco, B.D.G.M. Characterization of a two-peptide plantaricin produced by Lactobacillus plantarum MBSa4 isolated from brazilian salami. Food Control 2016, 60, 103–112. [Google Scholar] [CrossRef]
- Surono, I.S.; Collado, M.C.; Salminen, S.; Meriluoto, J. Effect of glucose and incubation temperature on metabolically active Lactobacillus plantarum from Dadih in removing microcystin-LR. Food Chem. Toxicol. 2008, 46, 502–507. [Google Scholar] [CrossRef]
- Tonon, T.; Lonvaud-Funel, A. Metabolism of arginine and its positive effect on growth and revival of Oenococcus oeni: Degradation of arginine by Oenococcus oeni. J. Appl. Microbiol. 2000, 89, 526–531. [Google Scholar] [CrossRef] [PubMed]
- Bech-Terkilsen, S.; Westman, J.O.; Swiegers, J.H.; Siegumfeldt, H. Oenococcus oeni, a species born and moulded in wine: A critical review of the stress impacts of wine and the physiological responses. Aust. J. Grape Wine Res. 2020, 26, 188–206. [Google Scholar] [CrossRef]
- Conway, T.; Schoolnik, G.K. Microarray expression profiling: Capturing a genome-wide portrait of the transcriptome: Genome-wide portrait. Mol. Microbiol. 2003, 47, 879–889. [Google Scholar] [CrossRef]
- Laalami, S.; Zig, L.; Putzer, H. Initiation of mRNA decay in bacteria. Cell. Mol. Life Sci. 2014, 71, 1799–1828. [Google Scholar] [CrossRef]
- Song, S.; Wood, T.K. ‘Viable but non culturable cells’ are dead. Environ. Microbiol. 2021, 23, 2335–2338. [Google Scholar] [CrossRef] [PubMed]
- Arvaniti, M.; Skandamis, P.N. Defining bacterial heterogeneity and dormancy with the parallel use of single-cell and population level approaches. Curr. Opin. Food Sci. 2022, 44, 100808. [Google Scholar] [CrossRef]
- Morton, D.S.; Oliver, J.D. Induction of carbon starvation-induced proteins in Vibrio vulnificus. Appl. Environ. Microbiol. 1994, 60, 3653–3659. [Google Scholar] [CrossRef] [PubMed]
- McGovern, V.P.; Oliver, J.D. Induction of cold-responsive proteins in Vibrio vulnificus. J. Bacteriol. 1995, 177, 4131–4133. [Google Scholar] [CrossRef] [PubMed]
- Federighi, M.; Tholozan, J.L.; Cappelier, J.M.; Tissier, J.P.; Jouve, J.L. Evidence of non-coccoid viable but non-culturable Campylobacter jejuni cells in microcosm water by direct viable count, CTC-DAPI double staining, and scanning electron microscopy. Food Microbiol. 1998, 15, 539–550. [Google Scholar] [CrossRef]
