All variables were evaluated for normality previous to multivariate analysis; no transformations were necessary. Four outliers were revealed in the open field data (Z ≥ 2.5) and each was replaced with the mean for that variable.
2.1. Sexual Motivation
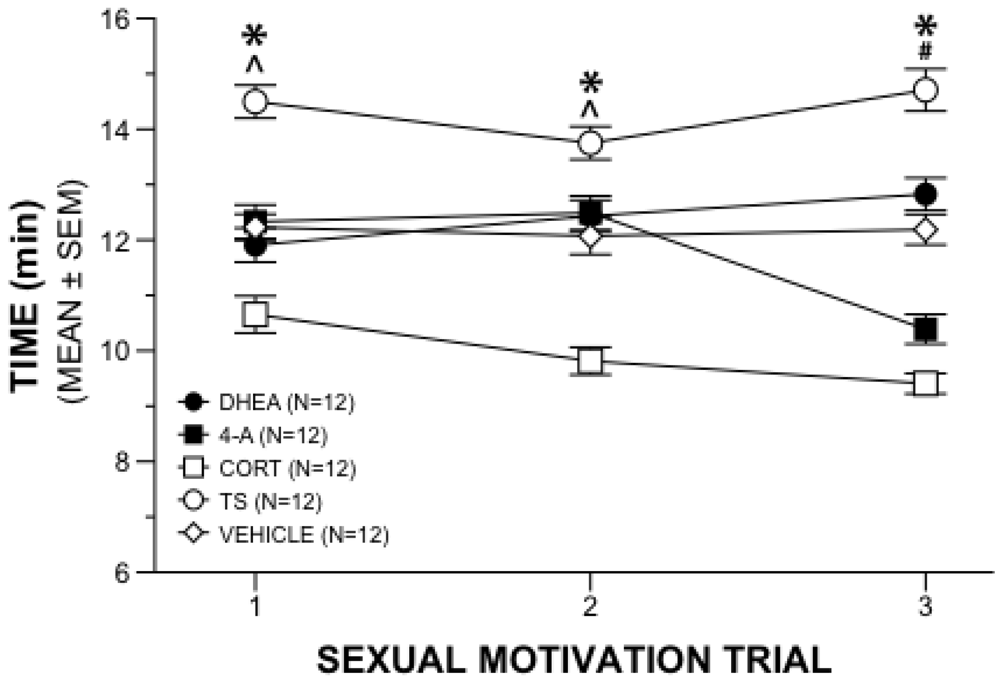
A repeated measures 5 × 3 ANOVA on the time near the receptive female in the sexual motivation paradigm revealed a significant interaction of drug and week (
F8,110 = 9.336,
p = 0.001). Further analyses with main effects allowed comparisons between treatment groups at each week of testing and within treatment groups across the three weeks. Between group analyses revealed time differences among the hormone treatments during the first week of sexual motivation testing (
F4,55 = 21.700,
p = 0.001). Subsequent comparisons with Tukey’s HSD tests (
p < 0.05) indicated that males administered TS spent more time near the estrous female than the other males, and males administered CORT spent less time near the estrous female than all other groups (
Figure 1). Androstenedione and DHEA did not differ from vehicle during the first week.
Figure 1.
Sexual Motivation Behavior. Mean (±SEM) time gonadally intact male rats given chronic steroid or vehicle supplement spent near estrous female during three weeks of 20-min sexual motivation trials. (*) TS significantly different from all other groups; (^) CORT significantly different from all other groups; (#) CORT significantly different from all other groups except 4-A.
Figure 1.
Sexual Motivation Behavior. Mean (±SEM) time gonadally intact male rats given chronic steroid or vehicle supplement spent near estrous female during three weeks of 20-min sexual motivation trials. (*) TS significantly different from all other groups; (^) CORT significantly different from all other groups; (#) CORT significantly different from all other groups except 4-A.
Those differences were mirrored in the second week of testing (F4,55 = 23.494, p = 0.001) in that TS animals spent more time near the estrous female, and the CORT animals spent less time near the estrous female, than all other groups. Comparisons of drug treatment at the third week of sexual motivation testing (F4,55 = 52.735, p = 0.001) revealed that males administered TS continued to spend more time near the estrous female than all other groups. The CORT group spent less time near the estrous female than other groups except for the 4-A animals. There were no differences between CORT and 4-A during the third week.
