Spatiotemporal Coupling of DNA Supercoiling and Genomic Sequence Organization—A Timing Chain for the Bacterial Growth Cycle?
Abstract
:1. Introduction
2. Coupling of Chromosomal Gene Order and Transcription
3. Gradients of Regulators
4. DNA Supercoiling Gradient and Genomic Sequence Organization
5. Role of Local Sequence Organization
6. Topological Domains
7. Modulation of the Transcriptional Supercoiling Response by NAPs
8. Coupling of DNA Structure to Function Using Two Types of DNA Information
9. Role for Changing Chromosome Configuration in Organizing Genomic Transcription
10. Coupling of DNA Topology, NAP Binding Effects, and Holoenzyme Sigma-Factor Composition: Major Regulatory Events during the E. coli Growth Cycle
11. Genomic Transcription during the Bacterial Growth Cycle Is Steered by Supercoil Energy
12. Spatially Shifting Superhelicity Optimum Determines the Temporal Gene Expression
13. The Temporal Gradient of Superhelicity Reflects the Gradient of Ion Composition and Intracellular pH
14. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Muskhelishvili, G.; Sobetzko, P.; Geertz, M.; Berger, M. General organisational principles of the transcriptional regulation system: A tree or a circle? Mol. BioSyst. 2010, 6, 662–676. [Google Scholar] [CrossRef] [PubMed]
- Muskhelishvili, G.; Travers, A. Integration of syntactic and semantic properties of the DNA code reveals chromosomes as thermodynamic machines converting energy into information. Cell. Mol. Life Sci. 2013, 70, 4555–4567. [Google Scholar] [CrossRef] [PubMed]
- Travers, A.; Muskhelishvili, G. Chromosomal Organization and Regulation of Genetic Function in Escherichia coli Integrates the DNA Analog and Digital Information. EcoSal Plus 2020, 9. [Google Scholar] [CrossRef] [PubMed]
- Macnab, R.M.; Koshland, D.E., Jr. The gradient-sensing mechanism in bacterial chemotaxis. Proc. Natl. Acad. Sci. USA 1972, 69, 2509–2512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berg, H.C.; Purcell, E.M. Physics of chemoreception. Biophys J. 1977, 20, 193–219. [Google Scholar] [CrossRef] [Green Version]
- Sobetzko, P.; Travers, A.; Muskhelishvili, G. Gene order and chromosome dynamics coordinate spatiotemporal gene expression during the bacterial growth cycle. Proc. Natl. Acad. Sci. USA 2012, 109, E42–E50. [Google Scholar] [CrossRef] [Green Version]
- Kosmidis, K.; Jablonski, K.P.; Muskhelishvili, G.; Hütt, M.-T. Chromosomal origin of replication coordinates logically distinct types of bacterial genetic regulation. NPJ Syst. Biol. Appl. 2020, 6, 1–9. [Google Scholar] [CrossRef]
- Travers, A.; Muskhelishvili, G. DNA supercoiling—A global transcriptional regulator for enterobacterial growth? Nat. Rev. Microbiol. 2005, 3, 157–169. [Google Scholar] [CrossRef]
- Scolari, V.F.; Sclavi, B.; Cosentino Lagomarsino, M. The nucleoid as a smart polymer. Front. Microbiol. 2015, 6, 424. [Google Scholar] [CrossRef] [Green Version]
- Pelletier, J.; Halvorsen, K.; Ha, B.Y.; Paparcone, R.; Sandler, S.J.; Woldringh, C.L.; Wong, W.P.; Jun, S. Physical manipulation of the Escherichia coli chromosome reveals its soft nature. Proc. Natl. Acad. Sci. USA 2012, 109, E2649–E2656. [Google Scholar] [CrossRef] [Green Version]
- Barbieri, M.; Chotalia, M.; Fraser, J.; Lavitas, L.M.; Dostie, J.; Pombo, A.; Nicodemi, M. Complexity of chromatin folding is captured by the strings and binders switch model. Proc. Natl. Acad. Sci. USA 2012, 109, 16173–16178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sobetzko, P.; Glinkowska, M.; Travers, A.; Muskhelishvili, G. DNA thermodynamic stability and supercoil dynamics determine the gene expression program during the bacterial growth cycle. Mol. BioSyst. 2013, 9, 1643–1651. [Google Scholar] [CrossRef] [PubMed]
- Benza, V.G.; Bassetti, B.; Dorfman, K.D.; Scolari, V.F.; Bromek, K.; Cicuta, P.; Lagomarsino, M.C. Physical descriptions of the bacterial nucleoid at large scales, and their biological implications. Rep. Prog. Phys. 2012, 75, 076602. [Google Scholar] [CrossRef] [PubMed]
- Travers, A.A.; Muskhelishvili, G. DNA thermodynamics shape chromosome organization and topology. Biochem. Soc. Trans. 2013, 41, 548–553. [Google Scholar] [CrossRef] [Green Version]
- Verma, S.C.; Qian, Z.; Adhya, S.L. Architecture of the Escherichia coli nucleoid. PLoS Genet. 2019, 15, e1008456. [Google Scholar] [CrossRef] [Green Version]
- Amemiya, H.M.; Schroeder, J.; Freddolino, P.L. Nucleoid-associated proteins shape chromatin structure and transcriptional regulation across the bacterial kingdom. Transcription 2021, 12, 182–218. [Google Scholar] [CrossRef]
- Raj, A.; van Oudenaarden, A. Nature, nurture, or chance: Stochastic gene expression and its consequences. Cell 2008, 135, 216–226. [Google Scholar] [CrossRef] [Green Version]
- Piras, V.; Selvarajoo, K. The reduction of gene expression variability from single cells to populations follows simple statistical laws. Genomics 2015, 105, 137–144. [Google Scholar] [CrossRef]
- Brinza, L.; Calevro, F.; Charles, H. Genomic analysis of the regulatory elements and links with intrinsic DNA structural properties in the shrunken genome of Buchnera. BMC Genom. 2013, 14, 73. [Google Scholar] [CrossRef] [Green Version]
- Bai, Y.; He, C.; Chu, P.; Long, J.; Li, X.; Fu, X. Spatial modulation of individual behaviors enables an ordered structure of diverse phenotypes during bacterial group migration. eLife 2021, 10, e67316. [Google Scholar] [CrossRef]
- Cashel, M.; Gallant, J. Two compounds implicated in the function of the RC gene of Escherichia coli. Nature 1969, 221, 838–841. [Google Scholar] [CrossRef] [PubMed]