- Suzina, N.E.; Mulyukin, A.L.; Dmitriev, V.V.; Nikolaev, Y.A.; Shorokhova, A.P.; Bobkova, Y.S.; Barinova, E.S.; Plakunov, V.K.; El-Registan, G.I.; Duda, V.I. The structural bases of long-term anabiosis in non-spore-forming bacteria. Adv. Space Res. 2006, 38, 1209–1219. [Google Scholar] [CrossRef]
- Pankratov, T.A.; Nikolaev, Y.A.; Yushina, Y.K.; Tikhonova, E.N.; El-Registan, G.I. Forms of bacterial survival in model biofilms. Coatings 2022, 12, 1913. [Google Scholar] [CrossRef]
- Krupyanskii, Y.F. Architecture of nucleoid in the dormant cells of Escherichia coli. Russ. J. Phys. Chem. B 2021, 15, 326–343. [Google Scholar] [CrossRef]
- Loiko, N.G.; Suzina, N.E.; Soina, V.S.; Smirnova, T.A.; Zubasheva, M.V.; Azizbekyan, R.R.; Sinitsyn, D.O.; Tereshkina, K.B.; Nikolaev, Y.A.; Krupyanskii, Y.F.; et al. Biocrystalline structures in the nucleoids of the stationary and dormant prokaryotic cells. Microbiology 2017, 86, 714–727. [Google Scholar] [CrossRef]
- Day, A.P.; Oliver, J.D. Changes in membane fatty acid composition during entry of Vibrio vulnificus into the viable but non culturable state. J. Microbiol. 2004, 42, 69–73. [Google Scholar]
- Signoretto, C.; Del Mar Lleò, M.; Tafi, M.C.; Canepari, P. Cell wall chemical composition of Enterococcus faecalis in the Viable but nonculturable state. Appl. Environ. Microbiol. 2000, 66, 1953–1959. [Google Scholar] [CrossRef] [PubMed]
- Mascher, T.; Helmann, J.D.; Unden, G. Stimulus perception in bacterial signal-transducing histidine kinases. Microbiol. Mol. Biol. Rev. 2006, 70, 910–938. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, T.; Scheide, D. The respiratory complex I of bacteria, Archaea and Eukarya and Its module common with membrane-bound multisubunit hydrogenases. FEBS Lett. 2000, 479, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Postnikova, O.A.; Shao, J.; Mock, N.M.; Baker, C.J.; Nemchinov, L.G. Gene Expression profiling in Viable but Nonculturable (VBNC) cells of Pseudomonas syringae pv. syringae. Front. Microbiol. 2015, 6, 01419. [Google Scholar] [CrossRef] [PubMed]
- Hua, J.; Ho, B. Is the Coccoid form of Helicobacter pylori viable? Microbios 1996, 87, 103–112. [Google Scholar] [PubMed]
