Sperm Incubation in Biggers–Whitten–Whittingham Medium Induces Capacitation-Related Changes in the Lizard Sceloporus torquatus
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Semen Collection and Incubation
2.3. Sperm Assessment
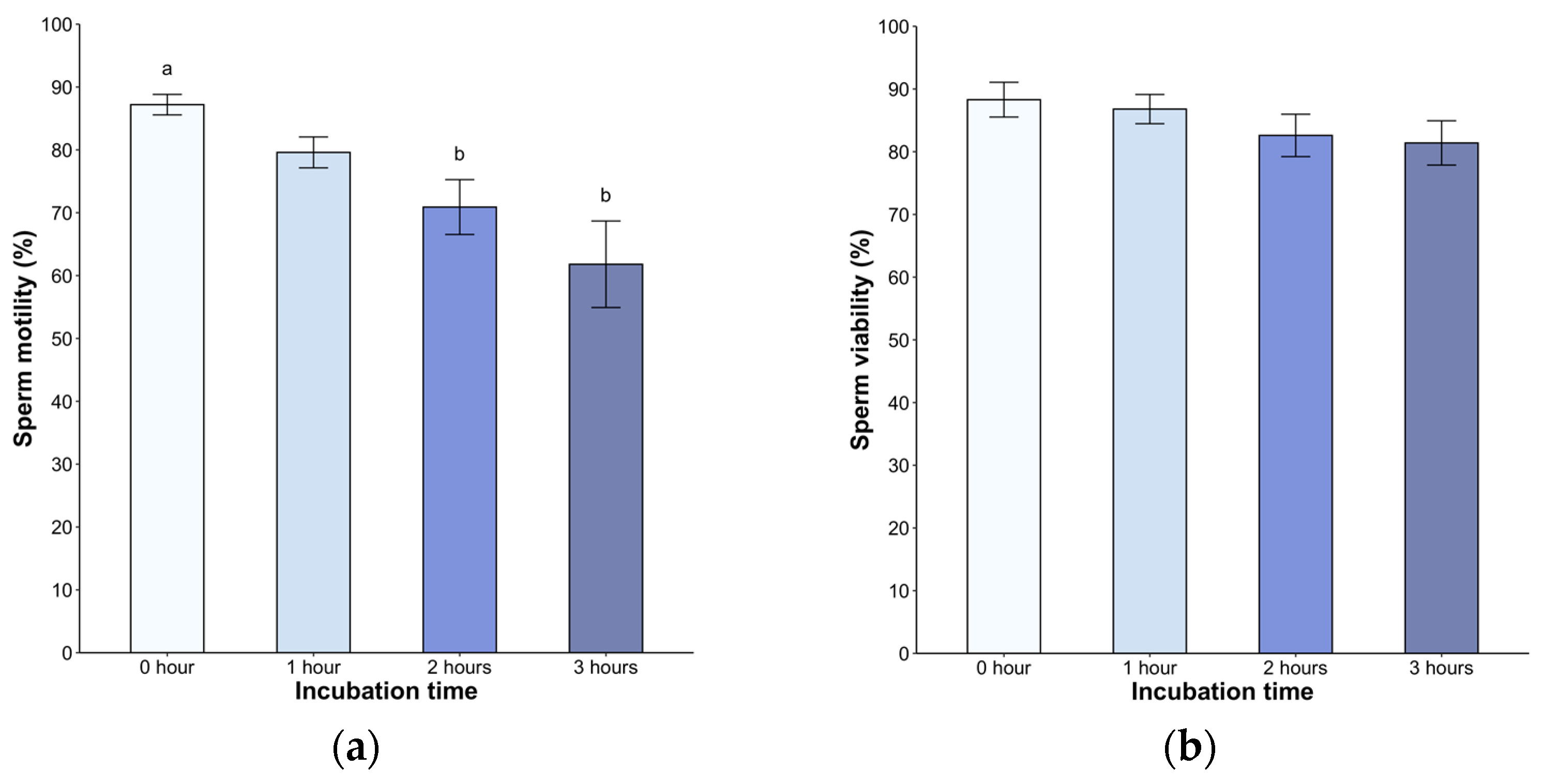
2.3.1. Sperm Motility
2.3.2. Sperm Viability
2.3.3. Capacitation Status
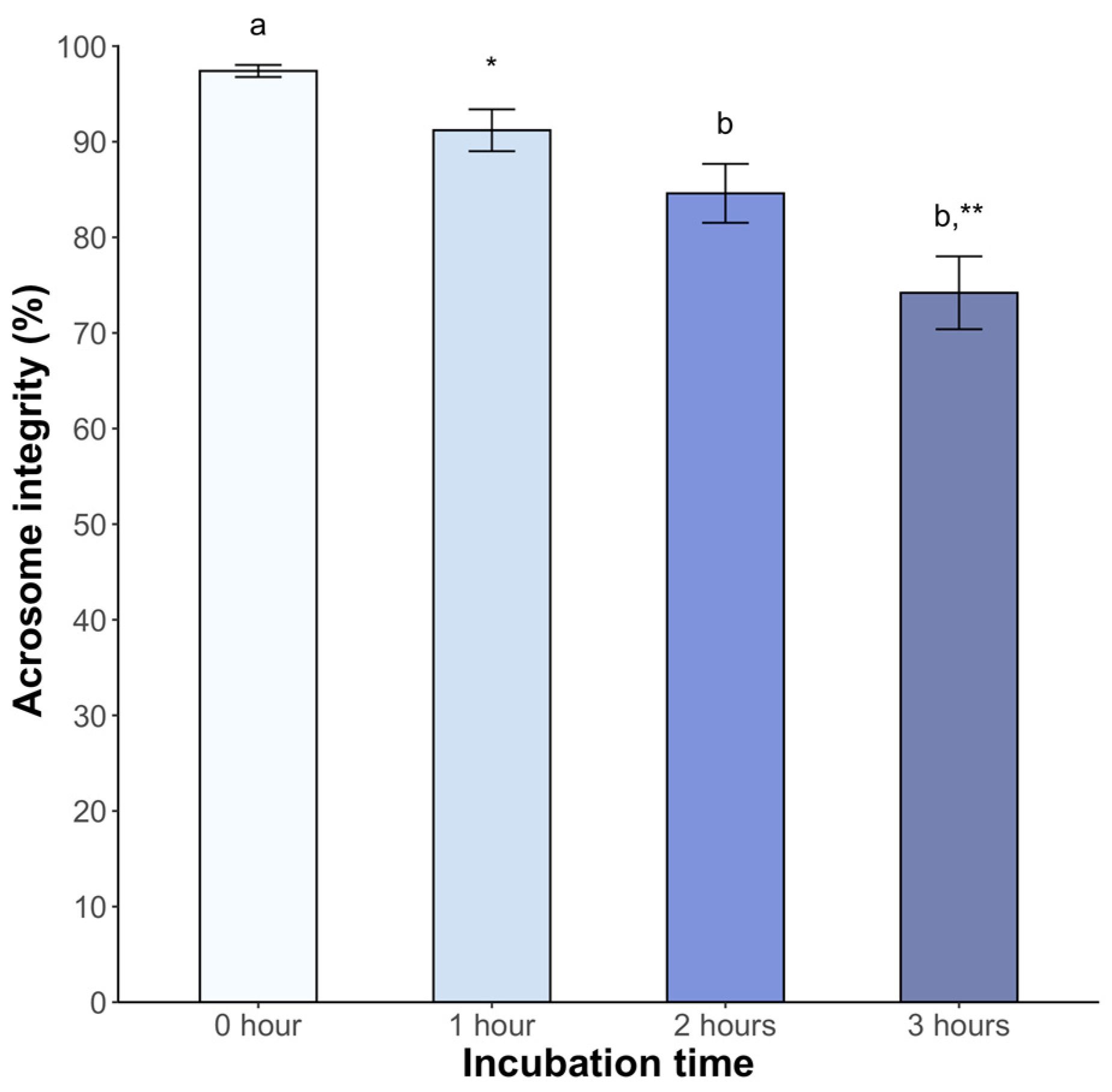
2.3.4. Acrosome Integrity
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fujihara, Y.; Miyata, H.; Ikawa, M. Factors controlling sperm migration through the oviduct revealed by gene-modified mouse models. Exp. Anim. 2018, 67, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Ickowicz, D.; Finkelstein, M.; Breitbart, H. Mechanism of sperm capacitation and the acrosome reaction: Role of protein kinases. Asian J. Androl. 2012, 14, 816–821. [Google Scholar] [CrossRef] [PubMed]
- Puga-Molina, L.C.; Luque, G.M.; Balestrini, P.A.; Marín-Briggiler, C.I.; Romarowski, A.; Buffone, M.G. Molecular basis of human sperm capacitation. Front. Cell Dev. Biol. 2018, 6, 72. [Google Scholar] [CrossRef] [PubMed]
- Stival, C.; Puga-Molina, L.C.; Paudel, B.; Buffone, M.G.; Visconti, P.E.; Krapf, D. Sperm capacitation and acrosome reaction in mammalian sperm. In Sperm Acrosome Biogenesis and Function During Fertilization, 1st ed.; Buffone, M.G., Ed.; Springer: Cham, Switzerland, 2016; Volume 220, pp. 93–106. [Google Scholar] [CrossRef]
- Lemoine, M.; Grasseau, I.; Brillard, J.P.; Blesbois, E. A reappraisal of the factors involved in in vitro initiation of the acrosome reaction in chicken spermatozoa. Reproduction 2008, 136, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Dosemane, D.; Bhagya, M. In vitro study of the spermatozoa motility in the lizard Eutropis carinata. Int. J. Zool. Res. 2015, 11, 89–95. [Google Scholar] [CrossRef]
- Martínez-Torres, M. Almacenamiento de espermatozoides en la vagina de la lagartija vivípara Sceloporus torquatus (Sauria: Prhynosomatidae). Acta Zool. Mex. 2009, 25, 497–506. [Google Scholar] [CrossRef]
