Sialyl-Tn in Cancer: (How) Did We Miss the Target?
Abstract
:1. Introduction
2. Sialyl-Tn Structure and Biosynthesis
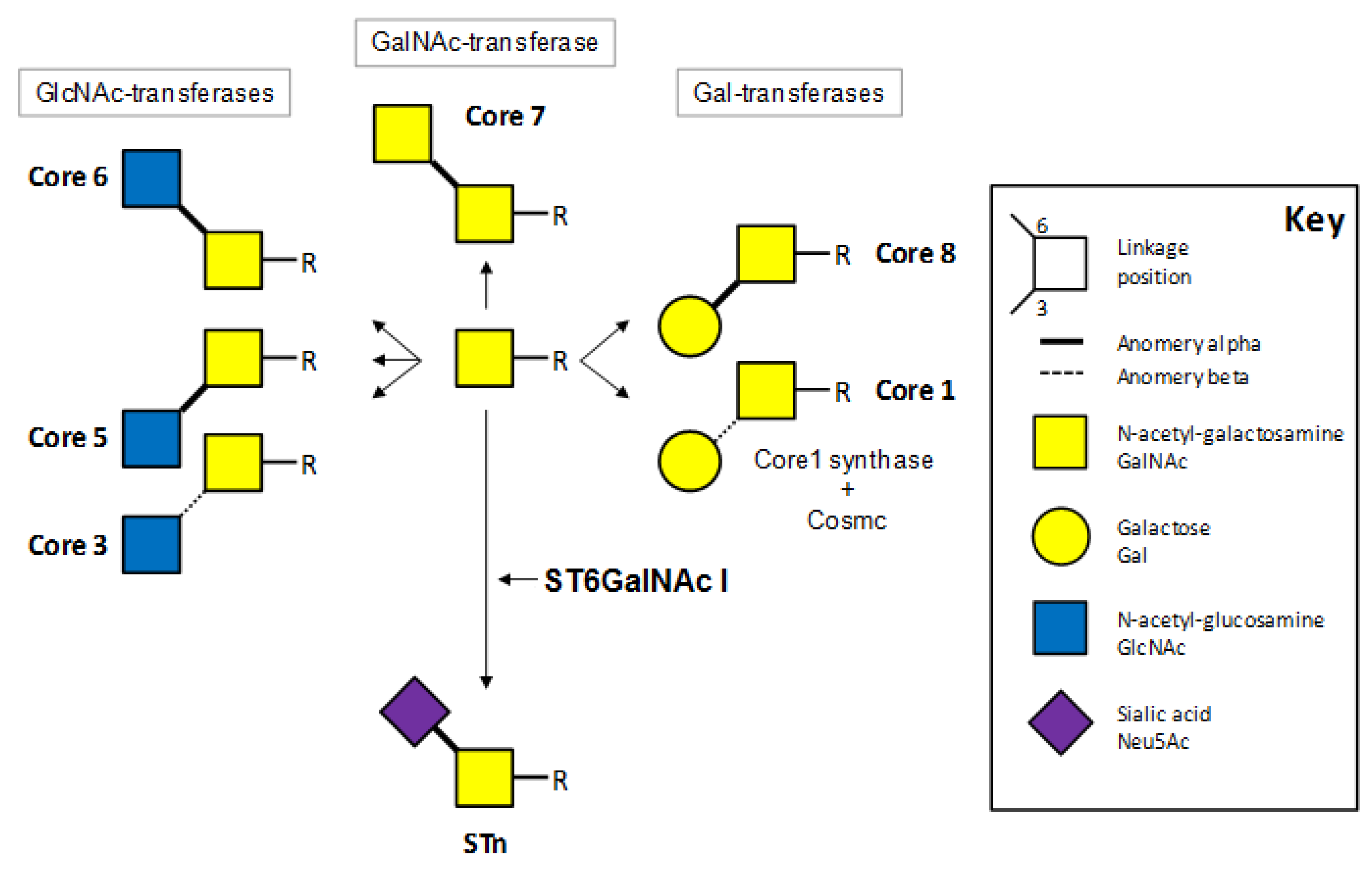
3. STn Immunodetection in Tissues and Tumors
3.1. Anti-STn Antibodies
| Monoclonal antibody | Immunogen | Isotype | Specificity | Ref. |
|---|---|---|---|---|
| B72.3 | Membrane fraction of breast cancer metastasis | Mouse IgG1 | Clustered STn bound to serine. Cross reacts with Tn clusters | [10,11] |
| MLS102 | LS 180 colonic cancer cells | Mouse IgG | Clustered STn, independently of the linkage to the peptide backbone. | [12,13] |
