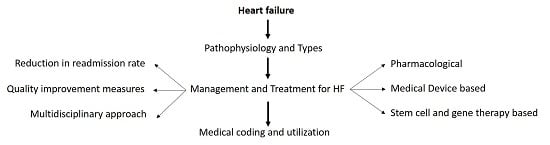
Heart Failure: Diagnosis, Management and Utilization
Abstract
:1. Introduction
1.1. Background
1.2. Classification of HFs
- Class I:
- HF does not cause limitations to physical activity; ordinary physical activity does not cause symptoms.
- Class II:
- HF causes slight limitations to physical activity; the patients are comfortable at rest, but ordinary physical activity results in HF symptoms.
- Class III:
- HF causes marked limitations of physical activity; the patients are comfortable at rest, but less than ordinary activity causes symptoms of HF.
- Class IV:
- HF patients are unable to carry on any physical activity without HF symptoms or have symptoms when at rest.
- Stage A:
- High risk of heart failure, but no structural heart disease or symptoms of heart failure;
- Stage B:
- Structural heart disease, but no symptoms of heart failure;
- Stage C:
- Structural heart disease and symptoms of heart failure;
- Stage D:
- Refractory heart failure requiring specialized interventions.
2. Clinical Presentation of HF
3. Diagnosis of HF
4. Predictors of Poor Outcome and High Mortality Rate
- Serum urea >15 mmol/L
- Systolic blood pressure <115 mmHg
- Serum creatinine >2.72 mg/dL (or 240 μmol/L)
- N-terminal pro-brain natriuretic peptide (NT-pro-BNP) >986 pg/mL
- Left ventricular ejection fraction <45%
- High NYHA functional class
- Reduced left ventricular ejection fraction
- Third heart sound
- Increased pulmonary artery capillary wedge pressure
- Reduced cardiac index
- Diabetes mellitus
- Reduced sodium concentration
- Raised plasma catecholamine and natriuretic peptide concentrations
5. Management of Heart Failure
5.1. In-Patient Management of HF
- Monitor oxygen, whether PaO2 < 60% or SaO2 < 90%.
- Provide noninvasive positive pressure ventilation (NIPPV) in the few cases with respiratory distress for respiratory support to avoid subsequent intubation.
- Use the following pharmacological agents depending on the precipitating factors and symptoms/signs for congestion:
- Diuretics (thiazides, loop diuretics and potassium sparing) (to reduce the edema by the reduction of blood volume and venous pressure) and salt restriction (to reduce fluid retention) in patients with current or previous heart failure symptoms and reduced left ventricular ejection fraction (LVEF) for symptomatic relief.
- Angiotensin-converting enzyme inhibitors (ACEIs) or angiotensin receptor blockers (ARBs) for neuro-hormonal modification, vasodilatation and improvement in LVEF (substitute them with hydralazine and/or nitrates in patients unresponsive to ACEIs and ARBs).
- Beta-adrenergic blockers for neuro-hormonal modification, improvement in symptoms and LVEF, survival benefit, arrhythmia prevention and control of ventricular rate.
- Aldosterone antagonists, as an adjunct to other drugs for additive diuresis, heart failure symptom control, improved heart rate variability, decreased ventricular arrhythmias, reduction in cardiac workload, improved LVEF and an increase in survival.
- Digoxin, which can lead to a small increase in cardiac output, improvement in heart failure symptoms and a decreased rate of heart failure hospitalizations.
- Anticoagulants, if applicable, to decrease the risk of thromboembolism.
- Inotropic agents to restore organ perfusion and reduce congestion in patients with heart failure with reduced ejection fraction, so as to increase in cardiac output and reduce neuro-humoral activation.
- Some other agents have been described under clinical trial (Table 2).
Discharge Criteria for HF Patients
- Exacerbating factors have been addressed and are under control
- Volume status has been optimized
- Diuretic therapy has been successfully transitioned to oral medication, with discontinuation of IV vasodilator and inotropic therapy if required for at least 24 h
- Oral therapy for chronic heart failure (HF), including angiotensin convertase enzyme inhibitors (ACEIs) and beta blockers (for patients with reduced LVEF), has been established with stable clinical status
- Patient and family education completed, including clear discharge instructions
- Left ventricular ejection fraction (LVEF) documented: echocardiography is the gold standard
- Smoking cessation (if applicable) counseling initiated
- Follow-up clinic visit scheduled within three days of discharge, usually for 7–10 days
- Oral medication regimen for heart failure has been established for 24 h
- No intravenous vasodilator or inotropic agent for at least 24 h
- Ambulation before discharge to evaluate the beneficial effect of therapy and restoration of functional capacity
- Plans for post-discharge management to prevent readmission (scale present in home, visiting nurse or telephone follow-up generally no longer than three days after discharge)
- Appropriate referral to a specialist for disease management of precipitant cause(s) if applicable
- Medication reconciliation, written plans for dietary sodium restriction and recommended activity level
- Follow-up by phone or clinic visit soon after discharge to reassess volume status
- Medication and dietary adherence
- Alcohol moderation and cessation of smoking
- Monitoring of body weight, electrolytes and renal function
- Consideration of referral for formal disease management
5.2. Out-Patient Management of HF [9,10]
- Comprehensive education and counseling individualized to the patient’s disease and socio-economic and educational level
- Education/promotion of self-care, including self-adjustment of diuretic therapy in appropriate patients with the help of a family member/caregiver
- Early attention to signs and symptoms of fluid overload
- Emphasis on behavioral strategies to increase adherence
- Optimization of medical therapy
- Vigilant follow-up after hospital discharge or after periods of instability
- Increased access to providers or healthcare/social services
- Assistance with social and financial concerns
6. Readmission
- (1)
- Medication noncompliance (21%–66%)
- (2)
- Smoking (60%)
- (3)
- Sodium- and fluid-restricted diet noncompliance (30%–44%)
- (4)
- Failure of documentation of discharge information and patient education (41%)
- (5)
- Co-morbidities (21%–34%) (hypertension, diabetes mellitus, metabolic syndrome and atherosclerotic disease, anemia, depression)
- (1)
- Medical predictors: severity of orthopnea, renal dysfunction, hemodynamic instability, high levels of Pro-BNP, hyponatremia and presence of co-morbidities.
- (2)
- Demographic predictors: male gender, advanced age, previous admission within six months, low median income, lack of psychosocial support, medication compliance and compliance to follow-up visits.
- Multidisciplinary HF clinics/centers (reduces all-cause readmission rates by 50%)
- Visiting nurse services and nurse specialist (reduces all-cause readmission rates by 37%)
- Physician-directed heart failure transitional care program (reduces all-cause readmission rates by 21%)
- Home tele-monitoring or structured phone calls (reduces all-cause readmission rates by 20%)
- Follow-up one-week post discharge (reduces all-cause readmission rates by 10%–15%)
- Transition care intervention home program (reduces all-cause readmission rates by 6%–12%)
7. Quality Improvement Strategies for HF
- Patient education:
- patient education about HF and strategies for its treatment.
- dietary counseling about sodium (2–3 gm/day; <2 gm/day may be considered in moderate to severe heart failure) and fluid restriction <2 L/day is considered when the fluid retention persists and when severe hyponatremia (serum Na <130 mEq/L) is present.
- healthy lifestyle changes (high fiber diet with vegetables; regular exercise in a tolerable amount under monitoring of a cardiac rehabilitation program; consuming alcohol in moderation and no smoking); especially, recent studies have advocated the importance of exercise training to HF patients via improvement in the skeletal muscle O2 delivery, while simultaneously correcting mitochondrial and contractile efficiency. The localized muscle training has been shown to improve convective and diffusive O2 transport in HF and, hence, is useful for patients with minimal lung reserve capacity; several variables, such as exercise type, duration, frequency, intensity, etc., need to be taken into consideration to best benefit from such training [56,65,66].
- efforts to improve patients’ compliance with medical regimens and interventions, such as phone calls, reminders and home nurse, to help patients remember to take the medications.
- understand the alarming signs and symptoms, such shortness of breath, excessive fatigue, swelling of feet/ankle, etc. [11].
- Arranging follow-up care: This includes assistance in scheduling the first follow-up appointment post-hospitalization along with re-enforcement of the importance of other follow-up visits. It also includes documentation of the date, time and location of the follow-up visit on the discharge plan, as well as sending reminders for subsequent appointments. One recent study has shown that it is possible to predict the readmission based on the response of the patients on the automated follow-up questionnaire [67].
