Prognostic Factors for Post-COVID-19 Syndrome: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Eligibility Criteria
2.3. Selection Process
2.4. Data Extraction
2.5. Risk of Bias Assessment
2.6. Data Synthesis
2.7. Patient and Public Involvement
3. Results
3.1. Study Selection
3.2. Study Characteristics
3.3. Quality of Included Studies
3.4. Factors Associated with PCS Risk
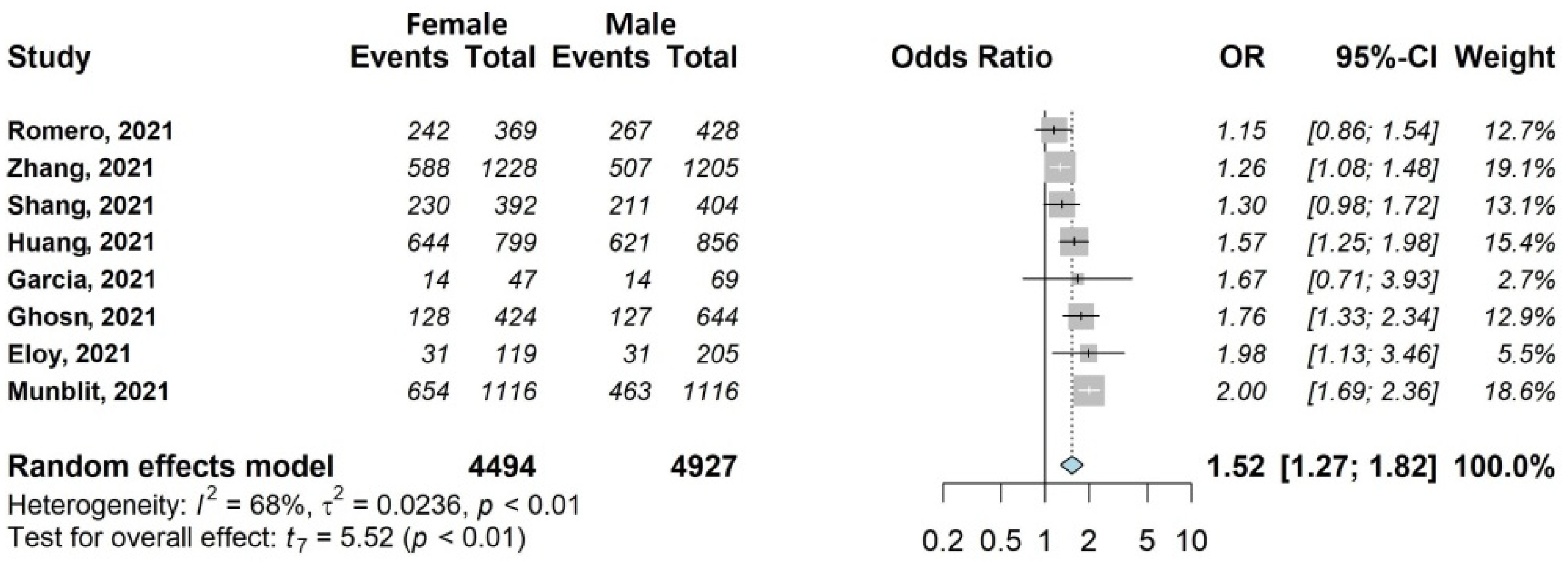
3.4.1. Any Symptoms
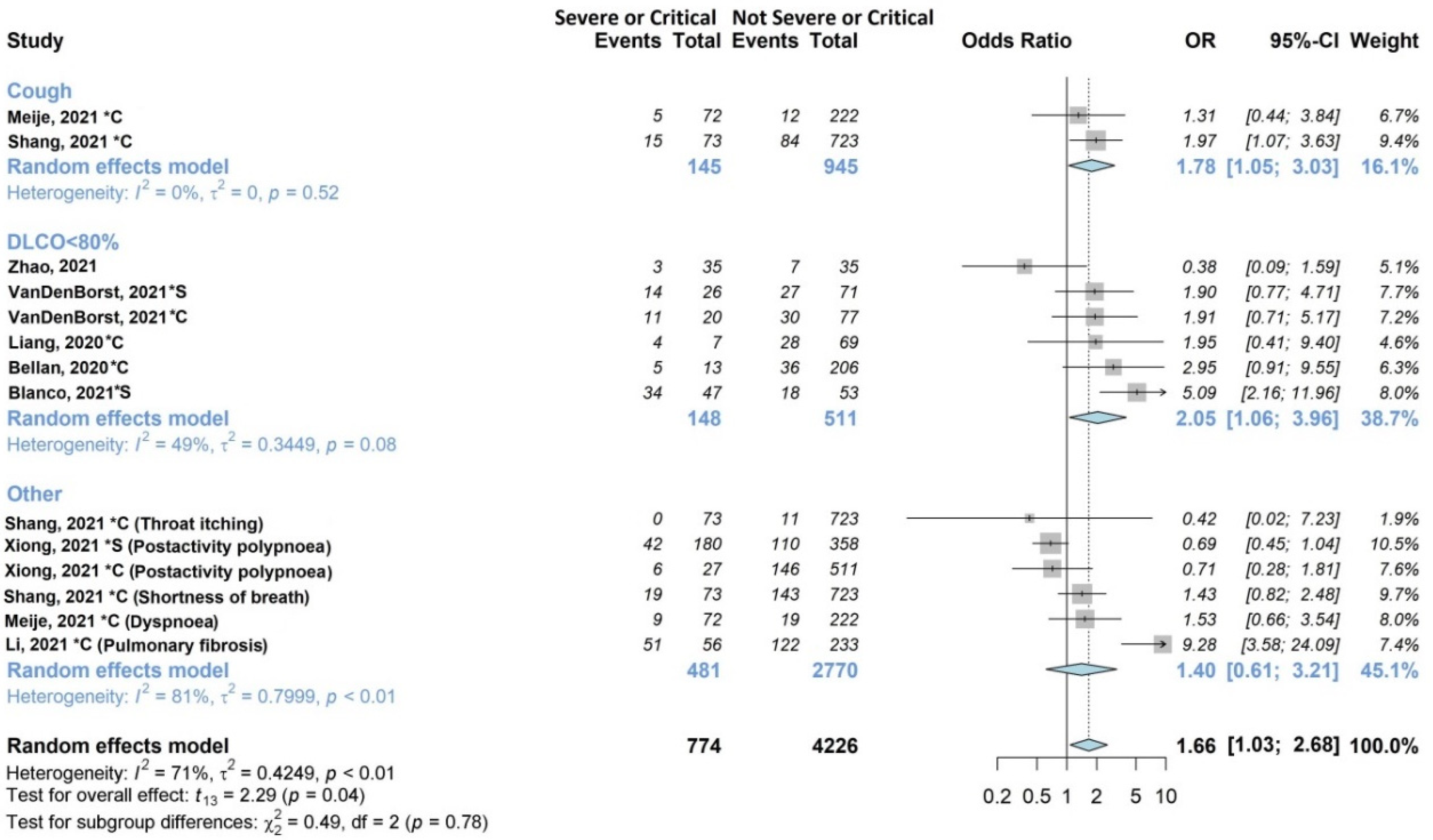
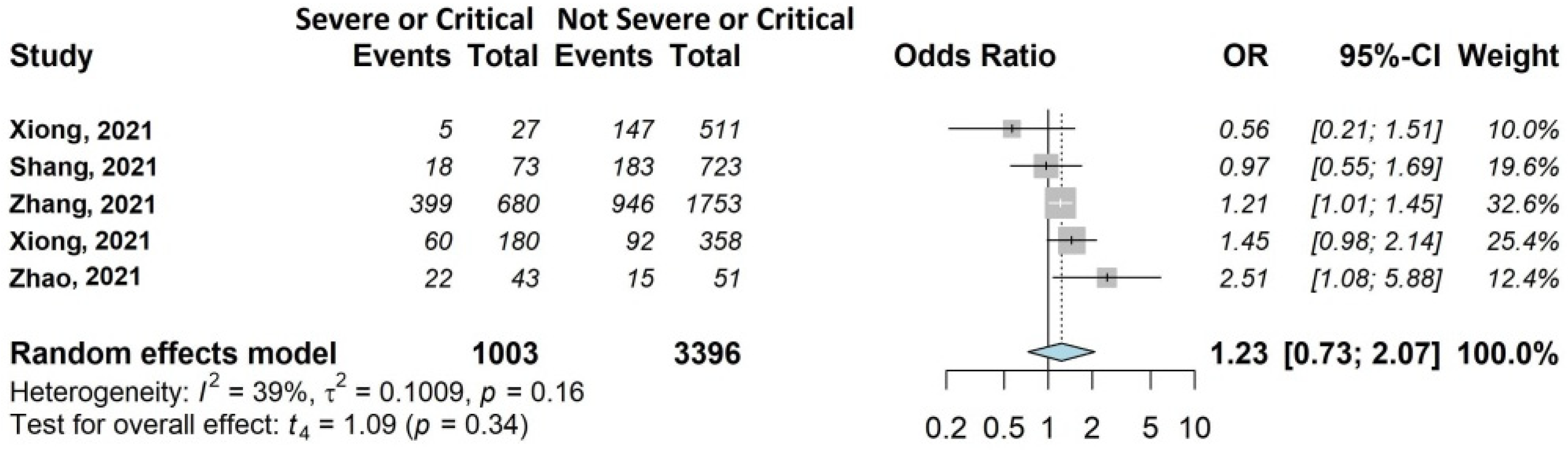
3.4.2. Respiratory Symptoms
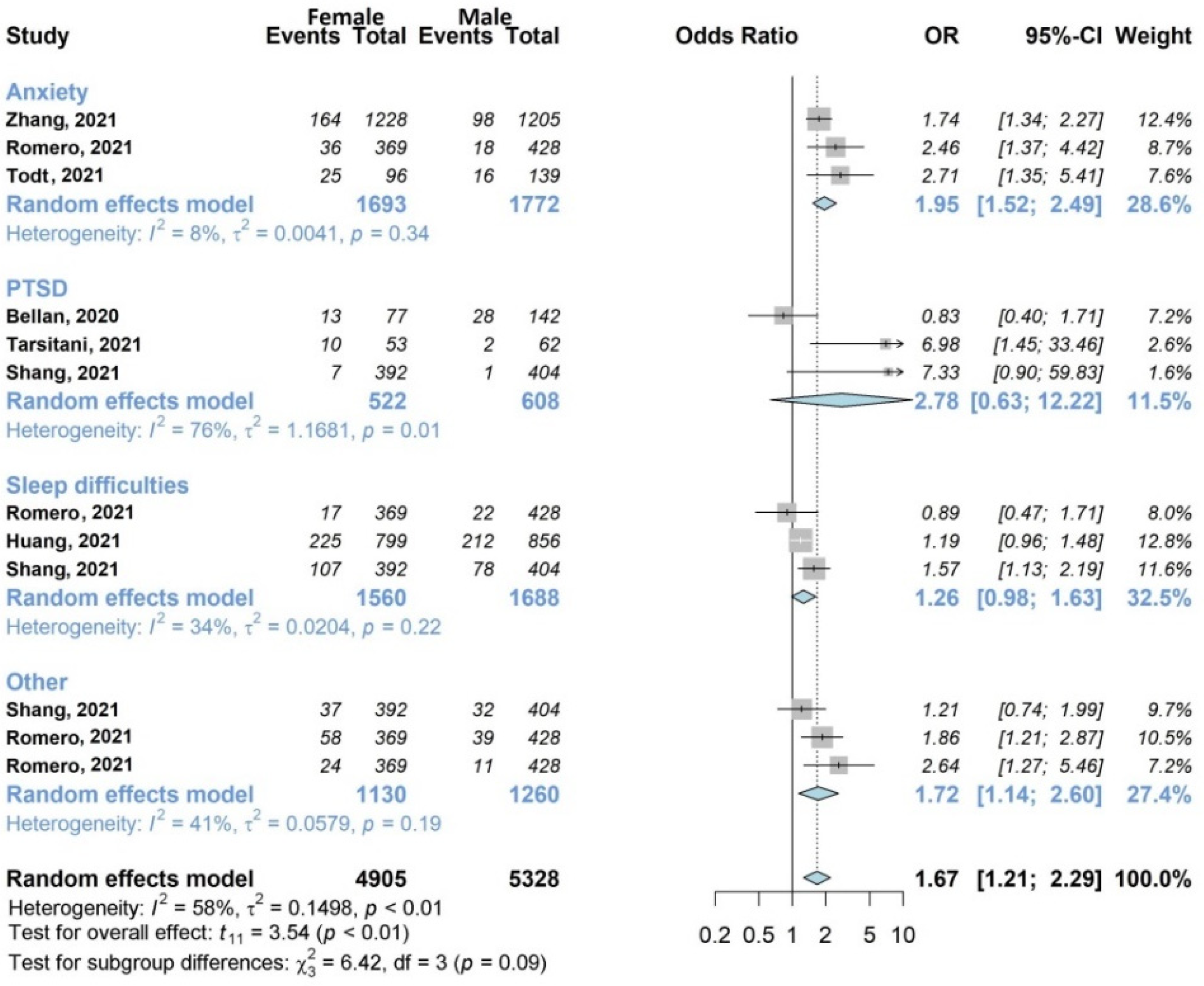
3.4.3. Mental Health Symptoms
3.4.4. Fatigue
4. Discussion
4.1. Main Findings
4.2. Strengths and Weaknesses of the Study
4.3. Implications for Future Research
4.4. Implications for Clinical Practice
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lopez-Leon, S.; Wegman-Ostrosky, T.; Perelman, C.; Sepulveda, R.; Rebolledo, P.A.; Cuapio, A.; Villapol, S. More than 50 long-term effects of COVID-19: A systematic review and meta-analysis. Sci. Rep. 2021, 11, 16144. [Google Scholar] [CrossRef] [PubMed]