- Ortega-León, A.M.; Cruz, M.V.; Zúñiga-Vega, J.J.; Castillo, R.C.; Méndez-de la Cruz, F.R. Sperm viability in the reproductive tract of females in a population of Sceloporus mucronatus exhibiting asynchronous reproduction. West N. Am. Nat. 2009, 69, 96–104. [Google Scholar] [CrossRef]
- Nixon, B.; Anderson, A.L.; Smith, N.D.; McLeod, R.; Johnston, S.D. The Australian saltwater crocodile (Crocodylus porosus) provides evidence that the capacitation of spermatozoa may extend beyond the mammalian lineage. Proc. R Soc. B 2016, 283, 20160495. [Google Scholar] [CrossRef]
- Depeiges, A.; Dacheux, J.L. Acquisition of sperm motility and its maintenance during storage in the lizard, Lacerta vivipara. Reproduction 1985, 74, 23–27. [Google Scholar] [CrossRef]
- Böhm, M.; Collen, B.; Baillie, J.E.M.; Bowles, P.; Chanson, J.; Cox, N.; Hammerson, G.; Hoffmann, M.; Livingstone, S.R.; Ram, M.; et al. The conservation status of the world’s reptiles. Biol. Conserv. 2013, 157, 372–385. [Google Scholar] [CrossRef]
- Sinervo, B.; Méndez-de la Cruz, F.R.; Miles, D.B.; Heulin, B.; Bastiaans, E.; Villagrán-Santa Cruz, M.; Lara-Resendiz, R.; Martínez-Méndez, N.; Calderón-Espinosa, M.L.; Meza-Lázaro, R.N.; et al. Erosion of Lizard Diversity by Climate Change and Altered Thermal Niches. Science 2010, 328, 894–899. [Google Scholar] [CrossRef] [PubMed]
- Comizzoli, P.; Holt, W.V. Recent Progress in Spermatology Contributing to the Knowledge and Conservation of Rare and Endangered Species. Ann. Rev. Anim. Biosci. 2022, 10, 469–490. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Cano, N.B.; Sánchez-Rivera, U.Á.; Álvarez-Rodríguez, C.; Dávila-Govantes, R.; Cárdenas-León, M.; Martínez-Torres, M. Sex steroids are correlated with environmental factors and body condition during the reproductive cycle in females of the lizard Sceloporus torquatus. Gen. Comp. Endocrinol. 