- Home tele-monitoring: This is a unique approach where the transmission of clinical parameters and symptoms of patients with HF at home to their healthcare provider, such as weight, blood pressure, heart rate, oxygen saturation, along with patients queries and questions regarding medications and symptoms and signs is conducted, thereby titrating the therapy based on the symptoms and signs. A few studies have shown that home tele-monitoring reduces mortality and hospitalizations, while in other studies, home tele-monitoring was found to be equivalent to telephone calls by a nurse [57].
- Transition home program: This helps patients to have a safe transition to home or to another healthcare setting, such as a skilled nursing facility, and includes thorough patient and caregiver education, enhanced individualized assessment of post-discharge needs, patient-centered communication with caregivers and a standardized process for further management of HF along with follow-up visits with healthcare professionals [59,60,68].
- Nurse assurance program: This program facilitates home service to follow-up on the patients with HF [58].
8. Standard and Novel Therapies for HF
8.1. Landmark Clinical Trials in the Management of HF
8.2. Role of Cardiac Rejuvenation Therapy in the Management of HF
9. Utilization and Medical Coding
10. Conclusions
Author Contributions
Conflicts of Interest
References
- Dassanayaka, S.; Jones, S.P. Recent Developments in Heart Failure. Circ. Res. 2015, 117, e58–e63. [Google Scholar] [CrossRef] [PubMed]
- Ohtani, T.; Mohammed, S.F.; Yamamoto, K. Diastolic stiffness as assessed by diastolic wall strain is associated with adverse remodeling and poor outcomes in heart failure with preserved ejection fraction. Eur. Heart J. 2012, 33, 1742–1749. [Google Scholar] [CrossRef] [PubMed]
- Zamani, P.; Rawat, D.; Shiva-Kumar, P.; Geraci, S.; Bhuva, R.; Konda, P.; Doulias, P.T.; Ischiropoulos, H.; Townsend, R.R.; Margulies, K.B.; et al. Effect of inorganic nitrate on exercise capacity in heart failure with preserved ejection fraction. Circulation 2015, 131, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Glean, A.A.; Ferguson, S.K.; Holdsworth, C.T.; Colburn, T.D.; Wright, J.L.; Fees, A.J.; Hageman, K.S.; Poole, D.C.; Musch, T.I. Effects of nitrite infusion on skeletal muscle vascular control during exercise in rats with chronic heart failure. Am. J. Physiol. Heart Circ. Physiol. 2015, 309, H1354–H1360. [Google Scholar] [CrossRef] [PubMed]
- Maeder, M.T.; Thompson, B.R.; Brunner-La Rocca, H.-P.; Kaye, D.M. Hemodynamic basis of exercise limitation in patients with heart failure and normal ejection fraction. J. Am. Coll. Cardiol. 2010, 56, 855–863. [Google Scholar] [CrossRef] [PubMed]
- Bhella, P.S.; Prasad, A.; Heinicke, K.; Hastings, J.L.; Arbab-Zadeh, A.; Adams-Huet, B.; Pacini, E.L.; Shibata, S.; Palmer, M.D.; Newcomer, B.R.; et al. Abnormal haemodynamic response to exercise in heart failure with preserved ejection fraction. Eur. J. Heart Fail. 2011, 13, 1296–1304. [Google Scholar] [CrossRef] [PubMed]
- Angadi, S.S.; Mookadam, F.; Lee, C.D.; Tucker, W.J.; Haykowsky, M.J.; Gaesser, G.A. High-intensity interval training vs. moderate-intensity continuous exercise training in heart failure with preserved ejection fraction: A pilot study. J. Appl. Physiol. (1985) 2015, 119, 753–758. [Google Scholar] [CrossRef] [PubMed]
- Paulus, W.J.; Tschöpe, C. A novel paradigm for heart failure with preserved ejection fraction: Comorbidities drive myocardial dysfunction and remodeling through coronary microvascular endothelial inflammation. J. Am. Coll. Cardiol. 2013, 62, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Yancy, C.W.; Jessup, M.; Bozkurt, B.; Butler, J.; Casey, D.E., Jr.; Drazner, M.H.; Fonarow, G.C.; Geraci, S.A.; Horwich, T.; Januzzi, J.L.; et al. 2013 ACCF/AHA guideline for the management of heart failure: A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J. Am. Coll. Cardiol. 2013, 62, e147–e239. [Google Scholar] [CrossRef] [PubMed]
- Watson, R.D.; Gibbs, C.R.; Lip, G.Y. ABC of heart failure. Clinical features and complications. BMJ Br. Med. J. 2000, 320, 236–239. [Google Scholar] [CrossRef]
- Lindenfeld, J.; Albert, N.M.; Boehmer, J.P.; Collins, S.P.; Ezekowitz, J.A.; Givertz, M.M.; Katz, S.D.; Klapholz, M.; Moser, D.K.; Rogers, J.G.; et al. HFSA 2010 Comprehensive Heart Failure Practice Guideline. J. Card. Fail. 2010, 16, e1–e194. [Google Scholar] [PubMed]
- Marti, C.N.; Georgiopoulou, V.V.; Kalogeropoulos, A.P. Acute heart failure: Patient characteristics and pathophysiology. Curr. Heart Fail. Rep. 2013, 10, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Poole, D.C.; Hirai, D.M.; Copp, S.W.; Musch, T.I. Muscle oxygen transport and utilization in heart failure: Implications for exercise (in)tolerance. Am. J. Physiol. Heart Circ. Physiol. 2012, 302, H1050–H1063. [Google Scholar] [CrossRef] [PubMed]
- Heidenreich, P.A.; Albert, N.M.; Allen, L.A.; Bluemke, D.A.; Butler, J.; Fonarow, G.C.; Ikonomidis, J.S.; Khavjou, O.; Konstam, M.A.; Maddox, T.M.; et al. Forecasting the impact of heart failure in the United States: A policy statement from the American Heart Association. Circ. Heart Fail. 2013, 3, 606–619. [Google Scholar] [CrossRef] [PubMed]
- Dunlay, S.M.; Shah, N.D.; Shi, Q.; Morlan, B.; VanHouten, H.; Long, K.H.; Roger, V.L. Lifetime costs of medical care after heart failure diagnosis. Circ. Cardiovasc. Qual. Outcomes 2011, 4, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Askoxylakis, V.; Thieke, C.; Pleger, S.T. Long-term survival of cancer patients compared to heart failure and stroke: A systematic review. BMC Cancer 2010, 10, 105. [Google Scholar] [CrossRef] [PubMed]
- Krumholz, H.M.; Lin, Z.; Keenan, P.S.; Chen, J.; Ross, J.S.; Drye, E.E.; Bernheim, S.M.; Wang, Y.; Bradley, E.H.; Han, L.F.; et al. Normand SL. Relationship between hospital readmission and mortality rates for patients hospitalized with acute myocardial infarction, heart failure, or pneumonia. JAMA J. Am. Med. Assoc. 2013, 309, 587–589. [Google Scholar] [CrossRef] [PubMed]
- Ni, H.; Xu, J. Recent trends in heart failure-related mortality: United States, 2000–2014. In NCHS Data Brief, 231; National Center for Health Statistics: Hyattsville, MD, USA, 2015. Available online: http://www.cdc.gov/nchs/data/databriefs/db231.htm (accessed on 4 January 2016). [Google Scholar]
- Roger, V.L. Epidemiology of heart failure. Circ. Res. 2013, 113, 646–659. [Google Scholar] [CrossRef] [PubMed]
- Anker, S.D.; von Haehling, S. Inflammatory mediators in chronic heart failure: An overview. Heart 2004, 90, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, U.; Frantz, S. How can we cure a heart “in flame”? A translational view on inflammation in heart failure. Basic Res. Cardiol. 2013, 108, 356. [Google Scholar] [CrossRef] [PubMed]
- Oikonomou, E.; Tousoulis, D.; Siasos, G.; Zaromitidou, M.; Papavassiliou, A.G.; Stefanadis, C. The role of inflammation in heart failure: New therapeutic approaches. Hell. J. Cardiol. 2011, 52, 30–40. [Google Scholar]