- Groff, D.; Sun, A.; Ssentongo, A.E.; Ba, D.M.; Parsons, N.; Poudel, G.R.; Lekoubou, A.; Oh, J.S.; Ericson, J.E.; Ssentongo, P.; et al. Short-term and long-term rates of postacute sequelae of SARS-CoV-2 infection: A systematic review. JAMA Netw. Open 2021, 4, e2128568. [Google Scholar] [CrossRef] [PubMed]
- Sigfrid, L.; Cevik, M.; Jesudason, E.; Lim, W.S.; Rello, J.; Amuasi, J.; Bozza, F.; Palmieri, C.; Munblit, D.; Holter, J.C.; et al. What is the recovery rate and risk of long-term consequences following a diagnosis of COVID-19? A harmonised, global longitudinal observational study protocol. BMJ Open 2021, 11, e043887. [Google Scholar] [CrossRef]
- Crook, H.; Raza, S.; Nowell, J.; Young, M.; Edison, P. Long covid-mechanisms, risk factors, and management. BMJ 2021, 374, n1648, Erratum in BMJ 2021, 374, n1944. [Google Scholar] [CrossRef]
- Yong, S.J. Long COVID or post-COVID-19 syndrome: Putative pathophysiology, risk factors, and treatments. Infect. Dis. 2021, 53, 737–754. [Google Scholar] [CrossRef] [PubMed]