- Jishage, M.; Kvint, K.; Shingler, V.; Nystrom, T. Regulation of sigma factor competition by the alarmone ppGpp. Genes Dev. 2002, 16, 1260–1270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zuo, Y.; Wang, Y.; Steitz, T.A. The mechanism of E. coli RNA polymerase regulation by ppGpp is suggested by the structure of their complex. Mol. Cell 2013, 50, 430–436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mechold, U.; Potrykus, K.; Murphy, H.; Murakami, K.S.; Cashel, M. Differential regulation by ppGpp versus pppGpp in Escherichia coli. Nucleic Acids Res. 2013, 41, 6175–6189. [Google Scholar] [CrossRef]
- Hengge, R. Stationary-phase gene regulation in Escherichia Coli. EcoSal Plus 2011, 4. [Google Scholar] [CrossRef] [PubMed]
- Potrykus, K.; Murphy, H.; Philippe, N.; Cashel, M. ppGpp is the major source of growth rate control in E. coli. Env. Microbiol. 2011, 13, 563–575. [Google Scholar] [CrossRef] [Green Version]
- Traxler, M.F.; Zacharia, V.M.; Marquardt, S.; Summers, S.M.; Nguyen, H.T.; Stark, S.E.; Conway, T. Discretely calibrated regulatory loops controlled by ppGpp partition gene induction across the ‘feast to famine’ gradient in Escherichia coli. Mol. Microbiol. 2011, 79, 830–845. [Google Scholar] [CrossRef] [Green Version]
- Makinoshima, H.; Nishimura, A.; Ishihama, A. Fractionation of Escherichia coli cell populations at different stages during growth transition to stationary phase. Mol. Microbiol. 2002, 43, 269–279. [Google Scholar] [CrossRef] [Green Version]
- Gangwe Nana, G.Y.; Ripoll, C.; Cabin-Flaman, A.; Gibouin, D.; Delaune, A.; Janniere, L.; Grancher, G.; Chagny, G.; Loutelier-Bourhis, C.; Lentzen, E.; et al. Division-Based, Growth Rate Diversity in Bacteria. Front. Microbiol. 2018, 9, 849. [Google Scholar] [CrossRef]
- Zhu, J.; Winans, S.C. Autoinducer binding by the quorum-sensing regulator TraR increases affinity for target promoters in vitro and decreases TraR turnover rates in whole cells. Proc. Natl. Acad. Sci. USA 1999, 96, 4832–4837. [Google Scholar] [CrossRef] [Green Version]
- Blot, N.; Mavathur, R.; Geertz, M.; Travers, A.; Muskhelishvili, G. Homeostatic regulation of supercoiling sensitivity coordinates transcription of the bacterial genome. EMBO Rep. 2006, 7, 710–715. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ayala, J.C.; Benitez, J.A.; Silva, A.J. A proteome-wide screen to identify transcription factors interacting with the Vibrio cholerae rpoS promoter. J. Microbiol. Methods. 2019, 165, 105702. [Google Scholar] [CrossRef] [PubMed]
- Rimsky, S.; Travers, A. Pervasive regulation of nucleoid structure and function by nucleoid-associated proteins. Curr. Opin. Microbiol. 2011, 14, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Ali Azam, T.; Iwata, A.; Nishimura, A.; Ueda, S.; Ishihama, A. Growth phase-dependent variation in protein composition of the Escherichia coli nucleoid. J. Bacteriol. 1999, 181, 6361–6370. [Google Scholar] [CrossRef] [Green Version]
- Talukder, A.A.; Ishihama, A. Growth phase dependent changes in the structure and protein composition of nucleoid in Escherichia coli. Sci. China Life Sci. 2015, 58, 902–911. [Google Scholar] [CrossRef] [Green Version]
- Rafiei, N.; Cordova, M.; Navarre, W.W.; Milstein, J.N. Growth Phase-Dependent Chromosome Condensation and Heat-Stable Nucleoid-Structuring Protein Redistribution in Escherichia coli under Osmotic Stress. J. Bacteriol. 2019, 201, e00469-19. [Google Scholar] [CrossRef]
- Ninnemann, O.; Koch, C.; Kahmann, R. The E. coli fis promoter is subject to stringent control and autoregulation. EMBO J. 1992, 11, 1075–1083. [Google Scholar] [CrossRef]
- Johansson, J.; Balsalobre, C.; Wang, S.Y.; Urbonaviciene, J.; Jin, D.J.; Sondén, B.; Uhlin, B.E. Nucleoid proteins stimulate stringently controlled bacterial promoters: A link between the cAMP-CRP and the (p)ppGpp regulons in Escherichia coli. Cell 2000, 102, 475–485. [Google Scholar] [CrossRef] [Green Version]
- Schneider, R.; Travers, A.; Muskhelishvili, G. The expression of the Escherichia coli fis gene is strongly dependent on the superhelical density of DNA. Mol. Microbiol. 2000, 38, 167–175. [Google Scholar] [CrossRef]
- Szafran, M.J.; Gongerowska, M.; Małecki, T.; Elliot, M.; Jakimowicz, D. Transcriptional Response of Streptomyces coelicolor to Rapid Chromosome Relaxation or Long-Term Supercoiling Imbalance. Front. Microbiol. 2019, 10, 1605. [Google Scholar] [CrossRef] [Green Version]
- Brandi, A.; Giangrossi, M.; Fabbretti, A.; Falconi, M. The hns Gene of Escherichia coli is Transcriptionally Down-Regulated by (p)ppGpp. Microorganisms 2020, 8, 1558. [Google Scholar] [CrossRef] [PubMed]
- Muskhelishvili, G.; Sobetzko, P.; Mehandziska, S.; Travers, A. Composition of Transcription Machinery and Its Crosstalk with Nucleoid-Associated Proteins and Global Transcription Factors. Biomolecules 2021, 11, 924. [Google Scholar] [CrossRef] [PubMed]
- Schneider, R.; Lurz, R.; Lüder, G.; Tolksdorf, C.; Travers, A.; Muskhelishvili, G. An architectural role of the Escherichia coli chromatin protein FIS in organising DNA. Nucleic Acids Res. 2001, 29, 5107–5114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Japaridze, A.; Muskhelishvili, G.; Benedetti, F.; Gavriilidou, A.F.; Zenobi, R.; De Los Rios, P.; Longo, G.; Dietler, G. Hyperplectonemes: A higher order compact and dynamic DNA self-organization. Nano Lett. 2017, 17, 1938–1948. [Google Scholar] [CrossRef] [PubMed]
- Ferrándiz, M.J.; Carreño, D.; Ayora, S.; de la Campa, A.G. HU of Streptococcus pneumoniae Is Essential for the Preservation of DNA Supercoiling. Front. Microbiol. 2018, 9, 493. [Google Scholar] [CrossRef] [Green Version]