- Tang, W.H.; Wang, Z.; Fan, Y.; Levison, B.; Hazen, J.E.; Donahue, L.M.; Wu, Y.; Hazen, S.L. Prognostic value of elevated levels of intestinal microbe-generated metabolite trimethylamine-N-oxide in patients with heart failure: Refining the gut hypothesis. J. Am. Coll. Cardiol. 2014, 64, 1908–1914. [Google Scholar] [CrossRef] [PubMed]
- Nagatomo, Y.; Tang, W.H. Intersections between Microbiome and Heart Failure: Revisiting the Gut Hypothesis. J. Card. Fail. 2015, 21, 973–980. [Google Scholar] [CrossRef] [PubMed]
- Maries, L.; Manitiu, I. Diagnostic and prognostic values of B-type natriuretic peptides (BNP) and N-terminal fragment brain natriuretic peptides (NT-pro-BNP). Cardiovasc. J. Afr. 2013, 24, 286–289. [Google Scholar] [CrossRef] [PubMed]
- Pfister, R.; Scholz, M.; Wielckens, K.; Erdmann, E.; Schneider, C.A. Use of NT-proBNP in routine testing and comparison to BNP. Eur. J. Heart Fail. 2004, 6, 289–293. [Google Scholar] [CrossRef] [PubMed]
- Simons, J.E.; Don-Wauchope, A.C. Evaluation of natriuretic peptide recommendations in heart failure clinical practice guidelines. Clin. Biochem. 2016, 49, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Maisel, A.; Hollander, J.E.; Guss, D.; McCullough, P.; Nowak, R.; Green, G.; Saltzberg, M.; Ellison, S.R.; Bhalla, M.A.; Bhalla, V.; et al. Primary results of the Rapid Emergency Department Heart Failure Outpatient Trial (REDHOT). A multicenter study of B-type natriuretic peptide levels, emergency department decision making, and outcomes in patients presenting with shortness of breath. J. Am. Coll. Cardiol. 2004, 15, 1328–1333. [Google Scholar] [CrossRef] [PubMed]
- Murtagh, G.; Canniffe, C.; Mahgoub, M.; Blake, L.; McCarroll, N.; Crowley, V.; Bennett, K.; Silke, B. Introduction of an NT-proBNP assay to an acute admission unit—A 2-year audit. Eur. J. Intern. Med. 2009, 20, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, T.; Fiuzat, M.; Felker, G.M.; O’Connor, C. Novel biomarkers in chronic heart failure. Nat. Rev. Cardiol. 2012, 9, 347–359. [Google Scholar] [CrossRef] [PubMed]
- Gaggin, H.K.; Januzzi, J.L., Jr. Biomarkers and diagnostics in heart failure. Biochim. Biophys. Acta 2013, 1832, 2442–2450. [Google Scholar] [CrossRef] [PubMed]
- van Kimmenade, R.R.; Januzzi, J.L., Jr. Emerging biomarkers in heart failure. Clin. Chem. 2012, 58, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Paterson, I.; Mielniczuk, L.M.; O’Meara, E.; So, A.; White, J.A. Imaging heart failure: Current and future applications. Can. J. Cardiol. 2013, 29, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Morbach, C.; Lin, B.A.; Sugeng, L. Clinical application of three-dimensional echocardiography. Prog. Cardiovasc. Dis. 2014, 57, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Butler, J. The emerging role of multi-detector computed tomography in heart failure. J. Card. Fail. 2007, 13, 215–226. [Google Scholar] [CrossRef] [PubMed]
- Upadhya, B.; Haykowsky, M.J.; Eggebeen, J.; Kitzman, D.W. Exercise intolerance in heart failure with preserved ejection fraction: More than a heart problem. J. Geriatr. Cardiol. 2015, 12, 294–304. [Google Scholar] [PubMed]
- Fonarow, G.C.; Yancy, C.W.; Heywood, J.T.; ADHERE Scientific Advisory Committee, Study Group, Investigators. Adherence to heart failure quality-of-care indicators in US hospitals: Analysis of the ADHERE Registry. Arch. Intern. Med. 2005, 165, 1469–1477. [Google Scholar] [CrossRef] [PubMed]
- Januzzi, J.L., Jr.; Sakhuja, R.; O’donoghue, M.; Baggish, A.L.; Anwaruddin, S.; Chae, C.U.; Cameron, R.; Krauser, D.G.; Tung, R.; Camargo, C.A., Jr.; et al. Utility of amino-terminal pro-brain natriuretic peptide testing for prediction of 1-year mortality in patients with dyspnea treated in the emergency department. Arch. Intern. Med. 2006, 166, 315–320. [Google Scholar] [CrossRef] [PubMed]
- West, R.; Liang, L.; Fonarow, G.C.; Kociol, R.; Mills, R.M.; O’Connor, C.M.; Hernandez, A.F. Characterization of heart failure patients with preserved ejection fraction: A comparison between ADHERE-US registry and ADHERE-International registry. Eur. J. Heart Fail. 2011, 13, 945–952. [Google Scholar] [CrossRef] [PubMed]
- Ouwerkerk, W.; Voors, A.A.; Zwinderman, A.H. Factors influencing the predictive power of models for predicting mortality and/or heart failure hospitalization in patients with heart failure. JACC Heart Fail. 2014, 2, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Pocock, S.J.; Wang, D.; Pfeffer, M.A.; Yusuf, S.; McMurray, J.J.; Swedberg, K.B.; Ostergren, J.; Michelson, E.L.; Pieper, K.S.; Granger, C.B. Predictors of mortality and morbidity in patients with chronic heart failure. Eur. Heart J. 2006, 27, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Tamargo, J.; López-Sendón, J. Novel therapeutic targets for the treatment of heart failure. Nat. Rev. Drug Discov. 2011, 10, 536–555. [Google Scholar] [CrossRef] [PubMed]
- Dickstein, K.; Cohen-Solal, A.; Filippatos, G.; McMurray, J.J.; Ponikowski, P.; Poole-Wilson, P.A.; Strömberg, A.; van Veldhuisen, D.J.; Atar, D.; Hoes, A.W.; et al. ESC guidelines for the diagnosis and treatment of acute and chronic heart failure 2008: The Task Force for the diagnosis and treatment of acute and chronic heart failure 2008 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association of the ESC (HFA) and endorsed by the European Society of Intensive Care Medicine (ESICM). Eur. J. Heart Fail. 2008, 10, 933–989. [Google Scholar] [PubMed]
- Abraham, W.T.; Adamson, P.B.; Bourge, R.C.; Aaron, M.F.; Costanzo, M.R.; Stevenson, L.W.; Strickland, W.; Neelagaru, S.; Raval, N.; Krueger, S.; et al. Wireless pulmonary artery haemodynamic monitoring in chronic heart failure: A randomised controlled trial. Lancet 2011, 377, 658–666. [Google Scholar] [CrossRef]
- Bui, A.L.; Fonarow, G.C. Home monitoring for heart failure management. J. Am. Coll. Cardiol. 2012, 59, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Guidi, G.; Pollonini, L.; Dacso, C.C.; Iadanza, E. A multi-layer monitoring system for clinical management of Congestive Heart Failure. BMC Med. Inform. Decis. Mak. 2015, 15 (Suppl. S3), S5. [Google Scholar] [CrossRef] [PubMed]
- Dharmarajan, K.; Hsieh, A.F.; Lin, Z.; Bueno, H.; Ross, J.S.; Horwitz, L.I.; Barreto-Filho, J.A.; Kim, N.; Bernheim, S.M.; Suter, L.G.; et al. Diagnoses and Timing of 30-Day Readmissions After Hospitalization for Heart Failure, Acute Myocardial Infarction, or Pneumonia. JAMA J. Am. Med. Assoc. 2013, 309, 355–363. [Google Scholar] [CrossRef] [PubMed]
- VanSuch, M.; Naessens, J.M.; Stroebel, R.J.; Huddleston, J.M.; Williams, A.R. Effect of discharge instructions on readmission of hospitalised patients with heart failure: Do all of the Joint Commission on Accreditation of Healthcare Organizations heart failure core measures reflect better care? Qual. Saf. Health Care 2006, 15, 414–417. [Google Scholar] [CrossRef] [PubMed]
- Donzé, J.; Lipsitz, S.; Bates, D.W.; Schnipper, J.L. Causes and patterns of readmissions in patients with common comorbidities: Retrospective cohort study. BMJ 2013, 347, f7171. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.S. The three-phase terrain of heart failure readmissions. Circ. Heart Fail. 2012, 5, 398–400. [Google Scholar] [CrossRef] [PubMed]
- Center for Medicare and Medicaid Services. Available online: www.CMS.gov (accessed on 22 February 2016).