- Asakura, H.; Ishiwa, A.; Arakawa, E.; Makino, S.; Okada, Y.; Yamamoto, S.; Igimi, S. Gene expression profile of Vibrio cholerae in the cold stress-induced viable but non-culturable state. Environ. Microbiol. 2007, 9, 869–879. [Google Scholar] [CrossRef]
- Abe, A.; Ohashi, E.; Ren, H.; Hayashi, T.; Endo, H. Isolation and characterization of a cold-induced nonculturable suppression mutant of Vibrio vulnificus. Microbiol. Res. 2007, 162, 130–138. [Google Scholar] [CrossRef]
- Lai, C.-J.; Chen, S.-Y.; Lin, I.-H.; Chang, C.-H.; Wong, H. Change of protein profiles in the induction of the viable but nonculturable state of Vibrio parahaemolyticus. Int. J. Food Microbiol. 2009, 135, 118–124. [Google Scholar] [CrossRef]
- Boaretti, M.; Del Mar Lleo, M.; Bonato, B.; Signoretto, C.; Canepari, P. Involvement of rpoS in the survival of Escherichia coli in the viable but non-culturable state. Environ. Microbiol. 2003, 5, 986–996. [Google Scholar] [CrossRef]
- Ramamurthy, T.; Ghosh, A.; Pazhani, G.P.; Shinoda, S. Current perspectives on Viable but Non-Culturable (VBNC) pathogenic bacteria. Front. Public Health 2014, 2, 00103. [Google Scholar] [CrossRef]
- Grove, A. MarR family transcription factors. Curr. Biol. 2013, 23, R142–R143. [Google Scholar] [CrossRef] [PubMed]
- López-Lara, L.I.; Pazos-Rojas, L.A.; López-Cruz, L.E.; Morales-García, Y.E.; Quintero-Hernández, V.; De La Torre, J.; Van Dillewijn, P.; Muñoz-Rojas, J.; Baez, A. Influence of rehydration on transcriptome during resuscitation of desiccated Pseudomonas putida KT2440. Ann. Microbiol. 2020, 70, 54. [Google Scholar] [CrossRef]
- Bai, K.; Xu, X.; Wang, X.; Li, Y.; Yu, C.; Jiang, N.; Li, J.; Luo, L. Transcriptional profiling of Xanthomonas campestris pv. campestris in Viable but nonculturable state. BMC Genom. 2023, 24, 105. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, Z.; Zhao, F.; Yang, H. Metabolome shifts triggered by chlorine sanitisation induce Escherichia coli on fresh produce into the viable but nonculturable state. Food Res. Int. 2023, 171, 113084. [Google Scholar] [CrossRef] [PubMed]
- Qi, Z.; Sun, N.; Liu, C. Glyoxylate cycle maintains the metabolic homeostasis of Pseudomonas aeruginosa in viable but nonculturable state induced by chlorine stress. Microbiol. Res. 2023, 270, 127341. [Google Scholar] [CrossRef] [PubMed]