2021, 314, 113921. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Rivera, U.Á. Análisis hormonal y citológico de la espermatogénesis mediante biopsia testicular en Sceloporus torquatus (Sauria: Phrynosomatidae). Bachelor’s Thesis, National Autonomous University of Mexico, Mexico City, Mexico, 2017. [Google Scholar]
- Martínez-Torres, M.; Sánchez-Rivera, U.Á.; Cruz-Cano, N.B.; Castro-Camacho, Y.J.; Luis, J.; Medrano, A. A non-invasive method for semen collection and evaluation in small and median size lizards. Reprod. Domest. Anim. 2019, 54 (Suppl. 4), 54–58. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Torres, M.; Álvarez-Rodríguez, C.; Luis, J.; Sánchez-Rivera, U.Á. Electroejaculation and semen evaluation of the viviparous lizard Sceloporus torquatus (Squamata: Phrynosomatidae). Zoo Biol. 2019, 38, 393–396. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Rivera, U.Á.; Medrano, A.; Cruz-Cano, N.B.; Alcántar-Rodríguez, A.; Dávila-Govantes, R.; Castro-Camacho, Y.J.; Martínez-Torres, M. Implementation of a method for sperm cryopreservation in sceloporine lizards. Conserv. Physiol. 2022, 10, coac068. [Google Scholar] [CrossRef] [PubMed]
- Campbell, L.; Cafe, S.I.; Upton, R.; Sean Doody, J.; Nixon, B.; Clulow, J.; Clulow, S. A model protocol for the cryopreservation and recovery of motile lizard sperm using the phosphodiesterase inhibitor caffeine. Conserv. Physiol. 2020, 8, coaa044. [Google Scholar] [CrossRef] [PubMed]
- Mattioli, M.; Barboni, B.; Lucidi, P.; Seren, E. Identification of capacitation in boar spermatozoa by chlortetracycline staining. Theriogenology 1996, 45, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Feria-Ortiz, M.; Nieto-Montes de Oca, A.; Ugarte, I.H. Diet and Reproductive Biology of the Viviparous Lizard Sceloporus torquatus torquatus (Squamata: Phrynosomatidae). J. Herpetol. 2001, 35, 104–112. [Google Scholar] [CrossRef]