- Rajaram, R.; Chung, J.W.; Kinnier, C.V.; Barnard, C.; Mohanty, S.; Pavey, E.S.; McHugh, M.C.; Bilimoria, K.Y. Hospital Characteristics Associated With Penalties in the Centers for Medicare & Medicaid Services Hospital-Acquired Condition Reduction Program. JAMA J. Am. Med. Assoc. 2015, 314, 375–383. [Google Scholar]
- Barlas, S. Hospitals Prepare for Medicare Cuts: The Need to Reduce the Federal Deficit Puts Seniors and Providers on Notice. Pharm. Ther. 2013, 38, 92–95. [Google Scholar]
- Desai, A.S.; Stevenson, L.W. Rehospitalization for heart failure: Predict or prevent? Circulation 2012, 126, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, M.B.; Schwartz, R.S.; Asher, C.R.; Navas, E.V.; Totfalusi, V.; Buitrago, I.; Lahoti, A.; Novaro, G.M. Predictors of 30-day readmission in patients hospitalized with decompensated heart failure. Clin. Cardiol. 2013, 36, 542–547. [Google Scholar] [CrossRef] [PubMed]
- Jolly, K.; Taylor, R.S.; Lip, G.Y.; Davies, M.; Davis, R.; Mant, J.; Singh, S.; Greenfield, S.; Ingram, J.; Stubley, J.; et al. A randomized trial of the addition of home-based exercise to specialist heart failure nurse care: The Birmingham Rehabilitation Uptake Maximisation study for patients with Congestive Heart Failure (BRUM-HF) study. Eur. J. Heart Fail. 2009, 11, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Inglis, S.C.; Clark, R.A.; McAlister, F.A.; Ball, J.; Lewinter, C.; Cullington, D.; Stewart, S.; Cleland, J.G. Structured telephone support or telemonitoring programmes for patients with chronic heart failure. Cochrane Database Syst. Rev. 2010, 8, CD007228. [Google Scholar] [PubMed]
- Butler, J.; Kalogeropoulos, A. Hospital strategies to reduce heart failure readmissions: Where is the evidence? J. Am. Coll. Cardiol. 2012, 60, 615–617. [Google Scholar] [CrossRef] [PubMed]
- Ota, K.S.; Beutler, D.S.; Gerkin, R.D.; Weiss, J.L.; Loli, A.I. Physician-directed heart failure transitional care program: A retrospective case review. J. Clin. Med. Res. 2013, 5, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Feltner, C.; Jones, C.D.; Cené, C.W.; Zheng, Z.J.; Sueta, C.A.; Coker-Schwimmer, E.J.; Arvanitis, M.; Lohr, K.N.; Middleton, J.C.; Jonas, D.E. Transitional care interventions to prevent readmissions for persons with heart failure: A systematic review and meta-analysis. Ann. Intern. Med. 2014, 160, 774–784. [Google Scholar] [CrossRef] [PubMed]
- Bekelman, D.B.; Plomondon, M.E.; Carey, E.P.; Sullivan, M.D.; Nelson, K.M.; Hattler, B.; McBryde, C.F.; Lehmann, K.G.; Gianola, K.; Heidenreich, P.A.; et al. Primary Results of the Patient-Centered Disease Management (PCDM) for Heart Failure Study: A Randomized Clinical Trial. JAMA Intern. Med. 2015, 175, 725–732. [Google Scholar] [CrossRef] [PubMed]
- Taylor, R.S.; Hayward, C.; Eyre, V.; Austin, J.; Davies, R.; Doherty, P.; Jolly, K.; Wingham, J.; Van Lingen, R.; Abraham, C.; et al. Clinical effectiveness and cost-effectiveness of the Rehabilitation Enablement in Chronic Heart Failure (REACH-HF) facilitated self-care rehabilitation intervention in heart failure patients and caregivers: Rationale and protocol for a multicentre randomised controlled trial. BMJ Open 2015, 5, e009994. [Google Scholar] [PubMed]
- Giamouzis, G.; Kalogeropoulos, A.; Georgiopoulou, V.; Laskar, S.; Smith, A.L.; Dunbar, S.; Triposkiadis, F.; Butler, J. Hospitalization epidemic in patients with heart failure: Risk factors, risk prediction, knowledge gaps, and future directions. J. Card. Fail. 2011, 17, 54–75. [Google Scholar] [CrossRef] [PubMed]
- Scalvini, S.; Giordano, A. Heart failure. Optimal postdischarge management of chronic HF. Nat. Rev. Cardiol. 2013, 10, 9–10. [Google Scholar] [CrossRef] [PubMed]
- Fleg, J.L.; Cooper, L.S.; Borlaug, B.A.; Haykowsky, M.J.; Kraus, W.E.; Levine, B.D.; Pfeffer, M.A.; Piña, I.L.; Poole, D.C.; Reeves, G.R.; et al. Exercise training as therapy for heart failure: Current status and future directions. Circ. Heart Fail. 2015, 8, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Hirai, D.M.; Musch, T.I.; Poole, D.C. Exercise training in chronic heart failure: Improving skeletal muscle O2 transport and utilization. Am. J. Physiol. Heart Circ. Physiol. 2015, 309, H1419–H1439. [Google Scholar] [CrossRef] [PubMed]
- Inouye, S.; Bouras, V.; Shouldis, E.; Johnstone, A.; Silverzweig, Z.; Kosuri, P. Predicting readmission of heart failure patients using automated follow-up calls. BMC Med. Inform. Decis. Mak. 2015, 15, 22. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R.; Huntley, A.; Mann, M.; Huws, D.; Paranjothy, S.; Elwyn, G.; Purdy, S. Specialist clinics for reducing emergency admissions in patients with heart failure: A systematic review and meta-analysis of randomised controlled trials. Heart 2013, 99, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Swedberg, K.; Komajda, M.; Böhm, M.; Borer, J.S.; Ford, I.; Dubost-Brama, A.; Lerebours, G.; Tavazzi, L.; SHIFT Investigators. Ivabradine and outcomes in chronic heart failure (SHIFT): A randomised placebo-controlled study. Lancet 2010, 376, 875–885. [Google Scholar] [CrossRef]
- Zannad, F.; McMurray, J.J.; Krum, H.; van Veldhuisen, D.J.; Swedberg, K.; Shi, H.; Vincent, J.; Pocock, S.J.; Pitt, B.; EMPHASIS-HF Study Group. Eplerenone in patients with systolic heart failure and mild symptoms. N. Engl. J. Med. 2011, 364, 11–21. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, C.M.; Starling, R.C.; Hernandez, A.F.; Armstrong, P.W.; Dickstein, K.; Hasselblad, V.; Heizer, G.M.; Komajda, M.; Massie, B.M.; McMurray, J.J.; et al. Effect of nesiritide in patients with acute decompensated heart failure. N. Engl. J. Med. 2011, 365, 32–43. [Google Scholar] [CrossRef] [PubMed]
- McMurray, J.J.; Packer, M.; Desai, A.S.; Gong, J.; Lefkowitz, M.; Rizkala, A.R.; Rouleau, J.L.; Shi, V.C.; Solomon, S.D.; Swedberg, K.; et al. Baseline characteristics and treatment of patients in prospective comparison of ARNI with ACEI to determine impact on global mortality and morbidity in heart failure trial (PARADIGM-HF). Eur. J. Heart Fail. 2014, 16, 817–825. [Google Scholar] [CrossRef] [PubMed]
- Goldhaber, J.I.; Hamilton, M.A. Role of inotropic agents in the treatment of heart failure. Circulation 2010, 121, 1655–1660. [Google Scholar] [CrossRef] [PubMed]
- Krishnamoorthy, A.; Felker, G.M. Fluid removal in acute heart failure: Diuretics versus devices. Curr. Opin. Crit. Care 2014, 20, 478–483. [Google Scholar] [CrossRef] [PubMed]
- Moss, A.J.; Hall, W.J.; Cannom, D.S.; Klein, H.; Brown, M.W.; Daubert, J.P.; Estes, N.A., 3rd; Foster, E.; Greenberg, H.; Higgins, S.L.; et al. Cardiac-resynchronization therapy for the prevention of heart-failure events. N. Engl. J. Med. 2009, 361, 1329–1338. [Google Scholar] [CrossRef] [PubMed]