- Akbarialiabad, H.; Taghrir, M.H.; Abdollahi, A.; Ghahramani, N.; Kumar, M.; Paydar, S.; Razani, B.; Mwangi, J.; Asadi-Pooya, A.A.; Malekmakan, L.; et al. Long COVID, a comprehensive systematic scoping review. Infection 2021, 49, 1163–1186. [Google Scholar] [CrossRef]
- Iqbal, F.M.; Lam, K.; Sounderajah, V.; Clarke, J.M.; Ashrafian, H.; Darzi, A. Characteristics and predictors of acute and chronic post-COVID syndrome: A systematic review and meta-analysis. EClinicalMedicine 2021, 36, 100899. [Google Scholar] [CrossRef]
- Michelen, M.; Manoharan, L.; Elkheir, N.; Cheng, V.; Dagens, A.; Hastie, C.; O’Hara, M.; Suett, J.; Dahmash, D.; Bugaeva, P.; et al. Characterising long COVID: A living systematic review. BMJ Glob. Health 2021, 6, e005427. [Google Scholar] [CrossRef]
- Nalbandian, A.; Sehgal, K.; Gupta, A.; Madhavan, M.V.; McGroder, C.; Stevens, J.S.; Cook, J.R.; Nordvig, A.S.; Shalev, D.; Sehrawat, T.S.; et al. Post-acute COVID-19 syndrome. Nat. Med. 2021, 27, 601–615. [Google Scholar] [CrossRef]
- National Institute for Health; Care Excellence. COVID-19 Rapid Guideline: Managing the Long-Term Effects of COVID-19. NICE Guideline (NG188). 2020. Available online: https://www.nice.org.uk/guidance/ng188 (accessed on 29 November 2021).
- International Prospective Register of Systematic Reviews (PROSPERO). PROSPERO. Available online: https://www.crd.york.ac.uk/prospero/ (accessed on 29 November 2021).
- Page, M.J.; Moher, D.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. PRISMA 2020 explanation and elaboration: Updated guidance and exemplars for reporting systematic reviews. BMJ 2021, 372, n160. [Google Scholar] [CrossRef]
- Riley, R.D.; Moons, K.G.M.; Snell, K.I.E.; Ensor, J.; Hooft, L.; Altman, D.G.; Hayden, J.; Collins, G.S.; Debray, T.P.A. A guide to systematic review and meta-analysis of prognostic factor studies. BMJ 2019, 364, k4597. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rayyan–Intelligent Systematic Review. Available online: https://www.rayyan.ai/ (accessed on 29 November 2021).