- Steinert, M.; Emödy, L.; Amann, R.; Hacker, J. Resuscitation of viable but nonculturable Legionella pneumophila Philadelphia JR32 by Acanthamoeba castellanii. Appl. Environ. Microbiol. 1997, 63, 2047–2053. [Google Scholar] [CrossRef] [PubMed]
- García, M.T.; Jones, S.; Pelaz, C.; Millar, R.D.; Abu Kwaik, Y. Acanthamoeba polyphaga resuscitates viable non-culturable Legionella pneumophila after disinfection. Environ. Microbiol. 2007, 9, 1267–1277. [Google Scholar] [CrossRef]
- Mukamolova, G.V.; Kaprelyants, A.S.; Kell, D.B. Secretion of an antibacterial factor during resuscitation of dormant cells in Micrococcus luteus cultures held in an extended stationary phase. Antonie Leeuwenhoek 1995, 67, 289–295. [Google Scholar] [CrossRef]
- Mukamolova, G.V.; Turapov, O.A.; Young, D.I.; Kaprelyants, A.S.; Kell, D.B.; Young, M. A Family of autocrine growth factors in Mycobacterium tuberculosis: Autocrine growth factors in Mycobacterium tuberculosis. Mol. Microbiol. 2002, 46, 623–635. [Google Scholar] [CrossRef]
- Zhao, R.; Chen, J.; Wang, Y.; Li, Y.; Kong, X.; Han, Y. Proteolytic activity of Vibrio harveyi YeaZ is related with resuscitation on the viable but non-culturable state. Lett. Appl. Microbiol. 2020, 71, 126–133. [Google Scholar] [CrossRef]
- Mukamolova, G.V.; Kaprelyants, A.S.; Young, D.I.; Young, M.; Kell, D.B. A bacterial cytokine. Proc. Natl. Acad. Sci. USA 1998, 95, 8916–8921. [Google Scholar] [CrossRef]
- Freestone, P.P.E.; Haigh, R.D.; Williams, P.H.; Lyte, M. Stimulation of Bacterial growth by heat-stable, norepinephrine-induced autoinducers. FEMS Microbiol. Lett. 1999, 172, 53–60. [Google Scholar] [CrossRef]
- Imazaki, I.; Nakaho, K. Temperature-upshift-mediated revival from the sodium-pyruvate-recoverable viable but nonculturable state induced by low temperature in Ralstonia solanacearum: Linear regression analysis. J. Gen. Plant Pathol. 2009, 75, 213–226. [Google Scholar] [CrossRef]
- Zhao, F.; Bi, X.; Hao, Y.; Liao, X. Induction of viable but nonculturable Escherichia coli O157:H7 by high pressure CO2 and its characteristics. PLoS ONE 2013, 8, e62388. [Google Scholar] [CrossRef]
- Gupte, A.R.; De Rezende, C.L.E.; Joseph, S.W. Induction and resuscitation of viable but nonculturable Salmonella enterica serovar typhimurium DT104†. Appl. Environ. Microbiol. 2003, 69, 6669–6675. [Google Scholar] [CrossRef]