| TKH2 | Ovine submaxillary mucin | Mouse IgG | Monomeric STn | [14,15] |
| HB-STn1 | Ovine submaxillary mucin | Mouse IgG1 | [16] |
3.2. Expression in Normal Tissues
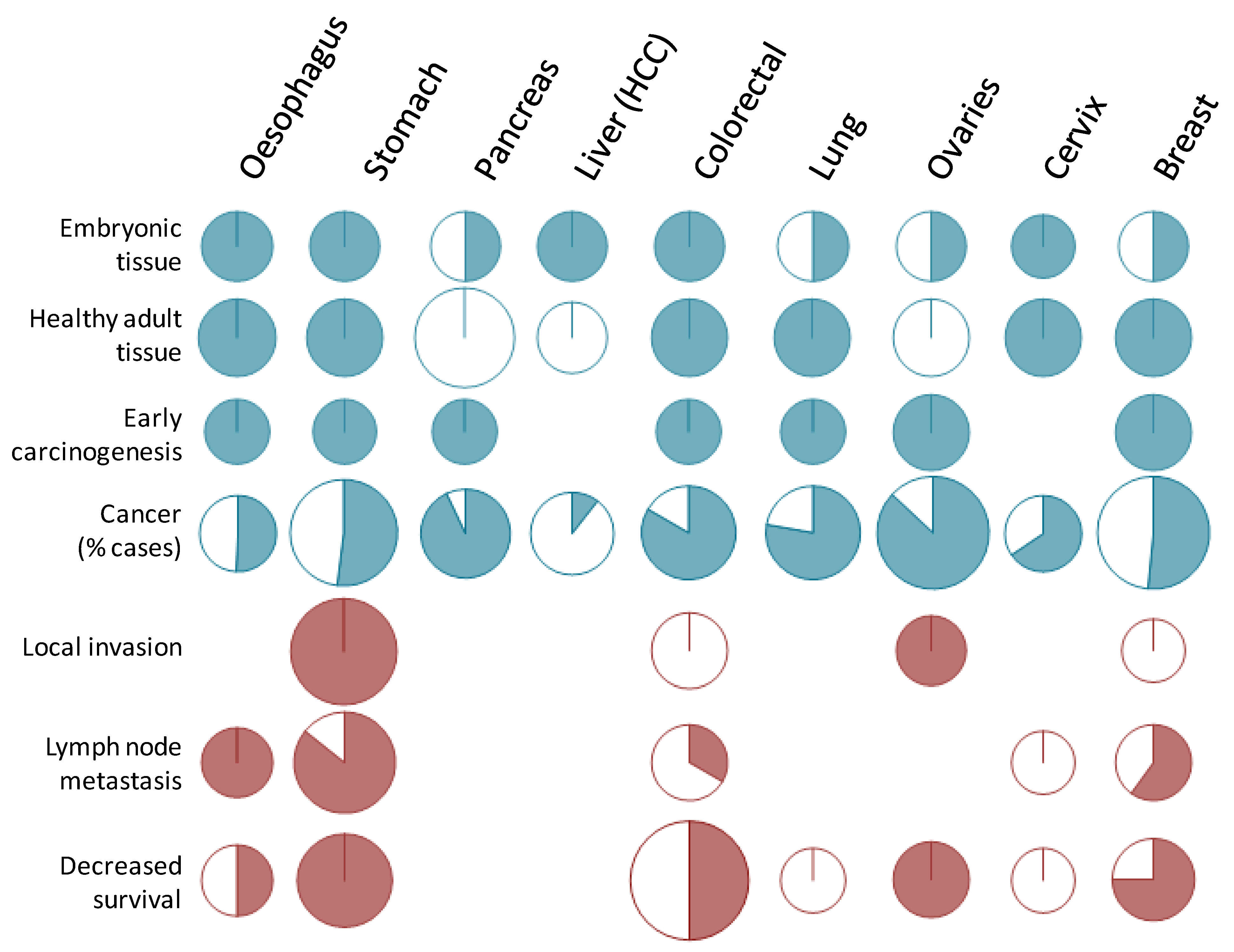
3.2.1. Fetal Tissues
3.2.2. Adult Healthy Tissues
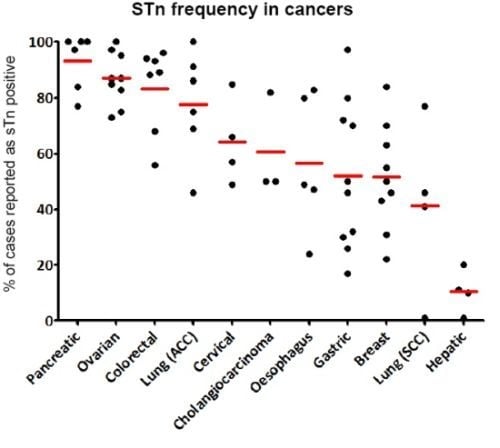
3.3. STn Expression in Cancers
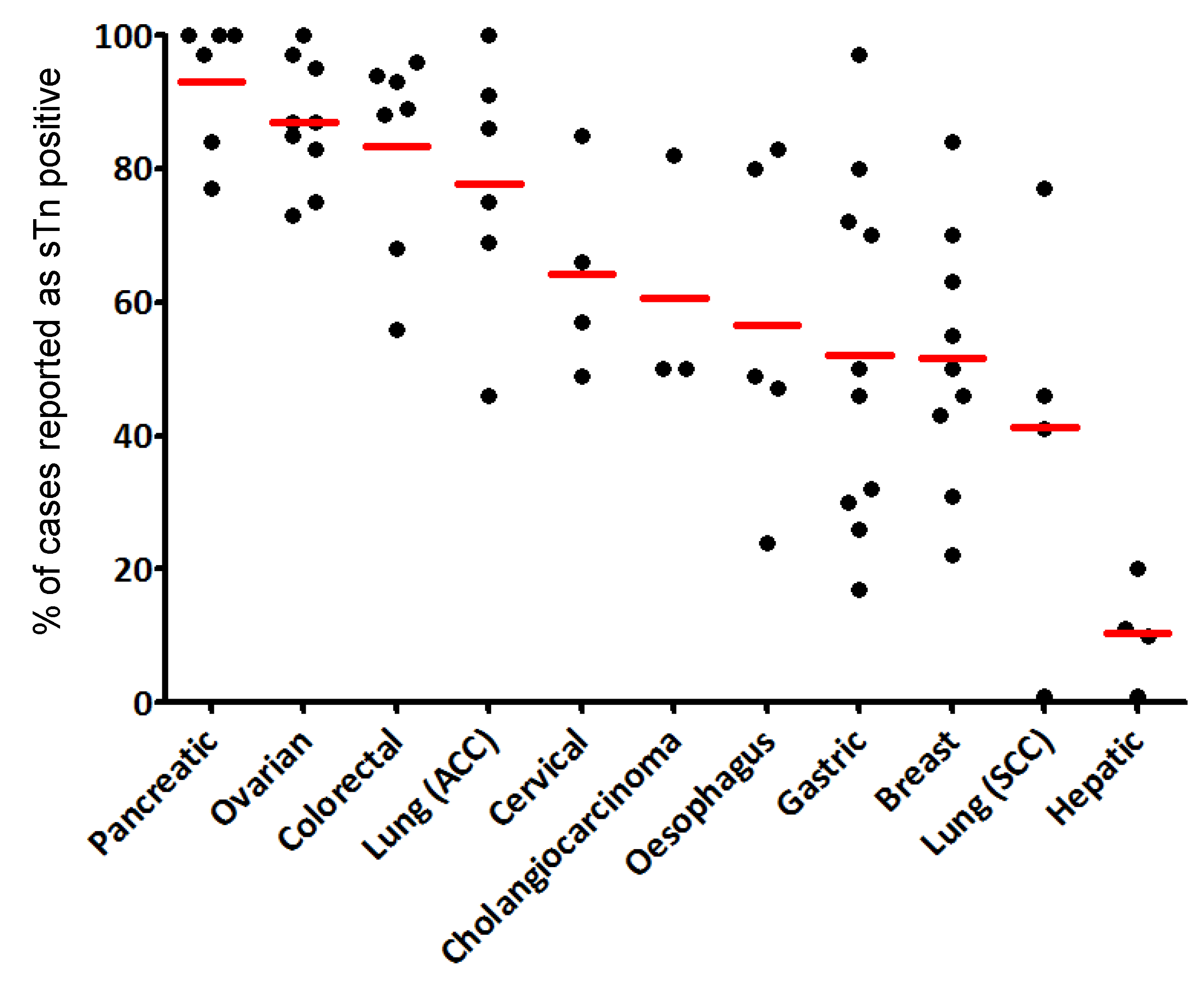
3.3.1. Early in Carcinogenesis
3.3.2. Cytological Types of Carcinomas
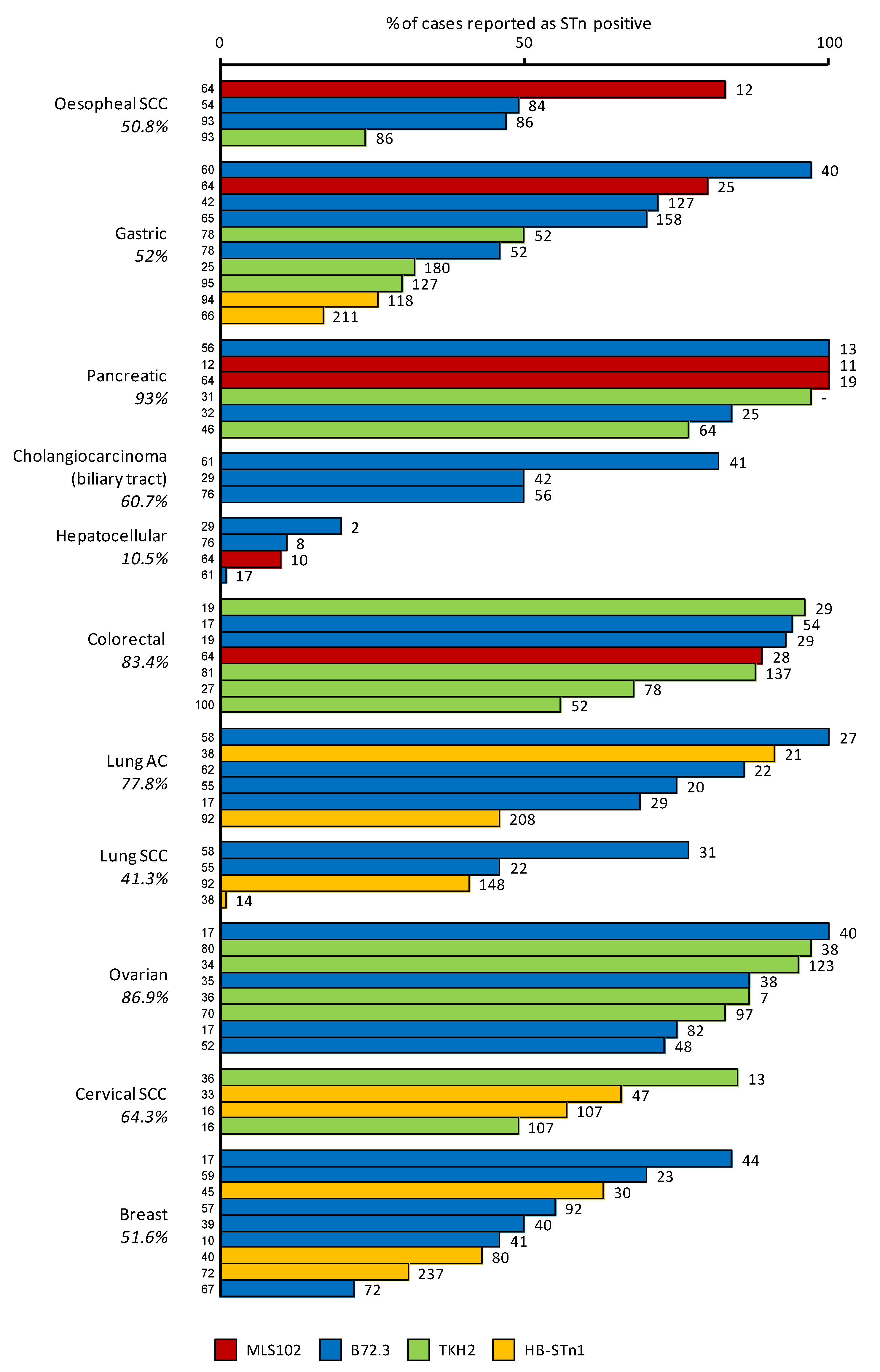
3.3.3. Cell Differentiation
3.3.4. Heterogeneity within Tumors
3.3.5. Association with Invasiveness
3.3.6. Detection in Serum
3.3.7. Prognosis Value of STn in Cancers
4. Immunotherapy Targeting STn
4.1. Theratope
4.1.1. Theratope in Clinical Trials
4.1.2. Highlights on the Theratope Mechanism
4.2. What Can Be Improved in Anti-STn Approaches?
4.2.1. Inducing Better Immune Responses
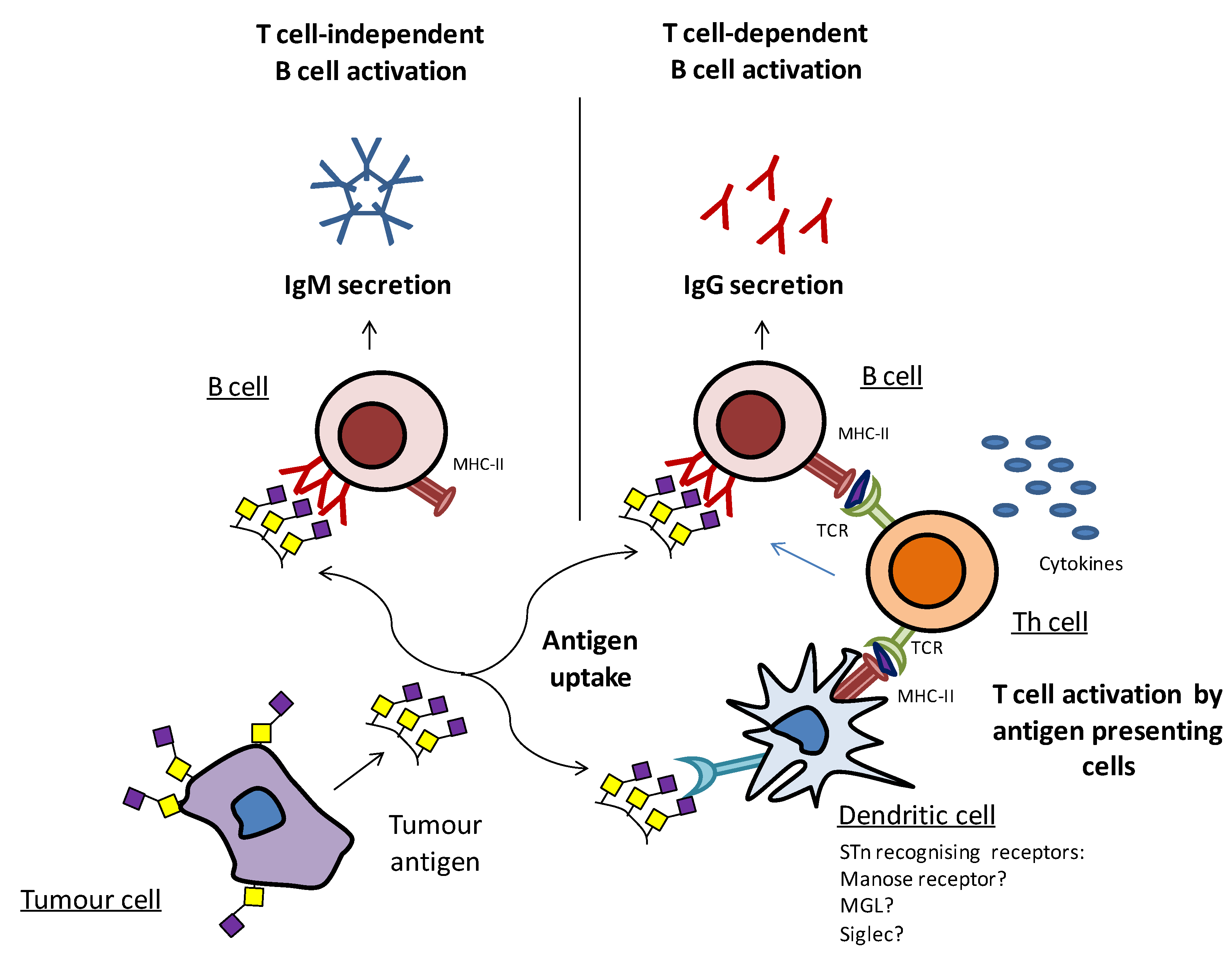
4.2.2. Co-targeting STn and Relevant Glycoproteins
4.2.3. Combining STn with other Cancer Associated Carbohydrates
4.2.4. Whom to Treat with Anti-STn Vaccine?
5. General Conclusion
Acknowledgments
References
- Rajpert-De Meyts, E.; Poll, S.N.; Goukasian, I.; Jeanneau, C.; Herlihy, A.S.; Bennett, E.P.; Skakkebaek, N.E.; Clausen, H.; Giwercman, A.; Mandel, U. Changes in the profile of simple mucin-type O-glycans and polypeptide GalNAc-transferases in human testis and testicular neoplasms are associated with germ cell maturation and tumour differentiation. Virchows Arch. 2007, 451, 805–814. [Google Scholar] [CrossRef]
- Marcos, N.T.; Pinho, S.; Grandela, C.; Cruz, A.; Samyn-Petit, B.; Harduin-Lepers, A.; Almeida, R.; Silva, F.; Morais, V.; Costa, J.; Kihlberg, J.; Clausen, H.; Reis, C.A. Role of the human ST6GalNAc-I and ST6GalNAc-II in the synthesis of the cancer-associated sialyl-Tn antigen. Cancer Res. 2004, 64, 7050–7057. [Google Scholar]
- Harduin-Lepers, A.; Vallejo-Ruiz, V.; Krzewinski-Recchi, M.A.; Samyn-Petit, B.; Julien, S.; Delannoy, P. The human sialyltransferase family. Biochimie 2001, 83, 727–737. [Google Scholar] [CrossRef]
- Sewell, R.; Bäckström, M.; Dalziel, M.; Gschmeissner, S.; Karlsson, H.; Noll, T.; Gätgens, J.; Clausen, H.; Hansson, G.C.; Burchell, J.; Taylor-Papadimitriou, J. The ST6GalNAc-I sialyltransferase localizes throughout the Golgi and is responsible for the synthesis of the tumor-associated sialyl-Tn O-glycan in human breast cancer. J. Biol. Chem. 2006, 281, 3586–3594. [Google Scholar]
- Marcos, N.T.; Bennett, E.P.; Gomes, J.; Magalhaes, A.; Gomes, C.; David, L.; Dar, I.; Jeanneau, C.; DeFrees, S.; Krustrup, D.; Vogel, L.K.; Kure, E.H.; Burchell, J.; Taylor-Papadimitriou, J.; Clausen, H.; Mandel, U.; Reis, C.A. ST6GalNAc-I controls expression of sialyl-Tn antigen in gastrointestinal tissues. Front Biosci. (Elite Ed.) 2011, 3, 1443–1455. [Google Scholar]
- Julien, S.; Adriaenssens, E.; Ottenberg, K.; Furlan, A.; Courtand, G.; Vercoutter-Edouart, A.-S.; Hanisch, F.-G.; Delannoy, P.; Le Bourhis, X. ST6GalNAc I expression in MDA-MB-231 breast cancer cells greatly modifies their O-glycosylation pattern and enhances their tumourigenicity. Glycobiology 2006, 16, 54–64. [Google Scholar]
- Ju, T.; Cummings, R.D. A unique molecular chaperone Cosmc required for activity of the mammalian core 1 beta 3-galactosyltransferase. Proc. Natl. Acad. Sci. USA 2002, 99, 16613–16618. [Google Scholar] [CrossRef]
- Ju, T.; Lanneau, G.S.; Gautam, T.; Wang, Y.; Xia, B.; Stowell, S.R.; Willard, M.T.; Wang, W.; Xia, J.Y.; Zuna, R.E.; Laszik, Z.; Benbrook, D.M.; Hanigan, M.H.; Cummings, R.D. Human tumor antigens Tn and sialyl Tn arise from mutations in Cosmc. Cancer Res. 2008, 68, 1636–1646. [Google Scholar]
- Yoo, N.J.; Kim, M.S.; Lee, S.H. Absence of COSMC gene mutations in breast and colorectal carcinomas. APMIS 2008, 116, 154–155. [Google Scholar] [CrossRef]
- Nuti, M.; Teramoto, Y.A.; Mariani-Costantini, R.; Hand, P.H.; Colcher, D.; Schlom, J. A monoclonal antibody (B72.3) defines patterns of distribution of a novel tumor-associated antigen in human mammary carcinoma cell populations. Int. J. Cancer 1982, 29, 539–545. [Google Scholar] [CrossRef]
- Reddish, M.A.; Jackson, L.; Koganty, R.R.; Qiu, D.; Hong, W.; Longenecker, B.M. Specificities of anti-sialyl-Tn and anti-Tn monoclonal antibodies generated using novel clustered synthetic glycopeptide epitopes. Glycoconj. J. 1997, 14, 549–560. [Google Scholar] [CrossRef]
- Ching, C.K.; Holmes, S.W.; Holmes, G.K.; Long, R.G. Comparison of two sialosyl-Tn binding monoclonal antibodies (MLS102 and B72.3) in detecting pancreatic cancer. Gut 1993, 34, 1722–1725. [Google Scholar] [CrossRef]
- Kurosaka, A.; Kitagawa, H.; Fukui, S.; Numata, Y.; Nakada, H.; Funakoshi, I.; Kawasaki, T.; Ogawa, T.; Iijima, H.; Yamashina, I. A monoclonal antibody that recognizes a cluster of a disaccharide, NeuAc alpha(2----6)GalNAc, in mucin-type glycoproteins. J. Biol. Chem. 1988, 263, 8724–8726. [Google Scholar]
- Kjeldsen, T.; Clausen, H.; Hirohashi, S.; Ogawa, T.; Iijima, H.; Hakomori, S. Preparation and characterization of monoclonal antibodies directed to the tumor-associated O-linked sialosyl-2—6 alpha-N-acetylgalactosaminyl (sialosyl-Tn) epitope. Cancer Res. 1988, 48, 2214–2220. [Google Scholar]
- Ogata, S.; Koganty, R.; Reddish, M.; Longenecker, B.M.; Chen, A.; Perez, C.; Itzkowitz, S.H. Different modes of sialyl-Tn expression during malignant transformation of human colonic mucosa. Glycoconj. J. 1998, 15, 29–35. [Google Scholar] [CrossRef]
- Terasawa, K.; Furumoto, H.; Kamada, M.; Aono, T. Expression of Tn and sialyl-Tn antigens in the neoplastic transformation of uterine cervical epithelial cells. Cancer Res. 1996, 56, 2229–2232. [Google Scholar]
- Thor, A.; Ohuchi, N.; Szpak, C.A.; Johnston, W.W.; Schlom, J. Distribution of oncofetal antigen tumor-associated glycoprotein-72 defined by monoclonal antibody B72.3. Cancer Res. 1986, 46, 3118–3124. [Google Scholar]
- Stanick, D.; Schuss, A.; Mishriki, Y.; Chao, S.; Thor, A.; Lundy, J. Reactivity of the monoclonal antibody B72.3 with fetal antigen: correlation with expression of TAG-72 in human carcinomas. Cancer Invest. 1988, 6, 279–287. [Google Scholar] [CrossRef]
- Itzkowitz, S.H.; Yuan, M.; Montgomery, C.K.; Kjeldsen, T.; Takahashi, H.K.; Bigbee, W.L.; Kim, Y.S. Expression of Tn, sialosyl-Tn, and T antigens in human colon cancer. Cancer Res. 1989, 49, 197–204. [Google Scholar]