- Hayden, J.A.; van Der Windt, D.A.; Cartwright, J.L.; Côté, P.; Bombardier, C. Assessing bias in studies of prognostic factors. Ann. Intern. Med. 2013, 158, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Dawes, A.J.; Maggard-Gibbons, M.; Maher, A.R.; Booth, M.J.; Miake-Lye, I.; Beroes, J.M.; Shekelle, P.G. Mental health conditions among patients seeking and undergoing bariatric surgery: A meta-analysis. JAMA 2016, 315, 150–163. [Google Scholar] [CrossRef] [PubMed]
- Lamberink, H.J.; Otte, W.M.; Geerts, A.T.; Pavlovic, M.; Ramos-Lizana, J.; Marson, A.G.; Overweg, J.; Sauma, L.; Specchio, L.M.; Tennison, M.; et al. Individualised prediction model of seizure recurrence and long-term outcomes after withdrawal of antiepileptic drugs in seizure-free patients: A systematic review and individual participant data meta-analysis. Lancet Neurol. 2017, 16, 523–531. [Google Scholar] [CrossRef] [Green Version]
- Hayden, J.A.; Wilson, M.N.; Riley, R.D.; Iles, R.; Pincus, T.; Ogilvie, R. Individual recovery expectations and prognosis of outcomes in non-specific low back pain: Prognostic factor review. Cochrane Database Syst. Rev. 2019, 25, CD011284. [Google Scholar] [CrossRef]
- Hayden, J.A.; Côté, P.; Steenstra, I.A.; Bombardier, C.; QUIPS-LBP Working Group. Identifying phases of investigation helps planning, appraising, and applying the results of explanatory prognosis studies. J. Clin. Epidemiol. 2008, 61, 552–560. [Google Scholar] [CrossRef]
- Veroniki, A.A.; Jackson, D.; Viechtbauer, W.; Bender, R.; Bowden, J.; Knapp, G.; Kuss, O.; Higgins, J.P.; Langan, D.; Salanti, G. Methods to estimate the between-study variance and its uncertainty in meta-analysis. Res. Synth. Methods 2016, 7, 55–79. [Google Scholar] [CrossRef] [Green Version]
- Axfors, C.; Janiaud, P.; Schmitt, A.M.; Van’t Hooft, J.; Smith, E.R.; Haber, N.A.; Abayomi, A.; Abduljalil, M.; Abdulrahman, A.; Acosta-Ampudia, Y.; et al. Association between convalescent plasma treatment and mortality in COVID-19: A collaborative systematic review and meta-analysis of randomized clinical trials. BMC Infect. Dis. 2021, 21, 1170. [Google Scholar] [CrossRef]
- IntHout, J.; Ioannidis, J.P.; Borm, G.F. The Hartung-Knapp-Sidik-Jonkman method for random effects meta-analysis is straightforward and considerably outperforms the standard DerSimonian-Laird method. BMC Med. Res. Methodol. 2014, 14, 25. [Google Scholar] [CrossRef] [Green Version]
- Brabaharan, S.; Veettil, S.K.; Kaiser, J.E.; Raja Rao, V.R.; Wattanayingcharoenchai, R.; Maharajan, M.; Insin, P.; Talungchit, P.; Anothaisintawee, T.; Thakkinstian, A.; et al. Association of hormonal contraceptive use with adverse health outcomes: An umbrella review of meta-analyses of randomized clinical trials and cohort studies. JAMA Netw. Open 2022, 5, e2143730. [Google Scholar] [CrossRef]
- Partlett, C.; Riley, R.D. Random effects meta-analysis: Coverage performance of 95% confidence and prediction intervals following REML estimation. Stat. Med. 2017, 36, 301–317. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwarzer, G.; Carpenter, J.R.; Rücker, G. Metaanalysis with R; Springer: Berlin/Heidelberg, Germany, 2015; Volume 4784, ISBN 978-3-319-21415-3. [Google Scholar]
- Bellan, M.; Soddu, D.; Balbo, P.E.; Baricich, A.; Zeppegno, P.; Avanzi, G.C.; Baldon, G.; Bartolomei, G.; Battaglia, M.; Battistini, S.; et al. Respiratory and psychophysical sequelae among patients with COVID-19 four months after hospital discharge. JAMA Netw. Open 2021, 4, e2036142. [Google Scholar] [CrossRef]
- Blanco, J.R.; Cobos-Ceballos, M.J.; Navarro, F.; Sanjoaquin, I.; Arnaiz de Las Revillas, F.; Bernal, E.; Buzon-Martin, L.; Viribay, M.; Romero, L.; Espejo-Perez, S.; et al. Pulmonary long-term consequences of COVID-19 infections after hospital discharge. Clin. Microbiol. Infect. 2021, 27, 892–896. [Google Scholar] [CrossRef]
- Eloy, P.; Tardivon, C.; Martin-Blondel, G.; Isnard, M.; Turnier, P.L.; Marechal, M.L.; Cabié, A.; Launay, O.; Tattevin, P.; Senneville, E.; et al. French COVID cohort study and investigators groups. Severity of self-reported symptoms and psychological burden 6-months after hospital admission for COVID-19: A prospective cohort study. Int. J. Infect. Dis. 2021, 112, 247–253. [Google Scholar] [CrossRef]