- Yang, D.; Wang, W.; Zhao, L.; Rao, L.; Liao, X. Resuscitation of viable but nonculturable bacteria promoted by ATP-mediated NAD+ synthesis. J. Adv. Res. 2023, in press. [Google Scholar] [CrossRef]
- Abisado, R.G.; Benomar, S.; Klaus, J.R.; Dandekar, A.A.; Chandler, J.R. Bacterial quorum sensing and microbial community interactions. mBio 2018, 9, e02331-17. [Google Scholar] [CrossRef]
- Hassett, D.J.; Ma, J.; Elkins, J.G.; McDermott, T.R.; Ochsner, U.A.; West, S.E.H.; Huang, C.; Fredericks, J.; Burnett, S.; Stewart, P.S.; et al. Quorum sensing in Pseudomonas aeruginosa controls expression of catalase and superoxide dismutase genes and mediates biofilm susceptibility to hydrogen peroxide. Mol. Microbiol. 1999, 34, 1082–1093. [Google Scholar] [CrossRef]
- Liao, H.; Zhong, X.; Xu, L.; Ma, Q.; Wang, Y.; Cai, Y.; Guo, X. Quorum-sensing systems trigger catalase expression to reverse the oxyR deletion-mediated VBNC state in Salmonella typhimurium. Res. Microbiol. 2019, 170, 65–73. [Google Scholar] [CrossRef]
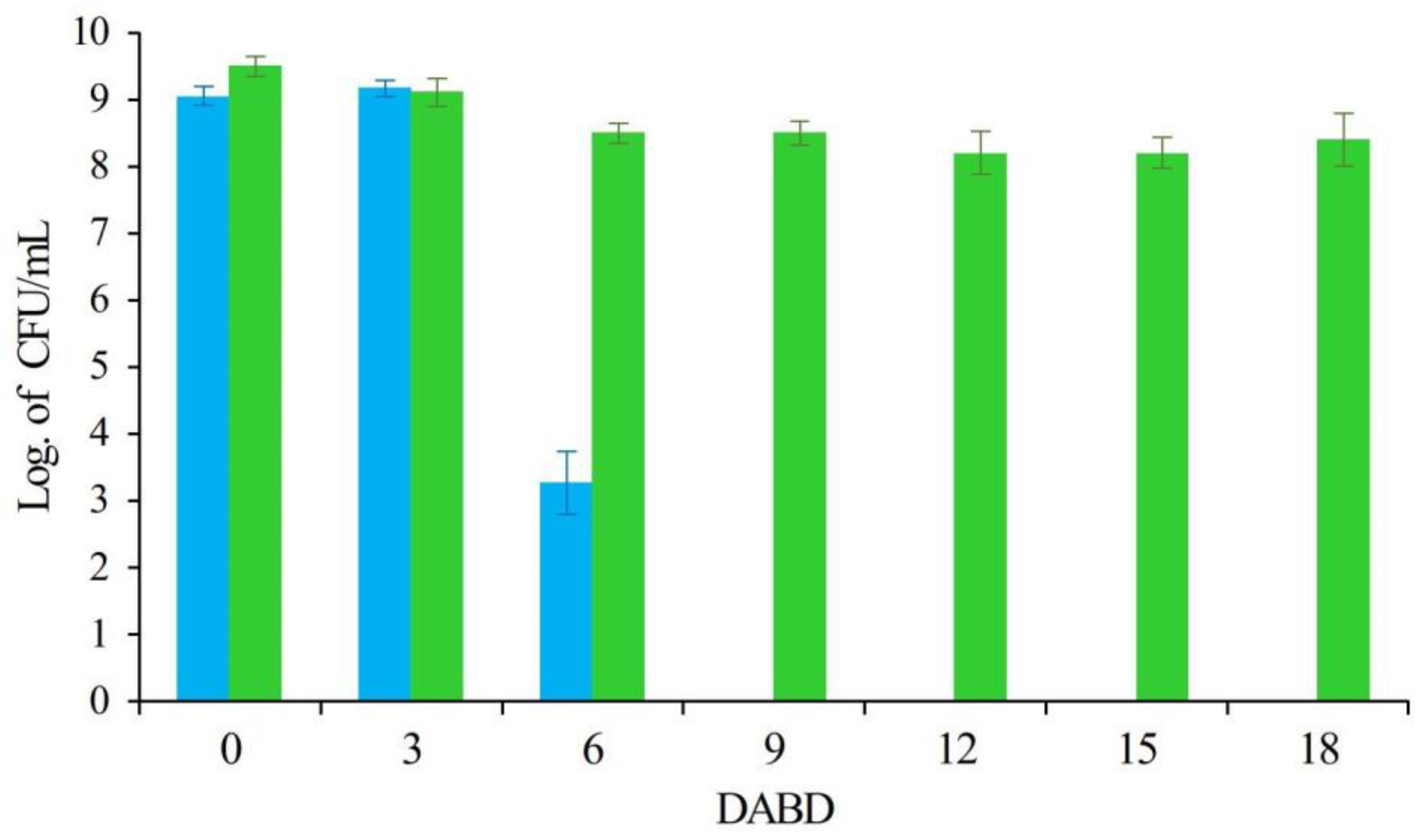
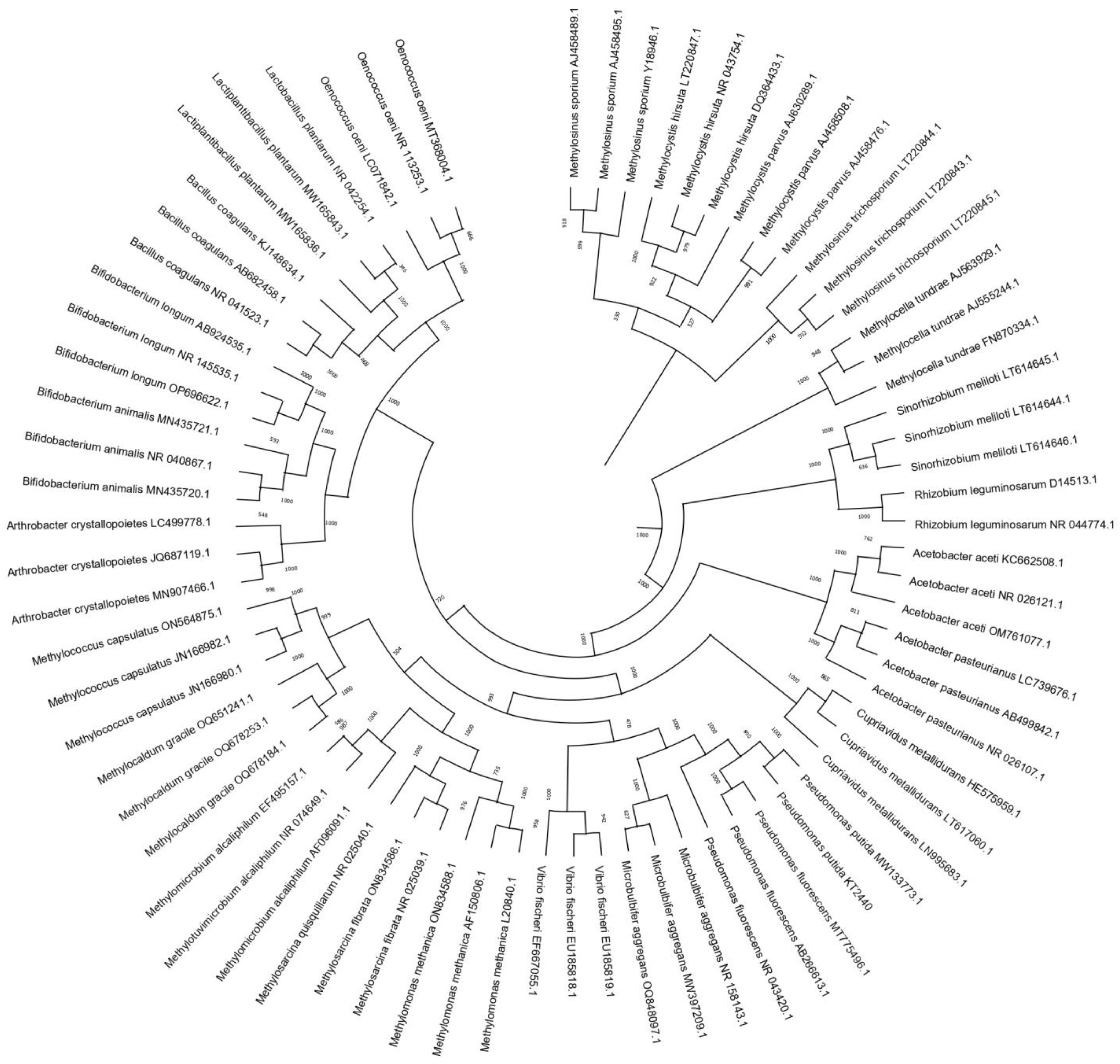
| Group of Bacteria | Species | Conditions that Induce VBNC State | References | |
|---|---|---|---|---|
| Proteobecteria | Alphaproteobacteria | Acetobacter aceti | Treatment with SO2 at a concentration of 30 and 50 mg/L | [46] |
| Acetobacter pasteurianus | High acid stress during fermentation | [47] | ||
| Methylosinus sporium | Freeze drying and cryopreservation (liquid nitrogen) | [25,30] | ||
| Methylosinus trichosporium | ||||
| Methylocystis hirsuta | ||||
| Methylocystis parvus | ||||
| Methylocella tundrae | ||||
| Rhizobium leguminosarum | Cupric sulfate to a concentration of 60 ppm | [48] | ||
| Sinorhizobium meliloti | Incubation at 25 °C in tap water (microcosm-water) Incubation under anoxic conditions in liquid microcosms Incubation in nitrocellulose filters at relative humidity of 22% for three days at 20 °C in the dark | [16] | ||
| Betaproteobacteria | Cupriavidus metallidurans | Incubation in artificial soil at 30 °C for 12 days, without any C source or H2O | [49] | |
| Gamaproteobacteria | Methylomonas methanica | Lyophilization and cryopreservation (liquid nitrogen) | [25,30] | |
| Methylosarcina fibrata | ||||
| Methylocaldum gracile | ||||
| Methylomicrobium alcaliphilum | ||||