- Naijian, H.R.; Kohram, H.; Shahneh, A.Z.; Sharafi, M.; Bucak, M.N. Effects of different concentrations of BHT on microscopic and oxidative parameters of Mahabadi goat semen following the freeze–thaw process. Cryobiology 2013, 66, 151–155. [Google Scholar] [CrossRef]
- Ortega-Morales, L.D.; Alcantar-Rodriguez, A.; Espejel, M.C.; Medrano, A. The effect of non-traditional cooling on dog sperm cryosurvival and ability to perform the acrosome reaction. Aust. J. Vet. Sci. 2019, 51, 73–82. [Google Scholar] [CrossRef]
- Sáez-Espinosa, P.; Huerta-Retamal, N.; Robles-Gómez, L.; Avilés, M.; Aizpurua, J.; Velasco, I.; Romero, A.; Gómez-Torres, M.J. Influence of in vitro capacitation time on structural and functional human sperm parameters. Asian J. Androl. 2020, 22, 447–453. [Google Scholar] [CrossRef]
- Bustani, G.S.; Baiee, F.H. Semen extenders: An evaluative overview of preservative mechanisms of semen and semen extenders. Vet. World. 2021, 14, 1220–1233. [Google Scholar] [CrossRef] [PubMed]
- Witte, T.S.; Schäfer-Somi, S. Involvement of cholesterol, calcium and progesterone in the induction of capacitation and acrosome reaction of mammalian spermatozoa. Anim. Reprod. Sci. 2007, 102, 181–193. [Google Scholar] [CrossRef]
- Kito, S.; Ohta, Y. Medium effects on capacitation and sperm penetration through the zona pellucida in inbred BALB/c spermatozoa. Zygote 2005, 13, 145–153. [Google Scholar] [CrossRef]
- McPartlin, L.A.; Littell, J.; Mark, E.; Nelson, J.L.; Travis, A.J.; Bedford-Guaus, S.J. A defined medium supports changes consistent with capacitation in stallion sperm, as evidenced by increases in protein tyrosine phosphorylation and high rates of acrosomal exocytosis. Theriogenology 2008, 69, 639–650. [Google Scholar] [CrossRef] [PubMed]
- Ho, H.; Suarez, S.S. Hyperactivation of mammalian spermatozoa: Function and regulation. Reproduction 2001, 122, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Suarez, S.S. Control of hyperactivation in sperm. Hum. Reprod. Update 2008, 14, 647–657. [Google Scholar] [CrossRef]