- Fernández-de-Las-Peñas, C.; Palacios-Ceña, D.; Gómez-Mayordomo, V.; Palacios-Ceña, M.; Rodríguez-Jiménez, J.; de-la-Llave-Rincón, A.I.; Velasco-Arribas, M.; Fuensalida-Novo, S.; Ambite-Quesada, S.; Guijarro, C.; et al. Fatigue and dyspnoea as main persistent post-COVID-19 symptoms in previously hospitalized patients: Related functional limitations and disability. Respiration 2021, 101, 132–141. [Google Scholar] [CrossRef]
- García-Abellán, J.; Padilla, S.; Fernández-González, M.; García, J.A.; Agulló, V.; Andreo, M.; Ruiz, S.; Galiana, A.; Gutiérrez, F.; Masiá, M. Antibody response to SARS-CoV-2 is associated with long-term clinical outcome in patients with COVID-19: A longitudinal study. J. Clin. Immunol. 2021, 41, 1490–1501. [Google Scholar] [CrossRef]
- Ghosn, J.; Piroth, L.; Epaulard, O.; Le Turnier, P.; Mentré, F.; Bachelet, D.; Laouénan, C.; French COVID Cohort Study and Investigators Groups. Persistent COVID-19 symptoms are highly prevalent 6 months after hospitalization: Results from a large prospective cohort. Clin. Microbiol. Infect. 2021, 27, 1041.e1–1041.e4. [Google Scholar] [CrossRef]
- Huang, C.; Huang, L.; Wang, Y.; Li, X.; Ren, L.; Gu, X.; Kang, L.; Guo, L.; Liu, M.; Zhou, X.; et al. 6-month consequences of COVID-19 in patients discharged from hospital: A cohort study. Lancet 2021, 397, 220–232. [Google Scholar] [CrossRef]
- Li, X.; Shen, C.; Wang, L.; Majumder, S.; Zhang, D.; Deen, M.J.; Li, Y.; Qing, L.; Zhang, Y.; Chen, C.; et al. Pulmonary fibrosis and its related factors in discharged patients with new corona virus pneumonia: A cohort study. Respir. Res. 2021, 22, 203. [Google Scholar] [CrossRef]
- Liang, L.; Yang, B.; Jiang, N.; Fu, W.; He, X.; Zhou, Y.; Ma, W.L.; Wang, X. Three-month follow-up study of survivors of coronavirus disease 2019 after discharge. J. Korean Med. Sci. 2020, 35, e418. [Google Scholar] [CrossRef] [PubMed]
- Meije, Y.; Duarte-Borges, A.; Sanz, X.; Clemente, M.; Ribera, A.; Ortega, L.; González-Pérez, R.; Cid, R.; Pareja, J.; Cantero, I.; et al. Hospital de Barcelona COVID-19 team. Long-term outcomes of patients following hospitalization for coronavirus disease 2019: A prospective observational study. Clin. Microbiol. Infect. 2021, 27, 1151–1157. [Google Scholar] [CrossRef] [PubMed]
- Munblit, D.; Bobkova, P.; Spiridonova, E.; Shikhaleva, A.; Gamirova, A.; Blyuss, O.; Nekliudov, N.; Bugaeva, P.; Andreeva, M.; DunnGalvin, A.; et al. Incidence and risk factors for persistent symptoms in adults previously hospitalized for COVID-19. Clin. Exp. Allergy 2021, 51, 1107–1120. [Google Scholar] [CrossRef]
- Romero-Duarte, Á.; Rivera-Izquierdo, M.; Guerrero-Fernández de Alba, I.; Pérez-Contreras, M.; Fernández-Martínez, N.F.; Ruiz-Montero, R.; Serrano-Ortiz, Á.; González-Serna, R.O.; Salcedo-Leal, I.; Jiménez-Mejías, E.; et al. Sequelae, persistent symptomatology and outcomes after COVID-19 hospitalization: The ANCOHVID multicentre 6-month follow-up study. BMC Med. 2021, 19, 129. [Google Scholar] [CrossRef] [PubMed]
- Shang, Y.F.; Liu, T.; Yu, J.N.; Xu, X.R.; Zahid, K.R.; Wei, Y.C.; Wang, X.H.; Zhou, F.L. Half-year follow-up of patients recovering from severe COVID-19: Analysis of symptoms and their risk factors. J. Intern. Med. 2021, 290, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Tarsitani, L.; Vassalini, P.; Koukopoulos, A.; Borrazzo, C.; Alessi, F.; Di Nicolantonio, C.; Serra, R.; Alessandri, F.; Ceccarelli, G.; Mastroianni, C.M.; et al. Post-traumatic stress disorder among COVID-19 survivors at 3-month follow-up after hospital discharge. J. Gen. Intern. Med. 2021, 36, 1702–1707. [Google Scholar] [CrossRef]
- Todt, B.C.; Szlejf, C.; Duim, E.; Linhares, A.O.M.; Kogiso, D.; Varela, G.; Campos, B.A.; Baghelli Fonseca, C.M.; Polesso, L.E.; Bordon, I.N.S.; et al. Clinical outcomes and quality of life of COVID-19 survivors: A follow-up of 3 months post hospital discharge. Respir. Med. 2021, 184, 106453. [Google Scholar] [CrossRef]