- Pistolesi, S.; Nuti, M.; Castagna, M.; Pingitore, R. Mammary fetal gland: identification of new oncofetal antigens by monoclonal antibodies B72.3, MM1.80 and 4.36. Tumori 2001, 87, 252–255. [Google Scholar]
- Baldus, S.E.; Hanisch, F.G.; Schwonzen, M.; Nakahara, Y.; Iijima, H.; Ogawa, T.; Peter-Katalinic, J.; Uhlenbruck, G. Monoclonal antibody SP-21 defines a sialosyl-Tn antigen expressed on carcinomas and K562 erythroleukemia cells. Anticancer Res. 1992, 12, 1935–1940. [Google Scholar]
- Cao, Y.; Stosiek, P.; Springer, G.F.; Karsten, U. Thomsen-Friedenreich-related carbohydrate antigens in normal adult human tissues: a systematic and comparative study. Histochem. Cell Biol. 1996, 106, 197–207. [Google Scholar] [CrossRef]
- Itoh, T.; Yonezawa, S.; Nomoto, M.; Ueno, K.; Kim, Y.S.; Sato, E. Expression of mucin antigens and Lewis X-related antigens in carcinomas and dysplasia of the pharynx and larynx. Pathol. Int. 1996, 46, 646–655. [Google Scholar] [CrossRef]
- Kirkeby, S.; Moe, D.; Bardow, A. MUC1 and the simple mucin-type antigens: Tn and Sialyl-Tn are differently expressed in salivary gland acini and ducts from the submandibular gland, the vestibular folds, and the soft palate. Arch. Oral Biol. 2010, 55, 830–841. [Google Scholar] [CrossRef]
- Kakeji, Y.; Maehara, Y.; Morita, M.; Matsukuma, A.; Furusawa, M.; Takahashi, I.; Kusumoto, T.; Ohno, S.; Sugimachi, K. Correlation between sialyl Tn antigen and lymphatic metastasis in patients with Borrmann type IV gastric carcinoma. Br. J. Cancer 1995, 71, 191–195. [Google Scholar] [CrossRef]
- Victorzon, M.; Nordling, S.; Nilsson, O.; Roberts, P.J.; Haglund, C. Sialyl Tn antigen is an independent predictor of outcome in patients with gastric cancer. Int. J. Cancer 1996, 65, 295–300. [Google Scholar] [CrossRef]
- Imada, T.; Rino, Y.; Hatori, S.; Takahashi, M.; Amano, T.; Kondo, J.; Suda, T. Sialyl Tn antigen expression is associated with the prognosis of patients with advanced colorectal cancer. Hepatogastroenterology 1999, 46, 208–214. [Google Scholar]
- Jass, J.R.; Allison, L.J.; Edgar, S.G. Distribution of sialosyl Tn and Tn antigens within normal and malignant colorectal epithelium. J. Pathol. 1995, 176, 143–149. [Google Scholar] [CrossRef]
- Cao, Y.; Karsten, U.; Otto, G.; Bannasch, P. Expression of MUC1, Thomsen-Friedenreich antigen, Tn, sialosyl-Tn, and alpha2,6-linked sialic acid in hepatocellular carcinomas and preneoplastic hepatocellular lesions. Virchows Arch. 1999, 434, 503–509. [Google Scholar] [CrossRef]
- Siddiki, B.; Ho, J.J.; Huang, J.; Byrd, J.C.; Lau, E.; Yuan, M.; Kim, Y.S. Monoclonal antibody directed against colon cancer mucin has high specificity for malignancy. Int. J. Cancer 1993, 54, 467–474. [Google Scholar] [CrossRef]
- Itzkowitz, S.; Kjeldsen, T.; Friera, A.; Hakomori, S.; Yang, U.S.; Kim, Y.S. Expression of Tn, sialosyl Tn, and T antigens in human pancreas. Gastroenterology 1991, 100, 1691–1700. [Google Scholar]
- Lyubsky, S.; Madariaga, J.; Lozowski, M.; Mishriki, Y.; Schuss, A.; Chao, S.; Lundy, J. A tumor-associated antigen in carcinoma of the pancreas defined by monoclonal antibody B72.3. Am. J. Clin. Pathol. 1988, 89, 160–167. [Google Scholar]
- Carrilho, C.; Cantel, M.; Gouveia, P.; David, L. Simple mucin-type carbohydrate antigens (Tn, sialosyl-Tn, T and sialosyl-T) and gp 230 mucin-like glycoprotein are candidate markers for neoplastic transformation of the human cervix. Virchows Arch. 2000, 437, 173–179. [Google Scholar] [CrossRef]
- Tashiro, Y.; Yonezawa, S.; Kim, Y.S.; Sato, E. Immunohistochemical study of mucin carbohydrates and core proteins in human ovarian tumors. Hum. Pathol. 1994, 25, 364–372. [Google Scholar] [CrossRef]
- Ghazizadeh, M.; Ogawa, H.; Sasaki, Y.; Araki, T.; Aihara, K. Mucin carbohydrate antigens (T, Tn, and sialyl-Tn) in human ovarian carcinomas: relationship with histopathology and prognosis. Hum. Pathol. 1997, 28, 960–966. [Google Scholar] [CrossRef]
- Numa, F.; Tsunaga, N.; Michioka, T.; Nawata, S.; Ogata, H.; Kato, H. Tissue expression of Sialyl Tn antigen in gynecologic tumors. J. Obstet. Gynaecol. (Tokyo 1995) 1995, 21, 385–389. [Google Scholar]
- Ogawa, H.; Ghazizadeh, M.; Araki, T. Tn and sialyl-Tn antigens as potential prognostic markers in human ovarian carcinoma. Gynecol. Obstet. Invest. 1996, 41, 278–283. [Google Scholar] [CrossRef]
- Hachiya, T.; Honda, T.; Kubo, K.; Sekiguchi, M. Expression patterns of type II pneumocyte apical surface glycoconjugates in lung adenocarcinoma cells. Virchows Arch. 1999, 434, 63–69. [Google Scholar] [CrossRef]
- Cho, S.H.; Sahin, A.; Hortobagyi, G.N.; Hittelman, W.N.; Dhingra, K. Sialyl-Tn antigen expression occurs early during human mammary carcinogenesis and is associated with high nuclear grade and aneuploidy. Cancer Res. 1994, 54, 6302–6305. [Google Scholar]
- Schmitt, F.C.; Figueiredo, P.; Lacerda, M. Simple mucin-type carbohydrate antigens (T, sialosyl-T, Tn and sialosyl-Tn) in breast carcinogenesis. Virchows Arch. 1995, 427, 251–258. [Google Scholar]
- Reed, W.; Bryne, M.; Clausen, H.; Dabelsteen, E.; Nesland, J.M. Simple mucins (T, sialosyl-T, Tn and sialosyl-Tn) are not diagnostic for malignant breast lesions. Anticancer Res. 1994, 14, 609–615. [Google Scholar]
- Baldus, S.E.; Zirbes, T.K.; Mönig, S.P.; Engel, S.; Monaca, E.; Rafiqpoor, K.; Hanisch, F.G.; Hanski, C.; Thiele, J.; Pichlmaier, H.; Dienes, H.P. Histopathological subtypes and prognosis of gastric cancer are correlated with the expression of mucin-associated sialylated antigens: Sialosyl-Lewis(a), Sialosyl-Lewis(x) and sialosyl-Tn. Tumour Biol. 1998, 19, 445–453. [Google Scholar] [CrossRef]
- Cao, Y.; Schlag, P.M.; Karsten, U. Immunodetection of epithelial mucin (MUC1, MUC3) and mucin-associated glycotopes (TF, Tn, and sialosyl-Tn) in benign and malignant lesions of colonic epithelium: apolar localization corresponds to malignant transformation. Virchows Arch. 1997, 431, 159–166. [Google Scholar] [CrossRef]
- Castagna, M.; Nuti, M.; Squartini, F. Mammary cancer antigen recognized by monoclonal antibody B72.3 in apocrine metaplasia of the human breast. Cancer Res. 1987, 47, 902–906. [Google Scholar]
- Schmitt, F.C.; Marinho, A.; Amendoeira, I. Expression of sialyl-Tn in fine-needle aspirates from mammographically detected breast lesions: a marker of malignancy? Diagn. Cytopathol. 1998, 18, 325–329. [Google Scholar] [CrossRef]