Within-group main effects provided a snapshot of changes in time near the receptive female over the 3 weeks of sexual motivation testing. Results were that, whereas the TS rats showed increased motivation, each of the three adrenal supplement groups were less interested in the receptive females over weeks. Specifically, animals administered 4-A (F2,54 = 23.876, p = 0.001) spent less time near the estrous female in week 3 compared to both weeks 1 and 2. Males administered DHEA (F2,54 = 3.515, p = 0.037) spent more time with the estrous female during week 3 when compared with week 1, but not week 2. Males administered CORT (F2,54 = 6.740, p = 0.002) spent less time near the estrous female during weeks 2 and 3 compared to week 1. Males receiving TS supplements (F2,54 = 5.036, p = 0.010) spent more time near the estrous female in week 3 when compared to week 2, although not to week 1. Vehicle control animals did not differ in this marker of sexual motivation across the three weeks of observation.
2.2. Urinary Marking and Open Field Activity
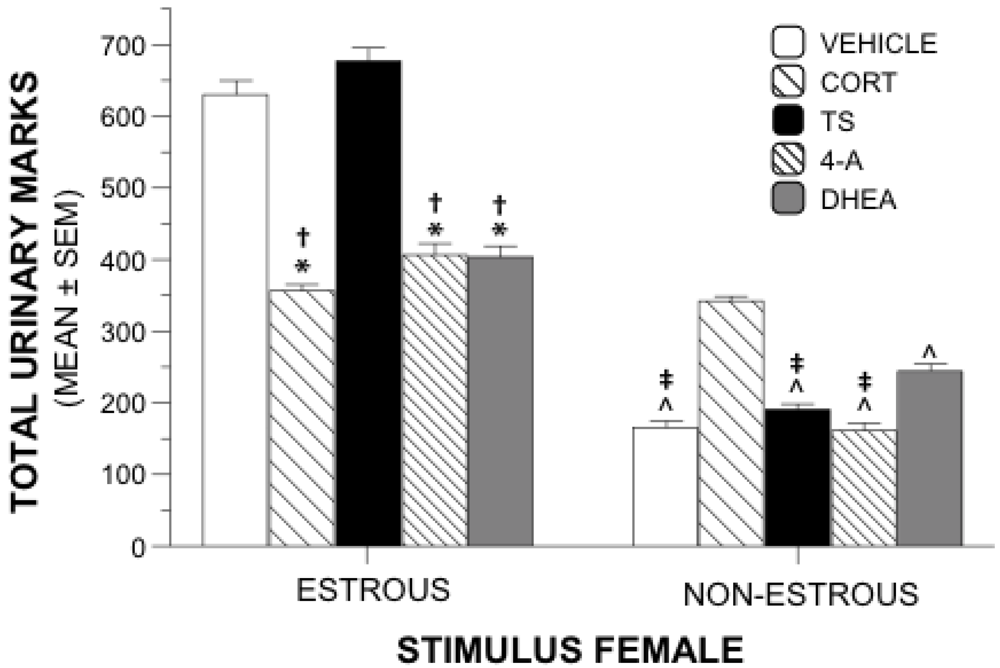
Total numbers of urinary marks near the estrous female or the nonestrous female were used as an additional measure of sexual motivation. A 5 × 2 repeated measures ANOVA revealed a significant interaction of drug and urinary marks (
F4,55 = 145.26,
p = 0.001). Main effects allowed comparisons between treatment groups of total marks near each stimulus female. Between-group analyses showed significant differences of total urinary marks near the estrous female (
F4.55 = 87.687,
p = 0.001). Post hoc tests using Tukey’s HSD (
p < 0.05) showed that animals receiving TS or vehicle marked significantly more near the estrous female than those animals receiving 4-A, DHEA, or CORT (
Figure 2). There was no difference in urinary marking between the vehicle and TS treatment groups, and the markings were similar among the 4-A, DHEA, and CORT groups.
Figure 2.
Urinary Marking Behavior. Mean (±SEM) total urinary marks near stimulus females during sexual motivation testing. Groups of males were gonadally intact and were administered supplements of steroid or vehicle during 3 weeks of testing. (*) Significantly different from TS; (†) Significantly different from vehicle; (^) Significantly different from CORT; (‡); Significantly different from DHEA.
Figure 2.