- van den Borst, B.; Peters, J.B.; Brink, M.; Schoon, Y.; Bleeker-Rovers, C.P.; Schers, H.; van Hees, H.W.H.; van Helvoort, H.; van den Boogaard, M.; van der Hoeven, H.; et al. Comprehensive health assessment 3 months after recovery from acute coronavirus disease 2019 (COVID-19). Clin. Infect. Dis. 2021, 73, e1089. [Google Scholar] [CrossRef]
- Wu, X.; Liu, X.; Zhou, Y.; Yu, H.; Li, R.; Zhan, Q.; Ni, F.; Fang, S.; Lu, Y.; Ding, X.; et al. 3-month, 6-month, 9-month, and 12-month respiratory outcomes in patients following COVID-19-related hospitalization: A prospective study. Lancet Respir. Med. 2021, 9, 747–754. [Google Scholar] [CrossRef]
- Xiong, Q.; Xu, M.; Li, J.; Liu, Y.; Zhang, J.; Xu, Y.; Dong, W. Clinical sequelae of COVID-19 survivors in Wuhan, China: A single-center longitudinal study. Clin. Microbiol. Infect. 2021, 27, 89–95. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, F.; Shen, Y.; Zhang, X.; Cen, Y.; Wang, B.; Zhao, S.; Zhou, Y.; Hu, B.; Wang, M.; et al. Symptoms and health outcomes among survivors of COVID-19 infection 1 year after discharge from hospitals in Wuhan, China. JAMA Netw. Open 2021, 4, e2127403. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Yang, C.; An, X.; Xiong, Y.; Shang, Y.; He, J.; Qiu, Y.; Zhang, N.; Huang, L.; Jia, J.; et al. Follow-up study on COVID-19 survivors one year after discharge from hospital. Int. J. Infect. Dis. 2021, 112, 173–182. [Google Scholar] [CrossRef]
- Barek, M.A.; Aziz, M.A.; Islam, M.S. Impact of age, sex, comorbidities and clinical symptoms on the severity of COVID-19 cases: A meta-analysis with 55 studies and 10014 cases. Heliyon 2020, 6, e05684. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Peng, F.; Xu, B.; Zhao, J.; Liu, H.; Peng, J.; Li, Q.; Jiang, C.; Zhou, Y.; Liu, S.; et al. Risk factors of critical & mortal COVID-19 cases: A systematic literature review and meta-analysis. J. Infect. 2020, 81, e16–e25. [Google Scholar] [CrossRef]
- Ahmed, A.; Ali, A.; Hasan, S. Comparison of epidemiological variations in COVID-19 patients inside and outside of china-a meta-analysis. Front. Public Health 2020, 8, 193. [Google Scholar] [CrossRef] [PubMed]
- Bai, F.; Tomasoni, D.; Falcinella, C.; Barbanotti, D.; Castoldi, R.; Mulè, G.; Augello, M.; Mondatore, D.; Allegrini, M.; Cona, A.; et al. Female gender is associated with “long COVID” syndrome: A prospective cohort study. Clin. Microbiol. Infect. 2021, in press. [Google Scholar] [CrossRef] [PubMed]
- Di Toro, A.; Bozzani, A.; Tavazzi, G.; Urtis, M.; Giuliani, L.; Pizzoccheri, R.; Aliberti, F.; Fergnani, V.; Arbustini, E. Long COVID: Long-term effects? Eur. Heart J. 2021, 23 (Suppl. E), E1–E5. [Google Scholar] [CrossRef]
- Lipworth, W.; Gentgall, M.; Kerridge, I.; Stewart, C. Science at warp speed: Medical research, publication, and translation during the COVID-19 pandemic. J. Bioethical Inq. 2020, 17, 555–561. [Google Scholar] [CrossRef]
- Sudre, C.H.; Murray, B.; Varsavsky, T.; Graham, M.S.; Penfold, R.S.; Bowyer, R.C.; Pujol, J.C.; Klaser, K.; Antonelli, M.; Canas, L.S.; et al. Attributes and predictors of long COVID. Nat. Med. 2021, 27, 626–631. [Google Scholar] [CrossRef]
- Callard, F.; Perego, E. How and why patients made long Covid. Soc. Sci. Med. 2021, 268, 113426. [Google Scholar] [CrossRef]



| First Author | Study Design | Country | Covid Patients Included, No | Age (Years) | Sex (% Female) | Follow-Up Length | Follow-Up Mode |
|---|---|---|---|---|---|---|---|
| Bellan et al. [27] | Ambidirectional study, single center | Italy | 238 | Median (IQR): 61 (50–71) | 96 (40.3%) | 3–4 months after discharge | Telephone interview and outpatient visit |
| Blanco et al. [28] | Longitudinal, prospective study, multicenter | Spain | 100 | >50 years: 69 (69%) | 36 (36%) | Median (IQR): 104 (89.25–126.75) after onset | Outpatient visit |
| Eloy et al. [29] | Longitudinal, prospective study, multicenter | France | 324 | Median (IQR): 61 (52–69) | 119 (37%) | 6 months after discharge | Outpatient visit |