- Huang, L.; Zhang, Z.; McMacken, R. Interaction of the Escherichia coli HU Protein with Various Topological Forms of DNA. Biomolecules 2021, 11, 1724. [Google Scholar] [CrossRef]
- Ro, C.; Cashel, M.; Fernández-Coll, L. The secondary messenger ppGpp interferes with cAMP-CRP regulon by promoting CRP acetylation in Escherichia Coli. PLoS ONE 2021, 16, e0259067. [Google Scholar] [CrossRef]
- Dorman, C.J.; Ni Bhriain, N.; Higgins, C.F. DNA supercoiling and environmental regulation of virulence gene expression in Shigella flexneri. Nature 1990, 344, 789–792. [Google Scholar] [CrossRef]
- Colgan, A.M.; Quinn, H.J.; Kary, S.C.; Mitchenall, L.; Maxwell, A.; Cameron, A.D.S.; Dorman, C.J. Negative supercoiling of DNA by gyrase is inhibited in Salmonella enterica serovar Typhimurium during adaptation to acid stress. Mol. Microbiol. 2018, 107, 734–746. [Google Scholar] [CrossRef] [Green Version]
- McClellan, J.A.; Boublikova, P.; Palecek, E.; Lilley, D.M. Superhelical torsion in cellular DNA responds directly to environmental and genetic factors. Proc. Natl. Acad. Sci. USA 1990, 87, 8373–8377. [Google Scholar] [CrossRef] [Green Version]
- Martis, B.S.; Forquet, R.; Reverchon, S.; Nasser, W.; Meyer, S. DNA Supercoiling: An Ancestral Regulator of Gene Expression in Pathogenic Bacteria? Comput. Struct. Biotechnol. J. 2019, 17, 1047–1055. [Google Scholar] [CrossRef] [PubMed]
- Travers, A.A.; Muskhelishvili, G.; Thompson, J.M. DNA information: From digital code to analogue structure. Philos. Trans. A Math. Phys. Eng. Sci. 2012, 370, 2960–2986. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sutthibutpong, T.; Matek, C.; Benham, C.; Slade, G.G.; Noy, A.; Laughton, C.; Doye, J.P.; Louis, A.A.; Harris, S.A. Long-range correlations in the mechanics of small DNA circles under topological stress revealed by multi-scale simulation. Nucleic Acids Res. 2016, 44, 9121–9130. [Google Scholar] [CrossRef] [PubMed]
- Muskhelishvili, G.; Travers, A.A.; Heumann, H.; Kahmann, R. FIS and RNA polymerase holoenzyme form a specific nucleoprotein complex at a stable RNA promoter. EMBO J. 1995, 14, 1446–1452. [Google Scholar] [CrossRef] [PubMed]
- Travers, A.; Muskhelishvili, G. DNA microloops and microdomains: A general mechanism for transcription activation by torsional transmission. J. Mol. Biol. 1998, 279, 1027–1043. [Google Scholar] [CrossRef]
- Sheridan, S.D.; Benham, C.J.; Hatfield, G.W. Inhibition of DNA supercoiling-dependent transcriptional activation by a distant B-DNA to Z-DNA transition. J. Biol. Chem. 1999, 274, 8169–8174. [Google Scholar] [CrossRef] [Green Version]
- Reverchon, S.; Meyer, S.; Forquet, R.; Hommais, F.; Muskhelishvili, G.; Nasser, W. The nucleoid-associated protein IHF acts as a ‘transcriptional domainin’ protein coordinating the bacterial virulence traits with global transcription. Nucleic Acids Res. 2021, 49, 776–790. [Google Scholar] [CrossRef]
- Guptasarma, P. Does replication-induced transcription regulate synthesis of the myriad low copy number proteins of Escherichia coli? Bioessays 1995, 17, 987–997. [Google Scholar] [CrossRef]
- Wang, M.; Zhang, J.; Xu, H.; Golding, I. Measuring transcription at a single gene copy reveals hidden drivers of bacterial individuality. Nat. Microbiol. 2019, 4, 2118–2127. [Google Scholar] [CrossRef]
- Yu, H.; Dröge, P. Replication-induced supercoiling: A neglected DNA transaction regulator? Trends Biochem. Sci. 2014, 39, 219–220. [Google Scholar] [CrossRef]
- Ahmed, S.M.; Dröge, P. Chromatin Architectural Factors as Safeguards against Excessive Supercoiling during DNA Replication. Int. J. Mol. Sci. 2020, 21, 4504. [Google Scholar] [CrossRef] [PubMed]
- Leng, F.; Amado, L.; McMacken, R. Coupling DNA supercoiling to transcription in defined protein systems. J. Biol. Chem. 2004, 279, 47564–47571. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhi, X.; Dages, S.; Dages, K.; Liu, Y.; Hua, Z.C.; Makemson, J.; Leng, F. Transient and dynamic DNA supercoiling potently stimulates the leu-500 promoter in Escherichia coli. J. Biol. Chem. 2017, 292, 14566–14575. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, L.F.; Wang, J.C. Supercoiling of the DNA template during transcription. Proc. Natl. Acad. Sci. USA 1987, 84, 7024–7027. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeong, K.S.; Ahn, J.; Khodursky, A.B. Spatial patterns of transcriptional activity in the chromosome of Escherichia Coli. Genome Biol. 2004, 5, R86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berger, M.; Farcas, A.; Geertz, M.; Zhelyazkova, P.; Brix, K.; Travers, A.; Muskhelishvili, G. Coordination of genomic structure and transcription by the main bacterial nucleoid-associated protein HU. EMBO Rep. 2010, 11, 59–64. [Google Scholar] [CrossRef] [Green Version]
- Muskhelishvili, G.; Travers, A. The regulatory role of DNA supercoiling in nucleoprotein complex assembly and genetic activity. Biophys Rev. 2016, 8, 5–22. [Google Scholar] [CrossRef] [Green Version]
- Balke, V.L.; Gralla, J.D. Changes in the linking number of supercoiled DNA accompany growth transitions in Escherichia Coli. J. Bacteriol. 1987, 169, 4499–4506. [Google Scholar] [CrossRef] [Green Version]
- Lal, A.; Dhar, A.; Trostel, A.; Kouzine, F.; Seshasayee, A.S.; Adhya, S. Genome scale patterns of supercoiling in a bacterial chromosome. Nat. Commun. 2016, 7, 11055. [Google Scholar] [CrossRef] [Green Version]