- Kim, G.E.; Bae, H.-I.; Park, H.-U.; Kuan, S.-F.; Crawley, S.C.; Ho, J.J.L.; Kim, Y.S. Aberrant expression of MUC5AC and MUC6 gastric mucins and sialyl Tn antigen in intraepithelial neoplasms of the pancreas. Gastroenterology 2002, 123, 1052–1060. [Google Scholar] [CrossRef]
- Hruban, R.H.; Goggins, M.; Parsons, J.; Kern, S.E. Progression model for pancreatic cancer. Clin. Cancer Res. 2000, 6, 2969–2972. [Google Scholar]
- Barresi, G.; Giuffrè, G.; Vitarelli, E.; Grosso, M.; Tuccari, G. The immunoexpression of Tn, sialyl-Tn and T antigens in chronic active gastritis in relation to Helicobacter pylori infection. Pathology 2001, 33, 298–302. [Google Scholar]
- Cohen, M.; Drut, R.; Cueto Rúa, E. SIALYL-Tn antigen distribution in Helicobacter pylori chronic gastritis in children: an immunohistochemical study. Pediatr. Pathol. Mol. Med. 2003, 22, 117–129. [Google Scholar] [CrossRef]
- Karlén, P.; Young, E.; Broström, O.; Löfberg, R.; Tribukait, B.; Ost, K.; Bodian, C.; Itzkowitz, S. Sialyl-Tn antigen as a marker of colon cancer risk in ulcerative colitis: relation to dysplasia and DNA aneuploidy. Gastroenterology 1998, 115, 1395–1404. [Google Scholar] [CrossRef]
- Ta, A.; Harpaz, N.; Bodian, C.; Roston, A.; Oberman, L.; Chen, A.; Itzkowitz, S. Sialyl-tn antigen expression in Crohn’s colitis. Inflam. Bowel Dis. 1997, 3, 254–259. [Google Scholar] [CrossRef]
- Federici, M.F.; Kudryashov, V.; Saigo, P.E.; Finstad, C.L.; Lloyd, K.O. Selection of carbohydrate antigens in human epithelial ovarian cancers as targets for immunotherapy: serous and mucinous tumors exhibit distinctive patterns of expression. Int. J. Cancer 1999, 81, 193–198. [Google Scholar] [CrossRef]
- Pinto, R.; Carvalho, A.S.; Conze, T.; Magalhães, A.; Picco, G.; Burchell, J.M.; Taylor-Papadimitriou, J.; Reis, C.A.; Almeida, R.; Mandel, U.; Clausen, H.; Söderberg, O.; David, L. Identification of new cancer biomarkers based on aberrant mucin glycoforms by in situ Proximity Ligation. J. Cell. Mol. Med. 2011, 16, 1474–1484. [Google Scholar]
- Flucke, U.; Zirbes, T.K.; Schröder, W.; Mönig, S.P.; Koch, V.; Schmitz, K.; Thiele, J.; Dienes, H.P.; Hölscher, A.H.; Baldus, S.E. Expression of mucin-associated carbohydrate core antigens in esophageal squamous cell carcinomas. Anticancer Res. 2001, 21, 2189–2193. [Google Scholar]
- López-Ferrer, A.; Barranco, C.; de Bolós, C. Differences in the O-glycosylation patterns between lung squamous cell carcinoma and adenocarcinoma. Am. J. Clin. Pathol. 2002, 118, 749–755. [Google Scholar] [CrossRef]
- Ching, C.K.; Holmes, S.W.; Holmes, G.K.; Long, R.G. Blood-group sialyl-Tn antigen is more specific than Tn as a tumor marker in the pancreas. Pancreas 1994, 9, 698–702. [Google Scholar] [CrossRef]
- Contegiacomo, A.; Alimandi, M.; Muraro, R.; Pizzi, C.; Calderopoli, R.; De Marchis, L.; Sgambato, A.; Pettinato, G.; Petrella, G.; De Filippo, M.R. Expression of epitopes of the tumour-associated glycoprotein 72 and clinicopathological correlations in mammary carcinomas. Eur. J. Cancer 1994, 30A, 813–820. [Google Scholar]
- Johnston, W.W.; Szpak, C.A.; Thor, A.; Schlom, J. Phenotypic characterization of lung cancers in fine needle aspiration biopsies using monoclonal antibody B72.3. Cancer Res. 1986, 46, 6462–6470. [Google Scholar]
- Lottich, S.C.; Johnston, W.W.; Szpak, C.A.; Delong, E.R.; Thor, A.; Schlom, J. Tumor-associated antigen TAG-72: correlation of expression in primary and metastatic breast carcinoma lesions. Breast Cancer Res. Treat. 1985, 6, 49–56. [Google Scholar] [CrossRef]
- Ohuchi, N.; Thor, A.; Nose, M.; Fujita, J.; Kyogoku, M.; Schlom, J. Tumor-associated glycoprotein (TAG-72) detected in adenocarcinomas and benign lesions of the stomach. Int. J. Cancer 1986, 38, 643–650. [Google Scholar] [CrossRef]
- Sasaki, M.; Yamato, T.; Nakanuma, Y. Expression of sialyl-Tn, Tn and T antigens in primary liver cancer. Pathol. Int. 1999, 49, 325–331. [Google Scholar] [CrossRef]
- Szpak, C.A.; Johnston, W.W.; Roggli, V.; Kolbeck, J.; Lottich, S.C.; Vollmer, R.; Thor, A.; Schlom, J. The diagnostic distinction between malignant mesothelioma of the pleura and adenocarcinoma of the lung as defined by a monoclonal antibody (B72.3). Am. J. Pathol. 1986, 122, 252–260. [Google Scholar]
- Ohshio, G.; Yoshioka, H.; Manabe, T.; Sakahara, H.; Yamabe, H.; Imamura, M.; Inoue, M.; Tanaka, N.; Nakada, H.; Yamashina, I. Expression of sialosyl-Tn antigen (monoclonal antibody MLS102 reactive) in normal tissues and malignant tumors of the digestive tract. J. Cancer Res. Clin. Oncol. 1994, 120, 325–330. [Google Scholar] [CrossRef]
- Miles, D.W.; Linehan, J.; Smith, P.; Filipe, I. Expression of sialyl-Tn in gastric cancer: correlation with known prognostic factors. Br. J. Cancer 1995, 71, 1074–1076. [Google Scholar] [CrossRef]
- Terashima, S.; Takano, Y.; Ohori, T.; Kanno, T.; Kimura, T.; Motoki, R.; Kawaguchi, T. Sialyl-Tn antigen as a useful predictor of poor prognosis in patients with advanced stomach cancer. Surg. Today 1998, 28, 682–686. [Google Scholar] [CrossRef]
- Imai, J.; Ghazizadeh, M.; Naito, Z.; Asano, G. Immunohistochemical expression of T, Tn and sialyl-Tn antigens and clinical outcome in human breast carcinoma. Anticancer Res. 2001, 21, 1327–1334. [Google Scholar]
- Leivonen, M.; Nordling, S.; Lundin, J.; von Boguslawski, K.; Haglund, C. STn and prognosis in breast cancer. Oncology 2001, 61, 299–305. [Google Scholar] [CrossRef]
- Lin, J.-C.; Liao, S.-K.; Lee, E.-H.; Hung, M.-S.; Sayion, Y.; Chen, H.-C.; Kang, C.-C.; Huang, L.-S.; Cherng, J.-M. Molecular events associated with epithelial to mesenchymal transition of nasopharyngeal carcinoma cells in the absence of Epstein-Barr virus genome. J. Biomed. Sci. 2009, 16, 105. [Google Scholar] [CrossRef]
- Davidson, B.; Berner, A.; Nesland, J.M.; Risberg, B.; Kristensen, G.B.; Tropé, C.G.; Bryne, M. Carbohydrate antigen expression in primary tumors, metastatic lesions, and serous effusions from patients diagnosed with epithelial ovarian carcinoma: evidence of up-regulated Tn and Sialyl Tn antigen expression in effusions. Hum. Pathol. 2000, 31, 1081–1087. [Google Scholar] [CrossRef]