Urinary Marking Behavior. Mean (±SEM) total urinary marks near stimulus females during sexual motivation testing. Groups of males were gonadally intact and were administered supplements of steroid or vehicle during 3 weeks of testing. (*) Significantly different from TS; (†) Significantly different from vehicle; (^) Significantly different from CORT; (‡); Significantly different from DHEA.
Between-group differences were also significant for total urinary marks near the nonestrous female (F4,55 = 78.022, p = 0.001). Tukey’s post hoc analysis revealed that the CORT animals marked more near the nonestrous female than all other groups. DHEA-treated animals marked near the nonestrous female significantly more than 4-A-, vehicle-, and TS- animals. Numbers of marks near the non-estrous female were similar among the 4-A, TS, and vehicle groups.
Within-group main effects revealed significant differences between marks near the estrous female and the nonestrous female within each treatment group (F4,55 = 39.134, p = 0.001). Males treated with 4-A, DHEA, TS, or vehicle marked significantly more near the estrous female than the nonestrous female (p = 0.001 for all comparisons). CORT-treated animals marked similarly near both stimulus females (p = 0.401).
Numbers of lines crossed in the open field were totaled across the three weeks of testing. A univariate ANOVA revealed no significant differences among groups (F4,55 = 0.654, p = 0.627).
2.3. Discussion
Results of the experiment revealed that supplements of 4-A or DHEA did not mimic the effects of TS supplements on sexual motivation in intact male rats. Both groups were significantly less interested than the TS supplemented group in a receptive female in each of three test sessions. Further, during the third week of testing, the 4-A animals showed similarly suppressed interest as the CORT group in remaining near the female. Examination of an additional marker of sexual motivation, urinary marking near the estrous female, over all weeks indicated that DHEA and 4-A marked similarly to the CORT animals and significantly less than both TS and vehicle control animals.
The present study is part of a research program on adrenal-testicular steroidal interactions using both castrated and intact males [
5,
6]. Here we used intact animals partly because of recent promotions of testosterone supplements in men experiencing perceived or actual low libido. Also, previous findings [
7,
8,
9] indicating that TS supplements can increase sexual motivation in intact rats provided a segue to the empirical question posed for the experiment. That is, will the adrenal precursors androstenedione and dehydroepiandrosterone induce a similar pattern of behavioral changes as TS? The results would also address the conceptual question of whether 4-A and DHEA serve only as precursors to TS or do the adrenal steroids have their own individual influence on brain and behavior. Inclusion of males receiving CORT supplements provided a control group for an adrenal steroid that often is found to suppress sexual behavior.
Findings from the experiment support the hypothesis that DHEA and 4-A have direct influences on behavior, and that they are not simply destined to influence function in an indirect manner by conversion to TS, E2, or DHT. If the adrenal steroids produced behavioral results similar to the TS group, it could be reasonably concluded that DHEA and 4-A lead to behavioral changes only after conversion to TS (indirect) and are without direct influences. Results from this experiment show that this is unlikely to be the case; DHEA and 4-A produced a behavioral footprint different than that of TS, indicating that they likely affect behavior directly and without requiring metabolic conversion. In agreement with previous findings [
10,
11] that increased levels of the stress hormone CORT accompany attenuated male sexual behavior, CORT-supplemented males represented a basement influence on sexual motivation. Unsurprisingly, TS treatment provided a “ceiling effect” for the measures of sexual motivation, resulting in the highest levels of anticipatory behavior [
12]. These findings also suggest that whether or not TS supplements suppressed testicular activity, they enhanced a TS-sensitive behavior [
8,
13].Results from the 4-A and DHEA treatment groups did not mimic this effect and in one instance were actually similar to CORT, providing support for 4-A and DHEA acting directly on the brain and sexual behavior. Decreased urine marking behavior indicates that DHEA and 4-A have their own direct suppressive effect on TS-like behaviors. The results also indicate that DHEA and 4-A are biologically active in ways that significantly affect behavior, indicating that any differences from TS are not attributable to the two simply having no effects,
i.e., replicating the control group.