- Cleland, J.G.; Mareev, Y. CRT for Heart Failure and ESRD: More Trials or More Thought Needed? J. Am. Coll. Cardiol. 2015, 66, 2630–2632. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Mai, L.; Cai, X.; Hu, Y.; Mai, W. Stem cell therapy for heart disease-Meta-analysis may be misleading. Int. J. Cardiol. 2016, 203, 351–352. [Google Scholar] [CrossRef] [PubMed]
- Jaski, B.E.; Jessup, M.L.; Mancini, D.M.; Cappola, T.P.; Pauly, D.F.; Greenberg, B.; Borow, K.; Dittrich, H.; Zsebo, K.M.; Hajjar, R.J. Calcium upregulation by percutaneous administration of gene therapy in cardiac disease (CUPID Trial), a first-in-human phase 1/2 clinical trial. J. Card. Fail. 2009, 15, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Jessup, M.; Greenberg, B.; Mancini, D.; Cappola, T.; Pauly, D.F.; Jaski, B.; Yaroshinsky, A.; Zsebo, K.M.; Dittrich, H.; Hajjar, R.J.; et al. Calcium Upregulation by Percutaneous Administration of Gene Therapy in Cardiac Disease (CUPID): A phase 2 trial of intracoronary gene therapy of sarcoplasmic reticulum Ca2+-ATPase in patients with advanced heart failure. Circulation 2011, 124, 304–313. [Google Scholar] [CrossRef] [PubMed]
- The Consensus Trial Study Group. Effects of enalapril on mortality in severe congestive heart failure. Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). N. Engl. J. Med. 1987, 316, 1429–1435. [Google Scholar]
- The SOLVD Investigators. Effect of enalapril on mortality and the development of heart failure in asymptomatic patients with reduced left ventricular ejection fractions. N. Engl. J. Med. 1992, 327, 685–691. [Google Scholar]
- Pitt, B.; Zannad, F.; Remme, W.J.; Cody, R.; Castaigne, A.; Perez, A.; Palensky, J.; Wittes, J. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N. Engl. J. Med. 1999, 341, 709–717. [Google Scholar] [CrossRef] [PubMed]
- Anonymous. The Cardiac Insufficiency Bisoprolol Study II (CIBIS-II): A randomised trial. Lancet 1999, 353, 9–13. [Google Scholar]
- Cohn, J.N.; Tognoni, G.; Valsartan Heart Failure Trial Investigators. A randomized trial of the angiotensin-receptor blocker valsartan in chronic heart failure. N. Engl. J. Med. 2001, 345, 1667–1675. [Google Scholar] [CrossRef] [PubMed]
- Publication Committee for the VMAC Investigators (Vasodilatation in the Management of Acute HF). Intravenous nesiritide vs. nitroglycerin for treatment of decompensated congestive heart failure: A randomized controlled trial. JAMA J. Am. Med. Assoc. 2002, 287, 1531–1540. [Google Scholar]
- Poole-Wilson, P.A.; Swedberg, K.; Cleland, J.G.; Di Lenarda, A.; Hanrath, P.; Komajda, M.; Lubsen, J.; Lutiger, B.; Metra, M.; Remme, W.J.; et al. Carvedilol Or Metoprolol European Trial Investigators. Comparison of carvedilol and metoprolol on clinical outcomes in patients with chronic heart failure in the Carvedilol or Metoprolol European Trial (COMET): Randomised controlled trial. Lancet 2003, 362, 7–13. [Google Scholar] [CrossRef]
- McMurray, J.J.; Ostergren, J.; Swedberg, K.; Granger, C.B.; Held, P.; Michelson, E.L.; Olofsson, B.; Yusuf, S.; Pfeffer, M.A.; CHARM Investigators and Committees. Effects of candesartan in patients with chronic heart failure and reduced left-ventricular systolic function taking angiotensin-converting-enzyme inhibitors: The CHARM-Added trial. Lancet 2003, 362, 767–771. [Google Scholar] [CrossRef]
- Granger, C.B.; McMurray, J.J.; Yusuf, S.; Held, P.; Michelson, E.L.; Olofsson, B.; Ostergren, J.; Pfeffer, M.A.; Swedberg, K.; CHARM Investigators and Committees. Effects of candesartan in patients with chronic heart failure and reduced left-ventricular systolic function intolerant to angiotensin-converting-enzyme inhibitors: The CHARM-Alternative trial. Lancet 2003, 362, 772–776. [Google Scholar] [CrossRef]
- Yusuf, S.; Pfeffer, M.A.; Swedberg, K.; Granger, C.B.; Held, P.; McMurray, J.J.; Michelson, E.L.; Olofsson, B.; Ostergren, J.; CHARM Investigators and Committees. Effects of candesartan in patients with chronic heart failure and preserved left-ventricular ejection fraction: The CHARM-Preserved Trial. Lancet 2003, 362, 777–781. [Google Scholar] [CrossRef]
- Konstam, M.A.; Gheorghiade, M.; Burnett, J.C., Jr.; Grinfeld, L.; Maggioni, A.P.; Swedberg, K.; Udelson, J.E.; Zannad, F.; Cook, T.; Ouyang, J.; et al. Efficacy of Vasopressin Antagonism in Heart Failure Outcome Study With Tolvaptan (EVEREST) Investigators. Effects of oral tolvaptan in patients hospitalized for worsening heart failure: The EVEREST Outcome Trial. JAMA J. Am. Med. Assoc. 2007, 297, 1319–1331. [Google Scholar] [CrossRef] [PubMed]
- McMurray, J.J.; Teerlink, J.R.; Cotter, G.; Bourge, R.C.; Cleland, J.G.; Jondeau, G.; Krum, H.; Metra, M.; O’Connor, C.M.; Parker, J.D.; et al. VERITAS Investigators. Effects of tezosentan on symptoms and clinical outcomes in patients with acute heart failure: The VERITAS randomized controlled trials. JAMA J. Am. Med. Assoc. 2007, 298, 2009–2019. [Google Scholar] [CrossRef] [PubMed]
- Kjekshus, J.; Apetrei, E.; Barrios, V.; Böhm, M.; Cleland, J.G.; Cornel, J.H.; Dunselman, P.; Fonseca, C.; Goudev, A.; Grande, P.; et al. Rosuvastatin in older patients with systolic heart failure. N. Engl. J. Med. 2007, 357, 2248–2261. [Google Scholar] [CrossRef] [PubMed]
- Torre-Amione, G.; Anker, S.D.; Bourge, R.C.; Colucci, W.S.; Greenberg, B.H.; Hildebrandt, P.; Keren, A.; Motro, M.; Moyé, L.A.; Otterstad, J.E.; et al. Results of a non-specific immunomodulation therapy in chronic heart failure (ACCLAIM trial): A placebo-controlled randomised trial. Lancet 2008, 371, 228–236. [Google Scholar] [CrossRef]
- Redfield, M.M.; Borlaug, B.A.; Lewis, G.D.; Mohammed, S.F.; Semigran, M.J.; Lewinter, M.M.; Deswal, A.; Hernandez, A.F.; Lee, K.L.; Braunwald, E.; et al. PhosphdiesteRasE-5 Inhibition to Improve CLinical Status and EXercise Capacity in Diastolic Heart Failure (RELAX) trial: Rationale and design. Circ. Heart Fail. 2012, 5, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Gheorghiade, M.; Böhm, M.; Greene, S.J.; Fonarow, G.C.; Lewis, E.F.; Zannad, F.; Solomon, S.D.; Baschiera, F.; Botha, J.; Hua, T.A.; et al. Effect of aliskiren on postdischarge mortality and heart failure readmissions among patients hospitalized for heart failure: The ASTRONAUT randomized trial. JAMA J. Am. Med. Assoc. 2013, 309, 1125–1135. [Google Scholar] [CrossRef] [PubMed]
- Study to Evaulate the Safety and Efficacy of IV Infusion Treatment with Omecamtiv Mecarbil in Subjects with Left Ventricular Systolic Dysfunction Hospitalised for Acute Heart Failure (ATOMIC-AHF). Available online: www.Clinicaltrials.gov (accessed on 25 February 2016).