- Kinney, A.Y.; Sahin, A.; Vernon, S.W.; Frankowski, R.F.; Annegers, J.F.; Hortobagyi, G.N.; Buzdar, A.U.; Frye, D.K.; Dhingra, K. The prognostic significance of sialyl-Tn antigen in women treated with breast carcinoma treated with adjuvant chemotherapy. Cancer 1997, 80, 2240–2249. [Google Scholar] [CrossRef]
- Miles, D.W.; Happerfield, L.C.; Smith, P.; Gillibrand, R.; Bobrow, L.G.; Gregory, W.M.; Rubens, R.D. Expression of sialyl-Tn predicts the effect of adjuvant chemotherapy in node-positive breast cancer. Br. J. Cancer 1994, 70, 1272–1275. [Google Scholar] [CrossRef]
- Takahashi, I.; Maehara, Y.; Kusumoto, T.; Yoshida, M.; Kakeji, Y.; Kusumoto, H.; Furusawa, M.; Sugimachi, K. Predictive value of preoperative serum sialyl Tn antigen levels in prognosis of patients with gastric cancer. Cancer 1993, 72, 1836–1840. [Google Scholar] [CrossRef]
- Nakagoe, T.; Sawai, T.; Tsuji, T.; Jibiki, M.-A.; Nanashima, A.; Yamaguchi, H.; Yasutake, T.; Ayabe, H.; Arisawa, K.; Ishikawa, H. Predictive factors for preoperative serum levels of sialy Lewis(x), sialyl Lewis(a) and sialyl Tn antigens in gastric cancer patients. Anticancer Res. 2002, 22, 451–458. [Google Scholar]
- Werther, J.L.; Tatematsu, M.; Klein, R.; Kurihara, M.; Kumagai, K.; Llorens, P.; Guidugli Neto, J.; Bodian, C.; Pertsemlidis, D.; Yamachika, T.; Kitou, T.; Itzkowitz, S. Sialosyl-Tn antigen as a marker of gastric cancer progression: an international study. Int. J. Cancer 1996, 69, 193–199. [Google Scholar] [CrossRef]
- Ma, X.C.; Terata, N.; Kodama, M.; Jancic, S.; Hosokawa, Y.; Hattori, T. Expression of sialyl-Tn antigen is correlated with survival time of patients with gastric carcinomas. Eur. J. Cancer 1993, 29A, 1820–1823. [Google Scholar]
- Imada, T.; Rino, Y.; Takahashi, M.; Hatori, S.; Shiozawa, M.; Amano, T.; Kondo, J. Serum CA 19-9, SLX, STN and CEA levels of the peripheral and the draining venous blood in gastric cancer. Hepatogastroenterology 1999, 46, 2086–2090. [Google Scholar]
- Ikeda, Y.; Mori, M.; Kamakura, T.; Saku, M.; Sugimachi, K. Immunohistochemical expression of sialyl Tn and sialyl Lewis(a) antigens in stromal tissue correlates with peritoneal dissemination in stage IV human gastric cancer. Eur. J. Surg. Oncol. 1995, 21, 168–175. [Google Scholar] [CrossRef]
- Ozaki, H.; Matsuzaki, H.; Ando, H.; Kaji, H.; Nakanishi, H.; Ikehara, Y.; Narimatsu, H. Enhancement of metastatic ability by ectopic expression of ST6GalNAcI on a gastric cancer cell line in a mouse model. Clin. Exp. Metastasis 2012, 29, 229–238. [Google Scholar]
- Davidson, B.; Gotlieb, W.H.; Ben-Baruch, G.; Kopolovic, J.; Goldberg, I.; Nesland, J.M.; Berner, A.; Bjåmer, A.; Bryne, M. Expression of carbohydrate antigens in advanced-stage ovarian carcinomas and their metastases-A clinicopathologic study. Gynecol. Oncol. 2000, 77, 35–43. [Google Scholar] [CrossRef]
- Itzkowitz, S.H.; Bloom, E.J.; Kokal, W.A.; Modin, G.; Hakomori, S.; Kim, Y.S. Sialosyl-Tn. A novel mucin antigen associated with prognosis in colorectal cancer patients. Cancer 1990, 66, 1960–1966. [Google Scholar] [CrossRef]
- Julien, S.; Lagadec, C.; Krzewinski-Recchi, M.-A.; Courtand, G.; Le Bourhis, X.; Delannoy, P. Stable expression of sialyl-Tn antigen in T47-D cells induces a decrease of cell adhesion and an increase of cell migration. Breast Cancer Res. Treat. 2005, 90, 77–84. [Google Scholar] [CrossRef]
- Motoo, Y.; Kawakami, H.; Watanabe, H.; Satomura, Y.; Ohta, H.; Okai, T.; Makino, H.; Toya, D.; Sawabu, N. Serum sialyl-Tn antigen levels in patients with digestive cancers. Oncology 1991, 48, 321–326. [Google Scholar] [CrossRef]
- Sato, T.; Nishimura, G.; Nonomura, A.; Miwa, K.; Miyazaki, I. Serological studies on CEA, CA 19-9, STn and SLX in colorectal cancer. Hepatogastroenterology 1999, 46, 914–919. [Google Scholar]
- Nanashima, A.; Yamaguchi, H.; Nakagoe, T.; Matsuo, S.; Sumida, Y.; Tsuji, T.; Sawai, T.; Yamaguchi, E.; Yasutake, T.; Ayabe, H. High serum concentrations of sialyl Tn antigen in carcinomas of the biliary tract and pancreas. J. Hepatobiliary Pancreat. Surg. 1999, 6, 391–395. [Google Scholar] [CrossRef]
- Inoue, M.; Ogawa, H.; Nakanishi, K.; Tanizawa, O.; Karino, K.; Endo, J. Clinical value of sialyl Tn antigen in patients with gynecologic tumors. Obstet. Gynecol. 1990, 75, 1032–1036. [Google Scholar]
- Kobayashi, H.; Terao, T.; Kawashima, Y. Serum sialyl Tn as an independent predictor of poor prognosis in patients with epithelial ovarian cancer. J. Clin. Oncol. 1992, 10, 95–101. [Google Scholar]
- Kudoh, K.; Kikuchi, Y.; Kita, T.; Tode, T.; Takano, M.; Hirata, J.; Mano, Y.; Yamamoto, K.; Nagata, I. Preoperative determination of several serum tumor markers in patients with primary epithelial ovarian carcinoma. Gynecol. Obstet. Invest. 1999, 47, 52–57. [Google Scholar] [CrossRef]
- Nakagoe, T.; Sawai, T.; Tsuji, T.; Jibiki, M.; Nanashima, A.; Yamaguchi, H.; Yasutake, T.; Ayabe, H.; Arisawa, K.; Ishikawa, H. Pre-operative serum levels of sialyl Tn antigen predict liver metastasis and poor prognosis in patients with gastric cancer. Eur. J. Surg. Oncol. 2001, 27, 731–739. [Google Scholar] [CrossRef]
- Maeda, K.; Chung, Y.; Onoda, N.; Nakanishi, I.; Nitta, A.; Arimoto, Y.; Yamada, N.; Kondo, Y.; Kato, Y.; Sowa, M. Serum sialyl-tn antigen level as a prognostic indicator in patients with gastric-cancer. Int. J. Oncol. 1994, 4, 129–132. [Google Scholar]
- Nakagoe, T.; Sawai, T.; Tsuji, T.; Jibiki, M.; Nanashima, A.; Yamaguchi, H.; Kurosaki, N.; Yasutake, T.; Ayabe, H.; Tagawa, Y. Prognostic value of circulating sialyl Tn antigen in colorectal cancer patients. Anticancer Res. 2000, 20, 3863–3869. [Google Scholar]
- D’Amico, T.A.; Aloia, T.A.; Moore, M.B.; Herndon, J.E., 2nd; Brooks, K.R.; Lau, C.L.; Harpole, D.H., Jr. Molecular biologic substaging of stage I lung cancer according to gender and histology. Ann. Thorac. Surg. 2000, 69, 882–886. [Google Scholar] [CrossRef]
- Ikeda, Y.; Kuwano, H.; Baba, K.; Ikebe, M.; Matushima, T.; Adachi, Y.; Mori, M.; Sugimachi, K. Expression of Sialyl-Tn antigens in normal squamous epithelium, dysplasia, and squamous cell carcinoma in the esophagus. Cancer Res. 1993, 53, 1706–1708. [Google Scholar]
- Takano, Y.; Teranishi, Y.; Terashima, S.; Motoki, R.; Kawaguchi, T. Lymph node metastasis-related carbohydrate epitopes of gastric cancer with submucosal invasion. Surg. Today 2000, 30, 1073–1082. [Google Scholar] [CrossRef]