- Visser, B.J.; Sharma, S.; Chen, P.J.; McMullin, A.B.; Bates, M.; Bates, D. Psoralen mapping reveals a bacterial genome supercoiling landscape dominated by transcription. Nucleic Acids Res. 2022, 50, 4436–4449. [Google Scholar] [CrossRef]
- Irobalieva, R.N.; Fogg, J.M.; Catanese, D.J., Jr.; Sutthibutpong, T.; Chen, M.; Barker, A.K.; Ludtke, S.J.; Harris, S.A.; Schmid, M.F.; Chiu, W.; et al. Structural diversity of supercoiled DNA. Nat. Commun. 2015, 6, 8440. [Google Scholar] [CrossRef] [PubMed]
- Ellison, M.J.; Fenton, M.J.; Ho, P.S.; Rich, A. Long-range interactions of multiple DNA structural transitions within a common topological domain. EMBO J. 1987, 6, 1513–1522. [Google Scholar] [CrossRef]
- Kelleher, R.J., 3rd; Ellison, M.J.; Ho, P.S.; Rich, A. Competitive behavior of multiple, discrete B-Z transitions in supercoiled DNA. Proc. Natl. Acad. Sci. USA 1986, 83, 6342–6346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aboul-ela, F.; Bowater, R.P.; Lilley, D.M. Competing B-Z and helix-coil conformational transitions in supercoiled plasmid DNA. J. Biol. Chem. 1992, 267, 1776–1785. [Google Scholar] [CrossRef]
- Kim, S.H.; Jung, H.J.; Lee, I.B.; Lee, N.K.; Hong, S.C. Sequence-dependent cost for Z-form shapes the torsion-driven B-Z transition via close interplay of Z-DNA and DNA bubble. Nucleic Acids Res. 2021, 49, 3651–3660. [Google Scholar] [CrossRef]
- Vlijm, R.; vd Torre, J.; Dekker, C. Counterintuitive DNA Sequence Dependence in Supercoiling-Induced DNA Melting. PLoS ONE 2015, 10, e0141576. [Google Scholar] [CrossRef]
- Jáuregui, R.; Abreu-Goodger, C.; Moreno-Hagelsieb, G.; Collado-Vides, J.; Merino, E. Conservation of DNA curvature signals in regulatory regions of prokaryotic genes. Nucleic Acids Res. 2003, 31, 6770–6777. [Google Scholar] [CrossRef] [Green Version]
- Olivares-Zavaleta, N.; Jáuregui, R.; Merino, E. Genome analysis of Escherichia coli promoter sequences evidences that DNA static curvature plays a more important role in gene transcription than has previously been anticipated. Genomics 2006, 87, 329–337. [Google Scholar] [CrossRef] [Green Version]
- Kravatskaya, G.I.; Chechetkin, V.R.; Kravatsky, Y.V.; Tumanyan, V.G. Structural attributes of nucleotide sequences in promoter regions of supercoiling-sensitive genes: How to relate microarray expression data with genomic sequences. Genomics 2013, 101, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Du, X.; Wojtowicz, D.; Bowers, A.A.; Levens, D.; Benham, C.J.; Przytycka, T.M. The genome-wide distribution of non-B DNA motifs is shaped by operon structure and suggests the transcriptional importance of non-B DNA structures in Escherichia coli. Nucleic Acids Res. 2013, 41, 5965–5977. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.H.; Ganji, M.; Kim, E.; van der Torre, J.; Abbondanzieri, E.; Dekker, C. DNA sequence encodes the position of DNA supercoils. Elife 2018, 7, e36557. [Google Scholar] [CrossRef] [PubMed]
- Borowiec, J.A.; Gralla, J.D. All three elements of the lac ps promoter mediate its transcriptional response to DNA supercoiling. J. Mol. Biol. 1987, 195, 89–97. [Google Scholar] [CrossRef]
- Auner, H.; Buckle, M.; Deufel, A.; Kutateladze, T.; Lazarus, L.; Mavathur, R.; Muskhelishvili, G.; Pemberton, I.; Schneider, R.; Travers, A. Mechanism of transcriptional activation by FIS: Role of core promoter structure and DNA topology. J. Mol. Biol. 2003, 331, 331–344. [Google Scholar] [CrossRef]
- Peter, B.J.; Arsuaga, J.; Breier, A.M.; Khodursky, A.B.; Brown, P.O.; Cozzarelli, N.R. Genomic transcriptional response to loss of chromosomal supercoiling in Escherichia coli. Genome Biol. 2004, 5, R87. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Forquet, R.; Pineau, M.; Nasser, W.; Reverchon, S.; Meyer, S. Role of the Discriminator Sequence in the Supercoiling Sensitivity of Bacterial Promoters. mSystems. 2021, 6, e0097821. [Google Scholar] [CrossRef]
- Klein, C.A.; Teufel, M.; Weile, C.J.; Sobetzko, P. The bacterial promoter spacer modulates promoter strength and timing by length, TG-motifs and DNA supercoiling sensitivity. Sci. Rep. 2021, 11, 24399. [Google Scholar] [CrossRef]
- Lamond, A.I.; Travers, A.A. Requirement for an upstream element for optimal transcription of a bacterial tRNA gene. Nature 1983, 305, 248–250. [Google Scholar] [CrossRef]
- Maurer, S.; Fritz, J.; Muskhelishvili, G.; Travers, A. RNA polymerase and an activator form discrete subcomplexes in a transcription initiation complex. EMBO J. 2006, 25, 3784–3790. [Google Scholar] [CrossRef] [Green Version]
- Postow, L.; Hardy, C.D.; Arsuaga, J.; Cozzarelli, N.R. Topological domain structure of the Escherichia coli chromosome. Genes Dev. 2004, 18, 1766–1779. [Google Scholar] [CrossRef] [Green Version]
- Stein, R.A.; Deng, S.; Higgins, N.P. Measuring chromosome dynamics on different time scales using resolvases with varying half-lives. Mol. Microbiol. 2005, 56, 1049–1061. [Google Scholar] [CrossRef] [Green Version]
- Cunha, S.; Woldringh, C.L.; Odijk, T. Restricted diffusion of DNA segments within the isolated Escherichia coli nucleoid. J. Struct. Biol. 2005, 150, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Hardy, C.D.; Cozzarelli, N.R. A genetic selection for supercoiling mutants of Escherichia coli reveals proteins implicated in chromosome structure. Mol. Microbiol. 2005, 57, 1636–1652. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Japaridze, A.; Zheng, X.; Wiktor, J.; Kerssemakers, J.W.J.; Dekker, C. Direct imaging of the circular chromosome in a live bacterium. Nat. Commun. 2019, 10, 2194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Le, T.B.; Imakaev, M.V.; Mirny, L.A.; Laub, M.T. High-resolution mapping of the spatial organization of a bacterial chromosome. Science 2013, 342, 731–734. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gerganova, V.; Maurer, S.; Stoliar, L.; Japaridze, A.; Dietler, G.; Nasser, W.; Kutateladze, T.; Travers, A.; Muskhelishvili, G. Upstream binding of idling RNA polymerase modulates transcription initiation from a nearby promoter. J. Biol. Chem. 2015, 290, 8095–8109. [Google Scholar] [CrossRef] [Green Version]