- Felker, G.M.; Teerlink, J.R.; Butler, J.; Hernandez, A.F.; Miller, A.B.; Cotter, G.; Davison, B.A.; Filippatos, G.; Greenberg, B.H.; Ponikowski, P.; et al. Effect of serelaxin on mode of death in acute heart failure: Results from the RELAX-AHF study. J. Am. Coll. Cardiol. 2014, 64, 1591–1598. [Google Scholar] [CrossRef] [PubMed]
- McMurray, J.J.; Packer, M.; Desai, A.S.; Gong, J.; Lefkowitz, M.P.; Rizkala, A.R.; Rouleau, J.L.; Shi, V.C.; Solomon, S.D.; Swedberg, K.; et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N. Engl. J. Med. 2014, 371, 993–1004. [Google Scholar] [CrossRef] [PubMed]
- Pieske, B.; Butler, J.; Filippatos, G.; Lam, C.; Maggioni, A.P.; Ponikowski, P.; Shah, S.; Solomon, S.; Kraigher-Krainer, E.; Samano, E.T. Rationale and design of the SOluble guanylate Cyclase stimulatoR in heArT failurE Studies (SOCRATES). Eur. J. Heart Fail. 2014, 16, 1026–1038. [Google Scholar] [CrossRef] [PubMed]
- Bardy, G.H.; Lee, K.L.; Mark, D.B.; Poole, J.E.; Packer, D.L.; Boineau, R.; Domanski, M.; Troutman, C.; Anderson, J.; Johnson, G.; et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N. Engl. J. Med. 2005, 352, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Whellan, D.J.; Ousdigian, K.T.; Al-Khatib, S.M.; Pu, W.; Sarkar, S.; Porter, C.B.; Pavri, B.B.; O’Connor, C.M.; PARTNERS Study Investigators. Combined heart failure device diagnostics identify patients at higher risk of subsequent heart failure hospitalizations: Results from PARTNERS HF (Program to Access and Review Trending Information and Evaluate Correlation to Symptoms in Patients with Heart Failure) study. J. Am. Coll. Cardiol. 2010, 55, 1803–1810. [Google Scholar] [PubMed]
- Assmus, B.; Fischer-Rasokat, U.; Honold, J.; Seeger, F.H.; Fichtlscherer, S.; Tonn, T.; Seifried, E.; Schächinger, V.; Dimmeler, S.; Zeiher, A.M. Transcoronary transplantation of functionally competent BMCs is associated with a decrease in natriuretic peptide serum levels and improved survival of patients with chronic postinfarction heart failure: Results of the TOPCARE-CHD Registry. Circ. Res. 2007, 100, 1234–1241. [Google Scholar] [CrossRef] [PubMed]
- Bolli, R.; Chugh, A.R.; D’Amario, D.; Loughran, J.H.; Stoddard, M.F.; Ikram, S.; Beache, G.M.; Wagner, S.G.; Leri, A.; Hosoda, T.; et al. Cardiac stem cells in patients with ischaemic cardiomyopathy (SCIPIO): Initial results of a randomised phase 1 trial. Lancet 2011, 378, 1847–1857. [Google Scholar] [CrossRef]
- Trachtenberg, B.; Velazquez, D.L.; Williams, A.R.; McNiece, I.; Fishman, J.; Nguyen, K.; Rouy, D.; Altman, P.; Schwarz, R.; Mendizabal, A.; et al. Rationale and design of the Transendocardial Injection of Autologous Human Cells (bone marrow or mesenchymal) in Chronic Ischemic Left Ventricular Dysfunction and Heart Failure Secondary to Myocardial Infarction (TAC-HFT) trial: A randomized, double-blind, placebo-controlled study of safety and efficacy. Am. Heart J. 2011, 161, 487–493. [Google Scholar] [PubMed]
- Perin, E.C.; Willerson, J.T.; Pepine, C.J.; Henry, T.D.; Ellis, S.G.; Zhao, D.X.; Silva, G.V.; Lai, D.; Thomas, J.D.; Kronenberg, M.W.; et al. Effect of transendocardial delivery of autologous bone marrow mononuclear cells on functional capacity, left ventricular function, and perfusion in chronic heart failure: The FOCUS-CCTRN trial. JAMA J. Am. Med. Assoc. 2012, 307, 1717–1726. [Google Scholar] [CrossRef] [PubMed]
- Hare, J.M.; Fishman, J.E.; Gerstenblith, G.; DiFede Velazquez, D.L.; Zambrano, J.P.; Suncion, V.Y.; Tracy, M.; Ghersin, E.; Johnston, P.V.; Brinker, J.A.; et al. Comparison of allogeneic vs autologous bone marrow–derived mesenchymal stem cells delivered by transendocardial injection in patients with ischemic cardiomyopathy: The POSEIDON randomized trial. JAMA J. Am. Med. Assoc. 2012, 308, 2369–2379. [Google Scholar] [CrossRef] [PubMed]
- Makkar, R.R.; Smith, R.R.; Cheng, K.; Malliaras, K.; Thomson, L.E.; Berman, D.; Czer, L.S.; Marbán, L.; Mendizabal, A.; Johnston, P.V.; et al. Intracoronary cardiosphere-derived cells for heart regeneration after myocardial infarction (CADUCEUS): A prospective, randomised phase 1 trial. Lancet 2012, 379, 895–904. [Google Scholar] [CrossRef]
- Vrtovec, B.; Poglajen, G.; Lezaic, L.; Sever, M.; Socan, A.; Domanovic, D.; Cernelc, P.; Torre-Amione, G.; Haddad, F.; Wu, J.C. Comparison of transendocardial and intracoronary CD34+ cell transplantation in patients with nonischemic dilated cardiomyopathy. Circulation 2013, 128, S42–S49. [Google Scholar] [CrossRef] [PubMed]
- Karantalis, V.; DiFede, D.L.; Gerstenblith, G.; Pham, S.; Symes, J.; Zambrano, J.P.; Fishman, J.; Pattany, P.; McNiece, I.; Conte, J.; et al. Autologous mesenchymal stem cells produce concordant improvements in regional function, tissue perfusion, and fibrotic burden when administered to patients undergoing coronary artery bypass grafting: The Prospective Randomized Study of Mesenchymal Stem Cell Therapy in Patients Undergoing Cardiac Surgery (PROMETHEUS) trial. Circ. Res. 2014, 114, 1302–1310. [Google Scholar] [PubMed]
- Bartunek, J.; Davison, B.; Sherman, W.; Povsic, T.; Henry, T.D.; Gersh, B.; Metra, M.; Filippatos, G.; Hajjar, R.; Behfar, A.; et al. Congestive Heart Failure Cardiopoietic Regenerative Therapy (CHART-1) trial design. Eur. J. Heart Fail. 2016, 18, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Alrefai, M.T.; Murali, D.; Paul, A.; Ridwan, K.M.; Connell, J.M.; Shum-Tim, D. Cardiac tissue engineering and regeneration using cell-based therapy. Stem Cells Cloning 2015, 8, 81–101. [Google Scholar] [PubMed]
- Blau, H.M.; Brazelton, T.R.; Weimann, J.M. The evolving concept of a stem cell: Entity or function? Cell 2001, 105, 829–841. [Google Scholar] [CrossRef]
- Dib, N.; Dinsmore, J.; Lababidi, Z.; White, B.; Moravec, S.; Campbell, A.; Rosenbaum, A.; Seyedmadani, K.; Jaber, W.A.; Rizenhour, C.S.; et al. One-year follow-up of feasibility and safety of the first U.S., randomized, controlled study using 3-dimensional guided catheter-based delivery of autologous skeletal myoblasts for ischemic cardiomyopathy (CAuSMIC study). JACC Cardiovasc. Interv. 2009, 2, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.N.; Geffner, L.; Vina, R.F.; Saslavsky, J.; Urschel, H.C., Jr.; Kormos, R.; Benetti, F. Surgical treatment for congestive heart failure with autologous adult stem cell transplantation: A prospective randomized study. J. Thorac. Cardiovasc. Surg. 2005, 130, 1631–1638. [Google Scholar] [CrossRef] [PubMed]
- Heldman, A.W.; DiFede, D.L.; Fishman, J.E.; Zambrano, J.P.; Trachtenberg, B.H.; Karantalis, V.; Mushtaq, M.; Williams, A.R.; Suncion, V.Y.; McNiece, I.K.; et al. Transendocardial mesenchymal stem cells and mononuclear bone marrow cells for ischemic cardiomyopathy: The TAC-HFT randomized trial. JAMA J. Am. Med. Assoc. 2014, 311, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Freyman, T.; Polin, G.; Osman, H.; Crary, J.; Lu, M.; Cheng, L.; Palasis, M.; Wilensky, R.L. A quantitative, randomized study evaluating three methods of mesenchymal stem cell delivery following myocardial infarction. Eur. Heart J. 2006, 27, 1114–1122. [Google Scholar] [CrossRef] [PubMed]
- Moss, J.A. Gene therapy review. Radiol. Technol. 2014, 86, 155–180. [Google Scholar] [PubMed]
- Williams, M.L.; Koch, W.J. Viral-based myocardial gene therapy approaches to alter cardiac function. Annu. Rev. Physiol. 2004, 66, 49–75. [Google Scholar] [CrossRef] [PubMed]
- Chira, S.; Jackson, C.S.; Oprea, I.; Ozturk, F.; Pepper, M.S.; Diaconu, I.; Braicu, C.; Raduly, L.Z.; Calin, G.A.; Berindan-Neagoe, I. Progresses towards safe and efficient gene therapy vectors. Oncotarget 2015, 6, 30675–30703. [Google Scholar] [PubMed]
- Svensson, E.C.; Marshall, D.J.; Woodard, K.; Lin, H.; Jiang, F.; Chu, L.; Leiden, J.M. Efficient and stable transduction of cardiomyocytes after intramyocardial injection or intracoronary perfusion with recombinant adeno-associated virus vectors. Circulation 1999, 99, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Giacca, M.; Zacchigna, S. VEGF gene therapy: Therapeutic angiogenesis in the clinic and beyond. Gene Ther. 2012, 19, 622–629. [Google Scholar] [CrossRef] [PubMed]
- Beeri, R.; Chaput, M.; Guerrero, J.L.; Kawase, Y.; Yosefy, C.; Abedat, S.; Karakikes, I.; Morel, C.; Tisosky, A.; Sullivan, S.; et al. Gene delivery of sarcoplasmic reticulum calcium ATPase inhibits ventricular remodeling in ischemic mitral regurgitation. Circ. Heart Fail. 2010, 3, 627–634. [Google Scholar] [CrossRef] [PubMed]
- Cannavo, A.; Liccardo, D.; Koch, W.J. Targeting cardiac β-adrenergic signaling via GRK2 inhibition for heart failure therapy. Front. Physiol. 2013, 4, 264. [Google Scholar] [CrossRef] [PubMed]
- Raake, P.W.; Tscheschner, H.; Reinkober, J.; Ritterhoff, J.; Katu, H.A.; Koch, W.J.; Most, P. Gene therapy targets in heart failure: The path to translation. Clin. Pharmacol. Ther. 2011, 90, 542–553. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. ICD-10 Second Edition Volume 2. Available online: http://www.who.int/classifications/icd/ICD-10_2nd_ed_volume2.pdf (accessed on 24 February 2016).