- Tsuchiya, A.; Kikuchi, Y.; Ando, Y.; Abe, R. Correlation between expression of sialosyl-T antigen and survival in patients with gastric cancer. Br. J. Surg. 1995, 82, 960–962. [Google Scholar] [CrossRef]
- Nakagoe, T.; Sawai, T.; Tsuji, T.; Jibiki, M.; Nanashima, A.; Yamaguchi, H.; Kurosaki, N.; Yasutake, T.; Ayabe, H. Circulating sialyl Lewis(x), sialyl Lewis(a), and sialyl Tn antigens in colorectal cancer patients: multivariate analysis of predictive factors for serum antigen levels. J. Gastroenterol. 2001, 36, 166–172. [Google Scholar] [CrossRef]
- Vierbuchen, M.J.; Fruechtnicht, W.; Brackrock, S.; Krause, K.T.; Zienkiewicz, T.J. Quantitative lectin-histochemical and immunohistochemical studies on the occurrence of alpha(2,3)- and alpha(2,6)-linked sialic acid residues in colorectal carcinomas. Relation to clinicopathologic features. Cancer 1995, 76, 727–735. [Google Scholar] [CrossRef]
- Lundin, M.; Nordling, S.; Roberts, P.J.; Lundin, J.; Carpelan-Holmström, M.; von Boguslawsky, K.; Haglund, C. Sialyl Tn is a frequently expressed antigen in colorectal cancer: No correlation with patient prognosis. Oncology 1999, 57, 70–76. [Google Scholar] [CrossRef]
- Nakagoe, T.; Sawai, T.; Tsuji, T.; Jibiki, M.; Nanashima, A.; Yamaguchi, H.; Yasutake, T.; Ayabe, H.; Arisawa, K. Preoperative serum level of CA19-9 predicts recurrence after curative surgery in node-negative colorectal cancer patients. Hepatogastroenterology 2003, 50, 696–699. [Google Scholar]
- Akamine, S.; Nakagoe, T.; Sawai, T.; Tsuji, T.; Tanaka, K.; Hidaka, S.; Shibasaki, S.-I.; Nanashima, A.; Yamaguchi, H.; Nagayasu, T.; Yasutake, T. Differences in prognosis of colorectal cancer patients based on the expression of sialyl Lewisa, sialyl Lewisx and sialyl Tn antigens in serum and tumor tissue. Anticancer Res. 2004, 24, 2541–2546. [Google Scholar]
- Soares, R.; Marinho, A.; Schmitt, F. Expression of sialyl-Tn in breast cancer. Correlation with prognostic parameters. Pathol. Res. Pract. 1996, 192, 1181–1186. [Google Scholar] [CrossRef]
- Kalaaji, A.N. Cytokine therapy in advanced melanoma. J. Drugs Dermatol. 2007, 6, 374–378. [Google Scholar]
- MacLean, G.D.; Reddish, M.A.; Bowen-Yacyshyn, M.B.; Poppema, S.; Longenecker, B.M. Active specific immunotherapy against adenocarcinomas. Cancer Invest. 1994, 12, 46–56. [Google Scholar] [CrossRef]
- Hadden, J.W. The immunology and immunotherapy of breast cancer: an update. Int. J. Immunopharmacol. 1999, 21, 79–101. [Google Scholar] [CrossRef]
- Holmberg, L.A.; Sandmaier, B.M. Theratope vaccine (STn-KLH). Expert. Opin. Biol. Ther. 2001, 1, 881–891. [Google Scholar] [CrossRef]
- Mitchell, M.S. Cancer vaccines, a critical review--Part II. Curr. Opin. Investig. Drugs 2002, 3, 150–158. [Google Scholar]
- Morse, M.A. Technology evaluation: Theratope, Biomira Inc. Curr. Opin. Mol. Ther. 2000, 2, 453–458. [Google Scholar]
- Miles, D.; Roché, H.; Martin, M.; Perren, T.J.; Cameron, D.A.; Glaspy, J.; Dodwell, D.; Parker, J.; Mayordomo, J.; Tres, A.; Murray, J.L.; Ibrahim, N.K. Phase III multicenter clinical trial of the sialyl-TN (STn)-keyhole limpet hemocyanin (KLH) vaccine for metastatic breast cancer. Oncologist 2011, 16, 1092–1100. [Google Scholar] [CrossRef]
- Ragupathi, G.; Howard, L.; Cappello, S.; Koganty, R.R.; Qiu, D.; Longenecker, B.M.; Reddish, M.A.; Lloyd, K.O.; Livingston, P.O. Vaccines prepared with sialyl-Tn and sialyl-Tn trimers using the 4-(4-maleimidomethyl)cyclohexane-1-carboxyl hydrazide linker group result in optimal antibody titers against ovine submaxillary mucin and sialyl-Tn-positive tumor cells. Cancer Immunol. Immunother. 1999, 48, 1–8. [Google Scholar] [CrossRef]
- MacLean, G.D.; Miles, D.W.; Rubens, R.D.; Reddish, M.A.; Longenecker, B.M. Enhancing the effect of THERATOPE STn-KLH cancer vaccine in patients with metastatic breast cancer by pretreatment with low-dose intravenous cyclophosphamide. J. Immunother. Emphasis. Tumor Immunol. 1996, 19, 309–316. [Google Scholar] [CrossRef]
- Miles, D.W.; Towlson, K.E.; Graham, R.; Reddish, M.; Longenecker, B.M.; Taylor-Papadimitriou, J.; Rubens, R.D. A randomised phase II study of sialyl-Tn and DETOX-B adjuvant with or without cyclophosphamide pretreatment for the active specific immunotherapy of breast cancer. Br. J. Cancer 1996, 74, 1292–1296. [Google Scholar] [CrossRef]
- MacLean, G.D.; Reddish, M.A.; Koganty, R.R.; Longenecker, B.M. Antibodies against mucin-associated sialyl-Tn epitopes correlate with survival of metastatic adenocarcinoma patients undergoing active specific immunotherapy with synthetic STn vaccine. J. Immunother. Emphasis. Tumor Immunol. 1996, 19, 59–68. [Google Scholar] [CrossRef]
- Reddish, M.A.; MacLean, G.D.; Poppema, S.; Berg, A.; Longenecker, B.M. Pre-immunotherapy serum CA27.29 (MUC-1) mucin level and CD69+ lymphocytes correlate with effects of Theratope sialyl-Tn-KLH cancer vaccine in active specific immunotherapy. Cancer Immunol. Immunother. 1996, 42, 303–309. [Google Scholar] [CrossRef]
- Holmberg, L.A.; Sandmaier, B.M. Vaccination with Theratope (STn-KLH) as treatment for breast cancer. Expert Rev. Vaccines 2004, 3, 655–663. [Google Scholar] [CrossRef]
- Mitchell, M.S. Immunotherapy as part of combinations for the treatment of cancer. Int. Immunopharmacol. 2003, 3, 1051–1059. [Google Scholar] [CrossRef]
- Limited, A.I. Cancer vaccine THERATOPE- Biomira. Drugs R. D. 2003, 4, 236–240. [Google Scholar] [CrossRef]
- Untch, M.; Ditsch, N.; Hermelink, K. Immunotherapy: new options in breast cancer treatment. Expert Rev. Anticancer Ther. 2003, 3, 403–408. [Google Scholar] [CrossRef]
- Ibrahim, N.K.; Murray, J.L. Clinical development of the STn-KLH vaccine (Theratope). Clin. Breast Cancer 2003, 3 Suppl. 4, S139–S143. [Google Scholar] [CrossRef]
- Miles, D.; Papazisis, K. Rationale for the clinical development of STn-KLH (Theratope) and anti-MUC-1 vaccines in breast cancer. Clin. Breast Cancer 2003, 3 Suppl. 4, S134–S138. [Google Scholar] [CrossRef]
- Gradishar, W.J.; Sahmoud, T. Current and future perspectives on fulvestrant. Clin. Breast Cancer 2005, 6 Suppl. 1, S23–S29. [Google Scholar] [CrossRef]
- Holmberg, L.A.; Oparin, D.V.; Gooley, T.; Lilleby, K.; Bensinger, W.; Reddish, M.A.; MacLean, G.D.; Longenecker, B.M.; Sandmaier, B.M. Clinical outcome of breast and ovarian cancer patients treated with high-dose chemotherapy, autologous stem cell rescue and THERATOPE STn-KLH cancer vaccine. Bone Marrow Transplant. 2000, 25, 1233–1241. [Google Scholar] [CrossRef]