- Jiang, X.; Sobetzko, P.; Nasser, W.; Reverchon, S.; Muskhelishvili, G. Chromosomal “stress-response” domains govern the spatiotemporal expression of the bacterial virulence program. mBio 2015, 6, e00353-15. [Google Scholar] [CrossRef] [Green Version]
- Ferrándiz, M.J.; Martín-Galiano, A.J.; Schvartzman, J.B.; de la Campa, A.G. The genome of Streptococcus pneumoniae is organized in topology-reacting gene clusters. Nucleic Acids Res. 2010, 38, 3570–3581. [Google Scholar] [CrossRef] [Green Version]
- Tupper, A.E.; Owen-Hughes, T.A.; Ussery, D.W.; Santos, D.S.; Ferguson, D.J.; Sidebotham, J.M.; Hinton, J.C.; Higgins, C.F. The chromatin-associated protein H-NS alters DNA topology in vitro. EMBO J. 1994, 13, 258–268. [Google Scholar] [CrossRef]
- Rochman, M.; Aviv, M.; Glaser, G.; Muskhelishvili, G. Promoter protection by a transcription factor acting as a local topological homeostat. EMBO Rep. 2002, 3, 355–360. [Google Scholar] [CrossRef] [Green Version]
- Malik, M.; Bensaid, A.; Rouviere-Yaniv, J.; Drlica, K. Histone-like protein HU and bacterial DNA topology: Suppression of an HU deficiency by gyrase mutations. J. Mol. Biol. 1996, 256, 66–76. [Google Scholar] [CrossRef]
- Berger, M.; Gerganova, V.; Berger, P.; Rapiteanu, R.; Lisicovas, V.; Dobrindt, U. Genes on a Wire: The Nucleoid-Associated Protein HU Insulates Transcription Units in Escherichia coli. Sci. Rep. 2016, 6, 31512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marr, C.; Geertz, M.; Hütt, M.-T.; Muskhelishvili, G. Dissecting the logical types of network control in gene expression profiles. BMC Syst. Biol. 2008, 2, 18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muskhelishvili, G.; Forquet, R.; Reverchon, S.; Meyer, S.; Nasser, W. Coherent Domains of Transcription Coordinate Gene Expression During Bacterial Growth and Adaptation. Microorganisms 2019, 7, 694. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meyer, S.; Reverchon, S.; Nasser, W.; Muskhelishvili, G. Chromosomal organization of transcription: In a nutshell. Curr.Genet. 2017, 64, 555–565. [Google Scholar] [CrossRef] [PubMed]
- De la Campa, A.G.; Ferrándiz, M.J.; Martín-Galiano, A.J.; García, M.T.; Tirado-Vélez, J.M. The Transcriptome of Streptococcus pneumoniae Induced by Local and Global Changes in Supercoiling. Front. Microbiol. 2017, 8, 1447. [Google Scholar] [CrossRef] [Green Version]
- Martín-Galiano, A.J.; Ferrándiz, M.J.; de la Campa, A.G. Bridging Chromosomal Architecture and Pathophysiology of Streptococcus pneumoniae. Genome Biol Evol. 2017, 9, 350–361. [Google Scholar] [CrossRef] [Green Version]
- Nigatu, D.; Henkel, W.; Sobetzko, P.; Muskhelishvili, G. Relationship between digital information and thermodynamic stability in bacterial genomes. EURASIP J. Bioinform. Syst. Biol. 2016, 2016, 4. [Google Scholar] [CrossRef] [Green Version]
- Whitson, P.A.; Hsieh, W.T.; Wells, R.D.; Matthews, K.S. Supercoiling facilitates lac operator-repressor-pseudooperator interactions. J. Biol. Chem. 1987, 262, 4943–4946. [Google Scholar] [CrossRef]
- Higgins, N.P.; Collier, D.A.; Kilpatrick, M.W.; Krause, H.M. Supercoiling and integration host factor change the DNA conformation and alter the flow of convergent transcription in phage Mu. J. Biol. Chem. 1989, 264, 3035–3042. [Google Scholar] [CrossRef]
- Pontiggia, A.; Negri, A.; Beltrame, M.; Bianchi, M.E. Protein HU binds specifically to kinked DNA. Mol. Microbiol. 1993, 7, 343–350. [Google Scholar] [CrossRef]
- Wei, J.; Czapla, L.; Grosner, M.A.; Swigon, D.; Olson, W.K. DNA topology confers sequence specificity to nonspecific architectural proteins. Proc. Natl. Acad. Sci. USA 2014, 111, 16742–16747. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Babbitt, G.A.; Schulze, K.V. Codons support the maintenance of intrinsic DNA polymer flexibility over evolutionary timescales. Genome Biol. Evol. 2012, 4, 954–965. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fisunov, G.Y.; Zubov, A.I.; Pobeguts, O.V.; Varizhuk, A.M.; Galyamina, M.A.; Evsyutina, D.V.; Semashko, T.A.; Manuvera, V.A.; Kovalchuk, S.I.; Ziganshin, R.K.; et al. The Dynamics of Mycoplasma gallisepticum Nucleoid Structure at the Exponential and Stationary Growth Phases. Front. Microbiol. 2021, 12, 753760. [Google Scholar] [CrossRef] [PubMed]
- Ohniwa, R.L.; Muchaku, H.; Saito, S.; Wada, C.; Morikawa, K. Atomic force microscopy analysis of the role of major DNA-binding proteins in organization of the nucleoid in Escherichia coli. PLoS ONE 2013, 8, e72954. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frenkiel-Krispin, D.; Ben-Avraham, I.; Englander, J.; Shimoni, E.; Wolf, S.G.; Minsky, A. Nucleoid restructuring in stationary-state bacteria. Mol. Microbiol. 2004, 51, 395–405. [Google Scholar] [CrossRef] [PubMed]
- Fraebel, D.T.; Mickalide, H.; Schnitkey, D.; Merritt, J.; Kuhlman, T.E.; Kuehn, S. Environment determines evolutionary trajectory in a constrained phenotypic space. eLife 2017, 6, e24669. [Google Scholar] [CrossRef]
- Muskhelishvili, G.; Travers, A. Order from the order: How a spatiotemporal genetic program is encoded in a 2-D genetic map of the bacterial chromosome. J. Mol. Microbiol. Biotechnol. 2014, 24, 332–343. [Google Scholar] [CrossRef]