- Mendes Cunha, A.T.; Faria, O.; Guimarães, A.L.S. Bovine Sperm Capacitation: Physiological Changes and Evaluations. JSM Invit. Fertil. 2017, 2, 1011. [Google Scholar] [CrossRef]
- Rathi, R.; Colenbrander, B.; Bevers, M.M.; Gadella, B.M. Evaluation of In Vitro Capacitation of Stallion Spermatozoa. Biol. Reprod. 2001, 65, 462–470. [Google Scholar] [CrossRef]
- Gadella, B.M.; Boerke, A. An update on post-ejaculatory remodeling of the sperm surface before mammalian fertilization. Theriogenology 2016, 85, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Rota, A.; Peña, A.I.; Linde-Forsberg, C.; Rodríguez-Martínez, H. In vitro capacitation of fresh, chilled and frozen–thawed dog spermatozoa assessed by the chlortetracycline assay and changes in motility patterns. Anim. Reprod. Sci. 1999, 57, 199–215. [Google Scholar] [CrossRef] [PubMed]
- Ded, L.; Dostalova, P.; Zatecka, E.; Dorosh, A.; Komrskova, K.; Peknicova, J. Fluorescent analysis of boar sperm capacitation process in vitro. Reprod. Biol. Endocrinol. 2019, 17, 109. [Google Scholar] [CrossRef]
- Kekäläinen, J.; Larma, I.; Linden, M.; Evans, J.P. Lectin staining and flow cytometry reveals female-induced sperm acrosome reaction and surface carbohydrate reorganization. Sci. Rep. 2015, 5, 15321. [Google Scholar] [CrossRef]
- Priyadarshana, C.; Setiawan, R.; Tajima, A.; Asano, A. Src family kinases-mediated negative regulation of sperm acrosome reaction in chickens (Gallus gallus domesticus). PLoS ONE 2020, 15, e0241181. [Google Scholar] [CrossRef] [PubMed]
- Soriano-Úbeda, C.; Romero-Aguirregomezcorta, J.; Matás, C.; Visconti, P.E.; García-Vázquez, F.A. Manipulation of bicarbonate concentration in sperm capacitation media improves in vitro fertilisation output in porcine species. J. Anim. Sci. Biotechnol. 2019, 10, 19. [Google Scholar] [CrossRef]