- Butts, C.A.; Mulder, K.E.; Fields, A.L.; Koski, S.; Scarfe, A.G.; Sawyer, M.B.; Au, H.-J.; Parker, J. Antibody responses to STn-KLH vaccine are produced in patients with metastatic colorectal cancer (CRC) receiving concurrent first-line chemotherapy (CX), ASCO Annual Meeting. In Proc. Am. Soc. Clin. Oncol., 2003 ASCO Annual Meeting, Chicago, IL, USA, 31 May –3 June; 2003; 22. abst. 684. [Google Scholar]
- Mayordomo, J.; Tres, A.; Miles, D.; Finke, L.; Jenkins, H. Long-term follow-up of patients concomitantly treated with hormone therapy in a prospective controlled randomized multicenter clinical study comparing STn-KLH vaccine with KLH control in stage IV breast cancer following first-line chemotherapy. In J. Clin. Oncol. (Post-Meeting Edition), 2004 ASCO Annual Meeting, New Orleans, LA, USA, 5–8 June, 2004; 22, p. 2603.
- Khanna, H.D.; Singh, S.; Agrawal, G.; Khanna, R.; Singh, L.P.; Khanna, R.; Khanna, S. Intra nuclear estrogen receptor status and its clinicopathological correlation in breast cancer patients. Cancer Biochem. Biophys. 1996, 15, 141–149. [Google Scholar]
- Martin, P.M.; Rolland, P.H.; Jacquemier, J.; Rolland, A.M.; Toga, M. Multiple steroid receptors in human breast cancer. III. Relationships between steroid receptors and the state of differentiation and the activity of carcinomas throughout the pathologic features. Cancer Chemother. Pharmacol. 1979, 2, 115–120. [Google Scholar] [CrossRef]
- Braun, D.P.; Crist, K.A.; Shaheen, F.; Staren, E.D.; Andrews, S.; Parker, J. Aromatase inhibitors increase the sensitivity of human tumor cells to monocyte-mediated, antibody-dependent cellular cytotoxicity. Am. J. Surg. 2005, 190, 570–571. [Google Scholar] [CrossRef]
- Julien, S.; Picco, G.; Sewell, R.; Vercoutter-Edouart, A.-S.; Tarp, M.; Miles, D.; Clausen, H.; Taylor-Papadimitriou, J.; Burchell, J.M. Sialyl-Tn vaccine induces antibody-mediated tumour protection in a relevant murine model. Br. J. Cancer 2009, 100, 1746–1754. [Google Scholar] [CrossRef]
- Herlyn, D.; Koprowski, H. IgG2a monoclonal antibodies inhibit human tumor growth through interaction with effector cells. Proc. Natl. Acad. Sci. USA 1982, 79, 4761–4765. [Google Scholar] [CrossRef]
- Blixt, O.; Bueti, D.; Burford, B.; Allen, D.; Julien, S.; Hollingsworth, M.; Gammerman, A.; Fentiman, I.; Taylor-Papadimitriou, J.; Burchell, J.M. Autoantibodies to aberrantly glycosylated MUC1 in early stage breast cancer are associated with a better prognosis. Breast Cancer Res. 2011, 13, R25. [Google Scholar] [CrossRef]
- Monti, P.; Leone, B.E.; Zerbi, A.; Balzano, G.; Cainarca, S.; Sordi, V.; Pontillo, M.; Mercalli, A.; Di Carlo, V.; Allavena, P.; Piemonti, L. Tumor-derived MUC1 mucins interact with differentiating monocytes and induce IL-10highIL-12low regulatory dendritic cell. J. Immunol. 2004, 172, 7341–7349. [Google Scholar]
- Lakshminarayanan, V.; Thompson, P.; Wolfert, M.A.; Buskas, T.; Bradley, J.M.; Pathangey, L.B.; Madsen, C.S.; Cohen, P.A.; Gendler, S.J.; Boons, G.-J. Immune recognition of tumor-associated mucin MUC1 is achieved by a fully synthetic aberrantly glycosylated MUC1 tripartite vaccine. Proc. Natl. Acad. Sci. USA 2012, 109, 261–266. [Google Scholar]
- Murphy, K.; Travers, P.; Walport, M.; Janeway, C. Janeway’s immunobiology; Garland Science: New York, NY, USA, 2012. [Google Scholar]
- Silva, Z.; Konstantopoulos, K.; Videira, P.A. The role of sugars in dendritic cell trafficking. Ann. Biomed. Eng. 2012, 40, 777–789. [Google Scholar] [CrossRef]
- Steinman, R.M.; Hawiger, D.; Nussenzweig, M.C. Tolerogenic dendritic cells. Annu. Rev. Immunol. 2003, 21, 685–711. [Google Scholar] [CrossRef]
- Adderson, E.E. Antibody repertoires in infants and adults: effects of T-independent and T-dependent immunizations. Springer Semin. Immunopathol. 2001, 23, 387–403. [Google Scholar] [CrossRef]
- Purcell, A.W.; McCluskey, J.; Rossjohn, J. More than one reason to rethink the use of peptides in vaccine design. Nat. Rev. Drug Discov. 2007, 6, 404–414. [Google Scholar] [CrossRef]
- Avci, F.Y.; Kasper, D.L. How bacterial carbohydrates influence the adaptive immune system. Annu. Rev. Immunol. 2010, 28, 107–130. [Google Scholar] [CrossRef]
- Liu, C.-C.; Ye, X.-S. Carbohydrate-based cancer vaccines: target cancer with sugar bullets. Glycoconj. J. 2012, 29, 259–271. [Google Scholar] [CrossRef]
- O’Boyle, K.P.; Zamore, R.; Adluri, S.; Cohen, A.; Kemeny, N.; Welt, S.; Lloyd, K.O.; Oettgen, H.F.; Old, L.J.; Livingston, P.O. Immunization of colorectal cancer patients with modified ovine submaxillary gland mucin and adjuvants induces IgM and IgG antibodies to sialylated Tn. Cancer Res. 1992, 52, 5663–5667. [Google Scholar]
- Lo-Man, R.; Vichier-Guerre, S.; Bay, S.; Dériaud, E.; Cantacuzène, D.; Leclerc, C. Anti-tumor immunity provided by a synthetic multiple antigenic glycopeptide displaying a tri-Tn glycotope. J. Immunol. 2001, 166, 2849–2854. [Google Scholar]
- Gaidzik, N.; Kaiser, A.; Kowalczyk, D.; Westerlind, U.; Gerlitzki, B.; Sinn, H.P.; Schmitt, E.; Kunz, H. Synthetic Antitumor Vaccines Containing MUC1 Glycopeptides with Two Immunodominant Domains-Induction of a Strong Immune Response against Breast Tumor Tissues. Angew. Chem. Int. Ed. Engl. 2011, 50, 9977–9981. [Google Scholar]
- Kagan, E.; Ragupathi, G.; Yi, S.S.; Reis, C.A.; Gildersleeve, J.; Kahne, D.; Clausen, H.; Danishefsky, S.J.; Livingston, P.O. Comparison of antigen constructs and carrier molecules for augmenting the immunogenicity of the monosaccharide epithelial cancer antigen Tn. Cancer Immunol. Immunother. 2005, 54, 424–430. [Google Scholar] [CrossRef]
- Kaiser, A.; Gaidzik, N.; Westerlind, U.; Kowalczyk, D.; Hobel, A.; Schmitt, E.; Kunz, H. A synthetic vaccine consisting of a tumor-associated sialyl-T(N)-MUC1 tandem-repeat glycopeptide and tetanus toxoid: induction of a strong and highly selective immune response. Angew. Chem. Int. Ed. Engl. 2009, 48, 7551–7555. [Google Scholar]
- Crespo, H.J.; Cabral, M.G.; Teixeira, A.V.; Lau, J.T.Y.; Trindade, H.; Videira, P.A. Effect of sialic acid loss on dendritic cell maturation. Immunology 2009, 128, e621–e631. [Google Scholar] [CrossRef]
- Videira, P.A. Departamento de Imunologia, Faculdade de Ciências Médicas, Universidade Nova de Lisboa, 1169-056 Lisboa, Portugal, 2012, Unpublished observation.