- Cameron, A.D.S.; Dillon, S.C.; Kröger, C.; Beran, L.; Dorman, C.J. Broad-scale redistribution of mRNA abundance and transcriptional machinery in response to growth rate in Salmonella enterica serovar Typhimurium. Microb. Genom. 2017, 3, e000127. [Google Scholar] [CrossRef]
- Carpentier, A.S.; Torrésani, B.; Grossmann, A.; Hénaut, A. Decoding the nucleoid organisation of Bacillus subtilis and Escherichia coli through gene expression data. BMC Genom. 2005, 6, 84. [Google Scholar] [CrossRef] [Green Version]
- Le Berre, D.; Reverchon, S.; Muskhelishvili, G.; Nasser, W. Relationship between the Chromosome Structural Dynamics and Gene Expression-A Chicken and Egg Dilemma? Microorganisms 2022, 10, 846. [Google Scholar] [CrossRef]
- Weng, X.; Bohrer, C.H.; Bettridge, K.; Lagda, A.C.; Cagliero, C.; Jin, D.J.; Xiao, J. Spatial organization of RNA polymerase and its relationship with transcription in Escherichia coli. Proc. Natl. Acad. Sci. USA 2019, 116, 20115–20123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krause, K.; Maciąg-Dorszyńska, M.; Wosinski, A.; Gaffke, L.; Morcinek-Orłowska, J.; Rintz, E.; Bielańska, P.; Szalewska-Pałasz, A.; Muskhelishvili, G.; Węgrzyn, G. The Role of Metabolites in the Link between DNA Replication and Central Carbon Metabolism in Escherichia coli. Genes 2020, 11, 447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schneider, R.; Travers, A.; Kutateladze, T.; Muskhelishvili, G. A DNA architectural protein couples cellular physiology and DNA topology in Escherichia Coli. Mol. Microbiol. 1999, 34, 953–964. [Google Scholar] [CrossRef] [PubMed]
- Sobetzko, P.; Glinkowska, M.; Muskhelishvili, G. GSE65244: Temporal Gene Expression in Escherichia coli. Gene Expression Omnibus. 2017. Available online: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE65244 (accessed on 20 June 2021).
- Hsieh, L.S.; Burger, R.M.; Drlica, K. Bacterial DNA supercoiling and [ATP]/[ADP]. Changes associated with a transition to anaerobic growth. J. Mol. Biol. 1991, 219, 443–450. [Google Scholar] [CrossRef]
- Van Workum, M.; Dooren, S.J.M.; Oldenburg, N.; Molenaar, D.; Jensen, P.R.; Snoep, J.L.; Westerhoff, H. DNA supercoiling depends on the phosphorylation potential in Escherichia coli. Mol. Microbiol. 1996, 20, 351–360. [Google Scholar] [CrossRef]
- Snoep, J.L.; van der Weijden, C.C.; Andersen, H.W.; Westerhoff, H.V.; Jensen, P.R. DNA supercoiling in Escherichia coli is under tight and subtle homeostatic control, involving gene-expression and metabolic regulation of both topoisomerase I and DNA gyrase. Eur. J. Biochem. 2002, 269, 1662–1669. [Google Scholar] [CrossRef]
- Sutormin, D.; Rubanova, N.; Logacheva, M.; Ghilarov, D.; Severinov, K. Single-nucleotide-resolution mapping of DNA gyrase cleavage sites across the Escherichia coli genome. Nucleic Acids Res. 2018, 47, 1373–1388. [Google Scholar] [CrossRef] [Green Version]
- Ball, C.A.; Osuna, R.; Ferguson, K.C.; Johnson, R.C. Dramatic changes in Fis levels upon nutrient upshift in Escherichia Coli. J. Bacteriol. 1992, 174, 8043–8056. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Bremer, H. Effects of Fis on ribosome synthesis and activity and on rRNA promoter activities in Escherichia Coli. J. Mol. Biol. 1996, 259, 27–40. [Google Scholar] [CrossRef]
- Bednar, J.; Furrer, P.; Stasiak, A.; Dubochet, J.; Egelman, E.H.; Bates, A.D. The twist, writhe and overall shape of supercoiled DNA change during counterion-induced transition from a loosely to a tightly interwound superhelix. Possible implications for DNA structure in vivo. J. Mol. Biol. 1994, 235, 825–847. [Google Scholar] [CrossRef]
- ten Heggeler-Bordier, B.; Wahli, W.; Adrian, M.; Stasiak, A.; Dubochet, J. The apical localization of transcribing RNA polymerases on supercoiled DNA prevents their rotation around the template. EMBO J. 1992, 11, 667–672. [Google Scholar] [CrossRef]
- Rivetti, C.; Guthold, M.; Bustamante, C. Wrapping of DNA around the E. coli RNA polymerase open promoter complex. EMBO J. 1999, 18, 4464–4475. [Google Scholar] [CrossRef] [Green Version]
- Kusano, S.; Ding, Q.; Fujita, N.; Ishihama, A. Promoter selectivity of Escherichia coli RNA polymerase Eσ70 and Eσ38 Holoenzymes. Effect of DNA supercoiling. J. Biol. Chem. 1996, 271, 1998–2004. [Google Scholar] [CrossRef] [Green Version]
- Minakhin, L.; Bhagat, S.; Brunning, A.; Campbell, E.A.; Darst, S.A.; Ebright, R.H.; Severinov, K. Bacterial RNA polymerase subunit omega and eukaryotic RNA polymerase subunit RPB6 are sequence, structural, and functional homologs and promote RNA polymerase assembly. Proc. Natl. Acad. Sci. USA 2001, 98, 892–897. [Google Scholar] [CrossRef] [Green Version]
- Gentry, D.; Xiao, H.; Burgess, R.; Cashel, M. The omega subunit of Escherichia coli K-12 RNA polymerase is not required for stringent RNA control in vivo. J. Bacteriol. 1991, 173, 3901–3903. [Google Scholar] [CrossRef] [Green Version]
- Claret, L.; Rouviere-Yaniv, J. Regulation of HU alpha and HU beta by CRP and FIS in Escherichia Coli. J. Mol. Biol. 1996, 263, 126–139. [Google Scholar] [CrossRef]
- Cabrera, J.E.; Jin, D.J. The distribution of RNA polymerase in Escherichia coli is dynamic and sensitive to environmental cues. Mol. Microbiol. 2003, 50, 1493–1505. [Google Scholar] [CrossRef]
- Schneider, R.; Travers, A.; Muskhelishvili, G. FIS modulates growth phase-dependent topological transitions of DNA in Escherichia Coli. Mol. Microbiol. 1997, 26, 519–530. [Google Scholar] [CrossRef]