| Fernández-de-Las-Peñas et al. [30] | Ambidirectional study, multicenter | Spain | 1142 | Mean (SD): 61 + 17 | 548 (48%) | 7 months after discharge | Telephone interview |
| García-Abellán et al. [31] | Longitudinal, prospective study, single center | Spain | 146 | Median (IQR): 64 (54–76) | 58 (40%) | 6 months after discharge | Outpatient visit |
| Ghosn et al. [32] | Longitudinal, prospective study, multicenter | France | 1137 | Median (IQR): 61 (51–71) | 424 (37%) | 6 months after admission | Outpatient visit |
| Huang et al. [33] | Ambidirectional study, single center | China | 1733 | Median (IQR): 57 (47–65) | 836 (48%) | Median (IQR): 186 (175–199) days after onset | Outpatient visit |
| Li et al. [34] | Longitudinal, prospective study, single center | China | 289 | Not available | Not available | From 90 to 150 days after onset | Outpatient visit |
| Liang et al. [35] | Ambidirectional study, single center | China | 76 | Mean (SD): 41.3 ± 13.8 | 55 (72%) | 3 months after discharge | Outpatient visit |
| Meije et al. [36] | Ambidirectional study, single center | Spain | 302 | Mean (SD): 68.8 (12.7) | 131 (43%) | 7 months after discharge | Telephone interview |
| Munblit et al. [37] | Ambidirectional study, multicenter | Russia | 2649 | Median (IQR): 56 (46–66) | 1353 (51.1%) | 6–8 months after discharge | Telephone interview |
| Romero-Duarte et al. [38] | Ambidirectional study, multicenter | Spain | 797 | Mean (SD): 63 (14.4) | 369 (46.3%) | 6 months after discharge | Outpatient visit and Telephone interview |
| Shang et al. [39] | Ambidirectional study, multicenter | China | 796 | Median (IQR): 62 (51–69) | 392 (49.2%) | 6 months after discharge | Telephone interview |
| Tarsitani et al. [40] | Ambidirectional study, single center | Italy | 115 | Median (IQR): 57 (48–66) | 67 (58%) | 3 months after discharge | Phone interview |
| Todt et al. [41] | Longitudinal, prospective study, single center | Brazil | 251 | Mean (SD): 53.6 (+14.9) | 101 (40.2%) | 3 months after discharge | Telephone interview |
| van den Borst et al. [42] | Ambidirectional study, single center | The Netherlands | 97 | Not available | 31 (32%) | Mean (SD): 13 (2.2) weeks after onset | Outpatient visit |
| Wu et al. [43] | Longitudinal, prospective study, single center | China | 83 | Median (IQR): 60 (52–66) | 36 (43%) | 3, 6, 9, and 12 months after discharge | Outpatient visit |
| Xiong et al. [44] | Longitudinal, prospective, controlled study, single center | China | 538 | Median (IQR): 52 (41–62) | 293 (54.5%) | Median (IQR): 97 (95–102) days after discharge | Telephone interview |
| Zhang et al. [45] | Ambidirectional study, multicenter | China | 2433 | Median (IQR): 60 (49–68) | 1228 (50.5%) | 1 year after discharge | Telephone interview |
| Zhao et al. [46] | Ambidirectional study, multicenter | China | 94 | Mean 48.11 | 40 (42.55%) | 1 year after discharge | Outpatient visit |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maglietta, G.; Diodati, F.; Puntoni, M.; Lazzarelli, S.; Marcomini, B.; Patrizi, L.; Caminiti, C. Prognostic Factors for Post-COVID-19 Syndrome: A Systematic Review and Meta-Analysis. J. Clin. Med. 2022, 11, 1541. https://doi.org/10.3390/jcm11061541
Maglietta G, Diodati F, Puntoni M, Lazzarelli S, Marcomini B, Patrizi L, Caminiti C. Prognostic Factors for Post-COVID-19 Syndrome: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine. 2022; 11(6):1541. https://doi.org/10.3390/jcm11061541
Chicago/Turabian StyleMaglietta, Giuseppe, Francesca Diodati, Matteo Puntoni, Silvia Lazzarelli, Barbara Marcomini, Laura Patrizi, and Caterina Caminiti. 2022. "Prognostic Factors for Post-COVID-19 Syndrome: A Systematic Review and Meta-Analysis" Journal of Clinical Medicine 11, no. 6: 1541. https://doi.org/10.3390/jcm11061541
APA StyleMaglietta, G., Diodati, F., Puntoni, M., Lazzarelli, S., Marcomini, B., Patrizi, L., & Caminiti, C. (2022). Prognostic Factors for Post-COVID-19 Syndrome: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine, 11(6), 1541. https://doi.org/10.3390/jcm11061541