- Heimburg-Molinaro, J.; Lum, M.; Vijay, G.; Jain, M.; Almogren, A.; Rittenhouse-Olson, K. Cancer vaccines and carbohydrate epitopes. Vaccine 2011, 29, 8802–8826. [Google Scholar] [CrossRef]
- Yin, Z.; Huang, X. Recent Development in Carbohydrate Based Anti-cancer Vaccines. J. Carbohydr. Chem. 2012, 31, 143–186. [Google Scholar]
- Clément, M.; Rocher, J.; Loirand, G.; Le Pendu, J. Expression of sialyl-Tn epitopes on beta1 integrin alters epithelial cell phenotype, proliferation and haptotaxis. J. Cell. Sci. 2004, 117, 5059–5069. [Google Scholar] [CrossRef]
- Schultz, M.J.; Swindall, A.F.; Bellis, S.L. Regulation of the metastatic cell phenotype by sialylated glycans. Cancer Meta. Rev. 2012. [Google Scholar]
- Dudley, M.E.; Wunderlich, J.R.; Yang, J.C.; Sherry, R.M.; Topalian, S.L.; Restifo, N.P.; Royal, R.E.; Kammula, U.; White, D.E.; Mavroukakis, S.A.; Rogers, L.J.; Gracia, G.J.; Jones, S.A.; Mangiameli, D.P.; Pelletier, M.M.; Gea-Banacloche, J.; Robinson, M.R.; Berman, D.M.; Filie, A.C.; Abati, A.; Rosenberg, S.A. Adoptive cell transfer therapy following non-myeloablative but lymphodepleting chemotherapy for the treatment of patients with refractory metastatic melanoma. J. Clin. Oncol. 2005, 23, 2346–2357. [Google Scholar]
- Hollingsworth, M.A.; Swanson, B.J. Mucins in cancer: protection and control of the cell surface. Nat. Rev. Cancer 2004, 4, 45–60. [Google Scholar] [CrossRef]
- Burdick, M.D.; Harris, A.; Reid, C.J.; Iwamura, T.; Hollingsworth, M.A. Oligosaccharides expressed on MUC1 produced by pancreatic and colon tumor cell lines. J. Biol. Chem. 1997, 272, 24198–24202. [Google Scholar]
- Madan, R.A.; Arlen, P.M.; Gulley, J.L. PANVAC-VF: poxviral-based vaccine therapy targeting CEA and MUC1 in carcinoma. Expert Opin. Biol. Ther. 2007, 7, 543–554. [Google Scholar] [CrossRef]
- Liu, M.; Acres, B.; Balloul, J.-M.; Bizouarne, N.; Paul, S.; Slos, P.; Squiban, P. Gene-based vaccines and immunotherapeutics. Proc. Natl. Acad. Sci. USA 2004, 101 Suppl. 2, 14567–14571. [Google Scholar]
- Bradbury, P.A.; Shepherd, F.A. Immunotherapy for lung cancer. J. Thorac. Oncol. 2008, 3, S164–S170. [Google Scholar] [CrossRef]
- Beatson, R.E.; Taylor-Papadimitriou, J.; Burchell, J.M. MUC1 immunotherapy. Immunotherapy 2010, 2, 305–327. [Google Scholar] [CrossRef]
- Pinto, R.; Carvalho, A.S.; Conze, T.; Magalhães, A.; Picco, G.; Burchell, J.M.; Taylor-Papadimitriou, J.; Reis, C.A.; Almeida, R.; Mandel, U.; Clausen, H.; Söderberg, O.; David, L. Identification of new cancer biomarkers based on aberrant mucin glycoforms by in situ proximity ligation. J. Cell. Mol. Med. 2012, 16, 1474–1484. [Google Scholar] [CrossRef]
- Yang, E.; Hu, X.F.; Xing, P.X. Advances of MUC1 as a target for breast cancer immunotherapy. Histol. Histopathol. 2007, 22, 905–922. [Google Scholar]
- Irimura, T.; Denda, K.; Iida, S.; Takeuchi, H.; Kato, K. Diverse glycosylation of MUC1 and MUC2: potential significance in tumor immunity. J. Biochem. 1999, 126, 975–985. [Google Scholar] [CrossRef]
- Grote, T.; Logsdon, C.D. Progress on molecular markers of pancreatic cancer. Curr. Opin. Gastroenterol. 2007, 23, 508–514. [Google Scholar] [CrossRef]
- Hanisch, F.-G.; Ninkovic, T. Immunology of O-glycosylated proteins: approaches to the design of a MUC1 glycopeptide-based tumor vaccine. Curr. Protein Pept. Sci. 2006, 7, 307–315. [Google Scholar] [CrossRef]
- Ninkovic, T.; Hanisch, F.-G. O-glycosylated human MUC1 repeats are processed in vitro by immunoproteasomes. J. Immunol. 2007, 179, 2380–2388. [Google Scholar]
- Xu, Y.; Gendler, S.J.; Franco, A. Designer glycopeptides for cytotoxic T cell-based elimination of carcinomas. J. Exp. Med. 2004, 199, 707–716. [Google Scholar] [CrossRef]
- Karsten, U.; Serttas, N.; Paulsen, H.; Danielczyk, A.; Goletz, S. Binding patterns of DTR-specific antibodies reveal a glycosylation-conditioned tumor-specific epitope of the epithelial mucin (MUC1). Glycobiology 2004, 14, 681–692. [Google Scholar] [CrossRef]
- Pedersen, J.W.; Blixt, O.; Bennett, E.P.; Tarp, M.A.; Dar, I.; Mandel, U.; Poulsen, S.S.; Pedersen, A.E.; Rasmussen, S.; Jess, P.; Clausen, H.; Wandall, H.H. Seromic profiling of colorectal cancer patients with novel glycopeptide microarray. Int. J. Cancer 2011, 128, 1860–1871. [Google Scholar] [CrossRef]
- Cazet, A.; Julien, S.; Bobowski, M.; Burchell, J.; Delannoy, P. Tumour-associated carbohydrate antigens in breast cancer. Breast Cancer Res. 2010, 12, 204. [Google Scholar] [CrossRef]
- Westerlind, U.; Kunz, H. Synthetic vaccines based on N- and O-glycopeptides-molecular tools for immunotherapy and diagnostics. In Carbohydrate Chemistry; Pilar Rauter, A., Lindhorst, T., Eds.; Royal Society of Chemistry: Cambridge, UK, 2010; Volume 36, pp. 1–37. [Google Scholar]
- Niederhafner, P.; Reinis, M.; Sebestík, J.; Jezek, J. Glycopeptide dendrimers, part III: a review. Use of glycopeptide dendrimers in immunotherapy and diagnosis of cancer and viral diseases. J. Pept. Sci. 2008, 14, 556–587. [Google Scholar] [CrossRef]
- Slovin, S.F.; Ragupathi, G.; Fernandez, C.; Diani, M.; Jefferson, M.P.; Wilton, A.; Kelly, W.K.; Morris, M.; Solit, D.; Clausen, H.; Livingston, P.; Scher, H.I. A polyvalent vaccine for high-risk prostate patients: “are more antigens better?”. Cancer Immunol. Immunother. 2007, 56, 1921–1930. [Google Scholar] [CrossRef]
- Gray, A.; Yan, L.; Kast, W.M. Prevention is better than cure: the case for clinical trials of therapeutic cancer vaccines in the prophylactic setting. Mol. Interv. 2010, 10, 197–203. [Google Scholar] [CrossRef]
- Jaini, R.; Kesaraju, P.; Johnson, J.M.; Altuntas, C.Z.; Jane-Wit, D.; Tuohy, V.K. An autoimmune-mediated strategy for prophylactic breast cancer vaccination. Nat. Med. 2010, 16, 799–803. [Google Scholar] [CrossRef]
- Altuntas, C.Z.; Jaini, R.; Kesaraju, P.; Jane-wit, D.; Johnson, J.M.; Covey, K.; Flask, C.A.; Dutertre, M.; Picard, J.-Y.; Tuohy, V.K. Autoimmune mediated regulation of ovarian tumor growth. Gynecol. Oncol. 2012, 124, 98–104. [Google Scholar] [CrossRef]
- Adams, M.; Borysiewicz, L.; Fiander, A.; Man, S.; Jasani, B.; Navabi, H.; Lipetz, C.; Evans, A.S.; Mason, M. Clinical studies of human papilloma vaccines in pre-invasive and invasive cancer. Vaccine 2001, 19, 2549–2556. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Julien, S.; Videira, P.A.; Delannoy, P. Sialyl-Tn in Cancer: (How) Did We Miss the Target? Biomolecules 2012, 2, 435-466. https://doi.org/10.3390/biom2040435
Julien S, Videira PA, Delannoy P. Sialyl-Tn in Cancer: (How) Did We Miss the Target? Biomolecules. 2012; 2(4):435-466. https://doi.org/10.3390/biom2040435
Chicago/Turabian StyleJulien, Sylvain, Paula A. Videira, and Philippe Delannoy. 2012. "Sialyl-Tn in Cancer: (How) Did We Miss the Target?" Biomolecules 2, no. 4: 435-466. https://doi.org/10.3390/biom2040435