- Broyles, S.S.; Pettijohn, D.E. Interaction of the Escherichia coli HU protein with DNA. Evidence for formation of nucleosome-like structures with altered DNA helical pitch. J. Mol. Biol. 1986, 187, 47–60. [Google Scholar] [CrossRef]
- Kraemer, J.A.; Sanderlin, A.G.; Laub, M.T. The Stringent Response Inhibits DNA Replication Initiation in E. coli by Modulating Supercoiling of oriC. mBio 2019, 10, e01330-19. [Google Scholar] [CrossRef] [Green Version]
- Antipov, S.; Tutukina, M.N.; Preobrazhenskaya, E.V.; Kondrashov, F.A.; Patrushev, M.V.; Toshchakov, S.V.; Dominova, I.; Shvyreva, U.S.; Vrublevskaya, V.V.; Morenkov, O.S.; et al. The nucleoid protein Dps binds genomic DNA of Escherichia coli in a non-random manner. PLoS ONE 2017, 12, e0182800. [Google Scholar] [CrossRef] [PubMed]
- Dadinova, L.A.; Chesnokov, Y.M.; Kamyshinsky, R.A.; Orlov, I.A.; Petoukhov, M.V.; Mozhaev, A.A.; Soshinskaya, E.Y.; Lazarev, V.N.; Manuvera, V.A.; Orekhov, A.S.; et al. Protective Dps-DNA co-crystallization in stressed cells: An in vitro structural study by small-angle X-ray scattering and cryo-electron tomography. FEBS Lett. 2019, 593, 1360–1371. [Google Scholar] [CrossRef] [PubMed]
- Hammel, M.; Amlanjyoti, D.; Reyes, F.E.; Chen, J.H.; Parpana, R.; Tang, H.Y.; Larabell, C.A.; Tainer, J.A.; Adhya, S. HU multimerization shift controls nucleoid compaction. Sci. Adv. 2016, 2, e1600650. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ohniwa, R.L.; Morikawa, K.; Kim, J.; Ohta, T.; Ishihama, A.; Wada, C.; Takeyasu, K. Dynamic state of DNA topology is essential for genome condensation in bacteria. EMBO J. 2006, 25, 5591–5602. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Lim, C.J.; Dröge, P.; Yan, J. Regulation of Bacterial DNA Packaging in Early Stationary Phase by Competitive DNA Binding of Dps and IHF. Sci. Rep. 2015, 5, 18146. [Google Scholar] [CrossRef] [Green Version]
- Graham, J.S.; Johnson, R.C.; Marko, J.F. Concentration-dependent exchange accelerates turnover of proteins bound to double-stranded DNA. Nucleic Acids Res. 2011, 39, 2249–2259. [Google Scholar] [CrossRef] [Green Version]
- Kamar, R.I.; Banigan, E.J.; Erbas, A.; Giuntoli, R.D.; Olvera de la Cruz, M.; Johnson, R.C.; Marko, J.F. Facilitated dissociation of transcription factors from single DNA binding sites. Proc. Natl. Acad. Sci. USA 2017, 114, E3251–E3257. [Google Scholar] [CrossRef] [Green Version]
- Tsai, M.Y.; Zhang, B.; Zheng, W.; Wolynes, P.G. Molecular Mechanism of Facilitated Dissociation of Fis Protein from DNA. J. Am. Chem. Soc. 2016, 138, 13497–13500. [Google Scholar] [CrossRef] [Green Version]
- Kasho, K.; Oshima, T.; Chumsakul, O.; Nakamura, K.; Fukamachi, K.; Katayama, T. Whole-Genome Analysis Reveals That the Nucleoid Protein IHF Predominantly Binds to the Replication Origin oriC Specifically at the Time of Initiation. Front. Microbiol. 2021, 12, 697712. [Google Scholar] [CrossRef]
- Flåtten, I.; Skarstad, K. The Fis protein has a stimulating role in initiation of replication in Escherichia coli in vivo. PLoS ONE 2013, 8, e83562. [Google Scholar] [CrossRef] [Green Version]
- Higgins, C.F.; Dorman, C.J.; Stirling, D.A.; Waddell, L.; Booth, I.R.; May, G.; Bremer, E. A physiological role for DNA supercoiling in the osmotic regulation of gene expression in S. typhimurium and E. Coli. Cell 1988, 52, 569–584. [Google Scholar]
- Hsieh, L.S.; Rouviere-Yaniv, J.; Drlica, K. Bacterial DNA supercoiling and [ATP]/[ADP] ratio: Changes associated with salt shock. J. Bacteriol. 1991, 173, 3914–3917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tse-Dinh, Y.C.; Qi, H.; Menzel, R. DNA supercoiling and bacterial adaptation: Thermotolerance and thermoresistance. Trends Microbiol. 1997, 5, 323–326. [Google Scholar] [CrossRef]
- Geertz, M.; Travers, A.; Mehandziska, S.; Sobetzko, P.; Janga, S.C.; Shimamoto, N.; Muskhelishvili, G. Structural coupling between RNA polymerase composition and DNA supercoiling in coordinating transcription: A global role for the omega subunit? mBio 2011, 2, e00034-11. [Google Scholar] [CrossRef] [Green Version]
- Bordes, P.; Conter, A.; Morales, V.; Bouvier, J.; Kolb, A.; Gutierrez, C. DNA supercoiling contributes to disconnect σS accumulation from σS-dependent transcription in Escherichia coli. Mol. Microbiol. 2003, 48, 561–571. [Google Scholar] [CrossRef]
- Vijayan, V.; Zuzow, R.; O’Shea, E.K. Oscillations in supercoiling drive circadian gene expression in cyanobacteria. Proc. Natl. Acad. Sci. USA 2009, 106, 22564–22568. [Google Scholar] [CrossRef] [Green Version]
- Crozat, E.; Philippe, N.; Lenski, R.E.; Geiselmann, J.; Schneider, D. Long-term experimental evolution in Escherichia coli. XII. DNA topology as a key target of selection. Genetics 2005, 169, 523–532. [Google Scholar] [CrossRef] [Green Version]
- Heyde, S.A.H.; Frendorf, P.O.; Lauritsen, I.; Nørholm, M.H.H. Restoring Global Gene Regulation through Experimental Evolution Uncovers a NAP (Nucleoid-Associated Protein)-Like Behavior of Crp/Cap. mBio 2021, 12, e0202821. [Google Scholar] [CrossRef]
- Yan, Y.; Xu, W.; Kumar, S.; Zhang, A.; Leng, F.; Dunlap, D.; Finzi, L. Negative DNA supercoiling makes protein-mediated looping deterministic and ergodic within the bacterial doubling time. Nucleic Acids Res. 2021, 49, 11550–11559. [Google Scholar] [CrossRef]
- Muskhelishvili, G.; Buckle, M.; Heumann, H.; Kahmann, R.; Travers, A.A. FIS activates sequential steps during transcription initiation at a stable RNA promoter. EMBO J. 1997, 16, 3655–3665. [Google Scholar] [CrossRef] [Green Version]