- Nixon, B.; Cafe, S.L.; Eamens, A.L.; De Iuliis, G.N.; Bromfield, E.G.; Martin, J.H.; Skerrett-Byrne, D.A.; Dun, M.D. Molecular insights into the divergence and diversity of post-testicular maturation strategies. Mol. Cell. Endocrinol. 2020, 517, 110955. [Google Scholar] [CrossRef] [PubMed]
- Vyklicka, L.; Lishko, P.V. Dissecting the signaling pathways involved in the function of sperm flagellum. Curr. Opin. Cell Biol. 2020, 63, 154–161. [Google Scholar] [CrossRef]
- Friesen, C.R.; Kahrl, A.F.; Olsson, M. Sperm competition in squamate reptiles. Phil. Trans. R. Soc. B. 2020, 375, 20200079. [Google Scholar] [CrossRef]
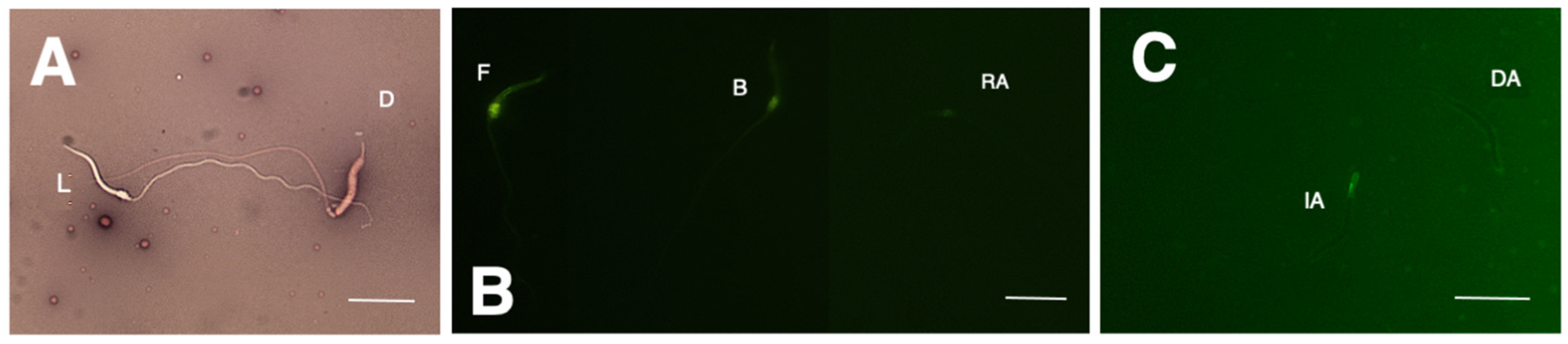

| Body Weight (g) | Snout–Vent Length (cm) | Vent–Tail Length (cm) | Number of Ejaculates | Total Semen Volume (μL) | Sperm Concentration (×106/mL) |
|---|---|---|---|---|---|
| 28.66 ± 4.2 (12.7–52.1) | 8.73 ± 0.5 (6.0–11.5) | 8.84 ± 0.6 (4.7–11.0) | 2.00 ± 0.4 (1.0–3.0) | 3.21 ± 1.31 (2.0–6.0) | 94.23 ± 19.2 (18.3–220.0) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez-Rivera, U.Á.; Cruz-Cano, N.B.; Medrano, A.; Álvarez-Rodríguez, C.; Martínez-Torres, M. Sperm Incubation in Biggers–Whitten–Whittingham Medium Induces Capacitation-Related Changes in the Lizard Sceloporus torquatus. Animals 2024, 14, 1388. https://doi.org/10.3390/ani14091388
Sánchez-Rivera UÁ, Cruz-Cano NB, Medrano A, Álvarez-Rodríguez C, Martínez-Torres M. Sperm Incubation in Biggers–Whitten–Whittingham Medium Induces Capacitation-Related Changes in the Lizard Sceloporus torquatus. Animals. 2024; 14(9):1388. https://doi.org/10.3390/ani14091388
Chicago/Turabian StyleSánchez-Rivera, Uriel Ángel, Norma Berenice Cruz-Cano, Alfredo Medrano, Carmen Álvarez-Rodríguez, and Martín Martínez-Torres. 2024. "Sperm Incubation in Biggers–Whitten–Whittingham Medium Induces Capacitation-Related Changes in the Lizard Sceloporus torquatus" Animals 14, no. 9: 1388. https://doi.org/10.3390/ani14091388