| Methylococcus capsulatus | ||||
| Microbulbifer aggregans | Incubation in modified artificial seawater (ASW) for 4 h at 30 °C | [50] | ||
| Pseudomonas fluorescens | Incubation in saline solution (NaCl 0.9% w/v) at 37 °C Exposure to benzalkonium chloride (BAC) | [51] | ||
| Pseudomonas putida KT2440 | Desiccation at 30 °C and 50% relative humidity | [4] | ||
| Vibrio fischeri | Incubation at 22 °C in nutrient-limited artificial seawater (ASW) | [52] | ||
| Terrabacteria | Actinobacteria | Bifidobacterium animalis subsp. lactis |
| [53] |
| Bifidobacterium longum | Storage in fermented foods | [54] | ||
| Arthrobacter albidus, reclassified as Sinomonas albida | Absence of resuscitation promoting factor (Rpf) protein in the culture medium | [55,56] | ||
| Firmicutes | Bacillus coagulans | Incubation at pH 2 for 24 h and subsequent incubation at 140 °C for 5 min | [57] | |
| Lactiplantibacillus plantarum | Treatment for 30 min at 100 °C or with 1 mol/L HCl Incubation in beer at 0 °C temperature | [20] | ||
| Oenococcus oeni | Sulfur dioxide and histidine decarboxylase activity in wines | [46] | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pazos-Rojas, L.A.; Cuellar-Sánchez, A.; Romero-Cerón, A.L.; Rivera-Urbalejo, A.; Van Dillewijn, P.; Luna-Vital, D.A.; Muñoz-Rojas, J.; Morales-García, Y.E.; Bustillos-Cristales, M.d.R. The Viable but Non-Culturable (VBNC) State, a Poorly Explored Aspect of Beneficial Bacteria. Microorganisms 2024, 12, 39. https://doi.org/10.3390/microorganisms12010039
Pazos-Rojas LA, Cuellar-Sánchez A, Romero-Cerón AL, Rivera-Urbalejo A, Van Dillewijn P, Luna-Vital DA, Muñoz-Rojas J, Morales-García YE, Bustillos-Cristales MdR. The Viable but Non-Culturable (VBNC) State, a Poorly Explored Aspect of Beneficial Bacteria. Microorganisms. 2024; 12(1):39. https://doi.org/10.3390/microorganisms12010039
Chicago/Turabian StylePazos-Rojas, Laura Abisaí, Alma Cuellar-Sánchez, Ana Laura Romero-Cerón, América Rivera-Urbalejo, Pieter Van Dillewijn, Diego Armando Luna-Vital, Jesús Muñoz-Rojas, Yolanda Elizabeth Morales-García, and María del Rocío Bustillos-Cristales. 2024. "The Viable but Non-Culturable (VBNC) State, a Poorly Explored Aspect of Beneficial Bacteria" Microorganisms 12, no. 1: 39. https://doi.org/10.3390/microorganisms12010039