- Hale, D. ACP Hospitalist: Billing and Coding. 2009. Available online: http://www.acphospitalist.org/archives/2009/01/coding.htm#sb1 (accessed on 24 February 2016).

| Myocardial Stress | Myocardial Injury | Matrix and Cellular Remodeling | Inflammation | Oxidative Stress | Neuro-Hormones | Vascular System | Cardio-Renal Syndrome |
|---|---|---|---|---|---|---|---|
| Natriuretic | Cardiac troponins | Osteopontin | C-reactive protein | Oxidized LDL | Nor-epinephrine | Homocysteine | Creatinine |
| peptides | High sensitivity cardiac troponins | Galectin-3 | sST2 | Myeloperoxidase | Renin | Adhesion molecules | Cystatin C |
| Mid-regional | Myosin light-chain kinase 1 | sST2 | Tumor necrosis factor | Urinary biopyrrins | Angiotensin-II | ICAM, P-selectin | NGAL |
| Pro-adrenomedullin | Heart-type fatty acid binding protein | GDF-15 | FAS (APO-1) | Urinary and plasma isoprostanes | Co-peptin | Endothelin | Trace protein |
| Neuregulin | Pentraxin 3 | MMPs | GDF-15 | Plasma malondialdehyde | Endothelin | Adiponectin | |
| sST2 | TIMPs | Pentraxin 3 | C-type natriuretic peptide | ||||
| Collagen propeptides | Adipokines | ||||||
| cytokines | |||||||
| Procalcitonin | |||||||
| Osteoprotegerin |
| Clinical Trial Name | Drug Class | Drugs | Condition | Phase | No. of Patients | Date | Outcome | References |
|---|---|---|---|---|---|---|---|---|
| CONSENSUS | ACE inhibitors (ACEis) | Enalapril vs. placebo | Severe congestive heart failure | Double-blinded multi-center RCT | 253 | 1987 | ACEi improved symptoms, reduced HF progression in NYHA IV and mortality | [80] |
| SOLVD | ACE inhibitors (ACEis) | Enalapril vs. placebo | Heart failure with ejection fractions of 0.35 or less and on drugs other than an angiotensin-converting enzyme inhibitor | Double-blinded multi-center RCT | 4228 | 1992 | ACEi in an asymptomatic LV dysfunction reduced incidence and hospitalization for HF | [81] |
| RALES | Aldosterone antagonists | Spironolactone vs. placebo | CCF (NYHA III and IV) | Double-blinded multi-center RCT | 1663 | 1999 | Spironolactone reduced hospitalization (35%), mortality (30%) and symptoms in NYHA III/IV | [82] |
| CIBIS-II | Beta blockers | Bisoprolol vs. placebo | HF (NYHA Classes III–IV) | Double-blinded multi-center RCT | 2647 | 1999 | All-cause mortality hospitalizations and sudden cardiac death were reduced by 50%. | [83] |
| ValHeFT | Angiotensin receptor blockers (ARBs) | Valsartan vs. placebo | Heart failure (NYHA II–IV) | Multicenter, double-blinded, parallel-group, placebo-controlled RCT | 5010 | 2001 | Valsartan improved symptoms and mortality in NYHA II+; no benefit when added to ACEi | [84] |
| VMAC | Recombinant form of human B-type natriuretic peptide Vs nitrates | Intravenous nesiritide vs. nitroglycerin vs. placebo | Acute decompensated HF | Randomized, double-blind trial | 489 | 2002 | Nesiritide improved hemodynamic function as assessed by measuring reduced pulmonary capillary wedge pressure (PCWP) | [85] |
| COMET | Beta blockers | Carvedilol vs. metoprolol | Heart failure (EF < 35%; Stage II–IV) | Multicenter, double-blind, parallel-group, RCT | 3029 | 2003 | Carvedilol decreased all-cause mortality by 6% as compared to metoprolol | [86] |
| CHARM (includes CHARM added/alternative/preserved) | Angiotensin receptor blockers (ARBs) | Candesartan +/− ACEis vs. placebo | Heart failure (EF < 40%; Stage II or IV); (EF < 40% on ACEi for added); (EF < 40% intolerant of ACEi for alternative); EF > 40% for preserved | Double-blinded multi-center RCT | 4576/2448 for added/2028 for alternative/30,233 for preserved | 2003 | Candesartan reduced death in HF; had added benefit in the presence of ACEi irrespective of ACEis dose; no benefit in preserved LV dysfunction | [87,88,89] |
| EVEREST | Vasopressin antagonists | Tolvaptan vs. placebo | Decompensated HF | Multi-center, double-blind, parallel-group, randomized controlled trial | 4133 | 2007 | Significant benefit on dyspnea, edema, body weight and serum sodium, but no improvement in cardio-vascular mortality or HF hospitalization | [90] |
| VERITAS | Endothelin receptor antagonist | Intravenous tezosentan vs. placebo | Acute HF | Randomized, double-blind trial | 1435 | 2007 | Tezosentan failed to improve symptoms or clinical outcomes in patients with acute heart failure | [91] |
| CORONA | Statin | Rosuvastatin vs. placebo | Congestive Cardiac Failure (CCF) (EF < 40%, NYHA II) | Multicenter, double-blind, randomized placebo-controlled trial | 5011 | 2007 | Rosuvastatin in statin-naive CCF patients reduced admissions, but not mortality | [92] |
| ACCLAIM | Device-based non-specific immuno-modulation therapy (IMT) | Celecade vs. placebo | NYHA II–IV HF | Double-blind, placebo-controlled study | 2426 | 2008 | Failed to demonstrate reduction in hospitalization or mortality, but proposed to be beneficial for the early stages of HF | [93] |
| SHIFT | Specific inhibitor of current in the sinoatrial node | Ivabradine vs. placebo | HF with LVEF 35% or lower with heart rate >70 in sinus rhythm | Double-blinded multi-center RCT | 6558 | 2010 | Ivabradine reduced CCF admissions and deaths, especially those with higher HR | [69] |
| EMPHASIS-HF | Aldosterone antagonists | Eplerenone vs. placebo | CCF (NYHA II and EF < 35%) | Double-blinded multi-center RCT | 2737 | 2011 | Eplerenone reduced mortality by 7% and symptoms in NYHA II | [70] |
| ASCEND-HF | Recombinant form of human B-type natriuretic peptide | Nesiritide infusion vs. placebo | HF | Double-blinded multi-center RCT | 7141 | 2011 | Improved the symptom of dyspnea, but no change in mortality | [72] |
| RELAX | cGMP-specific phosphodiesterase type 5 inhibitor | Sildenafil vs. placebo | Diastolic HF with NYHA II–III (LVEF > 50%) | Double-blinded multi-center RCT | 216 | 2012 | No improvement in health outcomes and exercise ability | [94] |
| ASTRONAUT | Renin inhibitor | Aliskiren vs. placebo | Decompensated HF | Multicenter, double-blind, randomized placebo-controlled trial | 1639 | 2013 | No additional benefit from the drug to standard therapy | [95] |
| ATOMIC-AHF | Cardiac-specific myosin activator | Omecamtiv mecarbil vs. placebo | ADHF with LVEF ≤ 40% | Multicenter, double-blind, randomized placebo-controlled trial | 614 | 2013 | Safe, but no change in the dyspnea symptoms | [96] |