- Sheridan, S.; Benham, C.J.; Hatfield, G.W. Activation of gene expression by a novel DNA structural transmission mechanism that requires supercoiling-induced DNA duplex destabilization in an upstream activating sequence. J. Biol. Chem. 1998, 273, 21298–21308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- González-Gil, G.; Kahmann, R.; Muskhelishvili, G. Regulation of crp transcription by oscillation between distinct nucleoprotein complexes. EMBO J. 1998, 17, 2877–2885. [Google Scholar] [CrossRef] [PubMed]
- Nasser, W.; Schneider, R.; Travers, A.; Muskhelishvili, G. CRP modulates fis transcription by alternate formation of activating and repressing nucleoprotein complexes. J. Biol. Chem. 2001, 276, 17878–17886. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Browning, D.F.; Cole, J.A.; Busby, S.J. Transcription activation by remodelling of a nucleoprotein assembly: The role of NarL at the FNR-dependent Escherichia coli nir promoter. Mol. Microbiol. 2004, 53, 203–215. [Google Scholar] [CrossRef]
- Duprey, A.; Muskhelishvili, G.; Reverchon, S.; Nasser, W. Temporal control of Dickeya dadantii main virulence gene expression by growth phase-dependent alteration of regulatory nucleoprotein complexes. Biochim. Biophys. Acta. 2016, 1859, 1470–1480. [Google Scholar] [CrossRef]
- Dorman, C.J. DNA supercoiling and transcription in bacteria: A two-way street. BMC Mol. Cell. Biol. 2019, 20, 26. [Google Scholar] [CrossRef] [Green Version]
- Fulcrand, G.; Zhi, X.; Leng, F. Transcription-coupled DNA supercoiling in defined protein systems and in E. coli topA mutant strains. IUBMB Life 2013, 65, 615–622. [Google Scholar] [CrossRef] [Green Version]
- Meyer, S.; Beslon, G. Torsion-mediated interaction between adjacent genes. PLOS Comput. Biol. 2014, 10, e1003785. [Google Scholar] [CrossRef] [Green Version]
- Sobetzko, P. Transcription-coupled DNA supercoiling dictates the chromosomal arrangement of bacterial genes. Nucleic Acids Res. 2016, 44, 1514–1524. [Google Scholar] [CrossRef]
- El Houdaigui, B.; Forquet, R.; Hindré, T.; Schneider, D.; Nasser, W.; Reverchon, S.; Meyer, S. Bacterial genome architecture shapes global transcriptional regulation by DNA supercoiling. Nucleic Acids Res. 2019, 47, 5648–5657. [Google Scholar] [CrossRef] [Green Version]
- Figueroa-Bossi, N.; Guérin, M.; Rahmouni, R.; Leng, M.; Bossi, L. The supercoiling sensitivity of a bacterial tRNA promoter parallels its responsiveness to stringent control. EMBO J. 1998, 17, 2359–2367. [Google Scholar] [CrossRef] [Green Version]
- SantaLucia, J., Jr. A unified view of polymer, dumbbell, and oligonucleotide DNA nearest-neighbor thermodynamics. Proc. Natl. Acad. Sci. USA 1998, 95, 1460–1465. [Google Scholar] [CrossRef] [Green Version]
- Chapman, A.G.; Fall, L.; Atkinson, D.E. Adenylate energy charge in Escherichia coli during growth and starvation. J. Bacteriol. 1971, 108, 1072–1086. [Google Scholar] [CrossRef] [Green Version]
- Schultz, S.G.; Solomon, A.K. Cation transport in Escherichia coli. I. Intracellular Na and K concentrations and net cation movement. J. Gen. Physiol. 1961, 45, 355–369. [Google Scholar] [CrossRef] [Green Version]
- Kashket, E.R.; Barker, S.L. Effects of potassium ions on the electrical and pH gradients across the membrane of Streptococcus lactis cells. J. Bacteriol. 1977, 130, 1017–1023. [Google Scholar] [CrossRef] [Green Version]
- Castanie-Cornet, M.P.; Penfound, T.A.; Smith, D.; Elliott, J.F.; Foster, J.W. Control of acid resistance in Escherichia coli. J. Bacteriol. 1999, 181, 3525–3535. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.Y.; Drlica, K.; Syvanen, M. Monovalent cations differ in their effects on transcription initiation from a sigma-70 promoter of Escherichia Coli. Gene. 1997, 196, 95–98. [Google Scholar] [CrossRef]
- Scolari, V.F.; Bassetti, B.; Sclavi, B.; Lagomarsino, M.C. Gene clusters reflecting macrodomain structure respond to nucleoid perturbations. Mol. Biosyst. 2011, 7, 878–888. [Google Scholar] [CrossRef] [Green Version]
- Kahramanoglou, C.; Seshasayee, A.S.; Prieto, A.I.; Ibberson, D.; Schmidt, S.; Zimmermann, J.; Benes, V.; Fraser, G.M.; Luscombe, N.M. Direct and indirect effects of H-NS and Fis on global gene expression control in Escherichia Coli. Nucleic Acids Res. 2011, 39, 2073–2091. [Google Scholar] [CrossRef]
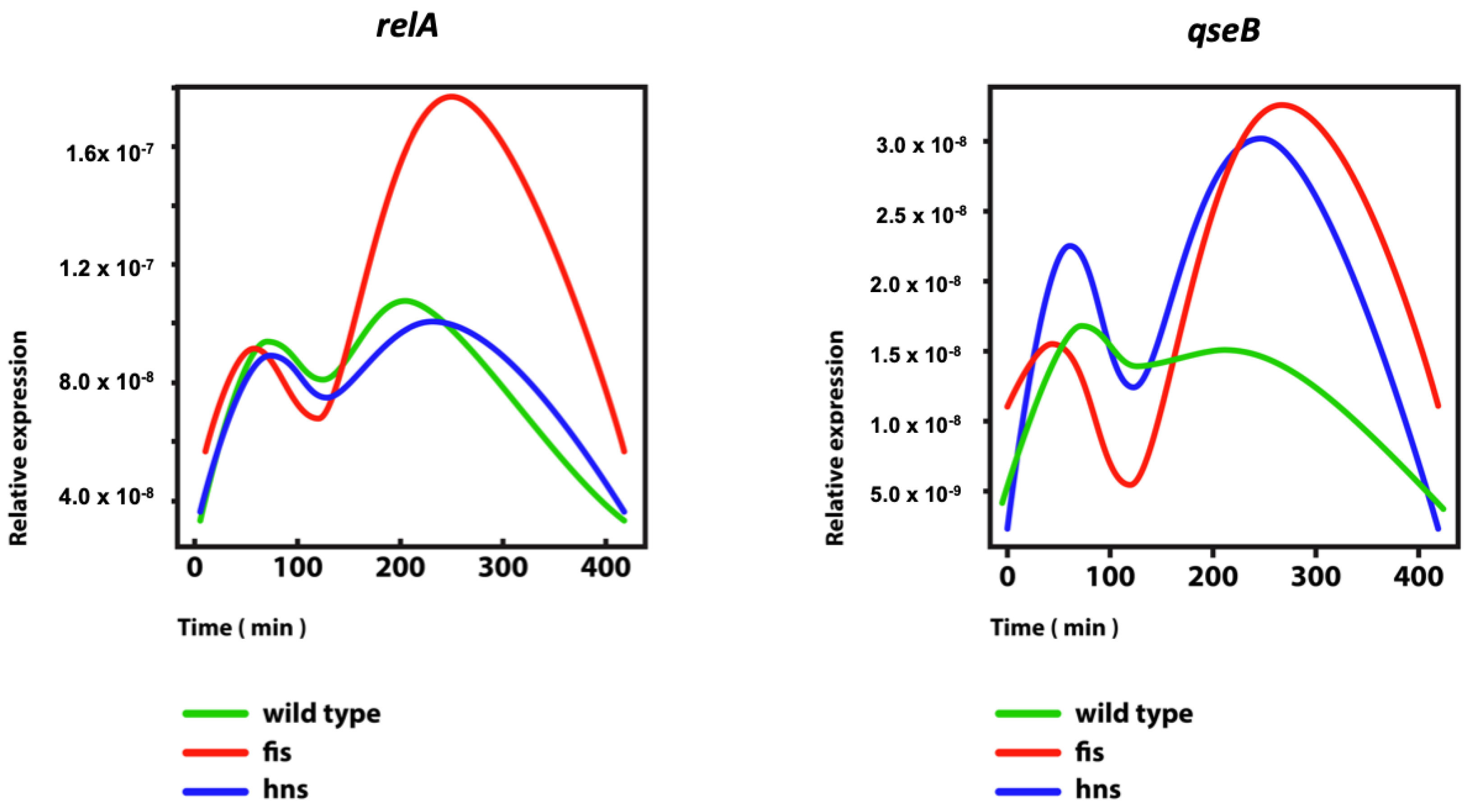
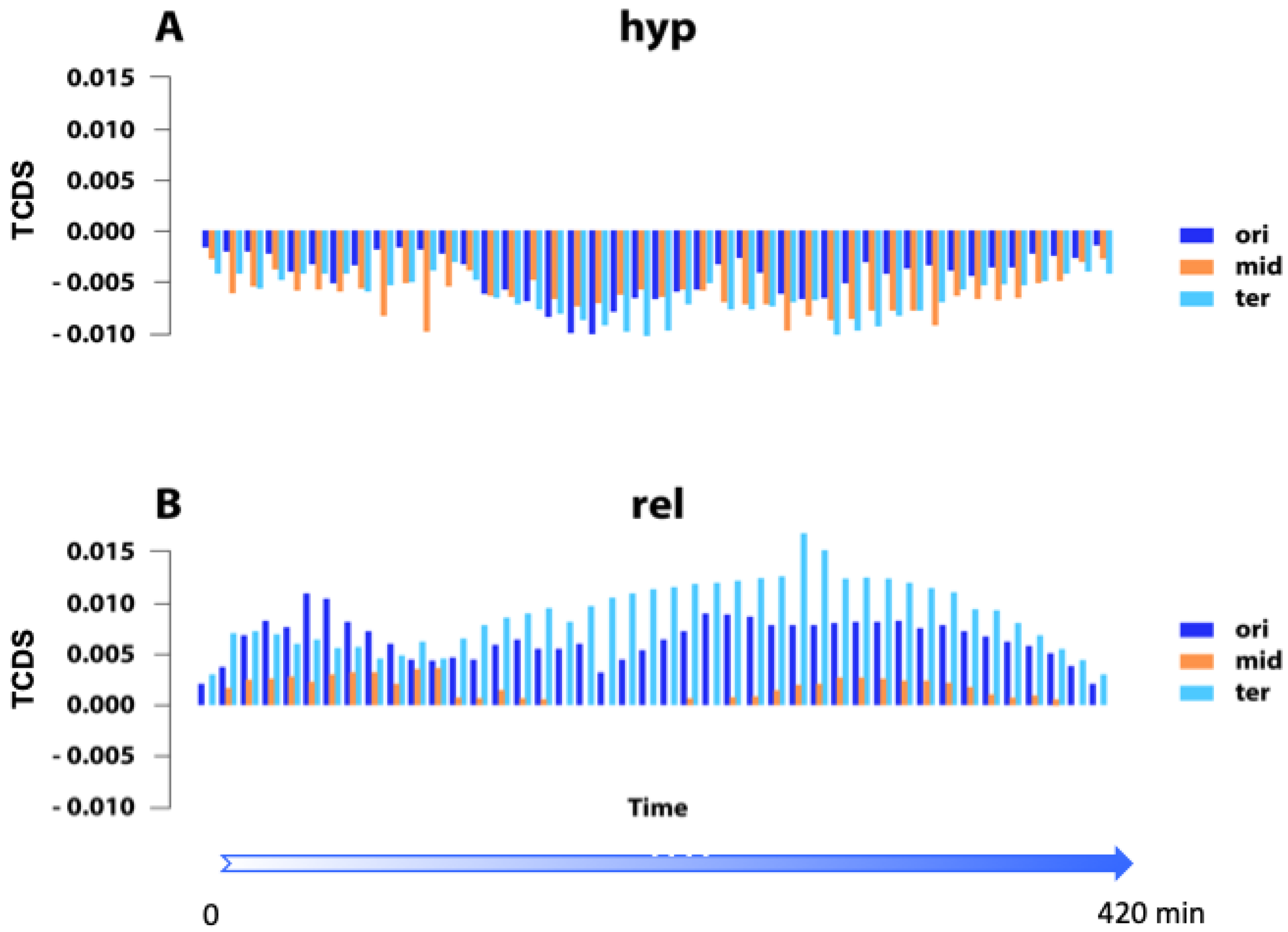
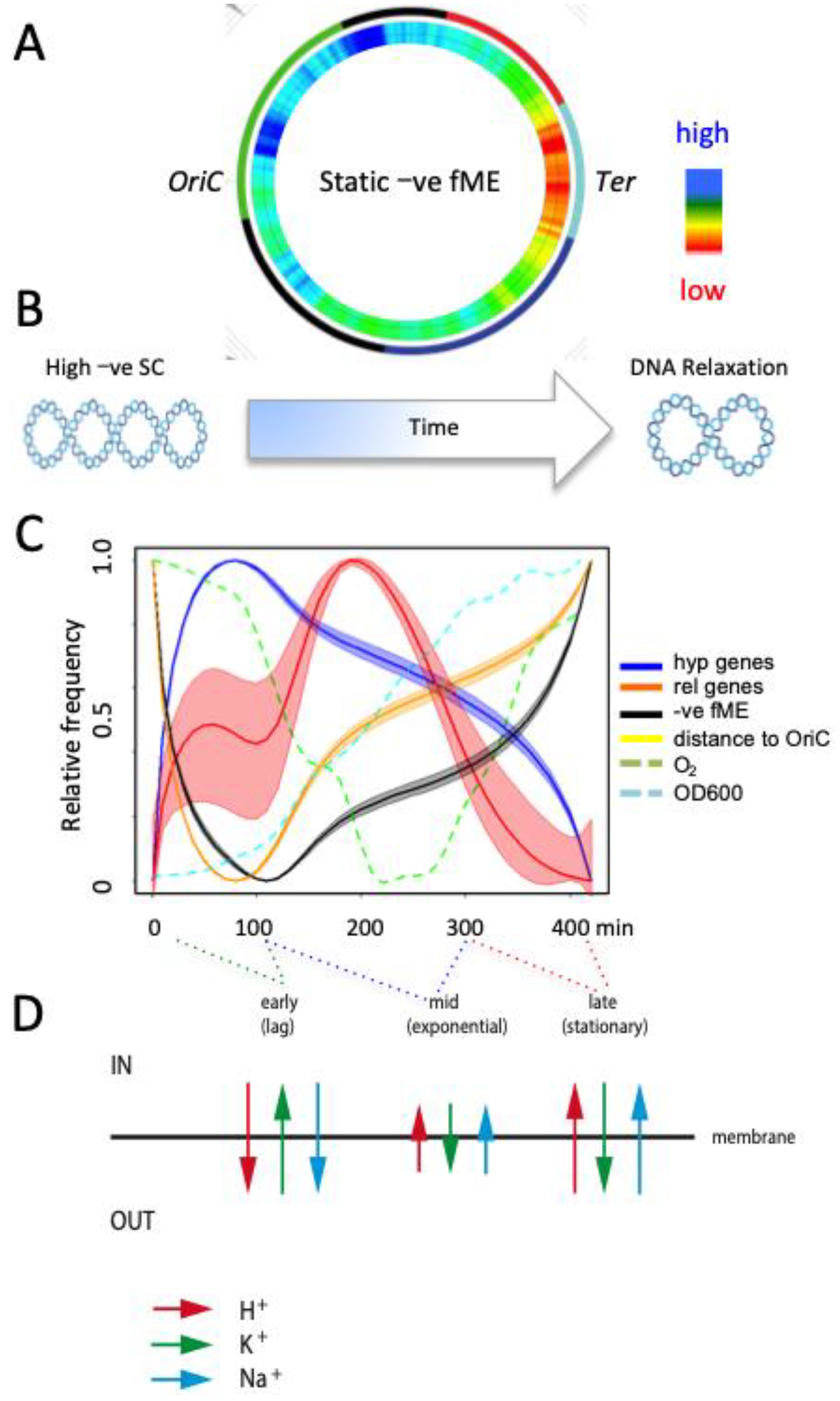
| Gene Promoter | Distance from OriC (bp) | Condition | Optimal * Superhelical Density (σ) | #References |
|---|---|---|---|---|
| hisR | 62,500 | In vitro | ~−0.08 to −0.1 | [172] |
| rrnAP1 | 120,000 | In vitro | ~−0.076 | [99] |
| fis | 508,500 | In vitro & in vivo | ~−0.07-to −0.08 | [39] |
| tyrT | 1,967,600 | In vitro | ~−0.05–0.06 | [83] |
| osmE | 2,097,900 | In vitro | ~−0.03–0.04 | [156] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muskhelishvili, G.; Sobetzko, P.; Travers, A. Spatiotemporal Coupling of DNA Supercoiling and Genomic Sequence Organization—A Timing Chain for the Bacterial Growth Cycle? Biomolecules 2022, 12, 831. https://doi.org/10.3390/biom12060831
Muskhelishvili G, Sobetzko P, Travers A. Spatiotemporal Coupling of DNA Supercoiling and Genomic Sequence Organization—A Timing Chain for the Bacterial Growth Cycle? Biomolecules. 2022; 12(6):831. https://doi.org/10.3390/biom12060831
Chicago/Turabian StyleMuskhelishvili, Georgi, Patrick Sobetzko, and Andrew Travers. 2022. "Spatiotemporal Coupling of DNA Supercoiling and Genomic Sequence Organization—A Timing Chain for the Bacterial Growth Cycle?" Biomolecules 12, no. 6: 831. https://doi.org/10.3390/biom12060831