An important consideration for the different behavioral results yielded from TS, 4-A, and DHEA treatment is the complex metabolic cascade of the steroids. Our recent review [
14] outlined the various possible pathways a steroid precursor can follow. One outcome is adrenal steroid conversion to TS, but another is diversion to estrogens. DHEA is converted to 4-A, which then can be metabolized into either TS or estrone (E1). If converted into E1, the next step in the metabolic cascade is estradiol. Further, TS itself serves as a direct precursor to E2. Therefore while it is entirely plausible that 4-A or DHEA supplement would result in increased TS and androgen-sensitive behaviors, it is equally plausible that such treatment would result in increased E2. The possibility of this secondary pathway to the estrogens, however, cannot explain our results. Both E1 and E2 bind the estrogen receptor (ER) in the periphery and the brain, and binding of the ER is likely one mechanism for male sexual behavior, including motivation [
12]. The estrogen receptor is also a likely mechanism for male urine marking behavior [
15]. Whether converted to TS or E2, the results would mimic TS supplements if DHEA and 4-A served as mere precursors. This is of particular interest when interpreting the results that chronic 4-A supplements suppress sexual motivation compared to the control group. Conversion to TS or E2 would likely lead to increased sexual motivation behavior, but chronic 4-A decreased motivation in the 4-A group of intact males. Although previous studies using various species, including rats, cite the ability of 4-A to reinstate sexual behavior following castration [
16,
17,
18,
19] there is little, if any, research explaining the effects of 4-A supplements administered to intact males. Our data indicate the possibility of a feedback, or down-regulating, mechanism following chronic 4-A treatment that has not been explained previously.
A concern for all hormone research is the issue of acute
versus chronic exposure. This is made apparent in the differences between the CORT-treated group presented here and that of a previous study [
20]. Males supplemented with CORT in the present study experienced suppressed sexual motivation compared to controls as early as 3 weeks into exposure. We cannot say how early this difference could be detected in our animals since we began testing at 3 weeks of exposure, but Retana-Marquez and colleagues indicate in their 1998 study that it would likely be after 8 days of exposure. Interestingly, in their study CORT was administered at a fixed dose (2 mg) higher than we used, yet after 8 days of exposure intact male rats did not experience suppressed sexual behavior. It could be speculated, therefore, that even a high dose of CORT supplement must be administered for longer than 8 days to affect sexual behavior. Future studies like ours may consider behavioral testing at multiple time points spaced closer together and earlier than the ones we used to better assess when differences can first be detected. As is likely apparent, the study just described illustrates another important issue in hormone research: dosing. Future studies on the behavioral effects of hormone supplements may benefit from using multiple levels of exposure rather than using the one we have found useful in our lab [
21].
An interesting result in the current study is that while TS increased, and CORT and 4-A decreased, time spent in the proximity of a receptive female compared to vehicle-treated animals, TS and vehicle induced similar urinary marking near a receptive female while DHEA, 4A, and CORT supplements led to less marking. Both time spent near a receptive female and urinary marking near a receptive female are established as measures of sexual motivation, but it may be that they differ in mechanism such that they are affected by hormones on a different time course. It is also possible that different levels of hormones are required to affect each behavior such that the levels of TS we administered were enough to increase time spent near a receptive female, but not enough to increase urinary marking. Similarly, it is possible that the levels of DHEA supplements we used were enough to decrease urinary marking near a receptive female, but not enough to decrease time spent near the same female. In this light, DHEA appears to exhibit a unique effect on overall sexual motivation; while DHEA-treated males spent a similar amount of time near estrous females as vehicle-only males, they exhibited significantly less urinary marking than vehicle males in this area. These issues can be clarified in future studies utilizing varying doses and exposure periods.
There are a few important limitations of the current study, some of which have been addressed above. In addition to those already mentioned, a significant limitation of our study is the lack of circulating hormone titers and reproductive organ weight data. Reproductive organ weights would provide a rudimentary measure of hormonal effect on the HPG axis, but circulating hormone measurements would provide definitive support for interpretation of the current results.
There are considerable numbers of reports that 4-A and, particularly, DHEA have other functions than those evaluated here. In the brain, DHEA is a neurosteroid that influences a variety of behaviors, including cognition and mood [
22]. Still, it is not clear if neurosteroids must be converted to TS or E2 and bind to the AR and ER receptor because no specific DHEA receptor has been identified. Recently, evidence has emerged that may solve the “DHEA receptor problem.” The suggestion is that the neurosteroids, including DHEA, could influence neural function via interaction with neuronal membranes [
23,
24,
25]. Our results provide evidence that the effects of DHEA and 4-A supplementation are not simply the results of conversion to TS. What seems more likely is that the extra DHEA and 4-A interact directly with neuronal membranes to produce their own sets of behaviors. If confirmed, it would add support for a direct influence of the androgenic adrenal steroids on brain activity.