| RELAX-AHF | Vasoactive peptide hormone | Serelaxin, recombinant human relaxin-2 vs. placebo | Acute HF | Randomized, placebo-controlled trial | 1161 | 2013 | Dyspnea relief and other symptoms of HF, but had no effect on hospital readmissions | [97] |
| PARADIGM-HF | Combination of ARB, valsartan and a neprilysin inhibitor prodrug sacubitril | Valsartan/sacubitril (LCZ696) vs. enalapril | NYHA functional Class II–IV (HFrEF and HFpEF) | Randomized study | 8442 | 2014 | Significant reductions in cardiovascular and all-cause mortality, as well as heart failure hospitalization | [72,98] |
| SOCRATES, including SOCRATES-REDUCED for LVEF ≤ 45 SOCRATES-PRESERVED for LVEF ≥ 45 | Oral cyclic guanosine monophosphate (cGMP) stimulator | Oral (cGMP) stimulator vericiguat (BAY 1021189) vs. placebo | HF with LVEF ≥ 45 and ≤ 45 | Double-blinded multi-center RCT | 456 | 2014 | Study completed, results awaited | [99] |
| NCT01919177 | Inorganic nitrates | Beet root vs. placebo | Heart failure with normal ejection fraction | randomized, double-blind, | 17 | 2015 | Increased exercise capacity by increasing exercise vasodilatory and cardiac output reserves | [3] |
| Defibrillator-based clinical trials | ||||||||
| SCD-HeFT | ICD vs. drug | ICD vs. amiodarone vs. placebo | CCF (NYHA II/III; LVEF < 35) | Double-blinded multi-center RCT | 2521 | 2005 | ICD significantly increased survival by 23%; amiodarone had no effect | [100] |
| MADIT-CRT | CRT | CRT with and without ICD | HF (NYHA I–II; EF < 30%; QRS > 130 ms) | Double-blinded multi-center RCT | 1820 | 2009 | CRT (added to ICD) slows the progression of heart failure in high-risk (QRS ≥ 130 ms, EF ≤ 3 0%), mildly symptomatic patients (NYHA I/II) | [75] |
| PARTNERS HF | HF device | Combined heart failure (HF) device guided diagnostic data to predict clinical deterioration of HF | CRT implantable cardioverter-defibrillators in HF patients | Observational study | 1024 | 2010 | Identifies patients at a higher risk of HF hospitalizations | [101] |
| Stem cell-based clinical trials | ||||||||
| TOPCARE-CHD | Bone marrow-derived mononuclear cells | intracoronary injection of functional BMMC vs. placebo | Ischemic HF | Single-center study randomized | 121 | 2007 | Improved cardiac function and suppression of NT-proANP and proBNP with BMMC, especially with cells with high functional capacity determined with the colony forming unit assay | [102] |
| SCIPIO | Cardiac stem cells | Intracoronary injection of in vitro expanded c-Kit+ CSC from myocardium vs. placebo | Ischemic HF with LVEF < 40% | Single-center study | 18 | 2011 | Significant improvement in myocardial performance, scar tissue reduction and LV systolic function | [103] |
| TAC-HFT | MSCs and BMMCs | Trans-endocardial injection of culture-expanded MSCs vs. whole BMMC vs. placebo | Ischemic cardio-myopathy with LVEF < 50% | Randomized, blinded, placebo-controlled study | 65 | 2011 | MSCs and BMMC were safe, but MSCs better for scar reduction and improved myocardial function than BMMCs | [104] |
| FOCUS-CCTRN | Bone marrow-derived mononuclear cells | Trans-endocardial injection of BMMC vs. placebo | Ischemic HF/NYHA II–III with LVHF < 45% | Randomized double-blind, placebo-controlled trial | 153 | 2012 | Failed to improve LVESV, maximal oxygen consumption or reversibility on SPECT | [105] |
| POSEIDON | Mesenchymal stem cells | Allogenic vs. autologous trans-endocardial injection of MSCs | Chronic ischemic left ventricular dysfunction with LVHF < 50% | Single-center study | 31 | 2012 | Both allo- and auto-MSCs were safe, reduced infarct size and improved ventricular remodeling | [106] |
| CADUCEUS | Cardiosphere-derived cells | Intracoronary administration of autologous CDCs vs. placebo | Ischemic HF, NYHF I with LVEF between 25% and 45% | Single-center study | 17 | 2012 | Safe and decreased scar size, increased viable myocardium and improved regional function of infarcted myocardium, but no significant improvement in EF | [107] |
| NOGA-DCM | Bone marrow-derived CD34+ cells | Trans-endocardial CD34+ vs. placebo | Non-ischemic cardiomyopathy with NYHA III and LVHF < 40% | Single-center study randomized | 33 | 2014 | Improved left ventricular function, decreased N-terminal pro-BNP and better exercise capacity with infusion of a high number of cells | [108] |
| PROMETHEUS | Mesenchymal stem cells | Intra-myocardial injection of autologous MSCs | Chronic ischemic cardiomyopathy undergoing CABG | Single-center study | 6 | 2014 | Scar reduction, improvement in myocardial perfusion, regional function and LVEF in patients undergoing CABG | [109] |
| CHART-1 | Cardiopoietic stem cells | bone marrow-derived and lineage-directed autologous cardiopoietic stem cells | Ischemic HF | Randomized, sham-controlled multicenter study | 240 | 2015 | Under progress | [110] |
| Gene therapy-based clinical trials | ||||||||
| CUPID-Phase I | Gene therapy | Antegrade epicardial coronary artery infusion of gene SERCA2a via an adeno-associated viral (AAV) vector | Advanced HF-NYHF III/IV (LVEF ≤ 30%) | Single-center study | 9 | 2008 | Safe and improvement in various parameters, such as exercise tolerance, LVEF, reduction of BNP levels | [78] |
| CUPID-Phase II | Gene therapy | Intracoronary adeno-associated virus type 1/sarcoplasmic reticulum Ca2+-ATPase vs. placebo | Advanced HF-NYHF III/IV (LVEF ≤ 30%) | Randomized, double-blind, placebo-controlled | 39 | 2011 | Improvement in various parameters, such as exercise tolerance, LVEF, reduction of BNP levels | [79] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Inamdar, A.A.; Inamdar, A.C. Heart Failure: Diagnosis, Management and Utilization. J. Clin. Med. 2016, 5, 62. https://doi.org/10.3390/jcm5070062
Inamdar AA, Inamdar AC. Heart Failure: Diagnosis, Management and Utilization. Journal of Clinical Medicine. 2016; 5(7):62. https://doi.org/10.3390/jcm5070062
Chicago/Turabian StyleInamdar, Arati A., and Ajinkya C. Inamdar. 2016. "Heart Failure: Diagnosis, Management and Utilization" Journal of Clinical Medicine 5, no. 7: 62. https://doi.org/10.3390/jcm5070062
APA StyleInamdar, A. A., & Inamdar, A. C. (2016). Heart Failure: Diagnosis, Management and Utilization. Journal of Clinical Medicine, 5(7), 62. https://doi.org/10.3390/jcm5070062